Potassium Arsenite: Deep Dive Into a Notorious Compound
Historical Development
Potassium arsenite crops up in chemistry stories tracing back centuries, and its presence lingers from the days when medicine and poison looked an awful lot alike. Apothecaries in Victorian England dolled out Fowler’s solution, a tonic once sold as a treatment for everything from psoriasis to malaria—though the dangers ended up outweighing the supposed cures. Arsenic itself appeared everywhere, in dyes, rat killers, and even in the sanctuary of households. Potassium arsenite played a part as a stand-alone chemical and a tool in early lab work, making its mark on research and industrial processes. After regulations stepped in mid-20th century, the darker side of its use—poisonings, chronic toxicity—pushed scientists to step back and rethink both policy and safety, lessons revisited every time someone works with compounds of arsenic today.
Product Overview
Potassium arsenite, often listed as KAsO2 or simply arsenous acid potassium salt, draws attention both for historic notoriety and stubborn persistence in labs. It doesn’t come off a shelf as a staple, and you won’t spot it bulk-packed with common industrial chemicals. As a white or grayish powder, its solubility, toxicity, and reactivity with acids and oxidizers keep it squarely classified as a hazardous material. Users remain few, but the compound still pops up among researchers analyzing arsenical behavior or those revisiting old synthetic routes. The name pops up in paperwork, on chemical lists, and among legacy stocks, usually carrying hazard warnings bright enough to get anyone's attention.
Physical & Chemical Properties
Potassium arsenite turns out as a crystalline solid, white to light gray, with a molecular weight pushing 147.92 g/mol. You’ll notice its ready ability to dissolve in water—one reason it slides so readily into biological systems, where it starts its mischief by interfering with cellular processes. The compound stays odorless; no tell-tale scent warns of danger, a trait that makes it more insidious in cases of exposure. Chemically, potassium arsenite relies on arsenic in its trivalent state (As(III)), unlike its more oxidized cousin potassium arsenate (As(V)). It has a melting point around 200°C, though decomposition starts far below, so heating leads not to a clean melt but release of arsenic-rich vapors and oxides. A reaction with acids kicks off liberation of toxic arsine gas, while oxidizing agents can shift arsenic to even more dangerous forms. Once these properties register, the need for airtight safety protocols becomes clear with just one glance at the compound.
Technical Specifications & Labeling
Labels on potassium arsenite containers do not mince words. Every bottle screams with hazard pictograms showing dead trees, crossbones, health hazard silhouettes, and clear text about acute toxicity, environmental damage, and necessary protective gear. Technical specs follow strict international standards—purity typically hovers around reagent grade, commonly above 98%. Documentation covers batch number, date of production, shelf-life (although storing the stuff invites regulatory scrutiny), and analytical certs. Transport, use, and disposal rules tie back to REACH and OSHA, calling for secondary containment, chemical fume hoods, and mandatory reporting if the chemical ever leaves the store room. No casual handling, no open benches.
Preparation Method
Chemists make potassium arsenite by dissolving arsenic trioxide (As2O3) in a solution of potassium hydroxide (KOH). The mixture reacts, forming potassium arsenite and water:
As2O3 + 2KOH + H2O → 2KAsO2·H2O
During this process, tight air extraction, full PPE, and rigorous waste control come into play. Even low vapor concentrations threaten health. Any careless procedure punishes with headaches, vomiting, and much worse, so modern labs rely on sealed systems, and automated dosing wherever possible. The product comes out as a solution or, with careful evaporation, a crystalline salt. Technicians run spectroscopy and titration to check product quality, never assuming purity just because the procedure ran by the numbers.
Chemical Reactions & Modifications
Potassium arsenite is reactive in a handful of classic inorganic chemistries. In the lab, it partners with acids to turn out volatile and violently toxic arsine gas, or it gets hit with oxidizers—like hydrogen peroxide—to convert it to less toxic but still-dangerous arsenates. In organoarsenic chemistry, this compound acts as a reducing agent, stripping oxygen from more stable arsenate forms. The instability of arsenites in air—where slow oxidation creeps in—poses storage headaches. Once you start modifying potassium arsenite, every step opens up risk management questions, from controlling exposures to ensuring byproducts don’t sneak out with the wastewater. Even when freshly prepared, it edges toward decomposition if left exposed. Proper technique involves not just glassware, but building-wide ventilation and years of trained instinct.
