Deep Dive into Polyhexamethylene Biguanide Hydrochloride: Historical Roots, Modern Value, and Critical Perspectives
Historical Development
The story of Polyhexamethylene Biguanide Hydrochloride starts in the mid-20th century, back when chemists and engineers searched for new disinfectants. This wasn't just about chasing innovative molecules—hospital-acquired infections worried people who saw the limits in old biocides. By the seventies and eighties, research labs across Europe began zeroing in on biguanide chemistry. Scientists chased safer alternatives to mercury-based and oxidizing disinfectants. Polyhexamethylene biguanide (PHMB) began standing out. Its chain structure and robust positive charge showed strong promise against bacteria, so development picked up. In the nineties, PHMB’s reputation as a water treatment biocide grew. Health standards also started leaning away from harsh oxidizers, spurring more demand for this polymeric disinfectant in wound care and surface cleaning. Today, pharmaceutical, personal care, textile, and water treatment factories see PHMB as much more than a relic. It is living history—a chemistry breakthrough whose value has only grown.
Product Overview
Polyhexamethylene Biguanide Hydrochloride doesn’t show up in everyday products by accident. On a basic level, this polymer hardly ever gets much media attention, yet it shows up in wound irrigation solutions, contact lens cleansers, disinfectant wipes, and swimming pool treatments. Sourcing matters. Purity standards differ between medical and industrial applications, so the manufacturing process adjusts to each use case. My experience in the medical supply field showed me how critical it is to match the product form to its task—whether a low-viscosity liquid for wound rinsing, a higher concentration for hard surface sanitation, or a fine powder for textile treatment. Consistency in its release properties and shelf life cements PHMB as a favorite in settings where reliable bio-barrier is essential. Knowing it won’t corrode most packaging or interact with other common materials only boosts its reputation for versatility.
Physical & Chemical Properties
On paper, PHMB hydrochloride looks straightforward: a white to off-white powder, easily soluble in water. Its long molecular chains pack a positive charge, letting it bind effectively with microbial membranes. That’s not just some abstract property. This positive charge, in practical terms, means PHMB disables a broad spectrum of bacteria and fungi at pretty low active concentrations. It holds together under a range of pH and temperature settings, giving it grit where many traditional disinfectants fail. People in the field swear by its calm in the face of organic soiling, especially in hospital environments buzzing with human activity. PHMB neither foams excessively nor leaves stubborn residues, both real-world strengths that keep hospital staff, janitors, and pool managers loyal to it.
Technical Specifications & Labeling
Any time you encounter PHMB commercially, you’ll spot certain tell-tale technical specs: active content (often 20% in aqueous form), pH range (typically 5 to 7 for most medical preparations), and shelf life (closest to two years under dry, shaded storage). Regulations slog through labels, listing recommended dilutions, expiry dates, and permissible uses. Labeling requirements stem from strict regional guidelines—the United States FDA, the European Medicines Agency, and China’s National Medical Products Administration all take differing approaches. But everywhere, they push for clarity. Product data sheets almost always include detail on purity (usually above 98%), trace contaminant levels, and manufacturing date. In my earlier days working on product launches, I kept running into hurdles when these certifications lagged behind production batches—clear evidence that paper trails and technical specs matter as much as the chemistry itself.
Preparation Method
Production starts with hexamethylenediamine reacting with biguanide hydrochloride under controlled, heated conditions. Additives adjust polymer chain length, which in turn affects the final product’s solubility and antimicrobial strength. Process engineers monitor timing, stir rates, and temperature using calibrated sensors. Once polymerization wraps up, the product runs through washing steps to strip unreacted small molecules. For anything destined for pharmaceutical use, an additional purification stage cuts trace impurities back down. Research teams constantly tinker with chain length, exploring how subtle tweaks affect activity against resistant strains. Technical teams know that even a slight dip in quality control means a batch fails regulatory hurdles, so batch records and chromatographic analysis run as constant fixtures in these facilities.
