Phosphorus Pentasulfide: A Deep Dive Into Its Modern Role and Ongoing Challenges
Tracking the Early Roots and Ongoing Growth
Phosphorus pentasulfide stepped onto the industrial stage more than a century ago. The chemical industries once operated with only a handful of such complex compounds, so this one caught attention for what it could do. As a kid visiting relatives working at fertilizer plants, I remembered seeing workers clad in thick gloves, talking about the “yellow stuff” that could make matches light—or break out trouble if left unchecked. Factories looked at its potential for new phosphorus-sulfur chemistry, drawing early patent filers and sparking waves of innovation. As research picked up in the mid-1900s, the compound found routes into both specialty and mass-produced goods, launching it into a permanent spot in manufacturing’s toolkit.
Getting to Know the Compound
Phosphorus pentasulfide appears as yellow-green crystals or flakes. Its solid form puts many at ease, but the story changes with friction or heat. You open a drum, and a sharp, foul odor rises—reminiscent of rotten eggs and garlic, a dead giveaway for sulfur compounds. It melts at around 288°C and boils a bit above 500°C, which makes handling under standard conditions manageable, but still risky. Water isn’t a friend to this compound, triggering a chemical scramble that belches out flammable hydrogen sulfide gas—and the noxious phosphoric acid clouds. In a warehouse, the metal containers keep it stable, but once dust spills or dampness creeps in, everyone’s on alert.
What Goes on the Label: Specs in Plain English
Look on a bag or barrel, and you’ll see Phosphorus Pentasulfide listed with a minimum purity of 99%. That level guarantees low contamination from phosphorous trioxide or other sulfur-rich byproducts. Industrial labeling shows warnings about reactivity with water, recommended storage temperatures (10-30°C), and clear instructions for airtight storage away from moisture. Containers must bear both hazard and handling symbols, not only for compliance but to prevent day-to-day slip-ups. Batch numbers and manufacturing dates help trace problems if they ever arise.
How it’s Made: From Simple Elements to Specialty Chemical
Manufacturers combine red phosphorus and molten sulfur using closed reactors. Temperatures reach past 300°C. The reaction isn’t quick; technicians keep an eye on pressure and off-gassing, since mistakes can mean toxic releases or runaway fires. Modern plants use gradual feed rates, scrubbing systems, and sensors to avoid sudden releases. Getting the product out calls for vacuum distillation, pulling away impurities and leaving a crisp, clean substance behind. These extra steps eat at margins but save headaches down the line when purity makes or breaks a process.
Reactions That Make a Difference
Phosphorus pentasulfide reacts with alcohols, amines, and a handful of hydrocarbons to introduce sulfur where it’s needed. In my college organic chemistry lab, I saw firsthand how it transformed simple esters into robust thioesters, the backbone for enzyme mimicry studies. Industry chemists push these reactions further: turning out lubricant additives, pesticides, and agents for rubber processing. Each modification alters how the sulfur-phosphorus bonds break and form, drawing out a range of specialty chemicals. Even the byproducts hold value or pose challenges, sometimes calling for capture and recycling methods that have only become common in recent decades.
What’s in a Name?
Phosphorus pentasulfide also goes under names such as P2S5, phosphoric sulfide, phosphorus(V) sulfide, and sometimes—less commonly—as diphosphorus pentasulfide. Safety sheets and shipping manifests might only list the CAS number 1314-80-3. Industries have nicknames depending on if they use it for matches, pesticides, or lubricant synthesis, but the underlying substance stays the same, bringing the same risks and benefits to production lines.
Meeting Demands: Safety and How It’s Handled
Handling phosphorus pentasulfide calls for strict safety protocols drawn from decades of lessons. At plants where it’s shipped in drums, workers suit up in gloves, goggles, and masks. Spills bring out sand or dry absorbents, never water, and dedicated ventilation ducts suck away stray fumes. Transport routes get planned to avoid exposure to rain or high humidity. Regulations now require thorough tracking and regular inspections. Despite these measures, human error still creeps in. Stories from long-timers in the chemical sector detail incidents where old habits—like grabbing a wet mop for clean-up—quickly turned into hazardous emergencies. Routine drills, clear labels, and encouraging open reporting change workplace culture, reducing those close calls.