Synonyms & Product Names
Potassium arsenite goes by a handful of names in catalogs and literature: arsenous acid, potassium salt; potassium meta-arsenite; and KAsO2. Some labels call it “potassic arsenite,” and in vintage lab books, it appears as Fowler’s solution (when in its original medicinal mixture with water and alcohol). Regulatory agencies sometimes call it potassium salt of arsenious acid or just “arsenite salt” in restricted substances lists. Whatever the name, the dangers travel right along with the change in vocabulary, so anyone handling it gets familiar with its aliases fast.
Safety & Operational Standards
Working with potassium arsenite means treating it as if it could cause acute poisoning every time. I learned in school that a mere dusting could spell disaster if inhaled or absorbed through skin. Chemical hygiene plans require gloves, goggles, and lab coats, with strict glove disposal—not the kind reused for a week. Operations go into certified fume hoods, and spill kits stand ready with absorbents specific to arsenic. I once saw a safety audit ding a lab because their arsenic spill protocol was tucked away in the wrong binder—these details matter. Waste goes in separate containers marked hazardous for heavy metals, locked up and collected by regulated disposal companies. Training drills drive home the need for immediate action on exposure, and OSHA watches over compliance, issuing penalties for lax standards. Medical monitoring follows staff who work with this and other arsenicals, checking blood arsenic levels and symptoms over time, a real-world reminder that the risks are not theoretical.
Application Area
These days, potassium arsenite turns up mainly in targeted research, legacy sample analysis, and forensic investigation. In the past, agriculture and medicine roped in potassium arsenite for fungicides and tonics, but modern regulations pulled those applications permanently, closing the book on casual use. Labs working on arsenic metabolism in plants or animals use it under tight controls to untangle how cells process toxic metals. Historic pesticide and paint analysis sometimes require reference samples or spike studies with potassium arsenite analogs. Forensic chemists, and occasionally environmental scientists, need real samples to calibrate detection methods for arsenic in groundwater and soil, learning from mistakes that once swept across the industry. No new everyday uses slip through regulation, but arsenite occasionally pops up in specialty syntheses where other reagents won’t work.
Research & Development
Innovation around potassium arsenite looks a lot different than other chemicals. Instead of scaling up use, most research today points toward safer substitutes, remediation, and new sensors to track low levels in the environment. Analytical labs work on better ways to tell arsenite from arsenate, since their toxicities differ. Crop scientists explore how food plants absorb, metabolize, or exclude arsenite, aiming to keep contamination out of food chains. Some medical researchers use it for cell studies, probing how trivalent arsenic disrupts DNA repair or triggers apoptosis. In the few cases where researchers still use potassium arsenite as a reagent (say, in redox-asymmetrical synthesis), teams try to minimize quantities or run reactions in closed microfluidic systems. Grant proposals on this topic earn scrutiny to make sure no one’s repeating mistakes of the past.
Toxicity Research
Potassium arsenite lands near the top of the charts for documented human toxicity. Its LD50 sits as low as 14 mg/kg in mammals. Early clinical cases, especially with Fowler’s solution, mapped out its cumulative effects: skin lesions, neuropathy, cardiovascular disease, and a strong link to multiple cancers. Researchers in the last decade sharpen understanding of molecular mechanisms—arsenite targets sulfhydryl groups, busting up essential enzymes and hijacking cell signaling. Chronic, low-level exposure triggers oxidative stress, damages DNA, and breaks down immune responses. Scientists working in environmental health maintain a keen interest in arsenite, as groundwater contamination issues persist in South Asia and other arsenic-rich regions. Social issues cross wires with biochemistry here, since some rural communities still suffer exposure despite decades of health campaigns.
Future Prospects
Potassium arsenite’s future doesn’t look promising in terms of new uses. Most signs point to tighter regulations, improved remediation technology, and development of arsenic-free alternatives for every niche where it used to appear. The R&D world centers more on cleanup than application. Scientists go after soil and water purification, new iron oxide filters, phytoremediation with hyperaccumulator plants, and low-cost sensors for fast, field-based detection. Companies that once handled potassium arsenite have pivoted toward safer chemistries. Looking forward, any revival would face both legal and societal roadblocks, plus a scientific consensus that the world can do better with less arsenic getting into circulation. As the world gets more aware of chemical safety and legacy pollution, potassium arsenite stays in the textbooks, not on the workbench, except under strictest controls.
What is Potassium Arsenite used for?