Chemical Reactions & Modifications
Looking at PHMB from a chemist’s perspective, this polymer stands up to most routine chemical stresses. Alkalis and moderate acids don't break down its structure, giving users leeway in storage and formulation. Direct sunlight or strong oxidizers degrade it eventually, but in realistic use scenarios—like pool maintenance or wound treatment—these factors rarely intrude. Modification techniques such as chain-length tailoring or introduction of co-polymers help expand its effectiveness against tough-to-kill spores or biofilm-forming bacteria. Lab teams dig into these modifications whenever new antimicrobial challenges crop up. And they never treat stability as a given—each molecular tweak gets subjected to months of stress testing, both in solution and as part of finished products. This adaptive approach helps stave off the slow march of resistance seen with more static disinfectant molecules.
Synonyms & Product Names
Plenty of people in hospital supply, textile, water treatment, and consumer goods circles recognize PHMB by its brand names as much as the scientific lingo. “Cosmocil CQ,” “Vantocil IB,” or simply “polyaminopropyl biguanide” mean the same active ingredient. When field technicians swap stories, these labels mix with technical synonyms depending on which regulatory territory the product crosses. In scientific papers and product handbooks, chemists don’t shy away from using its IUPAC name or abbreviations tailored for specific studies. Some markets blend PHMB with auxiliary ingredients then slap new proprietary names on the product, but technical sheets always trace back to that chain of biguanide repeat units first described decades ago.
Safety & Operational Standards
No industrial chemical gets widespread use without its fair share of regulatory oversight, and PHMB serves as a textbook case. Safety standards vary across regions, yet worker handling protocols converge on gloves, splash goggles, and well-ventilated mixing stations in bulk preparations. Material Safety Data Sheets flag the main risks: mild skin and eye irritation for workers handling concentrates, and rare allergic responses among patients exposed to undiluted solutions. The margin of safety in wound care and contact lens use brings some reassurance, but manufacturers and end users monitor for changing regulatory winds. For environmental safety, municipal water authorities and agriculture agencies keep tabs on disposal. Waste streams containing PHMB undergo treatment, as persistent molecules worry some researchers eyeing long-term ecological impacts. Each year, rulebooks inch forward based on new data—another reminder that real-world safety routines stay in step with ongoing research.
Application Area
Ask any nurse about wound rinsing options, and PHMB comes up almost immediately—a testament to its reliability for infection prevention. It’s a mainstay in moist wound dressings, surgical rinses, and non-stinging skin cleansers. Eye care professionals rely on PHMB-based solutions, especially for disinfecting reusable lenses. Water treatment plants add it to recirculating cooling towers and swimming pools where it cuts biofilm without damaging infrastructure. Textile processing plants use PHMB to treat athletic fabrics, medical linens, and air filters, banking on its long-term antibacterial persistence. I’ve seen food production spaces turn to PHMB where routine bleach use simply won’t do—sensitive surfaces, odor concerns, and long contact times all call for something less aggressive but still potent. This reach hasn’t come without pushback. Some agricultural users voice concern about accumulation, setting up a tension between short-term benefit and long-run environmental risks. Each field applies risk-benefit logic uniquely, relying on decades of operational data and evolving need.
Research & Development
Across universities and corporate labs, PHMB’s story keeps building. Microbiologists push to map out new resistance patterns, while process engineers chase more sustainable production methods. Many labs edge into “smart” delivery systems—embedding PHMB in wound gels, hydrogels, or nano-fibrous scaffolds for more targeted use. Regulatory scientists sift through toxicity studies, scrutinizing every animal model and in vitro result, aiming for a more precise read on long-term effects in both health and ecology. Academic collaborations often highlight alternative biguanide derivatives, picking apart the strengths and pitfalls compared with old standbys like chlorhexidine. High-impact journals keep publishing head-to-head trials, especially as emerging infectious diseases drive up demand for non-antibiotic antimicrobials. Development doesn’t stall at the bench—pilot plants and production lines test tweaks in real time, guided by fast feedback loops from both regulators and commercial partners.
