Potassium Bromate: Past, Present, and the Road Ahead
Historical Development
Potassium bromate came onto the chemical scene in the mid-1800s. A story unfolds from the early ambitions of baking scientists who tried to make bread rise fluffier and look whiter, especially in a time before modern additives changed the food industry. Bakers grabbed potassium bromate because it acted as a strong oxidizing agent, letting dough spring higher in the oven. As mass production of bread took off, bakeries needed reliable ways to improve product quality, especially in busy cities. Over decades, demand for processed foods rose, and so did potassium bromate’s popularity. Food manufacturers believed it improved consistency in baked goods. In regions with less strict food safety regulations, potassium bromate became a regular part of factory bread. Now, with global trade, some countries still allow its use, but others have phased it out due to safety concerns rooted in more recent scientific studies.
Product Overview
Potassium bromate is a white crystalline powder. It dissolves in water quickly and almost completely, mixing straight into dough. Factories add small, measured amounts. Its main chemical punch comes from its oxidizing nature, enhancing dough strength, elasticity, and shelf life in finished products. It keeps bread soft yet structured within industrial bakeries, where every loaf needs to look just right on the shelf. Beyond bread, its uses remain limited due to concerns about toxicity and close regulation. Throughout my time in pastry research, I came across colleagues who pointed out that in some countries, regulatory bodies insisted on labels if potassium bromate was in the recipe, driving bakers to seek out alternatives just to avoid complicated compliance steps.
Physical & Chemical Properties
This chemical shows up in storage rooms as small white granules or crystalline powder, almost odorless but nearly unmistakable for those who handle it often. On paper, potassium bromate has the molecular formula KBrO3, and weighs in at a molecular mass of about 167 g/mol. It melts around 350°C, though that rarely matters in baking setups. With its ability to act as a robust oxidizer, especially under the damp heat of baking, it interacts with dough proteins quickly and changes the physical texture of bread. While water dissolves it well, it proves less cooperative with organic solvents. Reactions speed up in warm and moist conditions, which fits bread-making perfectly. I remember a food chemist warning that potassium bromate could set off violent reactions when mixed carelessly with strong acids or reducing agents, so it calls for strict handling.
Technical Specifications & Labeling
Chemicals need to meet high purity standards for commercial use, especially in the food sector. Potassium bromate for bakeries comes with purity specifications often reaching above 99%. Manufacturers test batches for contaminants like heavy metals, because that can trigger regulatory headaches or worse, a public recall. Labeling rules set by agencies like the FDA and EFSA differ. In the U.S., its use in food is only allowed under strict limits and must be declared in the ingredients. In the European Union, food authorities banned potassium bromate outright, requiring bakers to drop it from recipes or face penalties. By the time it lands in a food manufacturing plant, it’s in sealed drums or special bags with hazard warnings and clear, regulated labeling. I’ve seen import/export headaches for companies who missed documentation or ended up with gray-market stock that didn’t meet these rules.
Preparation Method
Factories produce potassium bromate on a commercial scale by passing chlorine or bromine gas into a potassium hydroxide solution, triggering a chemical reaction that creates potassium bromate crystals. The process calls for skilled operators, good ventilation, and reliable equipment. After formation, the mixture is cooled so crystals can form, then filtered, washed, and dried. Any leftover chemicals or byproducts undergo proper treatment because regulations punish polluters harshly, especially for substances classified as hazardous. The method’s simplicity keeps costs down, but it places safety demands squarely on workers and managers, something my colleagues in chemical production never take for granted.
Chemical Reactions & Modifications
Potassium bromate acts as an oxidizer with a strong affinity for organic molecules, especially in the moist environments typical of dough. Inside bread, it helps form strong gluten networks by oxidizing sulfhydryl groups into disulfide bonds, giving the bread its signature chew and bounce. Outside the bakery, it reacts vigorously with reducing agents—like many sugars—and releases oxygen when heated or exposed to acidic conditions. That can be dangerous, as it increases combustion risks if stored alongside materials like paper, wood, or metal dusts. The chemical can be modified or substituted in certain processes by potassium iodate or other less controversial oxidizers; many researchers push for safer replacements. I sat on a panel years ago where food scientists debated whether reformulating bread recipes without potassium bromate really solved all quality issues. The consensus was that while change required investment in new technologies, the safety payoffs made the headache worthwhile.