Where It’s Used: Beyond the Lab
Phosphorus pentasulfide found roles in making gear oils that hold up under grinding pressure and heat. Factories rely on it for zinc dithiophosphate production—the secret behind lubricants that keep giant turbines spinning or car engines humming. It plays a role in manufacturing pest control products, especially in parts of the world where tropical farming faces relentless bugs. Specialty glass and matches still pull demand for the compound, though less so now with modern electronic ignition products cutting into that market. In research labs, it carves new paths for making sulfur-rich compounds, including next-generation medicines and polymer materials.
Pushing the Limits: The Research Race
Researchers keep finding new ways to stretch the use of phosphorus pentasulfide. It gets tested as a building block for lithium-ion battery electrolytes, in part because the mix of phosphorus and sulfur shows promise for high-energy-density designs. Some labs use its reactivity to tweak the properties of specialty rubbers, chasing better resistance to wear and ozone. There are ongoing questions about how additives derived from P2S5 might fend off corrosion in renewable energy infrastructure—solar and wind—where exposed steel needs robust, long-lasting coatings.
The Toxicity Tangle
Toxicologists started raising red flags about phosphorus pentasulfide’s breakdown products after early factory workers complained about headaches, nausea, and lung irritation. Hydrogen sulfide and phosphoric acid—the main culprits—cause both acute symptoms and long-term harm. Recent studies track exposure levels in workers, checking for early signs of respiratory or skin issues. Newer reports probe chronic exposure, focusing on low-level effects that weren’t on the radar a generation ago. Factories respond by tightening controls, adding more sensors, rotating workers, and sharing exposure data with local health authorities. These steps make a difference, but constant vigilance stays necessary as more subtle health links keep surfacing.
Looking Ahead: The Search for Smarter Solutions
Industry and science both chase alternatives—compounds with the same benefits but less environmental or health risk. Demand for safer, greener chemical routes pushes innovation in synthesis and storage. I’ve seen startups try biotechnological alternatives or recycling methods to recover phosphorus from waste. Digital monitoring brings real-time feedback, allowing for smarter controls. Researchers sift through natural analogues and new catalysts, hoping for breakthroughs that cut back on the need for such potent, hazardous intermediates. Despite the hurdles, the combined persistence of policy changes, worker advocacy, and ongoing R&D steadily shifts how phosphorus pentasulfide’s story unfolds—forging a legacy that keeps balancing usability against safety and sustainability.
What is Phosphorus Pentasulfide used for?
From Smelly Chemistry Class to Vital Industry Staple
I still remember walking into the chemistry lab in high school and catching a whiff of something sharp, almost garlicky—turns out, that was phosphorus pentasulfide. Back then, it felt like just another odd laboratory chemical. Outside those glass jars, it actually plays a big role much closer to daily life than most expect.
Phosphorus Pentasulfide in Lubricant Production
One of the most important markets for phosphorus pentasulfide hides inside car engines and heavy machinery. This chemical gets used as a building block for zinc dialkyldithiophosphate (ZDDP), an old but trusted additive that protects engines. ZDDP coats metal parts, helping them survive the grind and stress that comes from years of use. Before learning about it, I always thought engine oils were only about the base oils. Turns out, without phosphorus pentasulfide helping to create ZDDP, those engine components would wear out much faster, breaking down under pressure and heat.
Sulfur-Based Crop Treatments
Growing up in a farming family, keeping pests at bay felt like a never-ending challenge. Phosphorus pentasulfide shows up in agriculture too, especially in the production of certain pesticides and fungicides. Instead of just being a chemical floating around a lab, it gets transformed into ingredients that fight fungal infections and bugs. Some of these treatments have protected countless harvests and increased food supply during tough years. That comes with challenges, of course. Overuse of pesticides raises questions about long-term soil health, water safety, and worker exposure. Responsible handling and careful regulation go a long way to keeping benefits while limiting the risks.
