Phthalimide: Chemistry’s Unsung Backbone and Its Broad Reach
Tracing the Story: Phthalimide’s Historical Path
Ask anyone in a university organic chemistry lab what common chemical pops up in textbooks nearly every semester, and odds are, phthalimide gets a nod right along with urea and acetone. A little digging in the archives of 19th-century chemistry brings up Auguste Laurent and August Wilhelm von Hofmann, pioneers who set out to break benzene rings and build up early aromatic compounds. They managed to condense phthalic anhydride with ammonia, offering the world not only phthalimide, but the entire phthalimide-based chemistry toolbox. Many researchers over the years learned the ropes with this solid, white compound, finding it practically everywhere in synthetic labs, dye industries, and even as a bench test for new reaction techniques. If researchers hoped to tweak an amine or make a sneaky nucleophile, phthalimide usually got the call. In my own undergraduate days, the Gabriel synthesis served as the fabled rite of passage, with phthalimide acting as trusty launching pad turning alkyl halides into pure primary amines without useless byproducts—no mean feat.
Sizing Up Phthalimide: Product Overview, Form, and Specs
Phthalimide has this knack for showing up as an off-white powder, pretty dense and stable, drawing little attention to itself on a cluttered shelf. It clocks in with the chemical formula C8H5NO2, shaped by a benzene ring serving as anchor for two carbonyl groups and a nitrogen. This simple formula does a lot of heavy lifting. Drop it in organic solvents like acetone or hot water, and it breaks apart at about 238 degrees Celsius. Its melting point sometimes means you’ll spot it as crusty deposits after harsh reflux reactions. The firm crystalline structure gives it a solid shelf life, making it easy to store without much fuss. Watching a batch get filtered and weighed during a reaction feels almost too routine—until you realize just how many medicines and pigments rely on its presence.
Preparation in Practice: Making and Modifying Phthalimide
Most labs still turn to the tried-and-true method: heat phthalic anhydride with ammonia. This no-nonsense synthesis, first done in glass beakers well over a century ago, offers solid yields and no weird contaminants. Large-scale facilities tweak conditions—adding pressure or adjusting temperature steps—aiming for cleaner product or safer waste management. The chemical’s structure stands out for its ready participation in nucleophilic substitutions, especially during the Gabriel synthesis. Bring in a strong base like potassium hydroxide, and the imide opens up, letting chemists change the molecule in many ways, from N-alkylation to hydrolysis. Grab a good textbook and you’ll see it as a classic scaffold for turning plain aromatic rings into big-application chemicals.
Chemical Reactions and Synonyms in the Field
Phthalimide wears many hats with ease, acting as both a protecting group for amines and as a synthetic intermediate for all sorts of reactions. In the Gabriel synthesis, it quietly delivers pure primary amines where other routes flop. Add hydrazine, and it drops off like a spent delivery truck, leaving behind no unwanted baggage. Over the years, chemists coined several names—isoindoline-1,3-dione, o-phthalamide—though plain “phthalimide” covers most practical uses. Mix it with Suzuki couplings or metal catalysts, and you get new drugs, herbicides, or bright dyes that light up everything from textiles to food wrappers.
Safety and Lab Work: Handling With Respect
Every well-run chemistry department treats phthalimide with baseline respect. It rarely gives off fumes at room temperature, and it won’t burst into flames without extreme provocation. Still, it settles in the lungs and eyes if mishandled. Keeping dust down and skin untouched makes all the difference, and few sensible chemists work without gloves and goggles. Dry storage with a tight lid takes most of the drama out of using it. The compound does not jump off the page with acute toxicity, and routine lab ventilation deals with almost any risk. Researchers recognize that chronic overexposure can bring mild symptoms like irritation, but not enough to justify panic or special containment in a school lab or chemical plant.
