Potassium Bifluoride: Past Progress and Present Matters
Historical Roots and Modern Growth
The history of potassium bifluoride reaches back to the earliest years of industrial chemistry, where scientists hunted for sources of fluorine to tackle stubborn challenges in glass etching and metalworking. Its rise caught pace during the nineteenth century as both academia and industry caught on to the unique behaviour of bifluorides. Unlike more infamous fluoride salts, potassium bifluoride delivers properties that bridge the volatile with the workable. The ability to store and handle a reactive form of fluorine, without the nightmares of anhydrous hydrofluoric acid, drew attention rapidly. Factories producing glass, ceramics and aluminium relied on it to open doors that weren’t possible with common alkalis or acids. This compound gained a reputation for reliability wherever controlled aggression in chemistry was needed.
What Potassium Bifluoride Brings to the Table
Potassium bifluoride appears as a white crystalline salt that tends to cake if stored without due care. Sometimes overlooked for its lack of glamour, it’s the quiet workhorse of many industrial chemical rooms. Its formula, KHF2, tells the story in a nutshell: by adding an extra bit of hydrogen fluoride to potassium fluoride, you get a double salt with new tricks. Its solubility in water allows technicians to create solutions that challenge glass, metals, and minerals with precision. Potassium bifluoride combines caustic strength with a more manageable profile compared to pure HF, a fact that has saved more than a few eyebrows (and lives) over the years.
Physical and Chemical Characteristics
In the palm, dry potassium bifluoride feels gritty, much like common salt, though not advised to handle it bare-handed. Its crystals absorb water from the air, sometimes forming sticky lumps. Its melting point stands at a respectable temperature, no wilting at a steamy summer. Dissolved in water, the solution turns acidic and aggressive. In air, it won’t fume like some of the nastier fluorides, but it doesn’t lose its bite. Once in solution, potassium and bifluoride ions work together, releasing HF in controlled amounts. Their properties make them valuable in situations that demand a balance between reactivity and practicality, such as selective etching processes or in making certain metal alloys.
Technical Details and Labeling Realities
Laboratories tend to rely on its stability, so labeling often stresses the need to keep containers dry and tightly closed. Detailed technical literature highlights moisture sensitivity and the need for corrosion-resistant packaging, given the aggressive nature of hydrofluoric acid solutions that can form. Shipping regulations mark it as hazardous, giving extra paperwork to anyone moving more than a lab-sized jar. It doesn’t glow, fizz, or announce its presence, so clear hazard labels and safe handling procedures matter critically. Shelf-life varies, but stored out of humid environments in properly lined containers, it stays viable for years without much loss in potency.
Preparation and Sourcing
Making potassium bifluoride doesn’t call for exotic tricks. Mix potassium carbonate or potassium hydroxide with hydrofluoric acid, and you steer the chemistry toward KHF2. The manufacturing process requires tightly controlled conditions since hydrofluoric acid isn’t forgiving. Even with proper gear, the sharp fumes and corrosive splatters turn a routine synthesis into something that commands respect. Plant operators and chemists have learned to keep ventilation strong, personnel trained, and waste neutralization procedures ready on hand. The industry’s push toward greener, safer syntheses calls for less hazardous waste streams, though the raw ingredients themselves set a hard limit on what “safe” truly means.
How It Reacts and What Can Change
Potassium bifluoride stands out for its role as a fluorinating agent. Stirred into water, it yields up hydrofluoric acid, letting artisans and scientists attack silicates, ceramics, and oxides with directness. Its bifluoride ion brings extra punch, often outperforming single-fluoride compounds in dissolving glass or forming stable complexes with metals such as zirconium, titanium, or aluminum. It undergoes thermal decomposition at high temperatures, liberating hydrogen fluoride gas and leaving behind potassium fluoride as a residue. In organic synthesis, it serves as a gentle option for introducing fluorine atoms without torching the substrate, which broadens its appeal in complex molecule construction. Modifying the potassium ion or trading the acid for another base changes the picture, but for fluorine transfer reactions, the bifluoride stands nearly alone.