Toxicity Research
From the start, toxicologists eyed PHMB for risks and rewards. Results point to its relatively low acute toxicity—much less than chlorine or phenol-based disinfectants—but that summary leaves out some of the nuance. Chronic exposure studies, particularly in rodents, raised flags over possible liver effects when high doses linger. In wound care and contact lens cleansing, incidents of acute toxicity stay rare, showing why medical use continues to expand. Major toxicology reviews published in the past decade show cellular uptake without genotoxicity, but research teams watch carefully for allergic sensitization and subtle immune effects, especially in sensitive patients. The conversation changes when PHMB hits water treatment or broad-acre uses: here, aquatic toxicity trends upward. Some regulatory agencies limit concentrations in order to cap possible risk to fish or amphibians, bringing environmental toxicology into closer conversation with human safety. Long-term cohort studies among exposed workers track everything from respiratory effects to sensitization rates. These data—never frozen in time—fuel rolling reviews and regulatory tweaks.
Future Prospects
The years ahead look set for more growth, matched with new accountability. Pharmaceuticals and wound care products chart new uses, like spray-on barrier films and high-efficiency hydrogels. Textile researchers work to graft PHMB molecules onto synthetic fibers, aiming at permanent antibacterial socks, surgical scrubs, or HVAC filters. In pools and industrial water, stricter guidelines mean process engineers experiment with dosing, timing, and fully closed-loop systems to cut environmental loads. What stands out is the growing scrutiny: every innovation gets measured against tougher safety, waste, and environmental standards. As antibiotic resistance surges around the globe, non-antibiotic antimicrobials like this one draw closer inspection from health agencies, consumer advocates, and the general public. In my own experience speaking with regulatory scientists, it’s clear—checks and balances no longer count as a box-ticking exercise. They underpin business models and reputations more than ever. The coming wave of research will keep pushing for more selective, eco-friendly polymers. With every new breakthrough, the story of PHMB picks up another chapter—layered, gritty, and never dull for those living at the chemical and human frontier.
What is Polyhexamethylene Biguanide Hydrochloride used for?
The Hidden Strength in Disinfectants
Polyhexamethylene biguanide hydrochloride sounds like something you’d see on a chemistry exam, not in everyday life. Most people won’t spot this name on their household cleaners, because it goes by the shorthand PHMB. You’ll find it doing serious work in places that demand clean hands, clean surfaces, and clean wounds. My first encounter with PHMB came during a family emergency, when someone needed wound care supplies and I checked the label, looking for ingredients known to help prevent infections. There it was, sitting between more familiar names.
Guarding Against Germs in Healthcare and Home
Hospitals trust PHMB to protect their patients. It shows up in wound dressings, skin disinfectants, and rinses, helping stop bacteria right at the source. Surgeons and nurses understand the risks of letting even a small cut go untreated. With superbugs on the rise and antibiotic resistance spreading, PHMB gives healthcare pros another option to keep infections at bay. One study I read from the American Journal of Infection Control explains how PHMB’s approach blocks microbes from multiplying, and the evidence stacks up for burns and chronic wounds.
Some folks use PHMB-based sprays in their homes, too, especially if caring for kids or pets. It’s not bleach. It doesn’t eat away at fabric or leave a harsh smell. Instead, it offers a gentle way to keep things sanitary, earning its spot in everything from baby wipes to contact lens cleaning solutions. Regulatory bodies like the FDA and EPA set tough rules for products that go on your skin or in your eyes, and PHMB gets approval because it shows low toxicity with proper use.
Beyond Medical Use: Pools, Textiles, and More
PHMB’s trail leads beyond clinics. Most pool owners I know worry about chlorine irritating their skin. Times change, and PHMB jumped in as a chlorine-free option. Pool supply shops now carry PHMB as a water sanitizer. No heavy chemical scent, no faded swimsuits, but the same protection against algae and bacteria. Some textile and leather producers include PHMB in their process because it pushes back on unwanted mold and stains. For a fabric that needs to last, this makes a big difference.