Where Potassium Arsenite Shows Up
Potassium arsenite turns up in an unusual part of chemistry’s toolkit. Not too many folks outside scientific or agricultural circles bump into this compound, which mixes potassium, arsenic, and oxygen. Once, it found heavy use as an agricultural pesticide and fungicide. It helped farmers fight off pests and plant diseases, especially before safer or less toxic tools rolled out. The formula did its job well, but the health risks carried a long shadow.
In the lab, potassium arsenite still plays a role as a reagent in chemical experiments and analytical testing. Chemists use it to prepare other arsenic compounds, or as a reducing agent. Certain specialized chemical syntheses depend on its unique reactivity. Some older water treatment and wood preservation methods involved potassium arsenite, though most countries pulled back as data about arsenic toxicity grew stronger.
What Health Concerns Really Mean
My own learning in environmental science hammered home how exposure to compounds like potassium arsenite doesn’t just linger in textbooks. Arsenic builds up in soil and water. Once it’s there, it threatens entire communities. Chronic exposure can trigger cancer, skin problems, heart disease, and damage to the nervous system. Kids and elderly people face the highest risk. Soil residues stick around for years, raising ongoing challenges for anyone who works the land later.
The story of potassium arsenite shows how science keeps pace with new evidence about health. Regulatory agencies responded by banning or placing heavy limits on this chemical. The World Health Organization and the U.S. EPA both point to arsenic as a proven human carcinogen, based on decades of real-world effects and lab research. That knowledge shifted policies and forced chemical companies, farmers, and lab instructors to rethink what they use day-to-day.
Modern Agriculture and The Push for Safer Solutions
Farmers have always battled pests and tough soil conditions. Caught between the need for yield and the need to keep people safe, chemical choices matter. Decades ago, many didn’t have good options. Potassium arsenite solved certain problems, but left behind new ones. Over time, community-health studies and personal stories from growers laid out a picture where short-term gain didn’t outweigh the longer-term risks.
Shifting away from hazardous compounds isn’t easy. Farmers need reliable ways to fight blight and insects. Stronger rules, increased awareness, and government support for safer methods can push the process forward. Integrated pest management, natural fungicides, and strict testing for soil contamination reduce the need for toxic compounds. A few countries have created cleanup funds or transition programs to help smaller growers rebuild land damaged by past arsenic spraying.
Living With The Chemical Legacy
Every generation ends up wrestling with the legacies of past chemical decisions. For potassium arsenite, that legacy means enforcing bans, cleaning up contaminated grounds, and helping scientists innovate safer tools. Open data, public education, and a willingness to learn from honest mistakes make a real difference.
Even today, chemical safety relies on trust as much as on hard science. Families and workers living near old industrial lands deserve transparency about what’s in their water or soil. Finding solutions means governments, farmers, and communities have to talk honestly about risk, trade-offs, and the pace of change.
Is Potassium Arsenite hazardous to health?
What Potassium Arsenite Really Means for Health
Potassium arsenite rarely gets attention in daily conversation, but it played a role in agriculture and industry as a pesticide and preservative for decades. Today, people want to know what kind of danger it poses—not just to those working with it, but to anyone who may come into contact due to environmental pollution or old chemical stockpiles. Growing up in a farming area, I heard plenty about the risks of arsenic-based chemicals. Most local folks kept a healthy distance, knowing they weren’t just dealing with a regular fertilizer or weed killer.
Why Toxicity Creates Real-Life Health Issues
Science groups like the CDC and WHO call potassium arsenite a clear hazard. It’s made up of both potassium and arsenic, and that arsenic part is trouble. Exposure to arsenic compounds, no matter what element they’re paired with, attacks the body’s organs over time. I’ve seen old family friends develop nerve issues and skin lesions, and their doctors warned them about past chemical exposures, likely including arsenic products.
Drinking arsenic-contaminated water or breathing dust during pesticide application opens the door to poisoning. Chronic low-level exposure raises the odds for certain cancers—especially skin, lung, and bladder types. In the short run, someone breathing in enough potassium arsenite dust gets headaches, stomach cramps, and maybe even convulsions. Arsenic doesn’t always show up right away—it can take years for symptoms to appear, so people don’t always connect sick days with the hidden chemicals in their environment.
Strong Evidence: Not Just Theory
Researchers dug into what happens in towns with arsenic in the groundwater. Cancer rates rise, and immune systems weaken. That tracks with what I heard from families living along old industrial belts, who recounted cancers and mysterious illnesses after chemical use nearby. Potassium arsenite hasn’t been widely used for decades, but its legacy lingers. Old sites keep leaching toxins into soil and water, which explains strict international bans.
