Potassium Arsenate: Understanding Impact and Responsible Use
Historical Development
Potassium arsenate started drawing attention centuries ago, mainly in European chemistry labs. Researchers prized its unique ability to deliver arsenic in a soluble format. Alchemists once believed in mysterious powers hidden in these compounds, but modern science quickly revealed more practical and precarious truths. Through the 1800s, interest grew for its use in pigments, pest control, and glass manufacturing. Sadly, back then, many harnessed its toxic potential without any real oversight, which led to some tragic health incidents among factory workers. Over time, regulations began to tighten, not from sudden enlightenment but because communities witnessed the price of carelessness.
Product Overview
Potassium arsenate comes as a clear, colorless, or sometimes white crystalline powder. Chemists recognize it by its strong inorganic structure defined by formula K3AsO4. Years in the research field have shown that this compound dissolves well in water — a feature that brought plenty of industrial interest. In smaller amounts, it stabilizes mixtures or plays the role of a mordant in dyeing, though much of that utility faded as the danger became better known. In any lab, the unmistakable bitter-metallic taste and absence of odor allow it to stand out among other potassium salts.
Physical & Chemical Properties
At room temperature, potassium arsenate stands firm as a crystalline solid. Its high solubility in water lets it travel fast in solutions, which spells both convenience and risk. With a melting point generally above 800°C, the substance stays stable across a wide range of laboratory procedures. Its pH leans alkaline, so it can shift the properties of mixtures rapidly during experiments. From first-hand safety training, I have seen how quickly it can produce dense, persistent dust if handled poorly. That aspect alone raises alarms, as inhalation triples the chance of accidental poisoning.
Technical Specifications & Labeling
Every supply of potassium arsenate lands with strict labeling protocols. Genuine manufacturers stamp containers with warning icons reflecting its acute toxicity. You can expect hazard statements that leave no room for doubt about the risks involved in handling or storing the compound. Each batch comes with a Certificate of Analysis, containing purity percentages (often 98% or better) and detailed impurity profiles. Product data sheets provide information on molecular weight (around 246 g/mol), batch origin, and an emergency response guide. From experience, it's the small print about incompatibilities — strong acids, easily reduced metals — that separates a careful operator from someone inviting trouble.
Preparation Method
Chemists usually obtain potassium arsenate through a controlled reaction between arsenic acid and potassium carbonate. Workers add potassium carbonate slowly to avoid runaway reactions, keeping temperatures steady and using ventilation to carry away heat and rogue fumes. After the bubbling stops, the solution is filtered, then evaporated down to yield pure crystals. Larger operations often employ jacketed reactors and automated dosing to keep things safe and consistent. Those in research settings sometimes rely on smaller, batch-based approaches. In both industrial and laboratory settings, personal protective equipment is not negotiable—not after the well-documented poisoning incidents of earlier decades.
Chemical Reactions & Modifications
Potassium arsenate fits snugly into various chemical syntheses. Its main reaction partner is silver nitrate, where it forms silver arsenate — a classic test for the presence of arsenates. Mixing it with acids generates arsenic acid and potassium salts, often under controlled conditions due to the production of hazardous byproducts. Lab work frequently explores modifications, such as reducing potassium arsenate to more complex arsenic compounds for use in tracer studies. Over the years, research papers have documented countless routes to integrate potassium arsenate into new chemical frameworks, but the conversation always circles back to containment and personal safety.
Synonyms & Product Names
Chemists often refer to potassium arsenate as potassium orthoarsenate or tripotassium arsenate. In literature, trade names show up infrequently because most vendors stick to the basic IUPAC labeling for regulated substances. Historical texts might mention “Fowler’s Solution,” a medicinal preparation that, to modern eyes, feels reckless due to its arsenic load. Researchers also encounter terms like “arsenic acid, tripotassium salt” and “K3AsO4,” which reinforces the need for vigilance in identifying exactly which form of arsenate sits on the shelf.
