Terephthaloyl Chloride: Past, Present, and What Lies Ahead
The Road from Discovery to Utility
Looking at the story of terephthaloyl chloride shows how a single molecule can push entire industries forward. More than sixty years ago, chemists saw big promise in aromatic acid chlorides but couldn’t predict how far these compounds would reach. Early work, especially in Europe and the United States, centered around the promise of manipulating dicarboxylic acid chlorides. In the postwar era, as demand for stronger, lighter, and more versatile materials grew, terephthaloyl chloride emerged as a building block. It soon became unmistakable in the laboratories of those pioneering new polymers. This growth did not happen in isolation. Chemists built on work with benzene chemistry from the late 1800s, coming to realize the unique chemistry terephthaloyl chloride brings to the table. Instead of fading into patent archives, it marched right into chemical engineering handbooks and chemical plant operation manuals worldwide.
Getting to Know Terephthaloyl Chloride
No need to pull out a periodic table for this one: terephthaloyl chloride presents itself as a white to off-white crystalline solid. The faint, choking odor tells you quickly that this isn’t your average kitchen ingredient. Touching or breathing it isn’t something any reasonable person would do. The heart of this compound is a benzene ring with two acid chloride groups attached in the para position. It doesn’t just sit on the laboratory shelf; it reacts with diamines to create aromatic polyamides. Stories of high-performance fibers—think bulletproof vests and heat-resistant materials—begin here. Terephthaloyl chloride, by nature, holds a corrosive temperament. It stings on contact and fumes in humid air. That means clear technical labeling stays more than a matter of compliance. Labels need to shout clearly about hazards, risks, and proper handling, not bury the details in fine print. With something so reactive, accurate, up-to-date safety data makes all the difference.
Crafting the Compound: How It’s Made
Commercial synthesis of terephthaloyl chloride usually starts with terephthalic acid, itself a product that traces back to the petrochemicals under our feet. Chlorination using thionyl chloride pops up as a go-to method. For anyone spending time in an industrial facility, that often means bringing together solid terephthalic acid and liquid thionyl chloride, plus a dash of timing and temperature control. What comes out is a substance with a low melting point and a nasty habit of producing hydrogen chloride fumes. Workflows that keep out moisture and avoid accidental releases—ventilation, closed reactors, proper scrubbing—don’t just help the bottom line; they keep lungs and equipment in working order. This step, for all its apparent straightforwardness, has real environmental and safety impact. If one invests in cleaner technology or recycles chlorinating agents, the payoff extends past the immediate batch run.
Chemistry in Action: How It Reacts
What sets terephthaloyl chloride apart in the world of aromatic chlorides isn’t just its reactivity, but what that reactivity builds. The acid chloride groups command respect, attacking amines with a speed and intensity that make industrial-scale polymerizations viable. In those big spinning tanks, polycondensation with para-phenylenediamine creates poly(p-phenylene terephthalamide)—better known as Kevlar. Without this exact coupling, the lightweight, cut-resistant, and heat-tolerant properties of many fibers would be out of reach. Chemists and process engineers examine every variant reaction: swapping amines or modifying feedstocks shifts the performance of the resulting fibers. These modifications go hand-in-hand with research on making synthesis smoother and cleaner. Sometimes, switching to alternative solvents or milder acidic chlorides means less waste at the end of the process. Small tweaks make a huge difference—one batch at a time—for everyone from the factory worker to the emergency responder.
The Many Faces of One Chemical
Anyone leafing through technical documents or research papers might spot the name “terephthaloyl dichloride” almost as often as its more formal form. In Europe, trade names have cropped up, and researchers in Asia sometimes prefer the shorthand “TPC” as a label. Knowing the full list of synonyms does more than support regulatory compliance; it helps ensure no one loses track of a shipment or misreads a critical protocol. In my years translating between different regulatory schemes, I’ve seen missed connections stem from inconsistent naming, spurring avoidable accidents or inefficiencies. This mishmash of terminology still shows why clear communication remains as important as careful synthesis.