Hard numbers back this up. The EPA sets maximum contaminant levels for arsenic in water at 10 parts per billion, a figure that means even tiny doses over many years build up in the body. One sip or puff of dust probably won’t kill, but nobody wants to gamble when it comes to their own health—or their kids’ futures.
How People Can Limit Exposure
Avoiding potassium arsenite takes vigilance—especially around older homes, farmland, or storage sheds. Cleaning up old pesticides demands hazmat suits and trained teams, not just gloves and masks. Local governments need to test wells and public water, then share results honestly with everyone. Policies banning arsenic-based products work only if people follow disposal rules, and neighbors have to know what’s buried around their properties.
Safer alternatives now do the work arsenic once did, but protecting public health calls for ongoing education. Doctors working in rural areas should ask about chemical exposure histories, and schools must keep families in the loop about possible hazards. From my own view, nothing beats sharing real-world experiences—stories about sickness or recovery mean more than faceless warnings on a label.
The Bigger Picture
Potassium arsenite represents a classic case of a substance that once seemed helpful but now hangs around like a ghost, causing damage that’s hard to spot until it’s too late. People deserve to live and work without worrying about invisible poisons in their water, fields, and backyards. Open communication, tough enforcement, and investment in safer practices make a difference. My advice: respect the legacy, push for cleanups, and demand that health comes before convenience—no matter what chemical is on the label.
What are the storage requirements for Potassium Arsenite?
Why Worry About Storing Potassium Arsenite?
Potassium arsenite poses a serious hazard—its toxicity isn’t something to take lightly. Just a small amount of arsenic-based compounds like this can do real damage to people and the environment. I remember working in an old research lab where one bottle sat untouched for years. The label started to fade, the cap rusted, and nobody wanted to be the person to deal with what was almost forgotten but always dangerous.
Essential Storage Location and Setup
The right placement for potassium arsenite starts with isolation. Keep it in a locked, ventilated cabinet away from everyday foot traffic. A storage area needs solid ventilation—chemical fumes build up fast. Direct sunlight warms containers and raises pressure, so cool, dry spaces work best. Humidity triggers chemical reactions, corrosion, and can erode container seals. Basements often stay cool, but always check for any water leaks because moisture ruins packaging and could set off a chain of unsafe reactions.
Every expert I’ve spoken to points out the need for secondary containment. Think trays or tubs that catch leaks and spills. A stubborn crack in the bottom of a glass jar can turn into a crisis if undetected. Metal shelving tends to corrode over time, so chemical-resistant shelving wins in the long run. The cabinet should hold only compatible chemicals—no acids, no oxidizers, nothing that could start off a reaction if a small spill happens. Overcrowded shelves raise the risk of bottles tipping and breaking.
Labeling and Documentation: No Room for Guesswork
Potassium arsenite ought to carry a clear, permanent label. The hazard, the date of receipt, and the responsible person need to be easy to read, every single time. If the original label is lost, take five minutes and create a new one. In my first job, a missing label led to a whole shelf getting quarantined—nobody wanted to move or touch unknown bottles. Proper labeling saves hours and cuts down anxiety for everyone working near these materials.
Every chemical entering a storage cabinet gets logged in a central inventory list. This means quick tracking, easier inspections, and less chance of ignoring a forgotten bottle. Safety audits catch issues before they get out of hand. I’ve seen plenty of labs where regular checks would have prevented big, expensive cleanups later.
PPE, Spill Kits, Fire Protection: Safety Can’t Wait for Accidents
Personal protective equipment (PPE) stands next to every hazardous chemical storage. This goes beyond gloves and goggles—full splash protection works best. Handling potassium arsenite without the right gear risks skin absorption, which can have permanent health effects. Emergency showers and eye-wash stations belong within a short, quick walk.
Spills demand an immediate response. A spill kit built for toxic powders and liquids sits close by—complete with absorbent materials, not just basic towels. Fire protection is part of this setup as well. Although potassium arsenite itself does not burn, a fire nearby can release dangerous arsenic fumes. Class ABC extinguishers work well for most situations, but chemical safety training means knowing exactly how to respond if the room fills with smoke or the bottle breaks open.
Disposal and Long-Term Risk
Bottles don’t last forever. Inspect containers regularly for corrosion or damage. If crystals build up or liquid levels change, call a hazardous waste team. Avoiding the temptation to "just leave it alone" prevents bigger problems. Hazardous waste disposal isn’t cheap, but it’s far less costly than a contamination cleanup or health scare.