Alternate Names and Synonyms in Real-World Use
People in different trades and countries refer to this chemical as potassium hydrogen difluoride, potassium acid fluoride, or simply “bifluoride of potash”. In the literature, one bumps into KHF2 and potassium hydrogen fluoride just as often. Commercial catalogs use jargon that echoes safety guidelines, and confusion can follow unfamiliar translations. For anyone working internationally, cross-checking product codes and chemical synonyms guards against costly missteps.
Operational Discipline for Safety
Working around any fluorine compound puts a person’s caution to the test, but potassium bifluoride takes this to a new level. Contact with moisture on skin or eyes unlocks hydrofluoric acid, known for its ability to burrow through tissue and work nerve-deadening tricks that delay symptoms. Habits like using nitrile gloves, long sleeves, eye shields, and real ventilation draw a sharp line between routine tasks and emergency room visits. Training runs deeper than checklists: workers drill on neutralization procedures, recognize signs of exposure, and know where to find calcium gluconate gel. Modern industrial facilities write safety standards with potassium bifluoride near the top of the hazard list, striving to keep accidents rare and manageable.
Where Potassium Bifluoride Finds Work
Potassium bifluoride fills niches where skilled handling of fluoride chemistry matters. The glass industry relies on it for etching decorative patterns and marking graduations, creating sharp, smooth lines that stand up to years of use. Metals processing pays attention to its ability to clean and activate surfaces for subsequent coating or welding. In production of aluminum, especially refining and alloying, it plays a critical role in removing oxide layers. Chemical synthesis labs and pharmaceutical research choose KHF2 when seeking gentle fluorination steps, often avoiding harsher agents that would wreck already-built molecules. Its use in rust removal, stone cleaning, and electronics cleaning—although restricted by regulations—highlights its unmatched performance in stubborn jobs.
Research and Development Horizons
Academic and industrial researchers don’t treat potassium bifluoride as merely legacy technology. Teams investigate alternative synthesis routes to reduce hydrofluoric acid dependency, pushing for lower-impact production. Nanotechnology fields find dual benefit: the bifluoride’s chemistry etches silicon wafers with tolerance for micro- and nano-scale designs that fuel electronics and solar cells. In battery and energy research, fluorination presents possibilities for new electrode materials. Studies on mixed-metal bifluorides and advanced salt formulations explore higher selectivity, better handling, and longer shelf-life. Laboratories focus on minimizing waste and improving waste treatment strategies—pivotal for aligning with tighter environmental standards.
Toxicity—The Layer Beneath Utility
No matter how useful potassium bifluoride becomes, its toxic legacy can’t be dismissed. Medical literature details accidents where delayed treatment led to deep burns or electrolyte imbalances that threatened lives. Unlike caustic soda or acids that shout warnings with pain, HF-based exposures can numb nerves, masking injury until it’s severe. Standard procedures urge immediate washing and urgent neutralization, but prevention remains the only sure way to stay safe. Academic work continues on more effective treatments and better personal protective equipment, aiming to cut down on the number and severity of accidents.
Down the Road
The future for potassium bifluoride faces both headwinds and tailwinds. Industrial fluorine chemistry may pull back as regulations on toxic and persistent chemicals tighten. Researchers look for ways to capture the benefits of bifluoride chemistry, while reducing the environmental and health toll. New glass, metal, and composite materials may need gentler or more precise etching—pushing the demand for modified bifluorides with built-in safety features. The drive for sustainability motivates green chemistry advocates to coax advances out of old workhorses like KHF2. Its story stands as a lesson in balance—between reactivity and control, progress and responsibility. The next chapter depends on a blend of technical ingenuity and hard-learned discipline.
What is Potassium Bifluoride used for?
What Potassium Bifluoride Does in Industrial Life
Potassium bifluoride isn’t something most folks think about until they spot its name on a chemical barrel at work, or catch wind of it in a safety training. This white, crystalline salt plays a silent but essential role in several heavy-duty processes. In glass etching and cleaning, potassium bifluoride acts as a direct stand-in for hydrofluoric acid, breaking down silica and roughing up surfaces for better bonding. Industries pick it for tasks that need a punchy chemical but also call for a level of control that pure hydrofluoric acid just can’t give without big risks.
In aluminum production, potassium bifluoride walks the line between safety and performance. Smelters mix it into fluxes and electrolytes to help dissolve aluminum oxide, easing the movement of metal and pulling impurities out. I’ve seen workers swearing by the stuff when refining aluminum for aircraft parts, because it chops down on the energy needed and keeps metal quality up to snuff.