From Dyes and Drugs to Agriculture: Real-World Applications
Phthalimide gets to wear several hats outside the lab. In color chemistry, it lays groundwork for vibrant pigments, serving as a stepping stone toward indigo and phthalocyanines, which dye our fabrics and plastics. The agricultural sector leans on phthalimide-based fungicides such as captan and folpet to fight crop-killing mildew and rot. Even the drug industry owes a debt—look no further than thalidomide’s infamous history and its more responsible modern offshoots fighting cancer and auto-immune diseases. To most people, these industries look worlds apart, but a batch of phthalimide at the start often underpins their biggest successes. The reliability and adaptability of this compound turn it into a behind-the-scenes MVP, letting new research move from lab notebook into working products on the global stage.
Research Trends, Toxicity Insights, and Where Things Go From Here
Dig deeper and the record grows richer. Centuries after phthalimide’s first appearance, research continues to look at its potential risks and hidden benefits. While animal studies do not reveal high acute toxicity, chronic exposure or mishandling can still bring problems, especially if derivatives get out into the ecosystem. Some modern work looks at green chemistry—hopes for cleaner synthesis, easier recycling, or less waste. Universities and private labs alike wonder how to trim solvent use, push reactions under milder conditions, or reclaim spent materials safely. Meanwhile, breakthrough modifications to phthalimide’s ring structure have built a host of tailor-made molecules, spinning the chemistry into uses as diverse as high-performance polymers to molecular probes in biological assays. As drug developers hunt for new scaffolds, and material scientists seek more durable paints or flame retardants, phthalimide’s adaptability keeps it in the running.
Looking Down the Road: Phthalimide’s Place in Modern Chemistry
Few chemicals maintain relevance generation after generation without some fundamental strength, and phthalimide remains a testament to thoughtful design and practical utility. Its sturdy aromatic core, twin carbonyls, and easy-to-handle nature anchor a wide family of molecules that power sectors as different as pharmaceuticals and plastics. As a teacher and hands-on chemist, I still see it as a bridge between classic organic transformations and today’s green chemistry goals. As the demand for more sustainable lab practices grows, I expect the next wave of research will stretch phthalimide’s abilities into biomaterials, precision medicine, and safer crop protection—echoing its historical strengths while meeting ever-new challenges on the world stage.
What is phthalimide used for?
What Drives Interest in Phthalimide?
Phthalimide doesn’t show up in the headlines much, but chances are that the work it does touches many parts of our lives. Coming from humble roots as a white, crystalline compound, phthalimide plays a big part in the production of other chemicals and products people use daily, whether they realize it or not.
How Phthalimide Powers the World of Chemistry
Think about the complex web of the chemical industry. Phthalimide steps in as a crucial player in making medicines, dyes, and even agricultural products. Its biggest claim to fame comes from its use in the Gabriel synthesis, where it helps chemists create primary amines—building blocks for pharmaceuticals and agrochemicals.
Why is that important? Many modern medicines rely on finely tuned chemical structures made possible by primary amines. If you’ve ever taken over-the-counter cold medicine, pain relievers or even used sunscreen, you’ve probably benefited indirectly from the chemistry of phthalimide. The compound streamlines the process, keeping production efficient and costs under control.
Role in Agriculture and Public Health
Out on the farm, phthalimide does work as a starting material for fungicides such as folpet and captan. Fields treated with these protectants see fewer fungal diseases and bigger, healthier harvests. These effects roll down the food chain and can help keep produce plentiful at the supermarket.
But there’s always a risk when dealing with chemicals. Concerns over toxicity and environmental impact of some phthalimide-based fungicides have led to closer scrutiny and tighter rules in many countries. Researchers have pushed for safer alternatives and better monitoring, which is a step in the right direction. Responsible handling and careful regulation keep risks in check without pulling the rug from under growers who depend on efficient crop protection.
A Helping Hand in Dyes and Plastics
Dyes touch nearly every part of our lives, from the clothes we wear to the plastics in our homes. Phthalimide serves as a raw material for making phthalocyanine pigments. These pigments come with the kind of rich, stable colors that don’t fade after a few washes or hours in the sun. Manufacturers rely on this strength to maintain quality in textiles, coatings, and plastics.