Facing Risks and Looking for Balance
People worry about chemicals in daily life, and there’s conversation swirling around PHMB too. Some European regulators flagged concerns over long-term exposure or misuse, and studies look hard at any signs of toxicity with repeated contact. On the other hand, ignoring infection control would toss us back to a time when a simple scrape could threaten a life. Safety comes down to following directions and not assuming more is better.
We need transparent research, easy-to-read safety updates, and clear product labels. For those with sensitive skin or chronic wounds, guidance from a healthcare provider isn't optional – it’s a necessity. PHMB shines brightest when professionals help people use it safely and wisely. For as long as we need powerful infection fighters that don’t wreck the things we touch, it has a place in the toolkit.
Is Polyhexamethylene Biguanide Hydrochloride safe for skin contact?
Understanding Polyhexamethylene Biguanide Hydrochloride
Polyhexamethylene biguanide hydrochloride — often shortened to PHMB — shows up on ingredient labels for wound cleansers, contact lens solutions, and even some cosmetics. The name alone is a mouthful, which might raise eyebrows about safety. Safety with chemicals like PHMB goes beyond complicated names. It comes down to what is known from research, what dermatologists and regulators say, and what happens when real people use it daily.
How PHMB Works
PHMB is a disinfectant — it disrupts bacterial membranes, which slows the growth of unwanted microbes. Hospitals have turned to it in the fight against infections, and it pops up in cleansers meant to deal with open wounds. No disinfectant works without some potential for irritation, especially for those with extra-sensitive skin. Yet, compared to heavy-duty alternatives like alcohol or bleach, PHMB tends to be gentler in typical concentrations.
What the Research Shows
Medical research has looked at the usual doses used on human skin. Peer-reviewed studies in journals like “Contact Dermatitis” and “Journal of Hospital Infection” report that PHMB, at levels up to 0.1%, rarely causes skin reactions. One 2020 review, for example, found most people handled PHMB with little trouble, especially when used on wounds under medical supervision. Higher concentrations raise questions. The main risk points to possible irritation or an allergic response, especially for people who already have skin conditions. If redness, burning, or itching crops up, it makes sense to rinse the area and ask a healthcare provider.
What the Experts Say
Younger or compromised skin may react differently. Several European countries reviewed PHMB in the wake of worries about longer-term toxicity. The European Commission flagged that using PHMB at high levels and for long stretches might pose health risks, so they tightened controls on how much can go in products. The US Food and Drug Administration and Health Canada also limit PHMB’s use in certain personal care items.
For everyday products, keeping concentrations low has become the industry’s line. Manufacturers run their own safety testing, backed up by ophthalmologists and dermatologists. PHMB cleansers and sprays on store shelves tend to follow these stricter guidelines.
Practical Experience and Safer Use
In clinics, I have seen PHMB wound cleansers used on chronic ulcers. A nurse might swab a leg wound and watch for reactions over days or weeks. Usually, PHMB helpfully reduces infection risk without slowing healing. Problems seem rare, especially if the wound is rinsed and not left soaked. The same goes for PHMB wipes in gyms — most people use them without realizing what’s in them, and skin complaints seem less frequent than with harsh alcohol wipes.
Addressing Real-World Concerns
The internet sometimes raises alarms about PHMB being linked to cancer. Toxicological agencies looked for solid proof in animal studies, but doses that triggered concern were far above what a person would find in a wound gel or lens solution. Still, opinion shifts fast; some groups want even stricter limits.
Best advice for anyone allergic to preservatives: patch test a small spot before slathering it on. Store products away from heat and sunlight, since chemical changes can alter safety.
For those dealing with open wounds or sensitive eyes, listen to trusted professionals. If an ingredient feels uncomfortable, swap it out or ask about safer alternatives.
What are the side effects of using Polyhexamethylene Biguanide Hydrochloride?
