Phosphorus Pentachloride: A Deep Dive Into a Chemical Workhorse
Historical Development
Chemists began exploring phosphorus pentachloride over a century ago, drawn by its unique capability to convert all sorts of compounds through chlorination. Commercial production took off in the late 1800s when large-scale phosphorus chemistry emerged in Europe. Factories raced to supply new industries—pharmaceuticals, matches, dyes (many bright synthetic colors owe their existence to early phosphorus processes). I remember reading old chemistry texts that marveled at PCl5’s transformative powers and the confidence it gave to pioneers who wanted to push into new markets.
Product Overview
Phosphorus pentachloride shows up as a pale yellow-green, crystalline solid with an acrid bite. It goes by several names: phosphorus(V) chloride, PCl5, and sometimes “phosphoric chloride” in older literature. The global market mainly deals in drums or steel cans, moisture-free, since this compound reacts instantly with water. Companies list it as reagent grade or technical grade, depending on impurities (most specs offer >99% purity, though some applications tolerate less). In my work, even trace contamination throws off downstream synthesis, so knowing your supplier’s reputation matters.
Physical & Chemical Properties
Phosphorus pentachloride melts at around 160°C, though it decomposes before boiling. Its vapor fills the air with a choking, corrosive mist—something I learned quickly while handling it in the lab. Solid PCl5 sublimes at room temperature over time, which means leaving the lid off can coat an entire workspace with white fumes. It dissolves in carbon disulfide, benzene, and chloroform, but steers clear of water at any cost. Drop a crystal into a beaker of water, and clouds of hydrochloric acid rush out, leaving behind phosphoric oxychloride—a reaction that never fails to impress new students during demonstrations.
PCl5 holds a trigonal bipyramidal structure in the solid state, only to shift to ionic forms in polar solvents or melt. This flexibility in bonding stands at the root of its chemical prowess. Its reactivity comes from the drive to shed chlorine atoms, converting alcohols and acids into chlorinated analogs that power industry after industry. The chemical’s biting, irritating odor warns anyone nearby to keep their distance.
Technical Specifications & Labeling
Drums containing phosphorus pentachloride sport labels that leave no doubt: UN 1806, “Corrosive,” skull and crossbones, “Moisture sensitive.” Labels spell out the dangers—keep dry, call hazmat in event of spillage, restrict access by age and training. Regulatory compliance stands front and center. Shipping documents track batch numbers for every movement, a harsh lesson many manufacturers learned after near misses and accidents. Quality analysts test for chloride content, trace metals, and free amines before releasing material for export. Each shipment comes with a certificate analyzing purity, matching published standards to the decimal.
Preparation Method
Producing phosphorus pentachloride relies on direct chlorination of phosphorus trichloride under dry conditions. Factories gas PCl3 with dry chlorine, watching carefully for temperature spikes; overheating leads to side reactions, so temperature control technologies have evolved over decades to prevent costly losses or runaway reactions. Many seasoned operators trust batch methods for large volumes, where reaction vessels built of high-nickel alloys resist attack. In smaller labs, PCl5 grows in glass flasks, carefully chilled and dry. Simple in concept, the process risks severe burns, releases toxic gases, and calls for precise engineering.
Chemical Reactions & Modifications
Phosphorus pentachloride gets top billing as a chlorinating agent. Drop it onto carboxylic acids, and it substitutes the hydroxyl group for a chlorine, creating acid chlorides used in everything from pharmaceuticals to polymers. It converts alcohols into alkyl chlorides, a classic way to make building blocks for pesticides and solvents. In the dye industry, it turns aromatic amines into chloro-derivatives, opening doors to colorfast pigments. Sometimes, manufacturers blend PCl5 with Lewis acids to tweak its behavior, striving for selectivity in tough reactions. Its thirst for water means any contact with moisture kickstarts a cascade, yielding POCl3 and clouds of HCl. Experienced chemists respect its power—and its danger.
On the flip side, over-chlorination or rough handling degrades yields or produces tricky byproducts. I’ve seen a compound’s delicate structure buckle under the force of PCl5, so a steady hand and a sharp mind stay essential for new derivatives.
Synonyms & Product Names
Phosphorus pentachloride shows up on labels as “PCl5,” “phosphorus(V) chloride,” “phosphoryl chloride,” or “fuming phosphorus chloride.” In trade, it may also appear as “phosphorus perchloride” or “anhydrous phosphorus pentachloride.” Regulatory codes stick with “phosphorus pentachloride” for permits and hazard lists. Any buyer, chemist, or engineer using PCl5 quickly learns this variety—and checks all names before signing for a shipment.