Safety Matches and the Everyday Spark
I still get a kick out of striking a match and seeing that tiny flame leap up. Many safety matches rely on derivatives from phosphorus pentasulfide for their heads. This is where chemistry hooks up with everyday convenience—turning a tough, sulfur-based compound into something that plays a part in barbecues, campfires, or lighting candles at home. But it’s not just household fun. The production process demands strict attention to worker protection and ventilation because of the strong fumes. Manufacturing facilities must invest in training and equipment to keep everyone out of harm’s way, which points to another truth with specialty chemicals—proper safety protocols can mean the difference between a thriving business and a tragic accident.
Pushing for Cleaner, Safer Practices
Like a lot of industrial chemicals, phosphorus pentasulfide brings both opportunity and risk. In places I’ve worked, waste management and proper storage mattered just as much as the production itself. Fires from improper storage, chemical burns from accidental exposure, or air quality problems from fumes show up in accident reports every year. Recycling programs for chemical containers, investing in protective equipment, and regular environmental audits help companies stay ahead of trouble. Clear labeling on barrels and regular refresher courses for employees make a difference, too—isn’t much point to a fancy ventilation system if people don't know how to react to spills or leaks.
Building a Smarter Future in Chemical Manufacturing
Demand for phosphorus pentasulfide isn’t going away. So, industry can focus on smarter engineering controls, research greener alternatives, or even redesign processes to reduce emissions from start to finish. Anyone working in manufacturing or using the products downstream can push for transparency and safer options. That way, we get the benefits—better engines, more resilient crops, the comfort of a match on a rainy day—without carrying unnecessary risks into tomorrow’s world.
Is Phosphorus Pentasulfide hazardous?
Everyday Exposure Isn’t the Whole Story
Phosphorus pentasulfide rarely shows up in the kind of products most folks keep around the house. Instead, you’ll find it in spaces like industrial plants, where it plays a part in making lubricants and other chemicals. For those who work around it, the stuff comes with some real risks. Understanding those dangers means looking past what safety data sheets say and considering the gritty details in the shop or lab.
Why Hazards Aren’t Just “On Paper”
Contact with phosphorus pentasulfide gets worse than a simple chemical stain. Skin burns and eye injuries happen quickly. Inhaling the dust stings, irritates airways, and can land someone in the hospital. The worst part comes if the chemical hits water. It bursts into a violent reaction, throwing off hydrogen sulfide gas—that rotten egg smell everyone knows is more than just unpleasant. Breathing it in knocks out your sense of smell, so you might not even know you're in trouble until you’re dizzy or coughing hard.
Real-Life Risks for Real Workers
Years ago, in a fertilizer plant, I watched a minor spill turn into chaos. Someone used a wet mop, thinking cleanup was routine. Within moments, the staff had to clear out because of the gas filling the room. That lesson stuck—water and phosphorus pentasulfide never mix. OSHA has rules for this chemical for good reason, but rules on their own do little unless crews get training and gear.
Burns from even small splashes can stay with a worker for months. Inhaling dust just once without proper masks can force time off the job. Hydrogen sulfide carries extra weight—at crisis levels, the gas can kill in just a couple of breaths. Stories like these make the experts’ warnings real. Every safety meeting about this chemical has scars and close calls behind it.
Safe Handling Means Looking Out for Each Other
Hazard doesn’t end with individual cases. Phosphorus pentasulfide poses threats to whole buildings or communities if ignored. Spills can contaminate soil and water, and a leak in the wrong place threatens people blocks away. Training must match the risk. Experience says it works better to see training as a team effort: more experienced workers helping rookies spot trouble, or crews practicing quick evacuation until it feels automatic.
Companies need strong safety programs. Personal protective equipment matters—think face shields, gloves, and respirators that fit properly. Dry cleanup beats wet methods any day. Air monitors should be checked before someone says, “good enough.” Emergency plans must be more than a dusty binder—crew members should know them the way they know the back of their own hands. Only that kind of culture keeps risks down and workers safe.