Safety & Operational Standards
Rules governing potassium arsenate could fill volumes. Under the oversight of agencies like OSHA and the EPA in the United States, only highly trained individuals gain access to this compound. Protocol demands storage in secure, ventilated cabinets away from any acids, reducing agents, or easily oxidized materials. Operations rely on airtight gloves, double-layered protection, and immediate access to chemical spill kits and eyewash stations. Many labs run periodic drills on arsenic exposure symptoms—persistent headaches, confusion, muscle weakness—and have chelating agents on hand just in case. Disposal channels must account for groundwater safety, since arsenic contamination persists in the environment for generations.
Application Area
Most scientists today steer clear of potassium arsenate for routine applications. In earlier decades, its use as an insecticide and herbicide almost seemed progressive. Farmers blanketed fields in arsenic-based sprays, failing to anticipate the fallout decades later: toxic soils and cancer clusters in affected communities. Some niche uses survive — historical artifact preservation, specific dye processes, and a handful of analytical chemistry techniques. If you walk through a modern chemistry building, you’ll almost always find potassium arsenate locked tightly away, handled only when no safer alternative exists.
Research & Development
Modern research leans more toward finding ways to treat or remediate environments contaminated by arsenic than toward exploring new applications for potassium arsenate. Scientists dedicate real resources into soil science and filtration technologies meant to pull arsenic contamination out of ground and water. In medical research, focus shifts toward understanding arsenate’s interference with cell signaling and metabolic pathways. Biomedical teams explore novel chelation therapies in hopes of mitigating arsenic toxicity in exposed populations. Academic journals tell stories of how potassium arsenate still provides value as a model toxicant for investigating the mechanisms behind oxidative stress, DNA damage, and cellular apoptosis.
Toxicity Research
Toxicologists keep potassium arsenate constantly under their microscope. Data shows arsenate compounds disrupt cellular respiration by targeting enzymes essential for ATP production. Even minor exposure results in headaches, nausea, and neurological issues; chronic cases build up risk for skin lesions, organ failure, and various cancers. Studies on exposed populations — from miners to manufacturing workers — lay bare the dangers. Experience in research settings reminds me that safeguards are only as effective as the habits of the people using them. Medical case reports across continents back up the reality that acute arsenic poisoning leaves lasting scars on communities, making the push toward exposure prevention much stronger than the push for new industrial applications.
Future Prospects
Looking forward, demand for potassium arsenate in industry will likely continue to decline as regulatory pressures increase. Research will probably focus even more on environmental cleanup and on safer alternatives for any process where arsenate once figured. Phytoremediation projects, where plants help extract arsenic content from contaminated soils, inspire hope for communities sitting on the edge of old agricultural plots. Advances in chemistry may someday yield non-toxic analogs that serve the same laboratory functions. Until then, the path forward sits in education, strict controls, and unwavering respect for a substance capable of so much damage if left unchecked.
What is Potassium Arsenate used for?
Tracing Back Its Story
Anyone reading about potassium arsenate can’t avoid running into its long, checkered past. Labs used to rely on it as a source of arsenic and potassium for chemical reactions. Agriculture saw it as a weapon against insects and fungi back when safety barely registered in the public conversation. The white, tasteless powder did its job—and left behind an environmental headache. Its effectiveness came with a cost hardly anyone noticed until much later.
Modern Use Fades for a Reason
Nobody working in labs today reaches for potassium arsenate to solve daily problems. Most countries have shut the door on it as an agricultural chemical. People now know arsenic isn't just dangerous in large spills—a few grains can harm soil, contaminate groundwater, and stay in food grown years later. Chronic exposure links to skin, lung, and bladder cancers, plus developmental delays in children. That's why, if you find it in a storeroom, red flags go up fast.
Why Scientists Once Liked It
Potassium arsenate has a knack for interfering with biological processes. Arsenate substitutes for phosphate inside living cells, but it breaks chemical rhythms a body relies on. Early biochemists knew this made it a sharp tool for blocking enzymes in the lab. Teachers used it to show students what goes wrong when DNA or ATP gets twisted by the wrong element. Medical researchers once used these reactions to study illness, but modern ethics push us away from tools that leave poison on the workbench.