Safety and the Need for Smart Use
Any time a compound ends up in so many corners of industry, the spotlight turns to safety. Studies and regulations have set limits on exposure, especially for workers handling phthalimide directly. Masking, proper disposal, and ongoing monitoring all matter. The scientific community doesn’t sweep the risks under the rug; instead experts tackle them head-on, sharing findings and demanding accountability from manufacturers.
In my experience working with chemicals, open communication and access to up-to-date safety practices keep everyone safer. People deserve to know not just the benefits but also the possible downsides of the substances that shape their everyday world.
Looking Ahead
Phthalimide may never draw the crowds or spark debate like some hot-button issues, but its quiet utility keeps modern life running smoother. Researchers keep exploring greener pathways and safer substitutes where possible. An honest look at phthalimide not only highlights what’s possible in chemistry but also reminds us of the importance of balancing progress with precaution.
Is phthalimide hazardous or toxic?
A Closer Look at Phthalimide in Everyday Labs and Industry
Phthalimide. If you’ve stepped foot in an undergrad organic chemistry lab, you’ve seen it. In the industrial world, it’s there in the making of dyes, agricultural chemicals, and pharmaceuticals. People working with it often throw on gloves, work with decent ventilation, and get on with the job. The question about its safety or toxicity crops up in every chemical safety meeting, sometimes as a footnote, sometimes at the heart of the conversation for anyone worried about what they’re breathing in or touching.
What Science Says About Phthalimide
Looking up phthalimide in chemical safety databases pulls up a pretty clear picture. This white, often crystalline solid doesn’t fly under the radar with a bunch of scary hazard symbols. The GHS classifies it as an irritant, not a poison. Meaning, touch enough of it or let the dust fly on your skin or in your eyes, and you’ll feel it. Redness, a burning sensation, maybe tearing if it hits the eyes. Breathe in a lot of dust, and your throat and nose can get irritated. That’s not nothing, especially on a full day in the lab.
Long-term studies haven’t flagged it as a cancer risk for humans, nor do they point to significant problems with genetic mutations or reproductive toxicity—at least not under normal levels of exposure for people handling it using the recommended safety gear. Most agencies like the European Chemicals Agency (ECHA) and the US Environmental Protection Agency (EPA) put it well below the radar of “really bad actor” chemicals. Animal studies with huge doses show some toxic effects, but that’s the kind of dose you’d never see in a realistic workplace or classroom.
Is Phthalimide a Hidden Danger?
You get stories from old-timers in manufacturing who spent days surrounded by chemical powders without modern ventilation. Some talk about chronic coughs, not cancer. Phthalimide never showed up on lists of legendary workplace hazards. Lab staff, including myself during long syntheses and extractions, rarely reported anything worse than itchy hands or a runny nose if someone got careless with clean-up.
I’ve noticed some confusion sometimes because phthalimide’s relatives—like phthalic anhydride—carry more risk, particularly for asthma or allergic reactions. Mixing up names leads people to lump them all into the “toxic” category. Properly labeling and storing chemicals still matters, no matter how mild something looks on safety sheets.
Minimizing Risk and Moving Forward
The right way to work with phthalimide lines up with basic lab safety: gloves, goggles, keep the material off your skin, avoid breathing in dust. Good lab culture means dealing with any powder as if it mattered, not just the skull-and-crossbones stuff. If some hits your skin, wash up. If anyone in the workplace deals with bulk quantities, dust controls and regular clean-up routines matter more than ever.
Accurate information matters. One reason public trust in science waivers comes from half-heard stories about “toxic chemicals” with little detail. Talking about real risks, not horror stories or hand-waving, builds a better culture both among workers and in the public eye.