Potassium Bifluoride in Cleaning and Metalworking
Digging into cleaning, this chemical finds a seat at the table for removing stubborn scale, stains, and rust from metal surfaces. Factories rely on it to prep stainless steel — the chemical strips away oxide films so that pipes and tanks can get properly welded. Even jewelers reach for potassium bifluoride for brightening up silver and gold before final polishing. Anyone who’s got their hands dirty restoring old car parts or antique tools can tell you: it strips oxidation clean, but they’ve also got to respect its danger to skin and lungs.
What makes potassium bifluoride both useful and hazardous is its ability to deliver fluoride ions right to the chemical reaction. This sharp reactivity means it doesn’t always leave a margin for error, and you need safety gear and solid ventilation. Anyone who’s left a little white dust on a glove from potassium bifluoride knows the tingling, and that's just a warning shot before worse effects take hold.
Dangers and Considerations: Handling with Care
Potassium bifluoride doesn’t mess around with health. Direct contact leads to burns, and inhaling dust can scar lungs before you realize it. It’s not just errors in a plant, either. Even home hobbyists tinkering with glasswork could get burned from a careless sprinkle. I remember a case from an old shop where someone mixed a solution without gloves, and an hour later needed emergency treatment for skin exposure. Reports echo across industries — skin and respiratory injuries keep cropping up wherever potassium bifluoride isn’t handled with care.
Seeking Safer Practices and Alternatives
Strict protocol and protective gear form the backbone of safe potassium bifluoride use, from specialized gloves to fume extractors. Workers deserve training that covers what happens in less than ideal situations. Where possible, some industries switch to less aggressive chemicals, or retool their processes to avoid direct use of strong fluorides. It's encouraging to see companies exploring green chemistries, or turning to mechanical etching where feasible, cutting back on the risk without giving up on efficiency.
Potassium bifluoride isn’t leaving workshops and factories anytime soon. But the health hazards add a heavy weight, and remind us that behind every powerful chemical, protection and knowledge matter just as much as productivity.
Is Potassium Bifluoride hazardous to health?
Understanding Where Potassium Bifluoride Shows Up
Potassium bifluoride plays a big role in industries from glass etching to metal cleaning. It’s a white crystalline compound you’ll find in some cleaning products for bricks and in welding agents. It seems mundane, tucked away among bags of industrial chemicals or sold to folks looking to clean up an old masonry wall. Most people don’t hear about it until questions about chemical hazards pop up.
Health Hazards: The Real Issues
This compound poses real danger far beyond what its plain-white look might suggest. Anybody who’s worked in a shop or lab using potassium bifluoride knows to respect its power. Direct contact on your skin or in your eyes often brings sharp pain and quick burns. The sensation isn’t just irritation—potassium bifluoride can sneak through the skin and start attacking the tissue underneath. It isn’t only the outer layer that suffers. In some bad cases, this chemical keeps going until it hits bone. No shop towel or quick rinse can guarantee it’s gone.
Inhaling the dust or vapor proves hazardous, too. Airborne potassium bifluoride easily irritates the lungs, nose, and throat. Enough exposure leads to coughing, chest tightness, or nosebleeds. The longer the exposure, the higher the risk for chronic effects: folks who regularly inhale the dust on the job could end up facing long-term lung trouble.
Potassium bifluoride doesn’t stop with surface-level harm. Ingesting even small amounts leads to much larger issues. Nausea, vomiting, cramps, and diarrhea hit fast, but behind those symptoms sits its effect as a source of fluoride ions. Those ions hit hard on the body’s calcium reserves, sometimes throwing a person’s electrolyte balance dangerously off. That shift—especially if left unchecked—can trigger heart rhythm problems or even seizures.
Lessons from the Lab and Worksite
Nobody wants to end up in an emergency room over something they thought would clean up a stain. In my experience working in older workshops, new hires often treat bottles without respect until a close call or a health scare makes an impact. Safety data sheets and warnings only do so much; hands-on training tells the full story. Gloves, goggles, and face protection don’t just tick a box—they offer real defense.