Staying Safe: Standards in Real Life
Stories about chemical incidents often make headlines, but out in the supply chain, most operations follow a strict choreography. Regulatory standards for storage, handling, and transportation of terephthaloyl chloride come from a shared need for worker safety and public health. Corrosivity and pulmonary risks shape the rules: full PPE, chemical-resistant gloves, splash-proof goggles, and quick-drench emergency showers stand as non-negotiables. Closed handling systems and adequate ventilation shelter both people and the environment. I’ve watched teams rehearse spill response drills, not just to tick boxes, but because everyone in the room knows the feeling of exposure. Training, labeling, and clear Standard Operating Procedures don’t just serve the law—they give operators the faith that they’ll return home safe. Regulatory oversight has closed some of the old gaps, but vigilance still trumps complacency every time.
Far-Reaching Uses—From Fibers to Films
Terephthaloyl chloride lives at the intersection of function and innovation. Step into any facility turning out technical fibers, and you’ll find its fingerprint in aramid production for everything from military armor and firefighting suits to drive belts and reinforced hoses. It holds the line when heat or mechanical stress would shred lighter molecules. The electronics sector leans on its role in specialty films and high-strength composites, building lighter machines without cutting corners on durability or safety. I remember a colleague pointing out that sports equipment—tennis racquets, racing sails, even high-performance bikes—all carry the legacy of this chemical. In environmental engineering, more specialty membranes and filters now draw on aramid chemistry. It’s hard to overstate the reach, and yet, plenty of untapped uses wait in the wings. Process improvements and better integration with recycling streams could widen that scope even further.
Pushing the Boundary: Research and Development
Labs around the world continue to chase new ways to make and use terephthaloyl chloride. Academic groups scan for new diamines, targeting those rare pairings that yield better mechanical properties or more sustainable processes. It’s not just a race to boost numbers on tensile strength charts; researchers track each step for energy use, emissions, and waste as progress now wears a green tint. Startups and major chemical firms alike are rethinking reaction conditions—switching away from the harshest chlorinating agents, trying sealed-loop systems, or finding ways to recover monomer waste. Each new technical publication brings its own angle, but the shared thread is real: improvement in performance and cutbacks in environmental cost. In the past decade, the number of peer-reviewed papers focusing on low-waste or solvent-free methods has risen steadily. This pace only adds to the pressure on existing producers to adapt quickly—or risk falling behind.
Weighing the Risks: Toxicity and Health
Many see the promise in advanced materials but might not consider what such reactivity means for safety. Terephthaloyl chloride doesn’t pull its punches: researchers have repeatedly flagged its irritant and corrosive properties, especially for the respiratory tract, skin, and eyes. Toxicity studies in animals and on human cell cultures point to the danger of acute exposure—throat, lung, and eye damage, chemical burns, and, with enough contact, serious health setbacks. Working in regulated labs, I have seen the impact that proper hood design, work practices, and emergency planning can make in preventing incidents. Ongoing research now digs deeper into chronic risks, examining whether low-level exposure might affect workers over the long term. Industry must keep health front and center, as new findings demand more than compliance—they call for innovation in protective gear, monitoring, and process containment.
The Road Ahead
Every innovation built on terephthaloyl chloride spurs debate over resource use, waste, and safety. The future circles around greener chemistry: better raw materials, circular supply loops, and processes that minimize hazardous byproducts. The question has moved from “What can it make?” to “How clean and safe is the making?” Next-generation aramids with bio-based starting points—think plant-derived acids coupled with safer chlorinating agents—are in reach if today’s research delivers. Recycling spent polymers back into monomers, closing the resource loop, marks the next big leap. Regulatory action, public opinion, and cost discipline will steer this shift, but the rewards matter for both the environment and business resilience. Terephthaloyl chloride’s heritage in high-performance applications gives the industry a strong foundation—now it’s up to a new generation of chemists, engineers, and policy-watchers to push it further without repeating old mistakes.
What is terephthaloyl chloride used for?
Why Terephthaloyl Chloride Matters in Everyday Products
Spotting a material like terephthaloyl chloride in a news story rarely stirs excitement, but many everyday conveniences owe their practicality to this compound. Terephthaloyl chloride, known in the chemical world by its acronym TPC, plays a big part in creating strong, heat-resistant, and durable materials. From bulletproof vests to light yet tough luggage, its presence feels almost behind the scenes, yet its impact stretches far and wide.