A Closer Look at Side Effects
Polyhexamethylene biguanide hydrochloride usually turns up in disinfectants, wound care, and contact lens solutions. The promise is cleaner surfaces, fewer infections, and safer contact lens wear. It's a big win for controlling bacteria and keeping problem germs from spreading. But once you start putting something on your skin, in your eyes, or anywhere close to your body, you want to know what could go wrong.
Skin Reactions
I’ve seen folks with sensitive skin break out in redness and itching after using products with this chemical. No two people have identical skin, and allergies develop even in adulthood. Dryness shows up sometimes, especially after using strong solutions for wound cleaning. That stinging feeling can discourage patients from using it as directed, and skipping care risks infections. The European Centre for Disease Prevention and Control points out rare cases of more severe irritation, though most reactions stay mild and clear up with washing and stopping the product.
Eye Irritation
Contact lens wearers have a special relationship with this material since it cleans lenses and acts as a preservative in multipurpose solutions. Some people notice stinging or burning after rinsing their lenses. I’ve heard about blurred vision or a gritty feeling inside the lid, especially if solution remains on the lens. In rare cases, allergic conjunctivitis develops, making eyes red, swollen, and watery. The FDA collects these reports, and most improve after switching to a product with a different preservative.
Breathing and Inhalation Risks
Mist or vapors from wound irrigating solutions can irritate airways in confined spaces. Coughing, throat discomfort, and mild breathlessness sometimes follow. Hospitals and clinics take precautions to ventilate rooms during debridement procedures, keeping both staff and patients comfortable. Several studies in occupational health warn that repeated high-level exposure deserves careful attention, especially among those with asthma or chronic lung conditions.
Long-Term and Systemic Effects
So far, after years of use in hospitals and clinics, there’s no solid evidence of polyhexamethylene biguanide hydrochloride building up in the body or harming organs. Laboratory testing shows it doesn’t get absorbed through healthy skin in measurable amounts. Swallowing large amounts leads to stomach upset and, in serious cases, vomiting or diarrhea. For that reason, products come with safety caps and warnings to keep out of reach of young children.
Weighing the Benefits Against the Risks
Every time someone cleans a deep wound or disinfects contact lenses, the goal stays the same—kill germs before they hurt the patient. The risks stay low versus the dangers of infection. It still makes sense to check the label and stop using any product that causes reactions. Reporting problems to regulators helps companies and scientists keep watch for new concerns.
How to Stay on the Safe Side
Anyone with allergies or skin conditions gets a safety edge by testing on a small patch of skin first. Using eye products only as instructed, letting lenses air out, and rinsing thoroughly go a long way. Health professionals train on how to handle chemical splashes and maintain good ventilation. Parents lock away all disinfectants at home. Transparency from companies—about strength, purity, and exact formula—empowers everyone to use these products wisely.
How should Polyhexamethylene Biguanide Hydrochloride be stored?
What Happens In The Storage Room
Polyhexamethylene Biguanide Hydrochloride sounds like a mouthful, but for folks in healthcare and water treatment, it's a daily reality. It keeps swimming pools safe, freshens up contact lenses, and steps up in wound care. But even the toughest chemicals fall apart or cause harm if left sitting in the wrong spot. No one wants degraded disinfectant or, worse, a room full of accidental fumes.
Temperature: Not Too Hot, Not Too Cold
Experience in clinics and labs has shown me that chemicals don't forgive carelessness. This one stays potent when kept in a cool, dry place out of direct sunlight. A storeroom hitting above 30°C turns the product sluggish and unreliable. Start stacking bottles near radiators or sunny window ledges, and soon you notice changes in appearance and punch. Just last year, a friend running a pool shop mentioned losing a batch because it baked on the top shelf all summer. Routine checks saved the next supply; thermometers aren’t just for fridges, it turns out.