No one needs to sound alarms over phthalimide in normal use. Respect it, treat it like a typical chemical, and stay on top of new safety data if work routines change or unexpected symptoms crop up. A clear-eyed approach protects health without slowing down innovation or research.
How is phthalimide synthesized?
Why the Way We Make Phthalimide Matters
Staring at a white, crystalline solid called phthalimide, you see more than just another chemical. For chemists, this compound opens the door to a range of possibilities. Year after year, phthalimide shows up in textbooks and labs as a platform for making wide-ranging pharmaceuticals, agrochemicals, and dyes. That reputation rests on reliable, efficient production. As someone who’s spent afternoons in university labs and read crop protection studies, I know this stuff finds its way from glassware straight to real-world impact.
Phthalimide doesn’t grow on trees or show up in mineral deposits. Chemists synthesize it using practical, direct methods. One widely used route involves phthalic anhydride and ammonia. By carefully heating these two chemicals together, the reaction gives phthalimide along with a bit of water as a byproduct. Lab manuals and commercial setups like this method because it’s straightforward and doesn’t ask for exotic equipment. The process involves heating phthalic anhydride to about 130–150°C, then passing dry ammonia gas over the hot solid. After cooling, phthalimide crystals form and can get filtered out for later use.
The Chemistry Behind the Reaction
While many syntheses need precise conditions or fancy catalysts, this process avoids unnecessary complications. Phthalic anhydride is not expensive, and ammonia can be safely handled in professional environments. The reaction between them produces phthalimide directly, which saves time and reduces waste.
During graduate school, I handled glass tubes with solid phthalic anhydride clumps and passed ammonia through after preheating. The moment the reaction mix cooled down, white crystals started to appear. This simple transformation lets undergraduates learn real chemical synthesis without diving into hazardous territory, as the reactants stay manageable with basic personal protective gear.
Why the Synthesis Stands Out
Phthalimide has earned its reputation as a key intermediate thanks to this straightforward preparation. Gabriel synthesis of amines, which many organic chemists know by heart, wouldn’t exist without it. The process offers a safe, reproducible way to make phthalimide in both small and industrial batches.
On top of that, the byproduct, water, doesn’t leave behind harmful residues or pollutants. Large-scale manufacturers look for these efficiencies, especially as tighter regulations govern chemical waste. Here, cost and environmental responsibility go hand in hand.
Safety and Sustainability
Lab safety deserves more than a footnote. Working with ammonia does require a well-ventilated area and the use of goggles, gloves, and fume hoods. In my own teaching, reminding students to check for proper ventilation and quickly neutralize accidental spills was just as important as measuring reactants.
As green chemistry gains ground, fewer processes can claim low waste and minimal toxicity. This reaction’s profile fits neatly into sustainable practices. By designing processes around chemicals that don’t hang around in the environment or cause major health scares, chemists actively contribute to safer labs and cleaner products.
Room for Improvement and Future Directions
Scaling up doesn’t come without hurdles. Streamlining ammonia handling and further reducing the energy needed for heating remain challenges. Finding milder alternatives that lower the reaction temperature without sacrificing yield could help. Exploring catalysts or different reactors may bring those improvements within reach.
In a world full of buzzwords and new laboratory gadgets, this old-school synthesis of phthalimide stands as a reminder: Sometimes, the most effective solutions come from well-established methods, made even better with smart tweaks and a commitment to safety and sustainability.
What are the storage conditions for phthalimide?
How Safe Handling Protects Health and Product
Phthalimide doesn’t show up in daily conversations unless you spend time in chemistry labs or chemical plants. It pops up during lab syntheses, often as a reagent or intermediate. I’ve used it for making primary amines — it’s white and powdery, not the type of chemical you leave lying around without a care. Storage concerns go far beyond regulatory checkboxes: they keep people healthy and chemical stocks uncontaminated.
Understanding What Phthalimide Is
Most synthetic chemists recognize phthalimide’s strong role in organic reactions such as the Gabriel Synthesis. Once I spent hours wondering why phthalimide kept fine structure and purity over months, while other similar compounds clumped or discolored. The answer lies in how it gets stored and what happens if mistakes break routine.