Safety & Operational Standards
Safe use demands airtight PPE—acid-resistant gloves, full-face respirators, chemical-splash goggles, and work in certified fume hoods. Facilities need strict access control, emergency eyewash stations, and vented drums. OSHA and global agencies lay down exposure limits, mandating worker training. Spilled solid can strip varnish from floors, corrode metal tools, or release HCl gas dangerous to lungs and mucous membranes. Workers count every gram coming in or out. Plant protocols track exposure, and medical teams keep an eye on those with symptoms of acid exposure. In my own experience, ordinary latex gloves just crumble at the touch of PCl5; only specialized equipment stays up to the challenge.
Disposal plans require careful hydrolysis in alkaline conditions, capturing acid fumes through scrubbing towers before waste leaves any facility. Inspection teams check for leaks, corrosion, or any signs of compromised packaging. The line between safe practice and disaster runs razor-thin—everyone treats this chemical with respect.
Application Area
Phosphorus pentachloride drives innovation in more fields than most people expect. Chemical manufacturing leans on it for converting acids and alcohols to chlorides that form the backbone of many antibiotics, dyes, and surfactants. Agrochemical makers use it for pesticides and herbicides, often as a step in creating active ingredients that protect crops. Electronics companies shift volumes of PCl5 to treat circuit boards and etch semiconductors, its reactivity crucial for precise patterning. In specialty plastics and polymers, PCl5 modifies raw materials to introduce chlorine atoms, improving thermal resistance or blending properties. Even fragrance and flavor companies dip into PCl5 chemistry for rare ingredients.
Research teams scale down these reactions, chasing new ligands, complex catalysts, or next-gen battery electrolytes. My own work traced PCl5 as a direct precursor to phosphorus-based flame retardants—substances now heavily regulated but once used in hundreds of consumer products.
Research & Development
Laboratories worldwide dig into phosphorus pentachloride’s chemistry, searching for greener, safer alternatives or new transformations. Studies in synthesis tackle the challenge of reducing byproducts—getting single-step conversions with less waste and energy use. Nanotechnology researchers explore how PCl5 reacts with metal surfaces or carbon materials, hoping to unlock coatings that resist acids or tune semiconductor behaviors. Modern R&D chases selective chlorination for pharmaceuticals, aiming to cut out double reactions and tough purifications (laboratory experience proves that even a tiny improvement in specificity saves days of effort and hundreds of dollars). Some groups experiment with catalytic quantities of PCl5, recycling it through closed systems to keep emissions low while maximizing yield.
Universities—especially those with a tradition in synthetic organic chemistry—still run advanced classes on its use, comparing PCl5 with milder alternatives like thionyl chloride or phosphorus oxychloride. In roundtable discussions, chemists debate tradeoffs: process safety, cost, environmental persistence, and regulatory scrutiny drive innovation in this field.
Toxicity Research
Toxicology teams rank phosphorus pentachloride as highly hazardous to human health and the environment. Exposures by inhalation, skin contact, or ingestion trigger corrosive burns, respiratory distress, vomiting, and eye damage. The compound reacts in situ with body fluids, releasing hydrochloric acid and forming secondary toxic species. Chronic exposures link to pulmonary edema, bronchitis, or dermatitis. Animal studies establish lethal doses at a few hundred milligrams per kilogram, supporting safety managers’ insistence on strict isolation and personal protection.
Researchers monitor low-level exposure effects, linking incidents of acute irritation to poor industrial hygiene or faulty containment. Environmental teams worry about accidental releases—PCl5 hydrolyzes rapidly, but the acid byproducts devastate soil organisms and aquatic life. Cleanup requires immediate neutralization; delays amplify risk. In the early days of widespread phosphorus chemistry, lack of controls led to frequent injuries—a situation modern standards fight to prevent.
Future Prospects
Looking ahead, industries aim to cut the environmental and health hazards tied to phosphorus pentachloride without losing the sheer utility it provides. Green chemistry now calls for milder substitutes—safer reagents, less wasteful methods, and closed systems that capture emissions at the source. At the same time, the push for new drugs, advanced polymers, and nano-enabled devices means demand for effective chlorination isn’t fading. Factories in emerging economies ramp up output, spurred by electronics and agrochemical manufacturing, but face tougher competition and oversight. In my eyes, the companies that succeed will pair technical excellence in containment and recycling with ongoing investment in alternatives. Collaboration between academia, regulators, and major producers will shape the next chapter for PCl5—the race to balance productivity with sustainability has just begun.