With all the risks, potassium arsenite storage cannot become routine. Every step protects people and the surrounding community. A little extra effort makes the difference between safe chemical management and a dangerous situation nobody wants to face.
How should Potassium Arsenite be handled safely?
What Makes Potassium Arsenite Dangerous?
Potassium arsenite stands out as a potent poison. The compound carries a long track record of causing harm, both to those working around it and to the environment. Exposure can happen through inhalation, skin contact, or swallowing. My work in a lab has shown how even small lapses bring heavy risks, including cancer, skin irritations, and lasting nerve damage. Historical data report arsenic exposure has been linked to rates of skin, lung, and bladder cancers, as seen in both industrial accidents and prolonged, low-level chronic workplaces. Keeping clear of those dangers calls for more than reading warning labels.
Personal Protective Equipment: A Non-Negotiable
Stepping into a lab where potassium arsenite waits on the shelf, I never skip goggles and gloves. A sturdy lab coat and a fitted mask join the team. Simple soap and water do not clear up an accident—this compound lingers. Choice of gloves matters, too. Nitrile and neoprene last longer against arsenic solutions. Clothing should cover skin fully, leaving nothing open to a spill or a splash. In my experience, skipping these steps only creates regret, and stories circulate about colleagues who learned this the hard way.
Containment and Ventilation: Keeping Toxins Controlled
Potassium arsenite does not belong out in the open. Every time I open a container, I work under a fume hood. That strong airflow pulls vapors and dust away, stopping them from entering the lungs. Labs and industrial setups keep this equipment for a reason—simple fans do not offer enough protection. Sealed bottles with clear labels cut down mistakes, and spills require special absorbent pads and containers. No one trusts paper towels or regular trash cans for cleanup. Heavy fines follow poor storage, and the law backs this up across many countries.
Training and Awareness: Knowledge Saves Lives
No shortcut exists for training. Understanding the risk isn’t optional. Every person handling potassium arsenite should know first-aid steps, understand signs of poisoning, and recognize spills on the spot. I have seen seasoned chemists hold open talks on these topics, showing new staff what danger looks like in real life. Emergency plans hang on the wall in plain view and drills never feel like a waste of time. Even people with years of experience find something new during hands-on sessions.
Disposal and Environmental Responsibility
Potassium arsenite does not belong down any drain or in ordinary garbage. Special waste contractors know how to remove and destroy arsenic compounds without risk to water supplies or surrounding soils. I learned that simple mistakes can seep into groundwater, poisoning beyond the workplace. Sticking with certified disposal services—never home remedies or shortcuts—protects everyone downstream. Governments and regulators track how arsenic leaves facilities and punish missteps heavily, guided by hard data on environmental accidents.
Solutions and Safer Alternatives
Some processes still rely on potassium arsenite, but safer substitutes exist for most tasks. Where possible, labs switch to less toxic chemicals as a matter of policy. Companies invest in new technology and training to work with these alternatives. Stepwise removal of arsenic-based chemicals shows a track record of cutting health issues over time, backed up by studies from chemical safety boards. The push for safety has made workplaces less dangerous, reminding me no job is worth risking long-term health.
What is the chemical formula of Potassium Arsenite?
Understanding Potassium Arsenite
Potassium arsenite carries the chemical formula KAsO2. For folks who took high school chemistry, potassium stands for the letter K on the periodic table, and arsenic goes by As. Marrying these with oxygen, the compound shapes up as KAsO2. Most people working in agriculture, industry, or environmental testing might never bump into this formula outside a textbook, but its impact stretches far beyond the chemistry classroom.
More Than Just Letters and Numbers
Potassium arsenite slips into the spotlight mainly because of its historic use as an insecticide, herbicide, and wood preservative. In my college days, looking at the list of toxic substances in an environmental chemistry course, this name jumped out. The troubles tied to it come from arsenic — a notorious poison. Long-term exposure to arsenic-based compounds stirs up health disaster zones, from skin lesions to certain cancers.
Over the years, arsenic’s dangerous side has pressed regulators around the world to crack down on how potassium arsenite and similar chemicals get made, handled, and disposed. Some contaminated legacy sites in old farm areas still carry the chemical's footprint. I once volunteered in an environmental outreach to test groundwater outside an old agricultural belt, and more than a few test strips showed tell-tale traces of arsenic. Responsible stewardship today comes from respecting these lessons of the past.