Synonyms & Product Names
Chemists and manufacturers call potassium bromate by several names, including Bromic acid, potassium salt; E924; and KBrO3. On labels or in research papers, one sees it listed as potassium salt of bromic acid or even sometimes just its E number. Though the name changes from country to country, the chemical remains the same—and so do the risks. The branding doesn’t hide the controversy. In markets where regulations remain light, local versions appear under various trade names, sometimes causing confusion among importers and health inspectors alike.
Safety & Operational Standards
Because potassium bromate poses health risks, strict protocols surround its handling. Factories store it in cool, dry, well-ventilated areas far from combustible materials and acids. Workers wear gloves, goggles, and sometimes respirators to prevent breathing in dust, which can irritate airways or worse. Training programs drill employees on emergency measures, like how to handle spills and prevent fires. Occupational safety authorities in countries that still permit its use demand routine compliance checks. Regular air and residue testing limit workers’ long-term exposure, and many plants run internal audits on chemical stocks. In my years consulting on industrial safety, I saw that those who cut corners often suffered expensive accidents or regulatory blowback.
Application Area
Potassium bromate’s main application shows up in the large-scale baking industry, where it strengthens dough, boosts loaf volume, and enhances crumb texture, especially for sliced bread and sandwich loaves. Its role grew prominent in automated bakeries that rely on fast, standardized processes to deliver bread to supermarket shelves. It found less traction outside baked goods, because food chemists flagged its side effects, especially after testing linked it to potential health hazards. Globally, its use dropped off as awareness grew, but some bakeries in developing regions stick to it for cost reasons or due to slow regulatory response. Today, alternatives like ascorbic acid or enzyme-based oxidizers continue to carve out more market share, especially for health-focused consumers.
Research & Development
Scientific research on potassium bromate over the last few decades dug deep into both its bread-making benefits and its potential dangers. Academics and corporate partners funded studies to measure how much residual bromate remains in baked goods after cooking, hoping that modern ovens could destroy it all before bread reaches the customer. The results came back uneven. Some testing found traces in finished loaves, sparking media attention and renewed regulatory review. Research teams moved quickly to develop improved analytical methods—using high-performance liquid chromatography and mass spectrometry—to catch even small amounts. At food science conferences, I found myself at roundtable discussions on replacing potassium bromate without ruining bread quality. The push toward more natural, “clean label” formulations grew from consumer demand and industry reputation management, not just regulatory threat.
Toxicity Research
Health researchers found that potassium bromate can harm lab animals, leading to higher cancer risk in long-term studies. Cells exposed to potassium bromate often show genetic mutations, which put the chemical under scrutiny in toxicology circles. Although thorough baking can decompose most of the compound, trace amounts sometimes remain. These findings led regulatory agencies in the EU, China, Canada, and elsewhere to ban potassium bromate use in food. I remember cases where court orders forced manufacturers to recall whole batches of bread, causing not only financial losses but also public trust crises. Toxicologists kept pressing for tighter residue limits and modern detection tools, driving the industry away from old habits and toward safer options.
Future Prospects
Looking ahead, the future for potassium bromate in the food industry grows dimmer each year, as safer and less controversial alternatives take over. Consumer awareness and stricter rules fuel this trend. Regulatory bodies most likely will lower permissible residue levels even further or ban its use outright where it still lingers. In markets still holding onto the chemical, consumer advocacy drives education campaigns, pushing alternatives up the supply chain. Research focuses on enzymes and plant-derived oxidizers that offer similar dough properties without leaving toxic residues, reshaping the ingredient lists of commercial breads. In my years tracking food industry shifts, the move away from potassium bromate stands as a clear win for public health, even if it means more investment in retooling and staff training for bakeries around the world.
What is potassium bromate used for?