What is Phosphorus Pentachloride used for?
Crucial Role in Chemical Manufacturing
Phosphorus pentachloride stands out in the world of industrial chemistry, especially for anyone who's spent time in a lab or dealt with large-scale chemical production. I remember seeing it show up in the back rooms of a friend’s pharma business, stacked carefully with labels warning about its volatility. Its main role lands squarely in the hands of those producing chlorinating agents. If someone needs to turn an alcohol or a carboxylic acid into a chloride, this compound steps up to the job. Turnover of chlorine-containing products across the pharmaceutical world owes quite a bit to this pale yellow solid.
Companies find this stuff handy for making antibiotics, dyes, and plastic additives. For example, producing antibiotics like erythromycin gets easier and cleaner thanks to how Phosphorus Pentachloride swaps out oxygen for a chlorine atom, helping chemists manipulate molecules in ways nature rarely does. In dye manufacturing, it helps twist the structure of compounds so that pigments pop brighter and last longer. Even if you don’t see its name on a label, it’s working behind the scenes in the products you use every day.
Electronics and Beyond
Modern electronics count on this chemical for disk drives, semiconductors, and a swath of chips inside everything from laptops to electric cars. Semiconductor fabrication often calls for creative ways to shape and etch wafers, and here phosphorus pentachloride enters as a master of adding phosphorus into silicon. The result is faster, better circuits that drive the tech most folks now depend on.
Because this compound reacts strongly with water and organic material, it requires special handling, but that reactivity also makes it invaluable for advanced materials. Any step forward in battery tech, solar panels, or telecommunications cables often means someone out there is reaching for phosphorus pentachloride.
Environmental and Health Concerns
As much as I respect its utility, I’ve seen up close how working with such a reactive chemical needs sharp attention to health and safety. Breathing its vapors or touching it without solid equipment leads to burns, choking, and long-lasting health effects. In the production sites I visited, everyone dressed in thick gloves and well-sealed goggles, ready for the faintest whiff of trouble. Accidents don’t just hit lab workers; spills and improper storage can cause harmful releases into water and air.
Waste disposal creates another headache. You can’t just flush leftover phosphorus pentachloride or its byproducts into local sewage. Strict rules direct that waste to specialized treatment plants, and I’ve watched as companies struggle with costs and compliance. Stronger regulations, regular inspections, and investment into safer alternatives could ease the environmental load. Some outfits already turn toward less hazardous reagents or recycle their chlorine-based wastes, but across much of the industry, corners get cut to save money.
Looking Forward
So I’m convinced the future needs smarter choices in managing chemicals like this. Education for workers, tighter government oversight, and big investments in greener substitutions point toward safer workplaces and less pollution. Phosphorus pentachloride deserves its reputation for power, but too often, its risks get passed on to underpaid workers or unprepared communities. We can do better by making chemical safety a personal and public mission, not just a checkbox on a regulatory form.
What are the safety precautions when handling Phosphorus Pentachloride?
The Hazards Up Close
Phosphorus Pentachloride sounds like something from a high school chemistry set, but in the real world, it's a chemical that means business. You sense its bite quickly — it breaks down with water, gives off nasty fumes like hydrogen chloride, and can burn skin right down to the bone. There’s no reason to treat it lightly, even if some labs make it part of the daily routine.
Personal Experience and the Human Side
I spent a year working in a lab where phosphorus pentachloride came out for syntheses. Nobody grew careless, because it only took a tiny splash or waft to ruin your week. Once, a coworker cracked a stopper loose by mistake. The sour cloud that rose caught him off guard. Only fast moves and good training saved him from a trip to the hospital. He told me afterward, “That gas sticks with you. Your eyes, your lungs, your memory.”
Barrier Protection Matters
Nobody expects street clothes to stand up to phosphorous pentachloride. Thick, chemical-resistant gloves gave our hands real protection. I always kept my sleeves tight, with lab coats snapped up to the neck. Eye shields and sealed safety goggles did their part daily. The oddest thing you get used to is the face shield, but you never question it when working with chemicals that grab skin in seconds.