Why Chemical Literacy Counts
Learning a chemical formula like KAsO2 isn’t just memorization. Recognizing what those symbols represent gives everyone—including curious students and home gardeners—a leg up in understanding product labels and safety warnings. Imagine grabbing something from an old storage shed, only to realize the fine print warns about potassium arsenite as a hazardous ingredient. That quick recognition can prevent accidents, safeguard health, and stop contamination before it starts.
Government agencies and scientists keep track of chemicals like this down to the last decimal. In the States, the EPA restricts most uses of potassium arsenite these days, and international agreements keep it tightly regulated elsewhere. This move hasn’t only cut down on accidental poisonings—it’s also brought cleaner drinking water for rural communities hit hard by past practices.
Charting a Safer Path Forward
Facing environmental risks from chemicals like potassium arsenite calls for practical action. Farmers and property owners still discover barrels and containers with faded hazard symbols on old land. Simple steps can help: clear labeling, proper documentation of all chemical storage, and access to local hazardous waste disposal programs. Communities can spread the word, schools can add basic chemical safety to the curriculum, and local health departments can share warnings during well-testing days.
Shifts also happen in science and technology. Researchers are coming up with new, less toxic alternatives for the tasks arsenic-laced compounds once tackled. Some have found ways to rehabilitate soil and water using natural materials or bioremediation, relying on living organisms to clean up arsenic-contaminated sites. Real progress shows up not just in numbers on a chart but in families enjoying safe water straight from their taps.
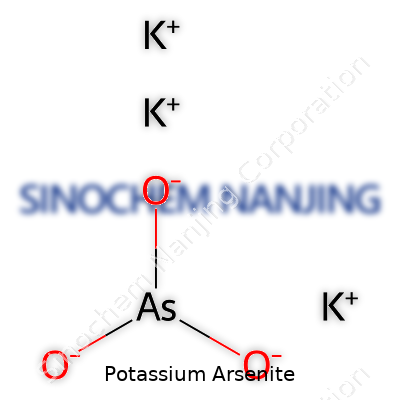
| Names | |
| Preferred IUPAC name | Potassium arsenite |
| Other names |
Arsenious acid, potassium salt Potassium metaarsenite Tripotassium arsenite |
| Pronunciation | /pəˈtæsiəm ˈɑːsənaɪt/ |
| Identifiers | |
| CAS Number | 10124-50-2 |
| Beilstein Reference | 1209263 |
| ChEBI | CHEBI:88221 |
| ChEMBL | CHEMBL1201601 |
| ChemSpider | 15323 |
| DrugBank | DB14649 |
| ECHA InfoCard | 03e9f001-5c4a-4ea4-bb51-0dc1c1e417bb |
| EC Number | 204-187-7 |
| Gmelin Reference | Gmelin 12298 |
| KEGG | C18714 |
| MeSH | D011125 |
| PubChem CID | 24856 |
| RTECS number | CG6475000 |
| UNII | A7TK2V3F78 |
| UN number | UN1559 |
| CompTox Dashboard (EPA) | DTXSID1020346 |
| Properties | |
| Chemical formula | KAsO₂ |
| Molar mass | 197.913 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 2.56 g/cm³ |
| Solubility in water | Soluble |
| log P | -0.31 |
| Vapor pressure | Negligible |
| Acidity (pKa) | ~9.0 |
| Basicity (pKb) | 11.5 |
| Magnetic susceptibility (χ) | -60.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.580 |
| Viscosity | Viscous liquid |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 151.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -666.6 kJ/mol |
| Pharmacology | |
| ATC code | V03AB03 |
| Hazards | |
| Main hazards | Fatal if swallowed, in contact with skin or if inhaled. Causes severe skin burns and eye damage. May cause cancer. May cause damage to organs through prolonged or repeated exposure. Very toxic to aquatic life with long-lasting effects. |
| GHS labelling | GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H350: May cause cancer. H373: May cause damage to organs through prolonged or repeated exposure. |
| Precautionary statements | P260, P262, P264, P270, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P311, P314, P330, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-0-A |
| Lethal dose or concentration | LD50 oral rat 14 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 14 mg/kg |
| NIOSH | SN4530000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Potassium Arsenite: 0.01 mg/m³ (as As) |
| REL (Recommended) | 0.2 mg As/m³ |
| IDLH (Immediate danger) | 5 mg As/m3 |
| Related compounds | |
| Related compounds |
Sodium arsenite Copper arsenite Lead arsenite Arsenic trioxide Potassium arsenate |

