Potassium Bisulfite: From Ancient Discovery to Modern Industry
Historical Development
Potassium bisulfite has stories that stretch back hundreds of years. Early chemists realized the power of sulfite salts long before organic chemistry had its place in textbooks. Winemakers found out early that a blend of potassium, sulfur, and water worked wonders at keeping their batches fresh. In the 1800s, as food science grew more rigorous, potassium bisulfite gained a steady reputation for preserving, cleaning, and stabilizing. Factories recognized the edge it gave in food, beverage, and chemical processes, keeping products safe from spoilage without changing flavor or appearance. Over time, production scaled up, and quality control became much tighter. Even now, the methods of making this salt borrow a lot from those old discoveries, just with better equipment and purer ingredients.
Product Overview
Potassium bisulfite shoots for a simple goal: offer reliable sulfur dioxide without the tricky business of handling a gas. Granular or powdery, it mixes easily into liquids, so food and wine makers get the benefits of antimicrobial and antioxidant action, but not the headache of specialized storage. It has a sharp, tangy bite if you ever taste a diluted sample, and it smells like a crisp mineral spring. Sold in food-grade bags and drums, most forms are stamped with certifications for food safety. Commercial users scan labels for the potassium content and the active SO2 value, since these numbers tell you how much "kick" the batch will add.
Physical & Chemical Properties
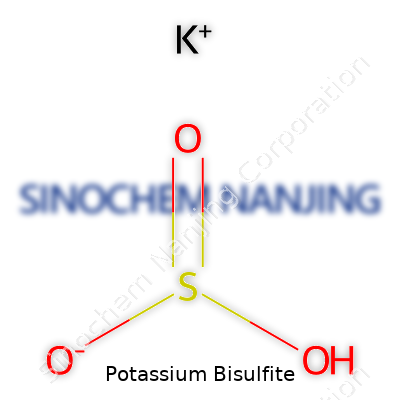
Potassium bisulfite (KHSO3) comes as a white crystalline powder. It feels gritty, much like refined salt, and dissolves well in water. The pH hovers in the acidic range, with strong reducing power thanks to the active sulfite. If you leave the substance exposed to air, it may begin picking up oxygen and transform partly into sulfate, cutting down its effectiveness. Store it wrong, and humidity gets inside, caking the powder and making dosage unpredictable. Chemically, its structure holds the sulfur atom wedged between three oxygen partners, with a potassium ion floating nearby. In air or on contact with acids, it starts releasing sulfur dioxide gas. This characteristic lines up with its main job in winemaking—protecting against unwanted microbes and oxidation.
Technical Specifications & Labeling
Product shipments carry strict technical data. A good label will list the percentage of pure KHSO3—usually between 97-99% for food and beverage use. Sulfur dioxide (SO2) content shows up on the specs, as that number tells the end user how much preservative power they'll get per gram. Moisture levels stay low, typically under 1%. Producers include batch numbers, production dates, shelf life (around two to three years if stored right), and safety warnings describing handling, mixing, and first aid. Certified suppliers carry third-party test results, often run by independent labs, showing heavy metal levels below strict legal limits, so food processors know nothing unexpected sneaks into the supply chain.
Preparation Method
Manufacturers prepare potassium bisulfite by bubbling sulfur dioxide directly through a chilled solution of potassium carbonate or potassium hydroxide. The acid gas reacts with the base until pH signals it's complete. Careful temperature control in the tanks keeps reaction rates balanced, so the solution doesn't lose potency. Afterward, workers concentrate and cool the liquid, letting crystals form and settle. Filtration removes leftover bits, and dryers finish the job. The dry product then heads to sterile packing lines, so it reaches customers with the guarantees needed for food-grade material. Environmental controls keep wasted sulfur dioxide out of the air and water, a concern that loomed larger as regulations toughened in recent decades.
Chemical Reactions & Modifications
Potassium bisulfite reacts whenever it meets strong acids, turning into potassium salts and releasing sulfur dioxide gas instantly. This release stands at the core of its use. In solution, it balances as an equilibrium mixture of bisulfite and sulfite ions, letting it soak up oxygen radicals and neutralize them before they do any damage. When used in wine, its chemical work stops the growth of wild yeasts and bacteria. Anyone working in food processing learns how modified usage—say, blending with ascorbic acid or buffering agents—can help fine-tune the antioxidant punch and make the most of limited regulatory allowances. In water treatment, bisulfite neutralizes chlorine and chloramine, keeping finished products safer for sensitive equipment and processes.