Potassium Bromate in Baking
Walk into a supermarket and check the label on a loaf of classic sandwich bread. Many loaves, especially mass-produced ones, have contained potassium bromate. This chemical works as a “flour improver.” It boosts dough strength, gives bread a better rise, and helps baked products keep their soft texture. Bread comes out looking fluffier, with a nice golden crust and stays soft longer. In other words, many baked goods in the past have relied on it to deliver that airy texture people expect from hamburger buns or packaged white bread.
Potassium bromate acts as an oxidizing agent. It helps the dough trap more air, which makes for smoother, bigger loaves. Bakeries have used it because it simplifies the process and delivers more consistent results, especially when using high-speed production. Many industrial bakeries turned to potassium bromate because the American public came to expect a certain standard: soft, voluminous loaves that keep for days.
The Health Concerns No One Should Ignore
Despite these practical baking benefits, potassium bromate brings risk into the kitchen. Research over the past few decades has pointed to health concerns—specifically, kidney and thyroid cancers in laboratory animals exposed over time. The International Agency for Research on Cancer (IARC) classifies potassium bromate as a possible human carcinogen. The body might not always convert all of the potassium bromate to a harmless form during baking. Residues can remain in finished bread.
Governments have responded. The European Union, Canada, China, Brazil, and India do not allow potassium bromate in food. Japan banned it almost 40 years ago. California lets consumers know by requiring warning labels on any foods using it. The rest of the United States still permits potassium bromate, although public pressure and lawsuits have prodded many large bakeries to stop using it.
What Happens Without Potassium Bromate?
People sometimes wonder how bread would change without this chemical. I remember talking to my grandmother about bread-baking in the 1950s. She worked with local flour and old recipes, producing small, dense loaves with a real chew. Nothing like the pillowy bread at the grocery store today. Those traditional recipes didn’t need potassium bromate. By adjusting the timing and mixing, or swapping ingredients like ascorbic acid (vitamin C), bakers can get reliable results without it. Ascorbic acid accomplishes a similar job—helping dough develop structure during mixing. The truth is, globally, bakers have adapted just fine. Many major supermarket loaves in the U.S. are now bromate-free because of public demand.
For people who bake at home, potassium bromate does not show up in any ingredient list at the neighborhood grocery. Home recipes rely on skill, patience, and time to build gluten and texture. Avoiding additives promotes trust in food, supports clean labeling, and lowers risk. Bakeries have more tools and knowledge now, and they can create consistent, appealing bread without this risky shortcut.
Simple Solutions for a Safer Loaf
Choosing bread without potassium bromate has gotten easier. Look for products labeled “bromate-free” or “unbleached flour.” Support bakeries committed to chemical-free baking. Industry can do more, too. Training for bakers, reformulating recipes with ascorbic acid or enzymes, and simple transparency around ingredients would improve consumer trust. Food safety should never take a back seat to convenience.
Bread has always been a staple, but people deserve real security in what they eat. It is time to recognize what works and move on from the shortcuts that do not match up with modern science or public expectation.
Is potassium bromate safe for consumption?
Knowing What Goes Into Bread
Growing up, I joined my family on weekends to bake bread together. We didn’t use anything fancy: flour, yeast, water, a bit of salt and sugar. The loaves had that warm, homemade scent and a crumbly crust. Once I started reading more about food production, I learned that some commercial bakeries add ingredients like potassium bromate to make bread rise taller and look whiter.
Potassium bromate acts as a flour improver. It strengthens dough and helps baked goods hold shape. The big question isn’t just about bread rising higher; it’s whether this additive should even be allowed at all. Studies link potassium bromate to serious health issues. Animal tests pointed toward a risk of cancer, with tumors found in the kidneys and thyroids of rats. The World Health Organization classified potassium bromate as a possible human carcinogen. This evidence has been enough for countries like the UK, Canada, Brazil, and the entire European Union to ban potassium bromate from food. The US, though, hasn’t followed suit.
The Problem with Residue
Bakeries and food producers claim potassium bromate breaks down completely during baking, so bread on the shelf won’t contain any. In reality, some products show trace amounts of bromate after baking, especially if baking times or temperatures aren’t consistent. Nobody checks every loaf or bagel for complete breakdown. That uncertainty means even careful eaters can wind up eating something that’s not safe.