Seeking Smarter Solutions
Thinking beyond quick fixes pays off. Substituting less hazardous chemicals, automating processes, or refining engineering controls matter most over the long haul. It’s not always cheap, but the alternative—fines, injuries, or worse—carries a heavier price. Industry experts share best practices to keep accidents rare and push toward safer chemical options where possible.
People who work around phosphorus pentasulfide earn a safer environment by staying honest about hazards and insisting on effective measures. No shortcut justifies a life put in danger. The chemistry may sound abstract in a book, but it stays grounded in real consequences every day in the field.
What is the chemical formula of Phosphorus Pentasulfide?
Unlocking Phosphorus Pentasulfide: More Than Just P2S5
Phosphorus pentasulfide goes by the formula P2S5. This chemical holds an almost greasy reputation in the fields of industry and agriculture. The formula looks simple on paper, but the stuff plays a big role in how we lubricate engines and grow food. It’s not one of those compounds that sits quietly on a shelf gathering dust. The world uses it for making lubricating oil additives, pesticides, and even certain kinds of flame retardants.
Digging Deeper: From Reactions to Real Life
One bag of phosphorus pentasulfide stands at the crossroad of chemistry and daily life. The compound appears when phosphorus reacts with sulfur vapors at high temperature. Those yellow-green flakes might not look exciting, but they change the game for farmers and manufacturers. Many farmers rely on pesticides made from phosphorus pentasulfide. This chemical plays a key part in keeping harmful insects off food crops, which feeds more people using less land.
In factories, phosphorus pentasulfide helps make additives called zinc dialkyldithiophosphates. These slow the wear and tear on engines, letting vehicles run longer without breaking down. Anyone who’s spent time under the hood knows engines don’t just need clean fuel—they need the right oil additives. P2S5 keeps those additives working hard, even under stress.
Health and Safety: Not as Simple as it Looks
Chemicals with formulas like this always bring some risk. Phosphorus pentasulfide isn’t something to leave lying around the kitchen. It reacts with water to release hydrogen sulfide gas, and nobody wants a whiff of rotten eggs or the headaches that follow. At the job site, handling this material requires protective equipment and training. Proper ventilation and sealed containers help keep both workers and nearby communities out of harm’s way.
In my work around chemical storage, we always talked about respect for P2S5—not out of fear, but because safety makes a difference between a smooth day and a trip to the emergency room. Training exists not just as a bureaucracy, but to send people home safe. The chemical industry’s track record still shows room to grow, but more companies have started focusing on thorough training and transparent processes.
Sustainability and the Road Ahead
As people push for greener lifestyles, phosphorus pentasulfide presents both a challenge and an opportunity. The pesticides stemming from it boost yields, but runoff poses issues for waterways. Alternative pest control methods and tighter regulations may lessen these effects. Engine additives made from P2S5 reduce mechanical waste, adding another layer to the sustainability conversation. When safe handling and responsible sourcing come together, the benefits of P2S5 spread beyond the factory or farm.
A simple formula like P2S5 shapes daily life more than many realize. Steak on the table and cars on the road both owe something to this tiny piece of chemical shorthand. The real challenge—responsible use—calls each of us to know the science and respect its power.
How should Phosphorus Pentasulfide be stored?
Why Safety Matters
Few chemicals in the industrial toolkit demand respect like phosphorus pentasulfide. Anyone involved in the world of mining, lubrication, or pesticides will recognize its yellow-green flakes at a glance. The stuff reacts with water to form toxic gases, which makes haphazard storage a recipe for disaster. Just looking at news stories about chemical accidents reminds me that even routine materials can spark chaos if treated lightly.
Common Sense Storage Steps
You can’t treat phosphorus pentasulfide like something benign. When I consulted safety teams at a fertilizer plant years ago, they hammered home the basics: keep this compound dry at all costs. Roof leaks or even a humid warehouse put everyone at risk. Even a plastic tarp can make a difference in an emergency, but permanent, sealed packaging always wins out. I remember a rainy night when a poorly covered drum caused a brief scare—a lesson that stuck with me.