Good Ventilation Is Not Optional
Most labs rely on fume hoods for handling this kind of stuff. I learned to test the airflow before every session, because even slight sluggishness in the system could turn a small spill into a dangerous exposure. Local exhaust systems draw fumes away from your face. It’s foolish to do without one, even for small batches. The people who skip this step don't last long in chemistry.
Storage and Segregation
Letting phosphorus pentachloride mix with moisture spells disaster. We stored it well away from any sinks or water jackets. The original containers, tightly sealed, with secondary containment trays, never left the dry chemical cabinet. Keeping incompatible chemicals apart — like water, alcohols, or anything that can react violently — is a simple, effective habit that shields everyone from trouble.
Clean-Up and Emergency Practice
No matter how careful you are, spills happen. Preparedness counts for more than bravado — spill kits loaded with absorbents, neutralizers, and PPE save the day. Training on quick evacuation, eye wash, and shower drills built confidence. I checked my routes to exits before every batch, even late at night. That practice felt reassuring, not paranoid.
Staying Informed and Alert
Nobody gets a free pass with chemicals like this. Reading the safety data sheets gave us real knowledge about symptoms and first aid — not just filler paperwork. Knowing the facts lets you recognize exposure and react quickly, because seconds matter when your lungs or eyes are on the line.
Chasing Better Practices
Advanced labs switched to automated systems that keep people hands-off during risky transfers. Double-checking labels, dates, and container integrity turns into a daily habit after you work with phosphorus pentachloride for a while. Experienced techs help teach safe habits — their stories remind us every mistake has real consequences. The best solution is sticking with solid training, honest attention, and a healthy respect for every strong chemical on your shelf.
How should Phosphorus Pentachloride be stored?
The Nature of the Chemical
Phosphorus pentachloride stands out as a strong chlorinating agent. It reacts with water and moisture, giving off hydrogen chloride gas, which stings the eyes, nose, and throat. I’ve seen how a small leak or even humidity in the air can release these fumes, making storage a serious concern. The reminder is, you can’t cut corners with chemicals that draw moisture like a magnet and turn the air toxic.
Container and Conditions Matter
Strong glass containers with airtight seals usually prove effective, but glass doesn’t mix well with sudden temperature changes or careless handling. Thick-walled polyethylene or fluoropolymer bottles offer a safer option for bulk storage. Metal containers—especially those with any moisture—are a disaster waiting to happen.
That sharp, suffocating gas you smell from an open container gives a clue why climate-controlled, dry storage matters. The area should stay at room temperature or cooler, always dry, and free of pockets where water can lurk. Keep the air moving gently, but not so much that you stir up dust or spread fumes in a mishap.
Stability Depends on Isolation
Phosphorus pentachloride picks fights with bases, oxidizers, and organic materials. Keeping it in the same storeroom as amines, alcohols, or even strong cleaning chemicals just multiplies the risk. I once saw a new tech stack cardboard stocks underneath their chemicals—moisture from the floor worked through and caused a leak. Paper, wood, and rags share the same fate if stored close by. Only non-reactive shelving and trays, made out of chemical-resistant plastic, belong anywhere near the shelves where it sits.
Labeling and Handling By the Book
Bold, clear labeling makes a difference during an emergency. Every bottle of phosphorus pentachloride must show hazard warnings and date of receipt. I’ve known chemists who only use color-coded systems and crosscheck logs weekly. Their caution isn’t paranoia; it’s a real safeguard when you can lose sight of when a batch arrived and when it’s expired.
Whenever I pulled a bottle from storage, I checked for clumps or crusts around the cap—a sign that moisture has crept in. The simple practice of wiping down containers before and after use prevents cross-contamination. Keeping gloves, goggles, and a fresh area for handling reduces panic during accidents. I remember how a sudden spill, if handled with calm and the right protocol, left everyone safe; those extra seconds you save by skipping PPE can cost much more.
Emergency Prep and Disposal
Anything that produces choking gas during a spill deserves a spill kit in arm’s reach: soda ash for neutralization, chemical-resistant coveralls, and plenty of ventilation. Local fire codes usually call for special fire extinguishers, not just water, since water triggers a reaction. Training everyone in your workspace, even the folks who never touch the chemical, pays off over time. Their muscle memory during a true emergency means fewer mistakes and less chaos.
For disposal, waiting isn’t a solution. Unused or outdated phosphorus pentachloride should go to hazardous waste experts. Pouring it down the drain or tossing it in the trash hands a problem to your community. Proper disposal helps keep air and water safer for everyone.