A well-run workplace sets up local exhaust ventilation, not just an open window. Clean surfaces and careful storage drop the chances of accidental spills. Good habits—like double-checking that gloves stay intact and hands get washed thoroughly—make accidents less likely.
Why All This Matters
Some hazards in life can’t be avoided. Potassium bifluoride, on the other hand, can and should be handled with care. Mistakes with this chemical lead to burns, lasting tissue damage, and sometimes hospitalization. People who spend their days around it—factory workers, tradespeople, lab techs—deserve to know what they’re up against, not just for their own safety but for their peace of mind.
Switching to safer alternatives for household use makes sense. On the industrial side, the solution comes from up-to-date training, real respect for the seriousness of chemical exposure, and regular safety checks—not just warnings on a label.
How should Potassium Bifluoride be stored?
Understanding the Risks Involved
Potassium bifluoride is not your average chemical. It’s used in glass etching, metal processing, and sometimes even pops up in laboratories that deal with fluorine chemistry. The big danger comes from its high reactivity with water and its ability to release hydrofluoric acid, a substance that can harm tissue, bone, and organs in surprisingly low amounts. Small mistakes, like keeping it in a humid spot or storing it with incompatible chemicals, can lead to much bigger problems.
Packing and Labeling: The Basics Matter
From personal experience in handling corrosive powders, I stick to containers that close tightly and don’t react with fluoride. High-density polyethylene (HDPE) or polypropylene jugs tend to hold up over time and don’t crumble when contacted by fumes. The original packaging usually works best, but if a container gets damaged, I reach for new, clean plastic containers. Glass should stay far away—hydrofluoric acid eats right through it.
Label everything properly and use clear, multilingual signage if the workplace has a diverse crew. It’s not paranoia to add warning stickers. Clear labeling saves confusion and avoids the kind of accidents caused by someone grabbing the wrong container in a hurry.
Where Should It Go?
Shelf space matters. I keep potassium bifluoride in a chemical cabinet, never near acids, organics, or stuff that’s flammable or moist. Chemical incompatibility charts do more than fill training time—they help cut down on risky storage mistakes. Any workplace that keeps this stuff has dedicated, ventilated storage, well away from food, drinks, and busy foot traffic. Better ventilation, fewer accidental inhalations.
Moisture and Temperature: The Silent Saboteurs
I’ve seen what happens when moisture sneaks in. The powder clumps, reacts, or starts releasing fumes, making a simple storage task a big problem. To fight moisture, I add desiccant packs inside cabinets. I always make sure the area stays cool—heat speeds up reactions that nobody wants. Windows nearby are a no-go because sunlight increases temperature swings and adds risk.
People: The Most Important Safety Factor
Storage rules work best when everyone buys in, so training means more than just a checked box. I’ve led teams where regular reviews of chemical handling keep these guidelines fresh. Workers need emergency instructions at arm’s reach. Spill kits with calcium gluconate gel (to treat hydrofluoric acid exposure) must stay near, not locked away.
Dealing with Waste and Old Stock
Old, damaged, or unknown potassium bifluoride never sits around long in a place I manage. We schedule hazardous waste pickups and avoid “stockpiling just in case”. Safe disposal costs money, but hospital bills and regulatory violations cost a lot more. Every employee gets clear instructions for reporting container damage or suspected leaks. No one wants mystery stains under a chemical cabinet.
Continuous Review and Community Responsibility
I’ve learned that what worked last year doesn’t always cut it now. Storage protocols need repeat reviews, based on updated safety data sheets and what regulators demand. Peer review and open communication across the team help spot oversights and keep everyone safer. Responsible chemical storage matters not just for the people inside the building, but for the community outside it too. Potassium bifluoride rewards careful attention, not shortcuts.
What precautions are needed when handling Potassium Bifluoride?
Getting Real About Potassium Bifluoride
Potassium bifluoride doesn’t get much attention outside labs and certain factories, but it deserves respect. This white crystal packs real bite—combining acid and fluoride risks. My stint working near chemical pipelines taught me fast that shortcuts around hazardous materials stack the odds against you. Potassium bifluoride can eat through skin, burn lungs, and break bones at the cellular level if you slip up. Knowing its nature keeps you out of the ER.