On top of moisture, you want to keep it away from open flames, sparks, or heated equipment. In most plants, that means keeping it in a separate, ventilated storage area away from traffic and high-energy activities. Some companies use concrete or brick rooms with well-fitted doors. Metal shelves work well as long as the container stays closed. I’ve seen warehouses rely on strict checklists, logging inspections so nothing gets overlooked.
Labeling and Spill Preparedness
Every barrel or drum needs clear hazard symbols and instructions, not just for regulatory reasons but for basic staff safety. People moving and using the material might not have chemistry backgrounds. Accidents happen most often because someone didn’t read a label and mixed the compound with the wrong thing. More than one friend in industry tells of people opening containers they shouldn’t have, simply because labeling faded or peeled away in the sun. Routine checks and relabeling prevent those moments.
Spill kits, neutralizing agents, and emergency showers make a big difference if something goes wrong. Nobody wants to scramble for supplies in a panic. I once saw a manager keep a “dry run” checklist and walk staff through what to do if a drum splits open. The teams who practice are the ones least likely to lose control during real incidents.
Legal and Ethical Considerations
Government guidelines exist for a reason. Agencies like OSHA, the EPA, and local fire marshals write some clear rules. I think it’s a mistake to treat those as mere “checkboxes.” Fines pale compared to the human cost of accidents. Whenever I spoke with chemical handlers after a close call, regret about not taking extra steps comes up every time.
Storage isn’t just about the product. It’s about the people, the community, and the reputation of everyone involved. In too many cases, corners get cut due to cost or ignorance—until someone pays for it. Good practice comes from a mix of regular training, routine audits, and real discussion with frontline workers. If folks are comfortable reporting near-misses or raising an eyebrow at sloppy storage, the whole workplace gets safer.
Thinking Ahead
Phosphorus pentasulfide sounds unremarkable until things go wrong. Proper storage habits—dry environments, sturdy containers, clear labeling, and staff training—keep people out of harm’s way. Seeing both close calls and best practices firsthand, I’ve learned that small, consistent efforts save more headaches than any after-the-fact fix. In the end, treating storage as a daily responsibility sets the tone for every other safety decision that follows.
What safety precautions are needed when handling Phosphorus Pentasulfide?
The Risks Everyone Should Know
Working with phosphorus pentasulfide brings some real dangers. This yellowish solid shows up in everything from the production of lubricating oil additives to pesticides. Even with that everyday use, this chemical doesn’t take many chances to cause injury. It reacts fiercely with water, even with small amounts of humidity, kicking out hydrogen sulfide and phosphoric acid fumes. These aren’t just unpleasant smells; both compounds can hurt your lungs, eyes, and skin. Eyes water, throats burn, noses sting. A single splash or breath of the wrong vapor lingers for a while—some workers talk about coughing fits and burning lungs that stick with them for days.
Explosive fire risk often gets brushed aside in busy shops. Phosphorus pentasulfide can catch fire if stored near oxidizers. Water-based fire fighting looks like a solution, but it’s the opposite—throwing water on phosphorus pentasulfide sends more hydrogen sulfide gas into the air, putting firefighters and workers at higher risk. Local evacuations sometimes follow accidents, and hospitals have treated more than a few workers for serious burns.
Real Steps to Safer Handling
No one likes wearing uncomfortable gear, but real protection matters here. Rubber gloves, face shields, chemical goggles, and aprons block splashes and dust better than regular cotton clothing. Supervisors tend to overlook fit-testing for respirators, but leaks render them useless, especially if a nearby spill releases vapors. Simple half-face masks don’t cut it; full-face, air-purifying cartridges rated for acidic gases stand a better chance. Some companies think fans or open windows do enough, but local exhaust ventilation works best by dragging fumes away before they hit a worker’s face.
Labeling and storage make a difference. Too many storage rooms squeeze incompatible chemicals into the same corner. Keeping phosphorus pentasulfide away from water, acids, and oxidizers lowers accident chances. Segregation goes beyond just a sign on the door; sturdy, sealed containers, moisture control packets, and regular checks for leaks prevent those “close calls” from turning into headlines. Blunt fact: forgotten containers and faded labels have caused real emergencies more than once.