Synonyms & Product Names
Potassium bisulfite doesn't always appear on labels under a single name. Some suppliers call it potassium hydrogen sulfite, others potassium acid sulfite, or just E228 for regulatory filings in food and beverage. Winemakers call it metabisulfite, though strictly that name points to the double salt. Some product lines use brand names, especially for high-purity or low-dust formulas. Chemical registries stick to Potassium Bisulfite or Potassium Hydrogen Sulfite, so cross-reference ensures buyers know what they’re actually getting. These variations spark confusion for buyers new to the market, making technical support and supply chain transparency crucial.
Safety & Operational Standards
Proper handling makes a big difference. Skin and eye contact stings, since the compound is strong enough to irritate tissues. Breathing in dust brings coughing and discomfort, and workers with asthma or sulfur allergies can have even worse reactions. Safety data sheets for potassium bisulfite lay out the basic kit: gloves, eye protection, and dust masks. Workspaces need good air flow, and storage stays cool, dry, and sealed from the air. In facilities where large quantities are handled, eye wash stations and safety showers remain non-negotiable. Disposal routes match regulations for sulfur compounds, so the waste stream avoids polluting groundwater. For the public, national agencies set food additive limits, and every processor runs batch-by-batch testing to meet those strict standards.
Application Area
Wine and juice producers reach for potassium bisulfite for both stability and shelf-life. It nips oxidation in the bud, keeps spoilage at bay, and lets subtle flavors develop without going stale. Fruit processors, breweries, and even some cheeses owe their flavor and safety to its invisible hand. In water plants, it scrubs traces of chlorine from municipal supplies destined for commercial use. Textile facilities use small doses to bleach wool without harsh reactions, and some labs turn to potassium bisulfite for prepping microscope slides or stabilizing dye mixtures. The range is wide, but food and beverage uses still take the lead by volume.
Research & Development
Academic researchers track advances in potassium bisulfite production and its alternatives. Food science labs map out how micro-dosing improves food safety with fewer chemical residues. Some work centers on keeping its benefits without triggering allergic reactions, which prompted regulatory pushback in parts of the world. Material technologists have tested encapsulated sulfite formulas, putting potassium bisulfite into coatings or controlled-release packets for more predictable behavior. Environmental engineering investigates clean-up systems to catch and recycle sulfur compounds from waste, driven by both regulation and the promise of circular manufacturing.
Toxicity Research
Toxicologists track exposure through diet, air, and skin. Most healthy adults break down sulfites efficiently, but sensitive groups—including those with asthma or enzyme deficiencies—can react with headaches, respiratory distress, or hives. Trace levels in food and drink seldom cause trouble for the general public, though cumulative exposure in industrial workers needs steady monitoring. Animal studies show no risk of cancer or genetic damage at normal dietary levels, supporting official chemical safety assessments. Long-term risk research now focuses on low-dose, chronic exposure to find any subtle health impacts missed in early studies.
Future Prospects
Potassium bisulfite holds its ground in a tightening field. Consumers want fewer additives, and regulators keep shrinking allowable concentrations. Scientists worldwide search for balancing acts: matching ancient preservation tricks with the demands of organic labels and allergen warnings. Demand remains steady where sulfite-sensitive microbes need curbing, especially in wine, but the next wave of products could leverage smarter delivery methods to cut totals and improve product purity. Process improvements keep waste and environmental impacts in check, carving out a space for potassium bisulfite with fewer side effects and greater peace of mind for processors, workers, and end users.
What is Potassium Bisulfite used for?
From Vineyards to Laboratories
Potassium bisulfite usually pops up in the world of winemaking. I remember walking into a small winery, smelling the sweet, sharp scent of fermenting grapes, and watching as a practiced hand reached for a bag labeled with this very chemical. Winemakers trust it as a tool to control the wild cards—bacteria and unwanted yeasts—that would try to spoil their hard work. Just a pinch in the crushed grape juice does wonders to keep the wine bright and true to its character.