Chemistry Lessons with a Cost
I’ve worked in university labs that held jars of dangerous compounds leftover from early twentieth-century teaching kits. You find potassium arsenate sitting among them, labeled with warnings half as big as they ought to be. The legacy isn't just a bottle on a shelf—cleanup of contaminated ground from past spills costs millions, and fear of arsenic exposure keeps some communities from using local wells. Facts speak clearly: the arsenic in potassium arsenate causes more harm than it ever solved in agriculture or research.
Real Solutions Start with Safer Choices
Modern chemists use less toxic alternatives. If anyone needs to mimic what arsenate does in a reaction, there's a good chance a synthetic analog or computer model can replace the hands-on hazard. In farming, advanced insecticides and integrated pest management step in. Cleanup depends on careful mapping, soil removal, and sometimes bioremediation using plants that take up arsenic—drawing toxins out of the ground over several years.
Learning from a Darker Past
Potassium arsenate’s legacy reminds us to stay cautious with chemicals whose risks we do not fully understand. Rules now force companies to deeply test pesticides and lab reagents for safety before letting them near food or public water. Industry, government, and academics must keep speaking up if shortcuts threaten the health of people or the land around them. Experience in research and community science shows that putting health first ends up saving time, money, and lives.
Is Potassium Arsenate hazardous or toxic?
Looking Beyond Chemistry Classrooms
Potassium arsenate pops up in science labs, older pesticides, and some industrial processes. Many folks might remember seeing the name in chemistry textbooks, but few recognize the real risk behind a simple bottle labeled K3AsO4. The truth feels stark: potassium arsenate brings with it the deeply rooted dangers of arsenic, a top-tier toxic substance.
Human Health Risks: Not Just A Theoretical Concern
Arsenic compounds like potassium arsenate have a reputation for good reason. Exposure to small doses over time or a single large dose leads to severe poisoning. People working with this stuff often deal with symptoms such as stomach pain, skin lesions, and nerve damage. Research connects long-term arsenic exposure to several types of cancer, including skin, bladder, and lung. According to the World Health Organization, more than 140 million people in 50 countries drink water contaminated with arsenic—proving these risks run far beyond chemistry labs or factories.
Workplace and Environmental Challenges
Factories and farms handled potassium arsenate more freely in the past. Some soils still carry the poison from decades ago, quietly affecting crops and groundwater. I used to spend weekends helping family on a farm where pesticide barrels from the seventies collected dust in a shed. These barrels leaked, leaving white crusts behind. We never thought twice about it until a neighbor’s soil test found arsenic levels twenty times higher than what health standards allow. Crops grown there had higher arsenic content in roots and stems, another sign the problem stays long after the chemical is gone.
Once potassium arsenate enters a river or field, it seeps deep, stubbornly resisting clean-up attempts. Fish absorb it, plants suffer, birds picking seeds get sick. Communities living on contaminated land face lifelong risks. In Bangladesh and some parts of the U.S., families drink from wells quietly tainted with arsenic.
Handling and Prevention: Not Just About Gloves and Labels
Putting on gloves and posting warning signs only get you so far. Real protection means policies with teeth and rapid testing. Industries that once used potassium arsenate have moved to safer alternatives in some regions. Strict regulations help, but enforcement often looks patchy. Some countries with less oversight still sell products containing this compound.
More public awareness can push industries and local governments to act. Many dangerous chemicals faded from use after people insisted on safety standards and regular monitoring. Regular testing of wells and soil, especially in places with an industrial history, can catch issues before they become crises. On the personal side, anyone working with older pesticides or lab chemicals should keep potassium arsenate off the shelf for good, following disposal guidelines carefully.