The Backbone of Kevlar and Modern Fabrics
All the talk about bulletproof vests brings Kevlar to mind. This material starts life as a liquid mixture involving terephthaloyl chloride. Kevlar finds purpose in body armor, military helmets, and protective gloves because it delivers lightweight strength. The strength comes from long, tightly packed polymer chains. Once I learned about the science behind these chains, it gave me new respect for the design of firefighter jackets and car tires, where split-second reaction times and reliability protect people.
Chemical plants use TPC with partners like p-phenylenediamine in processes called polycondensation. The result: polymer fibers that outshine steel in strength-for-weight. This means lighter vehicles, safer sports gear, and textiles that last longer under rough use.
Pharmaceuticals and Electronics Relate Too
It’s not just fabrics. Terephthaloyl chloride lends itself to the pharmaceutical world, where careful synthesis helps create new drugs. Electronics makers value specialty plastics built with TPC, since these materials resist harsh environments and insulate well. Circuit boards in smartphones use polyesters derived from this compound. Its versatility amazes me: one product working for safety, innovation, and medicine—all at once.
Environmental Costs and Forward Steps
Questions about environmental safety arise with strong chemicals like TPC. Manufacturing this stuff creates waste and byproducts, including hydrochloric acid. The challenge for companies: limit pollution and keep factory workers safe. Having spent time in factories, I’ve seen up close the maze of rules and ventilation systems that shield people from exposure. Yet, talk on the shop floor often turns to what more can go right: less toxic reagents, smarter chemistry, recycling programs for fabrics at the end of their life.
Regulators urge industry to rethink what goes into the water and air, pushing for improvements. Some companies now invest in “greener” routes, swapping traditional raw materials for those with smaller environmental footprints. There’s a demand for change, fueled by people wanting safer products and less waste.
Bridging Production and Innovation
It’s tempting to see terephthaloyl chloride as just another chemical, but its reach proves how chemistry shapes daily routines. Bulletproof materials, heat-resistant plastics, and modern drug development all come back to a few basic building blocks. Reining in the downsides—waste, pollution, worker risk—will take clever engineering and responsible practices.
Learning about materials like TPC brings a kind of curiosity I wish more people felt. It shows the balance between science, safety, and sustainability, all tangled together in the things we use every day.
What is the chemical formula of terephthaloyl chloride?
What Is Terephthaloyl Chloride’s Formula?
Terephthaloyl chloride may sound obscure outside laboratories, but its formula—C8H4Cl2O2—links to more aspects of daily life than most suspect. These eight carbon atoms, four hydrogens, two oxygens, and two chlorines come together in a perfect arrangement for manufacturing performance fibers and specialty plastics.
Not Just a Chemical, but an Industrial Workhorse
Few products show up as often in modern life as the results of terephthaloyl chloride’s unique chemistry. I’ve seen its true reach up close: my neighbor once worked for a polymer lab where they produced raw materials for protective gear. He explained how chemists rely on the dual chlorine atoms to react quickly and efficiently when spinning fibers like Kevlar. The entire process relies on the reliable reactivity of C8H4Cl2O2 with other chemicals, making it indispensable for industries seeking consistency and strength.
Numbers don’t lie here. Companies worldwide produce thousands of tons of this compound every year, largely to feed the demand for high-performance materials. In military and construction circles, products made with terephthaloyl chloride literally save lives through improved armor and stronger safety gear.
Safety, Sustainability, and Responsibility
Handling terephthaloyl chloride might look simple on a chemical equation, but in practice, its double dose of chlorine packs a punch. Exposure can cause severe irritation and more serious health problems without careful procedures. Over the years, industrial hygiene standards have grown more strict; now, ventilation, personal protective equipment, and training serve as the first lines of defense for workers.
Waste from large-scale processes demands respect, too. These halogenated chemicals resist easy breakdown, so firms have turned to closed-loop production systems and advanced filtration to capture every last molecule. The Environmental Protection Agency and similar organizations have published guidelines drawing on years of experience, nudging the industry toward lower emissions and recycling.
Early in my own teaching career, several chemistry students asked if there’s a greener option than using this chemical. No perfect fix has emerged, but research continues into bio-based alternatives and catalysts that work at lower temperatures, reducing energy use and risk. Investing in clean chemistry methods can minimize waste and help protect surrounding communities.