Moisture Ruins The Game
Dampness spells trouble. Moisture sneaks into loose caps or torn packaging and messes with the chemical makeup. I watched an assistant mop up a shelf after a leak—crystal clumps, an acrid smell, barrels rusting from inside out. Staff spent hours sorting contaminated stock from the usable, and sickening waste bills followed. Sealed lids and solid containers give the best shot at keeping the product intact. Old habits like using cracked jars or makeshift covers backfire in the long run. A little attention to tight seals and clean containers saves big headaches.
Keep Away From Acids And Iron
Mixing certain chemical groups triggers reactions that no one wants indoors. Polyhexamethylene Biguanide Hydrochloride clashes with acids, oxidizers, and anything iron-based. You don't want to store it next to battery fluid or rusty equipment. My supervisor set up plain labels and color-coded shelves for a reason—accidents from mix-ups can land people in the ER. A minor spill years ago corroded a metal drum before lunchtime and nearly shut the shop down with the fumes.
Lock And Label For Safety
Labels spell out hazards and prevent mistakes. I’ve seen new team members reach for the wrong drum before a proper label pointed out the danger. Locking storage—especially in schools and clinics—keeps curious hands away. Staff training helps, too; once a quarter, everyone runs through location checks and label updates. This isn’t just bureaucracy; it actually cuts emergency calls and inventory losses.
Smart Inventory Routines
FIFO—first stock in, first out—saves product and money. Outdated batches lose strength and may start to break down, creating new risks. In one busy city lab, a forgotten stash went brown and sticky, costing hours of cleanup. Barcode tracking and regular audits win over handwritten logbooks, especially as volumes grow. A digital trail keeps surprises to a minimum.
Conclusion
Those who handle Polyhexamethylene Biguanide Hydrochloride daily know that paying attention pays back. Decent temperature, dry air, good seals, clearly separated chemicals, and strong routines build trust and keep everyone safe. These habits fit any workplace, not just high-tech labs. Good storage means reliable results, and nobody has time to waste on preventable disasters.
Can Polyhexamethylene Biguanide Hydrochloride be used as a disinfectant?
Understanding the Role of PHMB in Disinfection
Polyhexamethylene biguanide hydrochloride doesn’t often come up in casual conversation, but this chemical deserves some attention. In healthcare and water management, cleaner surfaces mean fewer infections, and that truth never changes. Many disinfectants out there leave users worried about fumes, residue, or harsh action. PHMB pushes itself forward as a solution because it blends antimicrobial power with a lower risk of toxic effects, as shown by multiple studies published in environmental and medical journals.
Bacteria live everywhere—on hospital trays, pool changing rooms, or inside bandages. PHMB attacks the cell membranes of bacteria, causing the contents to leak out and rendering the microbe harmless. This action skips the need for high concentrations, making it less likely to cause skin irritation or corrosion. From my time training as a hospital volunteer, it always stood out how staff gravitated towards products less likely to cause chemical burns or lasting odors. This compound slips into that category after rounds of safety testing.
Why Hospitals Keep Reaching for Alternatives
Healthcare-associated infections create real concern. According to the World Health Organization, billions of dollars slip through the cracks every year from diseases picked up inside hospitals. Replacing traditional chlorine-based disinfectants always sparks debate. Chlorine can trigger asthma, damage fabrics, and leave behind a “chemical tang” that seems to stay longer than anyone wants. PHMB offers antimicrobial strength, especially against bacteria and some fungi, without reacting aggressively with other cleaners or creating noxious gases.
Nursing homes, school gyms, municipal water facilities, and dentist offices have already started to look at PHMB as a solid option. The chemical successfully reduces microbial contamination on surfaces, within wound dressings, and in swimming pools. The U.S. Food and Drug Administration allows its use in wound care because research proves it doesn’t slow healing as some older disinfectants do. Europe and Asia also adopt it in public swimming areas where eye irritation and skin allergic responses matter.