Building Solutions From Community Experience
There’s a lot of power in sharing stories—from retired farmhands warning neighbors of old chemical sheds, to doctors tracking unexplained symptoms back to arsenic-laced water. Community-level education paired with access to simple testing kits goes a long way. Drawing on these experiences, families and workers push for action on contamination.
Potassium arsenate’s risks might never vanish, but keeping them in check means staying alert, respecting the lessons of the past, and demanding both safety and accountability where it matters most.
What is the chemical formula of Potassium Arsenate?
The Backbone of Potassium Arsenate: Its Formula
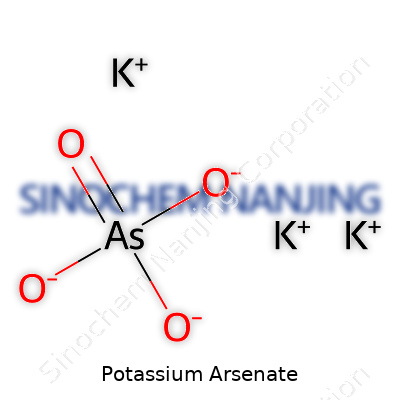
Potassium arsenate stands as a good example of how chemistry touches areas many people rarely think about. The formula for potassium arsenate is K₃AsO₄. That means every molecule contains three potassium atoms, one arsenic atom, and four oxygen atoms.
Digging into the Structure
Breaking it down, think of the K in the formula as potassium, As as arsenic, and O as oxygen. Potassium, on its own, shows up in bananas and supplements, but binds strongly when mixed with arsenic and oxygen. Arsenic is well-known for its dangerous side, but the way these atoms bond changes its properties. In potassium arsenate, arsenic sits at the center, forming a frame with oxygen, and potassium ions stay connected around the structure, balancing out the charge. This configuration is more than academic — it steers how the compound reacts in water and with other chemicals.
History and Uses: A Practical Look
Years ago, potassium arsenate showed up as a weed killer and an insecticide. Its ability to break down unwanted organisms looked like a solution for farmers for decades. I remember reading about older practices where orchard workers mixed this compound to protect crops from pests. That all changed once the health effects became clear. Exposure brings real risks. The World Health Organization and government agencies strictly regulate substances with arsenic for good reason — arsenic isn’t just toxic in high doses, it can build up in the body.
Right now, potassium arsenate rarely appears in mainstream agriculture because of those health dangers. Countries around the world tightened control, moving away from using it for food production. Scientific studies link chronic arsenic exposure to higher cancer risks, nerve problems, and disruptive effects on organs. These aren’t hypothetical worries; they’re based on decades of documented case reports and environmental monitoring.
Why Formula Knowledge Matters
Grasping the formula isn't just a trivia question for students cramming for exams. Knowing K₃AsO₄ helps people spot it on labels or in old chemistry kits, understand how it acts in the soil, and figure out what kinds of environmental cleanup might be needed. Chemical formulas aren't just numbers and letters; they’re blueprints for interaction. One potassium atom missing or an oxygen swapped out could change a compound’s toxicity, its behavior in water, and its long-term impact on the environment.
Managing Risks and Looking Ahead
Communities faced with contaminated land and old chemical stores have a tough job. The information about potassium arsenate’s formula plays a part in remediation plans. Cleaning up arsenic pollution draws on scientists’ understanding of its chemical form. Potassium ions, for instance, are harmless enough, but arsenate ions keep toxic power, especially in groundwater.
Safer, modern farming relies on alternatives to potassium arsenate. Research into biological pest control, crop rotation, and new synthetic compounds took off as regulations closed in on old-school arsenic solutions. When the dangers are known, people get motivated to invent something better. That’s what keeps chemistry relevant and useful in public health and environmental action.
Facts Backing Up the Caution
The Environmental Protection Agency classifies inorganic arsenic, including arsenate, as a Group A carcinogen. Drinking water rules limit arsenic because science shows even low amounts over years can escalate health problems. These facts stay important whether talking about a chemistry classroom or real-world contamination cases.
How should Potassium Arsenate be stored and handled?