Sticking to E-E-A-T — experience, expertise, authoritativeness, and trustworthiness — calls for clear facts. In 1999, California put potassium bromate on its Proposition 65 list of chemicals that can cause cancer. The FDA set limits on how much bakers could use, warning against excess. Even with warnings and suggested caps, some bakeries still get away with using it, since enforcement stays weak, and labeling doesn’t always mention bromate at all.
Why Ban a Risky Ingredient?
The food industry doesn’t lack safer options. Ascorbic acid (vitamin C) gives dough the same airy lift as bromate, without the danger. Bakeries in many countries continue turning out wonderful bread without potassium bromate. Keeping this chemical in food feels needless and outdated.
Consumers deserve simple, honest food. When food scientists, government panels, and international watchdogs have flagged a food additive, holding off on change puts people at risk. A slice of bread shouldn’t come with a side of worry.
How People Can Avoid Potassium Bromate
I look for labels that list ingredients or reach for bread from small bakeries that use natural recipes. Many health-conscious shoppers have switched to organic or make bread at home. Some US brands label “bromate-free,” though not all. Advocacy groups, including the Environmental Working Group, push for a ban and ask people to pressure lawmakers and brands.
In the end, food safety calls for trust. If an additive stands linked to health risks in credible studies, and if safer alternatives exist, holding onto it doesn’t make sense. More companies and countries should listen and make bakery shelves safe for all.
Why is potassium bromate banned in some countries?
Bread Isn’t Always Just Bread
I remember walking through bakeries as a kid, loving that freshly baked smell. Most folks don’t stop to ponder each ingredient in their daily loaf. I didn’t either. Years later, after getting interested in food safety, I found out about potassium bromate. Many countries ban it, but why? This isn’t about flour or yeast—it’s about what goes into making bread whiter, stronger, and softer. That’s the goal behind potassium bromate, used in commercial baking for decades. The trouble starts after the bread cools and hits the shelves.
Concerns About Cancer Risk
Plenty of research rings alarm bells about potassium bromate. The International Agency for Research on Cancer (IARC) points to clear evidence it causes cancer in animals, especially rats and mice. After hearing this, it’s hard to ignore the risk. The U.S. National Toxicology Program found tumors in several organs linked to exposure. For everyday people, it’s nearly impossible to measure how much residue might linger in finished bread. That uncertainty troubles scientists and consumers alike.
Global Moves Toward Safer Additives
Japan banned potassium bromate in the 1980s. The European Union, Canada, and China went the same way, citing risk to public health. They don’t want even trace amounts landing in lunchboxes or dinner rolls. For context, most bread today can turn out fluffy and appetizing using safer alternatives like ascorbic acid (vitamin C), which helps dough rise without risky chemicals. South Korea and Brazil also put consumer safety above convenience, forcing industry to switch to harmless compounds.
U.S. and Indian Rules Still Lag Behind
It surprises many that some places—including the United States and India—haven’t caught up. The U.S. Food and Drug Administration (FDA) allows potassium bromate in baking, but only in small amounts. Regulators say the chemical should break down completely during baking, but studies point to varying residual levels in finished products, especially in undercooked batches. In a busy commercial bakery, not every loaf gets the same attention, which means consumers have no real control over hidden risks.
Better Options Do Exist
Sue, an old friend who bakes for a living, tossed out potassium bromate over a decade ago. She uses vitamin C, and her customers have never noticed any difference in texture. Her sales haven’t taken a hit. This matches reports from bakeries across the EU and Canada—switching doesn’t impact bread quality in any meaningful way. It raises an obvious question: why keep using something with a question mark over its safety?
Path Forward for Industry and Consumers
Keeping bread safe shouldn’t be controversial. More people demand clean labels and fewer hidden chemicals. Food manufacturers could phase out potassium bromate without missing a beat. Governments who still permit it might reconsider by focusing on the mounting evidence. As someone cautious about what lands in my kitchen, I check labels for chemicals a loaf doesn’t need. If more people ask questions and push for change, safer bread could be in everyone's basket.