Tackling the Limits and Handling PHMB Risks
The benefits of PHMB keep drawing attention, but ignoring safety would be a mistake. No one should assume it’s perfect. Some countries want tighter control because lab research suggests potential links to genetic changes in animal cells, although studies don’t always reach the same conclusions. The European Chemicals Agency flagged it for further evaluation, asking for more research before wider use in cosmetics and personal care.
It comes down to responsible use, training for staff, and tight regulation. I’ve seen caretakers toss diluted solutions into drains without thinking about effects on aquatic ecosystems. Authorities should focus on careful labeling, proper disposal practices, and stricter monitoring. These small changes lower the odds of environmental or health problems over time. Teaching proper mixing, application, and storage makes a difference. Transparency keeps users confident and shields vulnerable patients from unnecessary risks.
Where to Go from Here
Every disinfectant brings trade-offs. PHMB stands tall for its bacterial action, surface friendliness, and reduced irritation risk compared to harsher chemicals. Scientists and regulators need to keep digging into long-term safety, especially for large-scale uses and potential environmental build-up. Uptake only works where trust and facts go hand in hand.
At its best, PHMB helps close the gap between strong infection control and gentle handling for both people and the environment, as long as everyone involved stays informed and cautious.
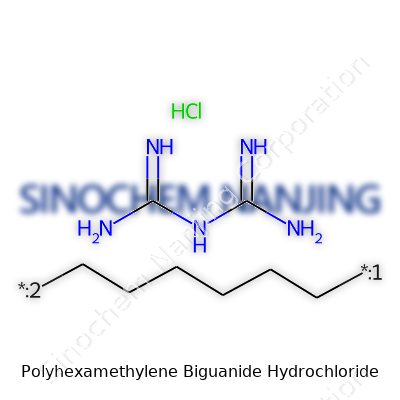
| Names | |
| Preferred IUPAC name | poly(hexane-1,6-diylbiguanide) hydrochloride |
| Other names |
PHMB Polyhexanide Polyaminopropyl Biguanide Poly (hexamethylene biguanide) hydrochloride |
| Pronunciation | /ˌpɒl.i.hɛk.səˌmɛθ.ɪˈliːn baɪˈɡwɑː.nɪd haɪˌdrɒk.ləˈraɪd/ |
| Identifiers | |
| CAS Number | 57028-96-3 |
| Beilstein Reference | 3087861 |
| ChEBI | CHEBI:88284 |
| ChEMBL | CHEMBL1200978 |
| ChemSpider | 20488464 |
| DrugBank | DB06828 |
| ECHA InfoCard | 100.115.691 |
| EC Number | 27083-27-8 |
| Gmelin Reference | 80938 |
| KEGG | C22183 |
| MeSH | D000077245 |
| PubChem CID | 71310047 |
| RTECS number | MU7176000 |
| UNII | W8U1WZ99SF |
| UN number | UN2821 |
| CompTox Dashboard (EPA) | DTXSID7020182 |
| Properties | |
| Chemical formula | (C8H17N5)n·xHCl |
| Molar mass | 340.93 g/mol |
| Appearance | White or almost white crystalline powder |
| Odor | Odorless |
| Density | 1.14 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -3.8 |
| Vapor pressure | <0.01 mmHg (20°C) |
| Acidity (pKa) | 4.5 |
| Basicity (pKb) | 4.2 |
| Magnetic susceptibility (χ) | -9.63×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.396 |
| Viscosity | Viscous liquid |
| Dipole moment | 4.07 D |
| Pharmacology | |
| ATC code | D08AJ10 |
| Hazards | |
| Main hazards | Irritating to eyes, respiratory system and skin. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | Flame, Exclamation Mark, Health Hazard |
| Signal word | Warning |
| Hazard statements | H302, H319, H335 |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313 |
| Lethal dose or concentration | LD50 oral rat > 2000 mg/kg |
| LD50 (median dose) | 35 mg/kg (rat, oral) |
| NIOSH | GBY6344000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Polyhexamethylene Biguanide Chlorhexidine Polyaminopropyl Biguanide Hexamidine Polyhexamethylene Guanidine |