Risks of Neglect
It’s tempting to downplay storage requirements when a material doesn’t smell or react violently. But even if phthalimide feels benign, moisture and heat still cause trouble. Damp conditions can break its molecular structure. Even modest exposure to air can attract enough water vapor to create problems. Over time, impurities introduce extra variables into lab results or production lines. Mishandling builds up risks, and exposure brings health dangers like irritation to the eyes, skin, and respiratory system. The best chemists I worked with always took these slow-burn hazards seriously.
Storing Phthalimide Right
I’ve seen the best results in dry, tightly sealed containers with clear labeling. Glass bottles with screw caps work well. Shelving phthalimide away from acids, bases, or reactive metals cuts down fire and chemical interaction hazards—especially important in busy storage areas where mix-ups happen. Some labs rely on desiccators; these remove lingering moisture and prevent caking. Locked chemical storage cabinets also help to reduce unauthorized access, keeping both people and inventory safe.
Beyond temperature, humidity control matters. Cool—meaning room temperature—is fine, but avoid ovens, steam pipes, or other sources of fluctuating heat. Any area subject to direct sunlight or drafts complicates storage, especially in buildings with older ventilation. At home labs or small operations, I try to mirror professional guidelines by borrowing silica gel packets and consistently checking expiration dates.
Clear Labeling and Good Habits
Simple habits build a safer culture around chemicals. I label each bottle with date received and date opened. Any mix-up or cross-contamination can mess up months of work and trigger safety incidents. Running checklist-based audits a couple times a year keeps standards consistent, even for experienced staff. It’s easy to dismiss procedures, but every seasoned chemist I know remembers a close call involving a poorly stored reagent.
Solutions for Industry and Home Labs
Busy workspaces sometimes cut corners, storing phthalimide alongside less stable reagents. Dedicated chemical shelves fix many problems. For industry settings, clear segregation by chemical class keeps audit trails cleaner and sharpens crisis response. Digital inventory systems give a heads-up on expiry dates or recall alerts. In smaller settings, even a simple spreadsheet can work wonders to reduce waste and improve safety.
Maintaining a dry environment calls for basic but effective tricks. Desiccators, silica packs, or sturdy airtight jars all add layers of protection. Rushing never pays off, especially when dealing with chemical powders. Good storage means fewer ruined batches and less risk to everyone in the building.
Can phthalimide be used in pharmaceutical applications?
What Drives Interest in Phthalimide?
Walking through the maze of organic chemistry, it’s not unusual to spot phthalimide on a lab bench. Its structure pops up in plenty of textbooks and old research papers, but its impact stretches far beyond academic interest. Chemists recognize phthalimide as a starting point for making several types of medicines, and for good reason. With its simple ring structure, phthalimide opens pathways to create new molecules for treating infection, seizure disorders, and cancer.
Phthalimide’s Role as a Versatile Starting Point
Few compounds match the flexibility phthalimide brings to synthesis. In drug development, chemists lean on it to introduce nitrogen groups or to build up more complex ring systems. Take thalidomide as a case: phthalimide serves as the foundation. Despite the drug’s tragic history with birth defects in the late 1950s, derivatives of thalidomide, such as lenalidomide and pomalidomide, have changed the lives of many with multiple myeloma.
Scientists use the Gabriel synthesis, which involves phthalimide, as a reliable route to make primary amines—those building blocks appear throughout drug design. This process offers precision and helps avoid unwanted side products that otherwise slow down discovery or complicate manufacturing.
Safety and Handling—Not Everything Is Perfect
Risks can’t be ignored. While phthalimide itself does not pose high toxicity in its pure form, poorly controlled processes or contamination with similar chemicals may raise safety flags. In my own lab years, careful handling and proper waste disposal kept exposure to a minimum. Regulatory agencies watch for compounds with harmful effects on people or the environment. Drug manufacturers and researchers face the challenge of balancing the benefits while reducing harm.