Sulfur-based preservatives like this one have held their place in cellars for centuries. Only in recent decades have we truly understood how they work, but tradition proved their usefulness long before the science could explain it. Potassium bisulfite, once added, releases sulfur dioxide. That gas acts fast, stopping the bad bugs without hurting the flavors winemakers chase after. Too much, though, and the bouquet can turn harsh—so experience and careful testing leads the way.
Extending Freshness in Food
I’ve seen the ingredient list on the package of dried fruit—the name might not stand out, but it keeps the colors vivid. Food processors lean on potassium bisulfite to keep products shelf-stable and appealing for longer stretches. Without this helper, a lot of shelf staples would look drab and turn long before we got them home. It cuts down the browning that comes naturally to apples, apricots, and other fruit. This preserves some natural sweetness and tang even months past picking season.
Concerns about sulfite sensitivity have made some companies label their packages more carefully, which helps folks who feel the side effects choose alternatives. For most of us, these preservatives just keep our pantries more reliable. The FDA keeps close tabs on food additives like potassium bisulfite. Only careful management and clear information keep its use responsible.
Helping Labs Get Accurate Results
In the lab, potassium bisulfite turns up on the shelf as a reducing agent. Chemists working on intricate tests use it to prevent oxidation, keeping samples fresh for accurate measurements. Back in college, our chemistry professor liked to remind us, “A reaction ruined by oxygen never gets a second chance.” He knew that stability matters in science—a slight error from air slipping into the mix can wreck weeks of work.
It’s not just grapes and science samples that benefit. Water treatment plants sometimes turn to potassium bisulfite to remove traces of chlorine. Chlorine takes care of germs, but the taste and extra residue don’t suit every application. This chemical neutralizes leftover chlorine so tap water tastes better.
Solutions for Health and Safety
People with asthma or sulfite allergy deserve more from food labels and stricter oversight. Stores and restaurants need to train staff better and respond quickly to customer questions. Regulators should impose routine audits and invest in new technology for even safer alternatives. On the production end, training workers to handle chemicals safely prevents both contamination and workplace accidents. Smaller producers making organic or “clean label” products might try fermentation techniques or vacuum-packing to replace preservatives entirely.
Why This Matters Now
As health awareness grows, clear labeling and safe limits matter more than convenience. Potassium bisulfite extends shelf life and protects food and drink quality, yet comes with responsibility. The choices made today ripple out to kitchen tables and vineyards everywhere. Solutions built on science, careful handling, and honest information point the way to confidence and safety in everything we eat and drink.
Is Potassium Bisulfite safe for consumption?
The Role of Potassium Bisulfite in Food and Drinks
Potassium bisulfite shows up most often on the label of your favorite wine or sometimes in dried fruits. Its main job is to keep foods from going off or changing color, and it helps winemakers control the process of fermentation. Potassium bisulfite releases sulfur dioxide when added to liquids. Sulfur dioxide keeps bacteria, mold, and unwanted yeasts in check, letting good microbes do their work. I’ve seen firsthand how chefs rely on this compound to save batches of homemade preserves from unpredictable spoilage.
What Science Says About Safety
Health authorities in the United States, Europe, and around the world have studied potassium bisulfite for years. The Food and Drug Administration (FDA) allows it in food and drink, setting a maximum level for safety. The European Food Safety Authority (EFSA) also gives potassium bisulfite the green light within strict limits. Both groups base their recommendations on research—the kind you see in peer-reviewed journals and years of lab testing.
I looked through several toxicology studies and regulatory reports. None of them found major risks for healthy people when eating or drinking moderate amounts. The acceptable daily intake is about 0.7 milligrams per kilogram of body weight for all sulfites. Most people don’t come close to hitting that limit through normal habits. One glass of wine or a serving of dried apricots rarely pushes anyone near the guideline. Most of the time, symptoms appear only above this limit.
Concerns for Sensitive Individuals
Not everyone reacts the same way to potassium bisulfite. Some people have sulfite sensitivity, which can bring on asthma-like symptoms, hives, or stomach issues. The FDA estimates that just under 1% of Americans, mainly those with asthma, may react badly to sulfites. In my own kitchen, I’ve seen dinner guests get a mild headache after a glass of wine, only to discover a link to sulfite preservatives like potassium bisulfite.