Defensive habits at work matter every day. Washing hands, changing out of soiled clothes, and using showers on site cuts down long-term health risks. Eye wash stations and emergency showers should stay uncluttered—no storage boxes resting in the way or dried out pipes. Quick access saves time in a crisis. Every team should hold regular drills and refreshers, since memory fades and new risks pop up. In my own experience, the best drill didn’t come from a manual but from a spill—everyone needs to know exactly where to run and what to grab.
Building a Culture of Care
Top workplaces encourage open reporting. Too often, fear of reprimand keeps workers quiet until a small leak turns into a major problem. Respect grows where everyone—new hire or veteran—gets taken seriously if they spot a crack, a smell, or a worry. OSHA standards don’t just exist on paper, either. Inspections and honest conversations about near-misses give teams a chance to adapt before a real emergency strikes.
Tough chemicals demand a real commitment from companies and workers. By focusing on prevention, clear training and gear, and open communication, companies can keep people safer. Sometimes the difference between a disaster and a close call comes down to one person choosing to double-check a glove or mop up a droplet before clocking out.
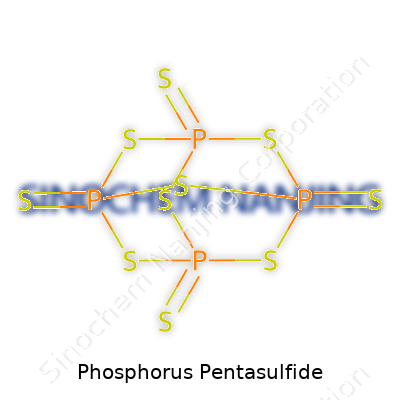
| Names | |
| Preferred IUPAC name | tetraphosphorus decasulfide |
| Other names |
Tetraphosphorus decasulfide Phosphorus(V) sulfide Phosphorus sulfide P2S5 |
| Pronunciation | /ˌfɒs.fə.rəs ˌpɛn.təˈsʌl.faɪd/ |
| Identifiers | |
| CAS Number | 1314-80-3 |
| Beilstein Reference | 1206827 |
| ChEBI | CHEBI:48878 |
| ChEMBL | CHEMBL1201649 |
| ChemSpider | 19013 |
| DrugBank | DB11162 |
| ECHA InfoCard | 100.029.254 |
| EC Number | 215-242-4 |
| Gmelin Reference | 113264 |
| KEGG | C18738 |
| MeSH | D010754 |
| PubChem CID | 24459 |
| RTECS number | RTECS TP0175000 |
| UNII | F5HNB5M831 |
| UN number | UN1340 |
| Properties | |
| Chemical formula | P2S5 |
| Molar mass | 222.27 g/mol |
| Appearance | Yellow crystalline solid |
| Odor | rotten eggs |
| Density | 2.09 g/cm³ |
| Solubility in water | Insoluble |
| log P | -0.21 |
| Vapor pressure | <0.1 hPa (20 °C) |
| Acidity (pKa) | -2.5 |
| Basicity (pKb) | 1.42 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.689 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 226.6 J⁄(mol·K) |
| Std enthalpy of formation (ΔfH⦵298) | -308.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1561.3 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB39 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H314, H410 |
| Precautionary statements | P223, P231+P232, P280, P370+P378, P402+P404, P501 |
| NFPA 704 (fire diamond) | 3-4-2-W |
| Flash point | 300 °C (572 °F) |
| Autoignition temperature | 300 °C (572 °F) |
| Lethal dose or concentration | LD50 (oral, rat): >6400 mg/kg |
| LD50 (median dose) | 4100 mg/kg (rat, oral) |
| NIOSH | WFJ85750 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Phosphorus Pentasulfide: "1 mg/m³ (as P2S5), 8-hour TWA (OSHA) |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
Phosphorus trisulfide Phosphorus sesquisulfide Phosphorus pentoxide |