What Innovation Looks Like
Advances in materials science often trace back to small tweaks in molecules. The simplicity and reactivity in the formula C8H4Cl2O2 make it the quiet engine behind stronger fibers and lighter composites, shaping everything from bulletproof vests to lightweight vehicles. Researchers keep searching for smarter synthesis methods, aiming for cost savings without compromising quality.
Communities and businesses have a role in pushing for research and making production processes as safe as possible. With open discussion, transparent standards, and willingness to invest in cleaner methods, the legacy of terephthaloyl chloride doesn’t have to end with the molecule itself.
How should terephthaloyl chloride be stored and handled safely?
Understanding the Hazards
Terephthaloyl chloride shows up in many industrial settings, especially where high-performance plastics or fibers like Kevlar come from. This chemical reacts strongly with water and even humid air, giving off hydrogen chloride gas in the process. I’ve worked in chemistry laboratories where the smallest mistake can fill the air with sharp odors, making your eyes and nose sting. That’s a warning you never ignore. Hydrogen chloride gas can burn skin, eyes, and lungs—this turns a simple spill into a serious emergency.
Keeping It Contained
Experience makes one careful. Terephthaloyl chloride comes as white flakes and crystals, but don’t let the plain look fool you. It eats through regular cardboard and melts through weak plastic. You want chemical-resistant containers—glass lined or heavy-duty polyethylene work well. Make sure the lid fits tight, sealing out air and moisture. Don’t keep it in open bins, even at arm’s length. I once saw a batch stored in a weak container, and humidity warped the bag and released vapors. That scare pushes home the point: reliable containers and dry, cool storage keep the risks low.
Setting Up a Good Storage Area
Keep terephthaloyl chloride away from acids, alkalis, metal powders, and especially water sources. I always look for a separate, locked cabinet labeled with strong hazard warnings. There’s a reason regulators push for this—they’ve seen what happens when incompatible chemicals share a cramped, humid space. You don’t want a drip or leak escalating into a full-on release.
Ventilation plays a major role. Dedicated chemical storage rooms run constant exhaust fans to pull out fumes and prevent toxic build-up. Shelves need spill trays. Anything that can catch and contain a leak helps emergency response crews go right to the source.
Using Personal Protective Equipment
I don’t trust my health to shortcuts, and neither should anyone else. Full chemical splash goggles stay on from start to finish. Long sleeves and nitrile or butyl rubber gloves shield skin from accidental contact. Face shields add a layer of certainty. Respirators with acid gas cartridges get pulled out if there’s even a hint of vapor. I learned early on to double-check gear before opening containers, not after exposure starts.
Dealing with Spills and Waste
Something always goes wrong at some point. Small spills should get covered with dry sand or an inert absorbent—never use water, as terephthaloyl chloride reacts violently and expands the problem. Properly trained techs scoop up and seal spill residues in corrosion-proof drums. Full suits and positive-pressure respirators are a must for any large release.
Disposing of this chemical takes professional waste management or an approved incinerator. Pouring it down any drain could trigger toxic fumes, so dedicated waste drums and scheduled pickups keep things safe and legal.
Training and Culture Make the Difference
No storage room works without people who understand why the rules matter. Regular drills on spills, up-to-date safety datasheets, and making sure everyone feels comfortable pointing out unsafe practices—these pieces stop problems before they start. Good habits survive budget cuts and staff changes. Clear labeling and simple emergency checklists stand as everyday reminders that safety grows from what you do, not just what you say.
Every step with terephthaloyl chloride demands respect, focus, and the right resources. Sticking to proven safe practices means no one has to learn the hard way.
What are the hazards and safety precautions associated with terephthaloyl chloride?
Looking at Terephthaloyl Chloride Risks Up Close
Terephthaloyl chloride is no stranger to anyone working in materials science or industrial chemistry. Folks in the business of making high-performance plastics and fibers call it TPC for short. You’ll likely recognize it from its role in spinning out aramid fibers like Kevlar, which catch bullets but offer big headaches in the plant. TPC is definitely not the kind of substance you want splashing on your skin or wafting through the lab without some preparation.