Clear Labeling and Informed Choices
Strict labeling rules help sensitive folks know what they’re consuming. U.S. law requires foods with more than 10 parts per million of sulfites to list them on the label. Most name-brand wines and packaged dry fruit do this on the ingredients list. Reading those labels has become second nature to many shoppers, especially families with allergies. Kids with asthma or those prone to allergic reactions need extra care and a close read of every ingredient panel.
Where to Go from Here
Producers could work to further reduce sulfite levels when safer ways exist to keep food shelf-stable. Wineries and food processors experiment with alternative methods—natural fermentation controls, better sealing technologies, and antioxidants from plants. Success with these new approaches may take time, though, as old processes change slowly. Shoppers who regularly face sulfite sensitivity already rely on organic wines and sulfite-free fruit snacks.
In my home, friends who avoid sulfites get a separate bottle of organic wine and fresh fruit when we gather. Plenty of safe options let us include everyone at the table. People with no medical concerns about sulfites can still enjoy their favorite foods and drinks by keeping an eye on ingredient lists and sticking to moderate servings.
How should Potassium Bisulfite be stored?
Understanding the Hazards
Potassium bisulfite often pops up in winemaking and food preservation. It helps prevent unwanted bacteria and reactions. Behind the scenes, storage mistakes can cause headaches – and sometimes, real danger. Like many chemicals, potassium bisulfite reacts with air and water in ways that surprise newcomers. Left exposed, it produces sulfur dioxide gas, which triggers coughing, eye discomfort, and can even spark asthma in people with lung issues. Keeping aware of this risk sits at the core of good storage.
Moisture and Air Exposure
Potassium bisulfite draws in water from the air. In humid environments, lumps form and gases get released. Growing up near a fermentation lab, I saw bags left open “for a minute” clump into bricks. Mold and strange smells followed. Proper storage starts with shutting out dampness. Use tightly sealed containers, not old cardboard. Desiccant packets add a little insurance.
Choosing a Container That Works
Chemicals leach through flimsy plastic after a while. Strong polyethylene or glass jars with accurate screw tops make a difference. Screwing the lid down firmly and double-checking the seal go further than just tossing the bag in a closet. Years ago, I visited a winery. Their crew used old food tubs; weeks of vapor had torn thin spots and started corrosion around metal shelving. They switched to chemical-grade storage barrels, which held up and slashed the risk of accidental gas leaks.
Temperature and Light
Heat speeds up chemical changes. Cool, dry spaces play a much bigger role than most realize. Sun streaming through windows or spotlights from overhead fixtures heat up containers and help start any chemical breakdown. Rooms that rarely hold onto heat preserve quality best. I’ve seen stockrooms near radiators where white powder turned yellow and lost its punch.
Labeling and Access
Part of safe storage means never hiding what something is. Clear, large labels matter more than people admit. Unmarked containers have caused close calls in food production and wine cellars alike. Safety data should sit somewhere nearby—wall posters work. Workers get a refresher on risks during daily routines this way. If children or pets live in the building, higher shelves or locked cabinets remove temptation and prevent tragedy.
Addressing Spills and Handling
Spills don’t take long to turn into bigger problems. Immediate cleanup with disposable gloves and plenty of ventilation limits health problems. Large spills need more—fans, and sometimes a fresh-air mask, depending on the amount. Waste shouldn’t go down drains or the regular trash bin. Look for local rules or chemical disposal services. People new to potassium bisulfite sometimes forget its potential to pollute groundwater, which then causes headaches for neighbors, not just the user.
Long-Term Storage Risks and Rotation
Packing shelves to the ceiling just to “stock up” guarantees problems down the line. Older inventory loses strength and risks clumping. Regularly using the oldest chemicals first (“first in, first out”) keeps the system working and reduces surprises. Keeping inventory logs saves money and improves safety. I saw one shop slash losses and cut unnecessary orders this way, simply by rotating more carefully.
Final Thoughts
Careful storage builds trust and limits emergencies. Tightly sealed, labeled, and protected from kids, potassium bisulfite does its job—without endangering health or reputation.