Practical Takeaways
Responsible storage means airtight containers, dry and cool rooms, chemical isolation, and vigilant labeling. Protective practices and quick emergency response aren’t overkill—they’re a daily habit. Regulations exist because too many strong chemicals have hurt unsuspecting users. Safety grows out of experience, not shortcuts.
What are the physical and chemical properties of Phosphorus Pentachloride?
A Glimpse at the Substance
Phosphorus pentachloride grabs attention even before anyone digs into its uses or chemistry. In the lab, its yellow-green crystals crumble easily, turning a bit darker if left in humid air. That change comes from simple contact with water vapor. This compound doesn’t just sit quietly on the shelf. I’ve watched it fume and release a sharp, choking odor that makes your eyes water if you get too close without proper protection. The stuff means business, demanding thick gloves and eye shields—there’s no room for carelessness.
Behavior in the Environment
Pentachloride falls apart fast around moisture. Water triggers a chemical reaction that turns it into phosphorus oxychloride and hydrochloric acid gas. Both pose hazards, not just for people but for equipment and the immediate environment. Even a sealed bottle sees slow breakdown, so safe storage matters a lot. I once noticed a slight hiss from a cap not shut tight—that’s all it took for the chemical smell to fill the room.
Reactivity and Industrial Uses
This compound rarely sits on a shelf for long. It helps make pesticides, flame retardants, and chemicals used in making plastics and dyes. Its real strength comes from snatching up water or pulling oxygen from other compounds. In organic chemistry labs, phosphorus pentachloride goes into reactions to switch out oxygen atoms in molecules for chlorine. I’ve seen synthetic chemists choose it for how quickly and directly it acts, though they tread carefully to avoid splashes and uncontrolled reactions.
Health Risks and Safety
Exposure to the vapors brings on coughing fits, eye pain, and even burns on the skin. Once, a forgotten glove hole meant dealing with an itchy red spot for days. Breathing in a small amount leaves a taste like metal in the mouth, plus a sore throat. Even a whiff in the hallway leaves no doubt something dangerous escaped its bottle. Emergency rinses and calcium gluconate cream remain on standby in every workplace where pentachloride enters the equation.
Handling Challenges
People in charge of labs and storage facilities can’t take shortcuts. Glass or fluoropolymer containers hold up best, since regular plastics and some metals break down from contact. Good ventilation matters more than almost anything else; fume hoods and gas detectors keep people safe. Every drum or bottle needs a label warning about violent reactions with water, and anyone in the area relies on routine training to keep sharp.
Reducing Risk
Fine-tuned storage control cuts down on leaks and accidents. Humidity sensors, tight seals, and regular inspections solve most problems before they start. I encourage anyone working with phosphorus pentachloride to learn signs of exposure and keep emergency gear within arm's reach. Respect for this compound stops injuries and lets researchers or workers get through each shift safely.
What happens when Phosphorus Pentachloride reacts with water?
Reaction in Real Time
Most people don’t spend their day thinking about phosphorus pentachloride, but anyone who steps into a lab storing this compound should know what can go wrong when a bit of water shows up. The reaction isn’t a gentle fizz or a slow transformation. Instead, it explodes into a drama that produces clouds of choking fumes. Phosphorus pentachloride, or PCl5, finds water irresistible in all the worst ways. Stir a little moisture into the mix and chlorine gas surges out. It also spits out phosphorus oxychloride, another biting compound. If the reaction keeps going, the final products swap again to yield phosphoric acid and more clouds of hydrochloric acid gas.
I’ve seen up close how careless handling around sinks or benches causes headaches for more than just the chemist mixing the vials. Chlorine’s not just irritating — inhaling the gas attacks the nose, throat, and lungs. The sharp, pungent odor signals danger even before your eyes start watering. Hydrochloric acid mist won’t just sting the skin; over time, it will corrode metalwork and punch holes in air filters. Experienced lab techs don’t leave PCl5 open anywhere humidity lingers. Keeping bottles tightly sealed and handling everything inside a fume hood protects people and equipment alike.
Importance Beyond the Lab
The havoc doesn’t stay confined to the chemistry classroom or research lab. Supplier warehouses and chemical plants stock this compound for making plastics, pharmaceuticals, dyes, and pesticides. Mishandled shipments have triggered local evacuations, turning neighborhoods into temporary no-go zones. In the wrong hands, these fumes can aggravate respiratory conditions and even trigger asthma attacks.