Health Dangers: How Terephthaloyl Chloride Messes With the Body
Breathing in terephthaloyl chloride dust or fumes irritates the nose, throat, and lungs. You might notice your eyes stinging just from a bit hanging in the air. Spills can burn the skin or eyes. This comes from the way TPC reacts—even moisture in your eyes or on your skin causes it to break down and make hydrochloric acid. Getting a whiff too often or handling this stuff without gloves or goggles invites real trouble. People have ended up in the emergency room with chemical burns or breathing problems because of it.
I once visited a small plant in Texas where a seasoned worker got doused on the forearm due to hasty drum handling. He washed up fast, but his skin blistered with a nasty burn, and he had to take two weeks off. That story sticks: Terephthaloyl chloride may seem like just another chemical powder to the untrained, but its bite runs deep.
Fire and the Environment
TPC burns, and it burns dirty. Flames feed off it and let loose toxic gases—hydrogen chloride and phosgene are the worst. These both pack a punch on your airways and can cause much bigger issues if an accident spills enough. In storage, even leaks can let the fumes build up, eat at metal shelving, or drift around until they hit an unlucky nose.
Out in the open, improper disposal lets terephthaloyl chloride stir up lasting problems. It poisons water and sets off chemical chain reactions. Nobody wants a slow-moving environmental mess, so controls and good habits matter as much as having the chemical in the first place.
What Real Safety Looks Like
Certified gloves, face shields, chemical-resistant aprons—that’s standard kit for anyone touching terephthaloyl chloride. Workspace ventilation needs to pull fumes away fast and keep air fresh. Respirators aren’t just for looks, either. Part of my job once meant checking that every storage drum was grounded and sealed. Even a small spill needs quick response: neutralizing with sodium bicarbonate, careful clean-up, everything bagged and labeled before disposal.
The most reliable defense always comes down to training. Every worker, from the front line to supervisors, has to learn the ropes. Regular safety drills reinforce what to do if something leaks or catches. Labeling stays clear, storage never crowds hot pipes or wet places, and emergency showers never go ignored.
Thinking Ahead: Safer Work Culture
In my experience, it always pays to run a tight ship. Third-party audits dig up problems before they hurt people. Smart inventory systems keep amounts small and easy to trace. Engineering controls, like sealed transfer systems and automatic shut-off valves, stop mistakes from turning into disasters. The more you invest in good practices, the less you worry about accidents biting back later.
Terephthaloyl chloride never turns friendly, but sticking to strict routines and treating it with caution keeps the workplace safe and the process off the nightly news.
Is terephthaloyl chloride available in different purities or grades?
Purity Levels in Focus
Terephthaloyl chloride doesn't arrive in a single, one-size-fits-all grade. Instead, it comes in several purities, usually described as technical, pure, or high-purity grades. The biggest impact of these variations lands on the final product’s reliability and safety, especially in chemical plants where downstream reactions leave no room for mistakes. Different grades come with different price tags, and those costs drive tough decisions in labs and factories. In a bottling plant, a chemist might pay more for a cleaner grade simply to cut down on unwanted by-products later on.
Real Stakes: Industries That Rely on Quality
Polymer manufacturing puts terephthaloyl chloride front and center, especially where high-strength fibers are concerned. DuPont’s Kevlar production, for instance, calls for very high purity because trace metals or water can set off unwanted reactions. Contaminants in the starting material tend to cascade through the process, making the fiber weak or causing buildup that wrecks expensive equipment. On a smaller scale, pharmaceutical research often asks for the purest substance available, since any impurity could throw off years of drug development and boost the risk of side effects.
Technical grade usually contains more residual acids and stays at the lower end of the cost spectrum. Lab workers sometimes choose it for early testing or processes where a little impurity won’t cause a disaster. High-purity or ultra-pure materials, on the other hand, cost more but save money in the long run by preventing ruined batches and cleanup headaches. Over the years I’ve seen chemists argue over whether it’s worth the upfront investment. Those who’ve had to toss out expensive batches after a trace contaminant derailed a synthesis often push to buy the clean stuff, no matter the cost.