Are there health risks associated with potassium bromate?
Bread on the Table, Chemicals in the Dough
Many people don’t know what goes into their daily slice of bread. Potassium bromate has lingered as a bread additive, used to strengthen dough and make loaves look fluffy. Food scientists say potassium bromate helps manufacturers cut costs and speed up baking. But the question won’t go away: does adding this chemical to baked goods endanger public health?
Health Concerns Based on Real Science
Potassium bromate has sparked concern for decades. Studies from as far back as the 1980s suggested a cancer risk in lab animals exposed to the chemical. The International Agency for Research on Cancer labeled it a possible human carcinogen. Japan and even the European Union have banned its use in food altogether. In the United States, potassium bromate remains legal, though California requires a warning label if it shows up in finished bread at significant amounts.
Evidence doesn’t just come from wonky animal studies. The U.S. Food and Drug Administration acknowledged the risk and advised bakers to use other dough improvers. But with little enforcement, bakers still find it in bags of flour. In low doses, most of the bromate converts to harmless bromide during baking, but tests have shown finished products can still contain traces.
Why Trust and Transparency Matter
Food should nourish, not cause worry. Potassium bromate’s story reminds us to take a closer look at food additives. Transparency from bakeries and food makers builds trust. Shoppers deserve clear labels, especially for ingredients flagged by cancer researchers.
Customers often say, “I’ve eaten bread for years and I’m fine.” True, most people won’t eat enough potassium bromate in a year to hit known risk levels. Still, scientists don’t ignore clear signals from animal studies. Banning potassium bromate in bread protects the most vulnerable — children, those with health conditions, and people who eat a lot of baked goods.
Safer Choices Exist
Other countries show it’s possible to bake soft, appealing bread without risky chemicals. Food companies can swap potassium bromate for safer options like ascorbic acid. Baking science keeps improving, making it easier to hit the same loaf quality without a side of worry. Even in the U.S., some big brands have dropped potassium bromate under pressure from consumers. Independent bakeries can do the same by being transparent with ingredients and seeking guidance from food safety agencies.
Solutions for the Future
Consumers have more power than they think. Ask local bakeries what goes into their products. Choose brands that leave out questionable additives. Decision-makers in government can help, too, by setting firm restrictions, not just suggestions, and backing food safety with clear evidence-based regulation.
No one wants to fear a staple as simple as bread. Potassium bromate’s risks have been known for a long time. Scientists keep sending up warning flares. With enough demand for change — from shoppers and food professionals both — bread can stay wholesome and safe, no risky shortcuts required.
How can I identify if a product contains potassium bromate?
Looking Beyond Labels
Potassium bromate slips quietly into many foods, especially baked goods. Many countries ban it, but the United States still allows its use in the baking industry. I remember peering at ingredient lists in my own kitchen, squinting to read every line, trying to keep my family away from unwanted chemicals. The truth is, potassium bromate is rarely front and center. Bakeries don’t always shout about it on their labels—sometimes it hides under unfamiliar names or buries itself on the list.
So, the first place most people check is the ingredient label. Potassium bromate appears under its own name, but sometimes just as “bromate” or similar variants. In smaller bakeries or imported baked goods, the label might not even mention every additive, which always frustrates me. Transparency should feel as basic as bread, yet it doesn’t always work out that way.
Learning the Laws Around Potassium Bromate
Countries like Canada, Brazil, and much of the European Union ban this additive because research links potassium bromate to cancer in animal studies. In the U.S., the FDA keeps it on the list of allowed additives but encourages bakers to switch to alternatives. California law goes a step further by requiring products with bromate to carry a warning label under Proposition 65. So, if you pick up a loaf while shopping in California and see a cancer warning, chances are bromate could be in it.
Worldwide, bread standards often follow local laws. Many global brands adapt their recipes to avoid bromate where it’s banned. If you travel or order international products, check the food regulations for that country or brand—the difference can surprise you.