What is the dosage of Potassium Bisulfite in winemaking?
The Role of Potassium Bisulfite
Potassium bisulfite carries a strong reputation among winemakers for safeguarding wine from spoilage and unwanted oxidation. Heading out to any local vineyard or talking with experienced hobbyists, the phrase “sulfite management” comes up as regularly as “fermentation.” Without a grasp of the proper dosage, even a simple homemade batch risks going bad, or worse, turning out with harsh chemical off-notes that can ruin the drinking experience.
Understanding the Numbers
Dosing potassium bisulfite isn’t about dumping a random scoop into the juice or must. This salt breaks down to release sulfur dioxide (SO₂), the compound doing the real preservative work. Most guides recommend somewhere between 30 to 70 parts per million (ppm) of free SO₂ for red wines and 20 to 40 ppm for whites, after fermentation wraps up. Sulfite calculators often pop up on respected winemaking forums, helping users account for variables like wine pH and storage practices. For context, a teaspoon of potassium bisulfite (about 6 grams) in 5 gallons of must typically delivers around 150 ppm of total SO₂—a hefty blast, usually suited for tackling fruit before fermentation or sanitizing equipment, not for stabilizing finished wine.
Personal Bottles, Personal Lessons
Nothing teaches as well as a lost batch. In my early days with Carignan grapes from a neighbor’s overgrown backyard, I followed vague instructions and ended up with a load of over-sulfited wine, sharp on the nose and punishing on the palate. Others run too light and wake up to bottles fizzing with unwanted ferment. Both mistakes underscore the need for precise measurement rather than guesswork or “eyeballing it.” The real key rests with test kits—titration kits show up on every well-equipped home winemaker’s shelf, because they measure free and total SO₂ accurately. A few drops of indicator and a color change give peace of mind far beyond any shortcut.
Why Getting It Right Matters
Sulfur dioxide doesn’t just protect wine; it shapes the way it ages and tastes. Too little, and a promising vintage drifts toward spoilage from bacteria or wild yeast. Too much, and the wine puts off drinkers sensitive to sulfites, or tastes artificial and overworked. According to research shared by UC Davis and other respected enology programs, maintaining free SO₂ in the recommended range dramatically cuts the risk of oxidation and microbial issues. This isn’t about chasing zero risk—wine always involves some gamble—but about stacking the odds toward bottles you can drink or share with pride.
Making Adjustment Easy
Rather than treating potassium bisulfite as a one-time fix, winemakers make a habit of checking SO₂ levels at several points: after fermentation, after rackings, and before bottling. Using online calculators or established tables keeps the math simple, and pH strips or meters help tweak dosages for acidic or soft wines. For folks without lab gear, established rules of thumb—1/4 teaspoon per six gallons every few months—work only as a starting place, not a rule. Because grapes, weather, and storage all change year by year, relying on observation and testing beats following a recipe every time.
A Safer, Cleaner Glass
Too often, conversation about wine preservatives drifts into rumor or fearmongering. Real-world winemakers, both in commercial cellars and backyard sheds, know that potassium bisulfite remains one of the safest, most controllable ways to protect a year’s labor in the bottle. Learning the right dosage and testing for SO₂ turns wine from a guessing game into something far more rewarding—and every great glass reflects the effort.
Are there any side effects or hazards associated with Potassium Bisulfite?
Why Potassium Bisulfite Matters in Everyday Life
Potassium bisulfite finds its way into winemaking, food processing, and water treatment. Its main job involves controlling microbial growth and acting as a preservative. Most people don’t think twice about additives like this, but once you work with it or have sensitivities, things start to look different. I learned that during my first experience helping a friend make homemade wine. The smell alone made me take a step back.
Physical Hazards and Handling Risks
This white, crystalline powder looks harmless. Yet direct exposure packs a punch. Potassium bisulfite causes skin and eye irritation. If it gets on your hands, you’ll probably feel an itching or burning sensation pretty quickly. Accidentally touching your face or rubbing your eyes after handling it can sting a lot more than most household substances. Friends who work in wineries have recounted stories about rashes after long bottling sessions. Gloves and goggles didn’t seem like overkill after hearing those stories.