Challenges in Sourcing
Access to consistently high-quality terephthaloyl chloride isn’t a given in every country. Sourcing relies on established supplies from chemical giants, such as those in Europe, Japan, or North America. Smaller producers in developing countries sometimes struggle with batch-to-batch variability, and this unpredictability can put manufacturers in tough spots. Unpredictable purity leads to wasted resources, especially if quality assurances can’t be met. When I worked with a local startup, we spent weeks testing batches from different suppliers just to find one consistent enough for scale-up. The real challenge came not from the chemistry itself, but from finding a source we could trust, time after time.
Fixing the Problem: What Works
Some labs use in-house testing, like HPLC or NMR, to check purity before a shipment goes into production. That’s a good safeguard, but it slows down delivery and needs specialized staff. These checks can prevent disaster, though, and most large companies stick with them even when costs rise. Big-name suppliers invest in stricter controls and skip suppliers who can’t provide detailed certificates of analysis. The most effective long-term solution often comes down to building tight relationships with trustworthy suppliers, setting clear specs, and double-checking every batch. For smaller operations without deep pockets, that may mean joining buying groups or pooling resources to land better deals on reliably pure compounds.
Looking Ahead
The market keeps demanding better materials, and innovation depends on solid, repeatable chemistry. Better manufacturing processes, stronger global standards, and improved supply chain transparency give buyers more confidence. Tech keeps evolving to let analysts catch problems early. At every step, the true cost of pure terephthaloyl chloride turns out to be way less than the hidden costs of starting over when something goes wrong.
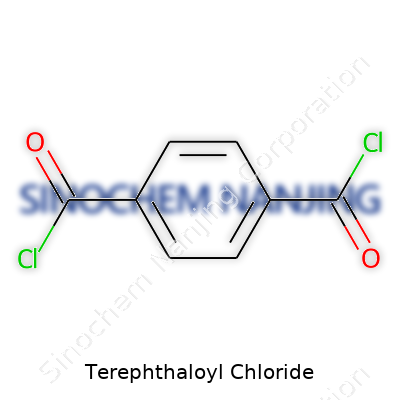
| Names | |
| Preferred IUPAC name | Benzene-1,4-dicarbonyl dichloride |
| Other names |
TPC Terephthalic acid dichloride 1,4-Benzenedicarbonyl chloride Terephthaloyl dichloride p-Phthaloyl chloride p-Benzene dicarbonyl chloride |
| Pronunciation | /ˌtɛrəfˈθæloɪl ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 100-20-9 |
| Beilstein Reference | 108969 |
| ChEBI | CHEBI:53288 |
| ChEMBL | CHEMBL135485 |
| ChemSpider | 6057 |
| DrugBank | DB08335 |
| ECHA InfoCard | 100.007.069 |
| EC Number | 204-825-9 |
| Gmelin Reference | Gmelin Reference: "138326 |
| KEGG | C06587 |
| MeSH | D014159 |
| PubChem CID | 8592 |
| RTECS number | KI4025000 |
| UNII | L1R7T4AMO8 |
| UN number | UN2475 |
| CompTox Dashboard (EPA) | DTXSID1039249 |
| Properties | |
| Chemical formula | C8H4Cl2O2 |
| Molar mass | 197.03 g/mol |
| Appearance | White to pale yellow crystalline powder |
| Odor | acidic |
| Density | 1.48 g/cm³ |
| Solubility in water | Reacts |
| log P | 2.52 |
| Vapor pressure | 0.03 mmHg (25°C) |
| Acidity (pKa) | -2.8 |
| Basicity (pKb) | -2.8 |
| Magnetic susceptibility (χ) | -54.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.630 |
| Viscosity | 0.97 cP (20°C) |
| Dipole moment | 2.85 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 333.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -704.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -859.2 kJ/mol |
| Pharmacology | |
| ATC code | No ATC code |
| Hazards | |
| Main hazards | Causes severe skin burns and eye damage. Causes serious eye damage. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H314, H317, H334, H335 |
| Precautionary statements | P260, P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P405, P501 |
| Autoignition temperature | > 243 °C (469 °F; 516 K) |
| Lethal dose or concentration | LD50 oral rat 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 679 mg/kg |
| NIOSH | GM2875000 |
| PEL (Permissible) | 5 mg/m3 |
| REL (Recommended) | 0.2 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Isophthaloyl chloride Phosgene Terephthalic acid |