Why Protective Gear Isn’t Just for Show
Forget the thin plastic gloves you grab for kitchen cleaning. Rubber or neoprene gloves, chemical-resistant apron, and a sturdy face shield count as your shield. Potassium bifluoride doesn’t care if your hands are tough. Tiny splashes find nicks and hangnails. I watched someone’s skin turn white inside ten minutes from a drop trickling under a loose cuff. Scary thing is, the fluoride in this stuff can sink deep—bone-deep—before pain even hits. Good gear protects you before you realize you need it.
Really Think About the Air You’re Breathing
Potassium bifluoride dust and mist drift easily, especially if you’re pouring or mixing it. Masks rated for acid gases and particulates (not the cheap blue ones) help stop breathing problems before they start. In factories, you’ll see extraction vents set up over work benches—nobody wants these fumes in their lungs. I’ve felt the burn after a mask slipped off for barely a minute. Local ventilation isn’t optional. If you deal with this substance in a tight spot, hold off until you can fix the airflow.
Storage: Not Just Another Jar on the Shelf
Moisture and potassium bifluoride—those two don’t mix well. It breaks down fast, sending out fumes and making the room dangerous without you even seeing the problem. Dry, well-labeled, airtight containers make mistakes much less likely. You won’t find it by the coffee in any warehouses I’ve worked in—this takes a locked cabinet, somewhere folks without training can’t wander. Double-checking labels and expiration dates kept plenty of my coworkers from headaches, both literal and legal.
Spills and Accidents: Quick Steps Matter
No chemical training prepares you for the feeling right after a spill—heart in your throat, a thousand things to do at once. Sand or special spill kits soak up potassium bifluoride. No water, no sweeping. Dry powders react ugly with moisture. Get those gloves and goggles back on quick, rope off the area, and start ventilation fans. I’ve walked through the sharp scent that hangs after a spill; even if you think you got it all, air the place out, then wait before cleaning up.
Training Makes All the Difference
People sometimes joke about the endless safety meetings. Then someone pours a powder, a breeze hits, and that “boring” training keeps hands and faces intact. Clear first aid steps, like rinsing skin with water or using calcium gluconate gel, turn emergencies into footnotes, not tragedies. Every shop and lab should post emergency contact numbers and first aid steps in plain view. Keeping antidotes handy, restocking safety gear, and reading through procedures together make a real difference.
Finding Room for Better Habits
Potassium bifluoride doesn’t forgive rushed work or missing masks. Proper gloves, masks, and routines keep people safe. People who deal with strong chemicals form habits—from double-checking lids to stepping back after a spill. Sharing stories with new faces or old hands keeps the lessons fresh, so nobody underestimates the risks.
Is Potassium Bifluoride soluble in water?
Not Just Salt: Potassium Bifluoride's Real Impact
Chemistry class memories might bring back images of dissolving salt in water. Take that lesson, move it up a notch, and you arrive at potassium bifluoride. Drop this compound in water, and it dissolves with impressive ease. The fact surprises some people, since its name doesn’t roll off the tongue at the supermarket. Everyday users and scientists alike have more reasons to care about its behavior than just passing a quiz. Potassium bifluoride matters far away from the lab bench: safety, environment, and industrial purpose all play a role here.
Simple Science, Serious Consequences
Reports from chemical databases and textbooks settle the question quickly: potassium bifluoride dissolves very well in water. This seems routine, but it shapes how people use and store it in workplaces. In high school, we dissolved sugar and salt to learn about solutions. With potassium bifluoride, things get a lot riskier. The resulting mixture releases hydrofluoric acid in solution. That acid is notorious. It can burn skin and damage bones. The water solubility of potassium bifluoride is more than a lab trick — it influences rules for personal protection and chemical handling.
Why It Matters in Industry and Life
Industry relies on potassium bifluoride’s ability to dissolve in water for glass etching, metal cleaning, and even some types of wood preservation. With water as the medium, workers get a fast, controlled chemical reaction. Manufacturing counts on this predictability. The downside comes from exactly the same feature — the potential for dangerous spills or improper disposal. I met a glassworker who explained how easy it is to underestimate fumes, especially when water makes the chemical so accessible and active.