Even opening a container in a small space can lead to coughing. Breathing in the dust or vapors triggers symptoms similar to sulfur dioxide inhalation – a burning throat, coughing, and, in high enough amounts, trouble breathing. Anyone with asthma or existing respiratory issues knows these effects get much worse. U.S. Centers for Disease Control and Prevention (CDC) lists potassium bisulfite as a respiratory hazard for a reason.
Food Sensitivities and Allergic Reactions
Food allergies get a lot of attention, but many people don’t realize sulfites in preservatives like potassium bisulfite can also cause reactions. The FDA requires labeling because even small traces might send sensitive individuals into a spiral of symptoms. My aunt, who has asthma, experienced shortness of breath after drinking a glass of wine treated with sulfites. For about 1% of the general population, sulfites can mean headaches, rashes, hives, or breathing problems. For asthmatics, those risks only grow.
Environmental Impact and Storage Practices
Potassium bisulfite isn’t friendly to the environment in large amounts. Dumping waste solutions down the drain harms aquatic life. Studies out of Europe found that high concentrations disrupt the balance in waterways, killing off insects and small fish. Careless storage also causes problems at home and in businesses. Leaving the lid loose invites moisture, which creates sulfur dioxide gas. That makes storage rooms harder to ventilate and puts workers at unnecessary risk.
Safe Use and Prevention are Possible
Taking some simple precautions makes all the difference. Gloves and splash-proof goggles keep direct contact to a minimum. Storing the powder away from heat and moisture can stop chemical reactions. Many winemakers now use lower concentrations, switching to natural alternatives if the recipe allows. For those with asthma or known sulfite sensitivities, reading labels and understanding what’s in food or drink remains the best defense. Community education—sharing real stories and symptoms—could spare someone else from serious harm. Instead of ignoring fine print, I now see labeling as a small price for peace of mind.
Looking Toward Better Options
Some industries have started testing new preservation methods. Biodegradable additives, lower levels of chemical preservatives, and more transparent labeling policies all push things in a safer direction. The science suggests it makes sense not just for sensitive individuals but anyone wanting safer food and workplaces. Lessons learned from potassium bisulfite come down to using caution, listening to those affected, and pushing for smarter solutions. People shouldn’t have to risk their health just to enjoy a meal or a glass of wine.
| Names | |
| Preferred IUPAC name | potassium hydrogen sulfite |
| Other names |
Potassium hydrogen sulfite Potassium metabisulfite Potassium acid sulfite Monopotassium sulfite |
| Pronunciation | /pəˈtæsiəm baɪˈsʌlfaɪt/ |
| Identifiers | |
| CAS Number | 7646-93-7 |
| Beilstein Reference | 3565085 |
| ChEBI | CHEBI:63045 |
| ChEMBL | CHEMBL1200882 |
| ChemSpider | 59466 |
| DrugBank | DB11136 |
| ECHA InfoCard | 100.033.972 |
| EC Number | 231-594-1 |
| Gmelin Reference | 563 |
| KEGG | C14149 |
| MeSH | D011096 |
| PubChem CID | 23665738 |
| RTECS number | TA4900000 |
| UNII | VSO7U50B1D |
| UN number | UN 2680 |
| Properties | |
| Chemical formula | KHSO3 |
| Molar mass | 120.16 g/mol |
| Appearance | White crystalline powder |
| Odor | Sulfur dioxide odor |
| Density | 1.34 g/cm³ |
| Solubility in water | Very soluble |
| log P | -4.2 |
| Acidity (pKa) | 7.0 |
| Basicity (pKb) | 7.2 |
| Magnetic susceptibility (χ) | Paramagnetic |
| Refractive index (nD) | 1.434 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 149.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -811.9 kJ/mol |
| Pharmacology | |
| ATC code | V03AB02 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes severe skin burns and eye damage. May cause respiratory irritation. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: "H302 Harmful if swallowed. H319 Causes serious eye irritation. |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P305+P351+P338, P330, P337+P313, P501 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Lethal dose or concentration | LD50 oral rat 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat) 2,000 mg/kg |
| NIOSH | VZ3150000 |
| REL (Recommended) | 350 mg/L |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Potassium metabisulfite Potassium sulfate Potassium sulfite Sodium bisulfite |