You won’t find this type of reaction making headlines every day, but industrial accidents linked to chlorine gas exposure often trace back to water leaks or poor training. An ounce of prevention, in my experience, does more than any cleanup operation. Staff need practical safety drills, not just a binder collecting dust in the back office. Emergency showers and stockpiles of neutralizing agents like sodium bicarbonate improve the odds when something does go wrong.
More Than a Chemistry Lesson
Digging into facts makes it clear why industries keep a close watch on phosphorus pentachloride. Roughly 800,000 metric tons of chlorine gas get released in chemical accidents worldwide every year, according to the United Nations Environment Programme. The U.S. Occupational Safety and Health Administration (OSHA) doesn’t mince words: exposure must stay below 1 ppm for chlorine gas over an 8-hour shift. Most local guidelines demand even stricter local rules.
In places where education lags, people cleaning up hazardous waste don’t always know what protective gear to grab. Even small spills, if not treated fast, can linger as a public health hazard. Kids playing near an abandoned warehouse or city workers repairing old plumbing run risks if chemicals sneak into the water supply.
Smarter, Safer Handling
It all boils down to respect — for the science behind the danger, and for the steps that keep everyone safe. Labeling bottles, checking lids before storing, inspecting workspaces every shift, and making sure everyone has access to training lowers the odds of a mess. Employers can set up safe disposal protocols and guarantee enough fume hoods run at full draw. No one earns a badge for risking their lungs, so clear checklists and up-to-date emergency plans matter far more than bravado or cost-cutting.
Phosphorus pentachloride shows how a little water in the wrong place turns routine chemistry into a hazard. Trust me, vigilance and respect keep chemistry exciting for the right reasons.
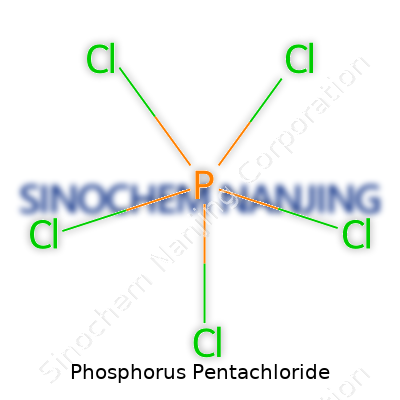
| Names | |
| Preferred IUPAC name | Phosphorane pentachloride |
| Other names |
Phosphorus(V) chloride Phosphoric chloride PCl5 |
| Pronunciation | /fɒsˈfɔːrəs ˌpɛntəˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 10026-13-8 |
| 3D model (JSmol) | `JSmol` string for **Phosphorus Pentachloride (PCl₅)**: ``` PCl5 ``` |
| Beilstein Reference | 358755 |
| ChEBI | CHEBI:24089 |
| ChEMBL | CHEMBL1376 |
| ChemSpider | 23015 |
| DrugBank | DB11378 |
| ECHA InfoCard | 05b1463a-8d0c-4bd0-bc13-6c473871b24b |
| EC Number | 233-060-3 |
| Gmelin Reference | 563 |
| KEGG | C01158 |
| MeSH | D010754 |
| PubChem CID | 24508 |
| RTECS number | SK6280000 |
| UNII | V8BQJ5QQ24 |
| UN number | UN1806 |
| Properties | |
| Chemical formula | PCl5 |
| Molar mass | 208.24 g/mol |
| Appearance | Yellowish-white crystalline solid |
| Odor | Pungent |
| Density | 2.1 g/cm³ |
| Solubility in water | Decomposes |
| log P | -1.7 |
| Vapor pressure | 1 mmHg (15.1°C) |
| Acidity (pKa) | -2 |
| Basicity (pKb) | Strong acid |
| Magnetic susceptibility (χ) | -59.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.645 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –374 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | −437.4 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB46 |
| Hazards | |
| GHS labelling | **GHS02, GHS05, GHS06, Danger, H290, H301, H314, H330, P220, P260, P264, P271, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P310** |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H290, H301, H314, H330 |
| Precautionary statements | P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P311, P321, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-W |
| Autoignition temperature | 160 °C (320 °F) |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LCLo human (inhalation): 1000 mg/m³/30m |
| LD50 (median dose) | 270 mg/kg (rat, oral) |
| NIOSH | SN1875000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Phosphorus Pentachloride: 0.1 ppm (1 mg/m³) |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | IDLH: 5 ppm |
| Related compounds | |
| Related compounds |
Phosphorus trichloride Phosphoryl chloride Phosphorus pentafluoride |