Environmental Stakes Are Real
Anyone tossing potassium bifluoride into water without thinking risks more than personal injury. High solubility means runoff can slip into wastewater systems. Even small releases may threaten aquatic life, since fluoride ions remain toxic in certain concentrations. Scientists and regulators keep studying water samples for traces, especially near factories or labs using the compound. Safety protocols rise to another level with chemicals that go straight into water — and with good reason.
Rethinking Chemical Safety and Education
I still remember my chemistry teacher showing how much respect even a harmless-looking solution deserves. Potassium bifluoride offers a good example for emphasizing real-world safety. Knowing its strong solubility should encourage both experienced professionals and beginners to use gloves, goggles, and proper ventilation every time. Emergency showers and spill kits turn from afterthoughts to mainstays in places that keep potassium bifluoride close by.
Learning From Everyday Chemistry
Widespread industrial use comes with responsibility. Proper signage and ongoing education keep people alert to risks. Some workplaces adopt smart labeling systems and regular audits for handling chemicals like potassium bifluoride. Planners set up designated disposal routines, minimizing the chances that the stuff lands in the wrong place. On the consumer side, even hobbyists tinkering with glass etching should read the warning labels and stay within recommended guidelines. With the ease potassium bifluoride dissolves, there’s no room for shortcuts.
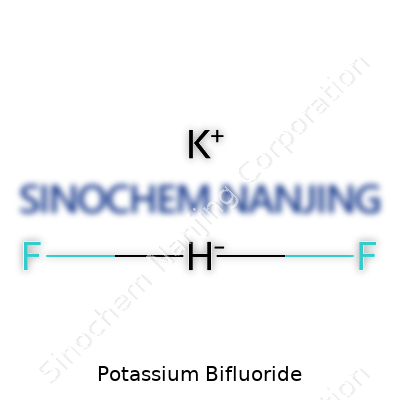
| Names | |
| Preferred IUPAC name | potassium;difluoride |
| Other names |
Acid potassium fluoride Bifluoridopotassium Potassium hydrogen difluoride Potassium hydrogen fluoride Potassium hydrofluoride Potassium fluoride hydrofluoride |
| Pronunciation | /pəˈtæsiəm baɪˈflʊəraɪd/ |
| Identifiers | |
| CAS Number | 7789-29-9 |
| Beilstein Reference | 358792 |
| ChEBI | CHEBI:88061 |
| ChEMBL | CHEMBL1201886 |
| ChemSpider | 43708 |
| DrugBank | DB14290 |
| ECHA InfoCard | 03f0fa9396c2-40ae-97dd-6e6c39a52e81 |
| EC Number | 231-575-6 |
| Gmelin Reference | Gmelin Reference: **1739** |
| KEGG | C18635 |
| MeSH | D011099 |
| PubChem CID | 24515 |
| RTECS number | TT2975000 |
| UNII | E9AJ6Q3U76 |
| UN number | UN1811 |
| Properties | |
| Chemical formula | KHF2 |
| Molar mass | 78.10 g/mol |
| Appearance | White crystalline solid |
| Odor | pungent |
| Density | 2.50 g/cm³ |
| Solubility in water | 100 g/100 mL (20 °C) |
| log P | -1.39 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 1.57 |
| Basicity (pKb) | 10.5 |
| Magnetic susceptibility (χ) | -39.5e-6 cm³/mol |
| Refractive index (nD) | 1.363 |
| Dipole moment | 1.92 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 128.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -697.99 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -850 kJ/mol |
| Pharmacology | |
| ATC code | V03AB29 |
| Hazards | |
| Main hazards | Toxic if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301, H314 |
| Precautionary statements | P234, P260, P264, P270, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P330, P363, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | Health: 3, Flammability: 0, Instability: 1, Special: - |
| Autoignition temperature | 510 °C (950 °F; 783 K) |
| Lethal dose or concentration | LD50 oral rat 100 mg/kg |
| LD50 (median dose) | LD50 (median dose): 100-215 mg/kg (oral, rat) |
| NIOSH | KWG197 |
| PEL (Permissible) | 2.5 mg/m³ |
| REL (Recommended) | 6 mg/m³ |
| IDLH (Immediate danger) | 500 mg/m3 |
| Related compounds | |
| Related compounds |
Hydrogen fluoride Potassium fluoride |