Types of Food to Watch
Potassium bromate helps dough rise, especially in mass-produced breads and buns—think supermarket white bread, hamburger buns, and pizza dough. Artisanal bakeries usually skip it since traditional methods give bread better texture naturally. Whenever I shop, I find the more generic the bread or roll, the more likely it includes questionable additives. Packaged baked goods from large brands should make you look twice.
What Works Instead of Potassium Bromate?
You don’t need bromate for good bread. Many bakeries use ascorbic acid (vitamin C), enzymes, or gluten to create the same fluffy results. During some of my own experiments baking sourdough at home, I never needed any sketchy powders from a chemical supplier. Good flour, time, and care can do wonders. If a product’s label lists ascorbic acid or enzymes but no bromate, the company already made a safer choice.
Practical Steps for Shoppers
For anyone wanting to avoid potassium bromate, keep it simple: read labels closely, favor products from bakeries that avoid chemical dough improvers, and support brands that list all ingredients clearly. Reach out to companies and ask—sometimes a quick email gets results, especially from local bakeries. If you’re unsure about something, let curiosity lead. I’ve even gone so far as to call supermarket hotlines while standing in the bakery aisle.
Real Change Takes Transparency and Action
The only surefire way to keep potassium bromate out of your diet is to stay vigilant and support changes in labeling laws. While we wait for those laws to catch up, relying on a mix of personal knowledge, careful shopping, and honest communication with food producers makes a real difference. Nothing beats bread made with simple ingredients, especially when you know what’s inside.
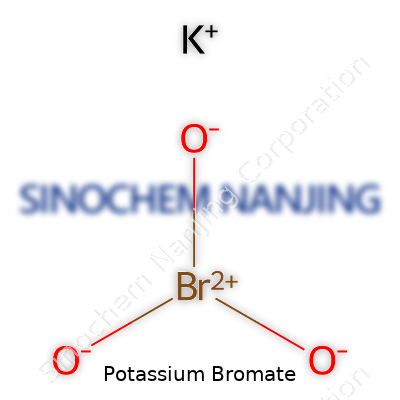
| Names | |
| Preferred IUPAC name | Potassium bromate |
| Other names |
Bromic acid, potassium salt Potassium bromate(V) Potassium salt of bromic acid UN 1484 |
| Pronunciation | /pəˈtæsiəm ˈbroʊ.meɪt/ |
| Identifiers | |
| CAS Number | 7758-01-2 |
| Beilstein Reference | 3598225 |
| ChEBI | CHEBI:73692 |
| ChEMBL | CHEMBL1431052 |
| ChemSpider | 5762 |
| DrugBank | DB11449 |
| ECHA InfoCard | 100.009.593 |
| EC Number | 231-829-8 |
| Gmelin Reference | Gmelin Reference: **14568** |
| KEGG | C14326 |
| MeSH | D011180 |
| PubChem CID | 23668152 |
| RTECS number | EF8774000 |
| UNII | 3P5SQ47T7J |
| UN number | UN1484 |
| Properties | |
| Chemical formula | KBrO3 |
| Molar mass | 167.00 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 3.27 g/cm³ |
| Solubility in water | 7.5 g/100 mL (25 °C) |
| log P | -0.77 |
| Vapor pressure | Negligible |
| Magnetic susceptibility (χ) | +39.0e-6 |
| Refractive index (nD) | 1.495 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −811.3 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -390 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AN01 |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, may cause cancer, irritating to eyes, skin, and respiratory system. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02, GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H315, H319, H335, H350, H372 |
| Precautionary statements | P210, P220, P221, P260, P264, P273, P280, P301+P310, P305+P351+P338, P306+P360, P308+P311, P311, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0-OX |
| Autoignition temperature | **> 380°C (716°F)** |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 1870 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 1870 mg/kg |
| NIOSH | ST07920 |
| PEL (Permissible) | 0.1 mg/m³ |
| REL (Recommended) | 0.02 mg/kg bw |
| IDLH (Immediate danger) | IDHL: 30 mg/m3 |
| Related compounds | |
| Related compounds |
Sodium bromate Potassium chloride Potassium bromide |