The Nature of Potassium Arsenate
Potassium arsenate doesn’t draw crowds outside a lab, but anyone who works with chemicals knows this one deserves respect. This compound, used in research and industry, has a bad rep for a reason: it’s highly toxic, carcinogenic, and has the potential to cause serious harm with small exposures. In my early days working in a chemistry department, the first step before touching a container of potassium arsenate was always to check your own habits—rushing, improvising, and skipping steps invite disaster.
Why Labeling and Isolation Matter
Labels matter more than people sometimes admit. Once I watched a new technician pull down a poorly marked bottle, which could have ended in exposure if I hadn’t stopped her. Every bottle of potassium arsenate deserves a clear hazard warning. The label should scream “toxic” and should display the skull and crossbones sign. Storerooms aren’t casual places for this stuff; potassium arsenate belongs in a locked cabinet, separated from acids and reducing agents. I've seen labs dedicate a special shelf just for inorganic arsenic compounds, far from anything reactive or flammable. Nobody wants cross-contamination or accidental mixing—these events can turn an average workday into a trip to the emergency room or worse.
Protective Gear Isn’t Optional
You can always spot an experienced chemist by their respect for gloves, goggles, and lab coats. Thin latex gloves will not stand up to potassium arsenate. Work requires thicker, chemical-resistant gloves and a fitted respirator if powders are moving through the air. I learned the hard way to double-check for leaks or tears; a little hole can mean a lot of trouble. Safety showers and eye-wash stations need to be in the same room as the chemical—distance costs valuable seconds in an accident.
Smart Storage Strategies
Potassium arsenate does best in tight, non-reactive containers. Plastic with strong lids works, but glass remains the gold standard for most chemicals—in my lab, only glass bottles get the honor. The container should never mingle with food or drinks, and, in fact, shouldn’t leave the chemical storage room. Doors stay locked, not just to keep items from “walking off,” but to prevent any unsuspecting person from making contact.
Handling Spills And Disposal
Chemical spills aren’t the time to improvise with paper towels and water. Years ago, I saw a seasoned scientist rush to contain a minor spill with proper absorbent pads, full-face protection, and a spill kit. Quick, confident movements and no heroics kept the whole lab safe. Used material needs containment in sealed, labeled bags for hazardous waste pickup—no shortcuts, no pretending it’s just household trash. In my experience, keeping a detailed spill log helps pinpoint patterns and correct bad habits early.
Reducing the Risk: Training and Oversight
The best storage protocols only work if people stick to them. Routine training makes rules into habits—refreshers at every onboarding and before any big project roll-out. Some labs assign “chemical buddies” to spot-check each other. This peer oversight creates accountability and culture change, more effective than a wall poster ever could. Potassium arsenate reminds everyone that chemical safety isn’t optional—it’s an everyday discipline that protects lives.
What are the safety precautions for using Potassium Arsenate?
Understanding the Danger
Potassium arsenate sits on a long list of chemicals that don’t belong anywhere near uninformed hands. It’s toxic on multiple fronts—swallowing it, breathing in dust, or getting it on the skin can start a chain of health problems including cancer, nerve damage, and stomach pain that stays with you. As someone who has worked in labs handling dangerous substances, I’ve seen more harm come from casual attitudes than from the chemicals themselves.
Respecting Personal Protection
No job with potassium arsenate kicks off without suiting up. I always pick nitrile or neoprene gloves because thin latex rips far too easily. A face shield blocks splashes, and a lab coat goes over regular clothes. When moving powder or mixing solutions, a respirator with high-efficiency filters makes a difference, stopping toxic particles from slipping into lungs. Splash goggles often seem like overkill, until that first near-miss.
Relying on Preparation, Not Luck
Before touching the bottle, I double check the labels and the container’s integrity. Labeling can wear off. Damaged caps let out dust and vapor that hang in the air just waiting for an unsuspecting hand. Every serious chemical storage room uses clear, well-maintained warning signs. Air flows out through a fume hood, never back into the room. Setting up near the door, instead of a crowded area, means a speedy exit if needed.