Innovation: Seeing Beyond Old Boundaries
Researchers search for new ways to use familiar chemicals with better results and fewer risks. Some teams experiment by tweaking phthalimide structures, hunting for a new drug that works without old side effects. Others improve manufacturing by swapping out outdated solvents and improving recovery methods, cutting costs and cutting waste.
Data is key. By gathering toxicity data and environmental records, teams can fine-tune their approach. Public awareness also pushes drug companies to find greener solutions, making safety and sustainability top priorities.
Potential for New Treatments
Interest in phthalimide will not fade soon. Every year, new reports highlight drug molecules designed around its ring. Patents stack up, and clinical trials move forward. Many of these new drugs aim to fight cancer or control immune system problems. As resistant infections challenge doctors, chemists chase innovative antibiotics. Some of these candidates have roots in phthalimide chemistry.
Pushing Forward Responsibly
Looking back on my own struggles with older lab methods, I understand the pressure to keep up with regulations, refine synthesis, and avoid outdated mistakes. Fact-checking potential risks pays off for both the scientist and the patient. Using phthalimide in pharmaceuticals delivers real benefits—when the work keeps ethics, safety, and sustainability front and center. Today’s choices set the tone for tomorrow’s treatments.
| Names | |
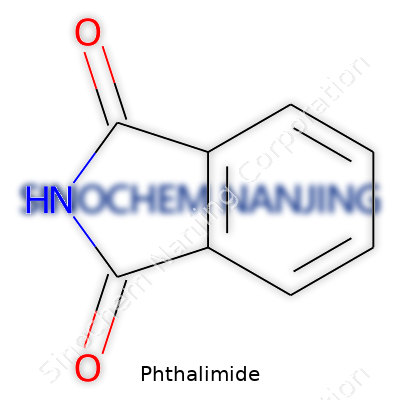
| Preferred IUPAC name | 1H-isoindole-1,3(2H)-dione |
| Other names |
1H-Isoindole-1,3(2H)-dione Isoindoline-1,3-dione N-Phthalimide Phenylene dicarboximide |
| Pronunciation | /ˈθæl.ɪ.maɪd/ |
| Identifiers | |
| CAS Number | 85-41-6 |
| 3D model (JSmol) | `phthalimide.mol::JSmol` |
| Beilstein Reference | 82126 |
| ChEBI | CHEBI:29009 |
| ChEMBL | CHEMBL1576 |
| ChemSpider | 1196 |
| DrugBank | DB12733 |
| ECHA InfoCard | 100.013.840 |
| EC Number | 204-419-1 |
| Gmelin Reference | 202159 |
| KEGG | C01743 |
| MeSH | D010764 |
| PubChem CID | 6780 |
| RTECS number | TI1575000 |
| UNII | U0A9K6GMPD |
| UN number | UN2214 |
| Properties | |
| Chemical formula | C8H5NO2 |
| Molar mass | 147.13 g/mol |
| Appearance | White to off-white crystalline powder |
| Odor | Odorless |
| Density | 1.21 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 0.86 |
| Vapor pressure | <0.01 mmHg (20°C) |
| Acidity (pKa) | 8.3 |
| Basicity (pKb) | 7.68 |
| Magnetic susceptibility (χ) | -49.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.654 |
| Dipole moment | 0.000 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 143.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -452.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2548.7 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P280, P304+P340, P305+P351+P338, P312 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 286°C |
| Autoignition temperature | Autoignition temperature: 610°C |
| Lethal dose or concentration | LD50 (oral, rat): 6400 mg/kg |
| LD50 (median dose) | LD50 (median dose): 6400 mg/kg (oral, rat) |
| NIOSH | JN9625000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.4 mg/m³ |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Phthalic anhydride Phthalic acid N-Hydroxyphthalimide Saccharin Sulfanilamide |