Managing Spills Without Panic
Every lab I’ve worked in keeps spill kits close—never hidden on a top shelf, always within arm’s reach. The protocol stays simple: isolate the spill, use absorbent material, and collect everything for hazardous waste disposal. Scrubbing with water doesn’t cut it, since it can spread arsenic further and turn a small spill into a bigger hazard. Calling in the site’s safety officer, not handling it solo, keeps egos out of the equation.
Ventilation Isn’t Optional
Good ventilation means more than cracking open a window. I've felt the sting in my nose after working in poorly ventilated spaces, and the discomfort doesn't go away fast. Fume hoods keep the air moving and direct toxic vapors out, away from people. Everyone in the room breathes easier, literally.
Disposal: No Shortcuts
Tipping potassium arsenate down the drain is illegal for a reason. Municipal water treatment doesn’t break down arsenic nearly enough. Every drop goes into a sealed, labeled container for collection by hazardous waste services. Paper towels, gloves, and any disposables also join the waste bin, not the regular trash. Lab techs new to this work learn quickly how serious it gets if traces slip into common waste.
Training and Vigilance
Training never ends in a setting with such high-stakes risk. I’ve attended annual refresher sessions where old hands swap stories with newcomers, discussing mistakes and successes. That culture of openness makes people stop and think twice. Safety data sheets—those thick documents taped above every chemical bench—are more than paperwork; they’re reality checks.
Why These Precautions Matter
Those health stories in the toxicology books—arsenic poisoning, nervous system breakdown, lasting organ damage—aren’t fiction. Following safety steps isn’t about ticking boxes. It springs from real incidents, remembered losses, and respect for everyone working nearby. That’s how workplaces avoid yet another cautionary tale in a safety training slideshow.
| Names | |
| Preferred IUPAC name | potassium trioxidoarsenate |
| Other names |
Arsenic acid dipotassium salt Dipotassium arsenate Potassium hydrogenarsenate |
| Pronunciation | /pəˈtæsiəm ˈɑːsəneɪt/ |
| Identifiers | |
| CAS Number | 7784-41-0 |
| Beilstein Reference | 1309616 |
| ChEBI | CHEBI:131378 |
| ChEMBL | CHEMBL1201601 |
| ChemSpider | 17224 |
| DrugBank | DB13724 |
| ECHA InfoCard | 100.019.045 |
| EC Number | 231-903-0 |
| Gmelin Reference | 8209 |
| KEGG | C01882 |
| MeSH | D011007 |
| PubChem CID | 24449 |
| RTECS number | CA9625000 |
| UNII | 7Z3530888X |
| UN number | UN1559 |
| Properties | |
| Chemical formula | K3AsO4 |
| Molar mass | 222.00 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 2.39 g/cm³ |
| Solubility in water | Soluble |
| log P | -0.77 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 2.19 |
| Basicity (pKb) | 2.54 |
| Magnetic susceptibility (χ) | -51.0e-6 cm³/mol |
| Refractive index (nD) | 1.530 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 230.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1596 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3159.9 kJ/mol |
| Pharmacology | |
| ATC code | S01AX10 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; carcinogenic; causes damage to organs; environmental hazard |
| GHS labelling | GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H300 + H330: Fatal if swallowed or inhaled. H350: May cause cancer. H373: May cause damage to organs through prolonged or repeated exposure. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P264, P270, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P310, P330, P391, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-0-0-ACID |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 14 mg/kg |
| LD50 (median dose) | LD50: 14 mg/kg (oral, rat) |
| NIOSH | SN4550000 |
| PEL (Permissible) | PEL: 0.01 mg/m³ |
| REL (Recommended) | 0.2 mg/m³ |
| IDLH (Immediate danger) | 5 mg/m3 |
| Related compounds | |
| Related compounds |
Arsenic acid Arsenous acid Arsenic trioxide Arsenic pentoxide Potassium arsenite Sodium arsenate Sodium arsenite |

