Phosphonic Acid: Unlocking Chemistry’s Silent Workhorse
Historical Development
Walking through the corridors of chemical history, phosphonic acid doesn’t command headlines like ammonia or phosphoric acid, but its roots run deep. Over a century ago, researchers first pieced together its structure and observed its unique behavior in water. Early chemists, driven by an urge to explore phosphorus-containing molecules, gradually recognized phosphonic acid as more than just an academic curiosity. In the 20th century, as industrial chemistry boomed, the need grew for compounds that could improve water treatment or stabilize metal ions—roles where phosphonic acid found a welcome home. Once synthetic methods became reliable, production scaled up, and phosphonic acid quietly slipped into applications as diverse as agriculture, cleaning formulations, and even pharmaceutical syntheses.
Product Overview
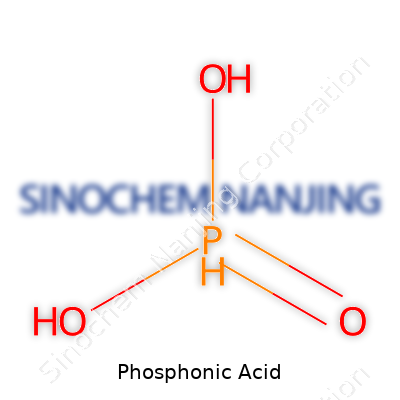
Phosphonic acid sits between two worlds: strong enough to matter in industry, subtle enough to fly under most radars. Its formula, H3PO3, packs three hydrogens and a phosphorus atom surrounded by oxygen. Chemists who spend years behind fume hoods recognize its biting, almost sour aroma, and its strong affinity for water. In labs and factories, it often appears as a clear, syrupy liquid or a white crystalline solid. Companies ship it in containers built to resist corrosion, mindful of what happens if phosphonic acid leaks onto metal surfaces or concrete. Most label drums with hazard warnings, because once it splashes, it bites through skin and stains surfaces. Unlike showier acids, this one earns its respect quietly, not through volatility or splash, but with quiet effectiveness and dependability.
Physical and Chemical Properties
At room temperature, phosphonic acid typically forms deliquescent crystals that soak up water from the air. This property makes storage challenging in any damp or poorly sealed warehouse. Melting occurs around 73°C, lower than many mineral acids, turning it into a dense, colorless liquid. In water, phosphonic acid dissolves quickly, producing a strongly acidic solution. Unlike phosphoric acid’s three ionizable hydrogens, phosphonic acid gives up two protons easily, while the third clings tightly, thanks to the molecule’s unique arrangement. This stubbornness influences every reaction it enters, giving users more control over acidity or reactivity. In terms of chemical stability, it resists standard oxidizers but can decompose at high temperatures, giving off irritating fumes containing phosphorus oxides. Its ability to chelate, or bind, metal ions stands out, proving useful in cleaning agents, water conditioners, and chemical syntheses that need to control metal content.
Technical Specifications & Labeling
Users can’t afford to skip safety standards. Typical commercial phosphonic acid arrives at over 99% purity, but trace water and related phosphorus acids sometimes sneak in if the process isn’t tightly controlled. Labels highlight risks: corrosive to eyes and skin, harmful if swallowed, and environmentally tricky if discharged untreated. International transport relies on standardized labeling, usually following GHS or REACH guidelines. Warehouse teams know storage temperature matters, since even a minor leak can corrode shelves and flooring. For bulk buyers, companies provide certificates of analysis listing heavy metal levels and impurities—confidence that the acid won’t sabotage sensitive chemical processes or damage equipment.
Preparation Method
Most modern factories synthesize phosphonic acid through hydrolysis of phosphorus trichloride in water, a process that sounds simple but rewards precision. As phosphorus trichloride meets water, the reaction generates heat and fumes of hydrochloric acid. Expert handlers moderate temperature and add water in controlled amounts to avoid dangerous pressure spikes. After the reaction, distillation or crystallization purifies the crude acid. Some specialized routes might use oxidation of phosphine, but industry prefers the directness of phosphorus trichloride hydrolysis. Anyone who’s worked at the interface of glassware and corrosive fumes knows the artistry lies in careful control—never dumping reagents together, always watching for runaway reactions and keeping neutralization protocols close at hand.
Chemical Reactions and Modifications
Phosphonic acid’s chemical life revolves around its active hydroxyl and P-H bonds. Its family tree includes esters and salts, many of which show up in specialty cleaning products or water treatment chemicals. Reacting phosphonic acid with alcohols under acidic conditions gives phosphonate esters—molecules that find their way into herbicides or additives that control metal corrosion. The acid itself reacts with metals, forming stable phosphonate-metal complexes, prized for their ability to control hardness in water and prevent scale buildup in boilers. These complexes have carved a niche in places where municipal and industrial water systems intersect. In pure chemistry labs, phosphonic acid doubles as a reducing agent, donating electrons to speed up key synthetic steps. Time after time, chemists exploit its versatility, modifying the basic acid with organic or inorganic groups to tailor its reactivity and keep up with evolving demands in agriculture and manufacturing.
Synonyms and Product Names
Few chemicals walk around wearing as many hats as phosphonic acid. Literature and trade catalogs sometimes list it as phosphorous acid, but that name often refers to its technical grade or certain hydrated forms. Walk through the shelves, and synonyms like dihydroxyphosphine oxide or H3PO3 rear their heads. Across the globe, regulatory agencies and suppliers rely on CAS 10294-56-1 for clarity, since so many similar-sounding acids crowd the market. In agriculture, labeling may highlight its role as a precursor for glyphosate or other selective agents, giving it a subtle presence in debates about food security and herbicide resistance. The pharmaceutical sector points to analogs and derivatives, where names pile up almost as fast as the applications.
Safety and Operational Standards
Anyone who's ever spent time in a chemical warehouse or manufacturing line understands why strict safety standards protect workers and the environment from phosphonic acid’s hazards. Contact burns, respiratory irritation, and the risk of eye damage mean operators favor gloves, eye protection, and well-ventilated spaces. Spills must get cleaned immediately—no time for procrastination, since the acid seeps into concrete and rusts steel. Disposal follows hazardous waste rules: neutralize first, dilute second, always document. Regulatory boards demand up-to-date training for handlers, periodic audits of storage and labeling, and quick access to neutralizing agents. The safest facilities build emergency response into their culture, rehearsing what to do if a drum leaks or a pump fails mid-transfer. Environmental risk looms if waste streams reach rivers or soil untreated, an issue plant managers take to heart knowing fines and reputational damage follow any negligence.
Application Area
Few chemicals move so easily between sectors as phosphonic acid. Water treatment specialists know it for its ability to lock down calcium and magnesium, preventing tough mineral scale in pipes and boilers. Cleaning product manufacturers blend it with surfactants and builders to lift stubborn stains or neutralize hard water’s effects in laundry cycles. In agriculture, phosphonic acid or its derivatives go into fertilizer blends, sometimes finding itself in the middle of arguments about crop safety and regulatory oversight. Some industrial chemists value it for catalysis or as a mild reducing agent, streamlining tricky synthetic steps. Sectors like oil recovery and mining lean on phosphonic acid to maintain process fluids and keep metal ions in check. For the pharmaceutical industry, research chemists push its reactivity to build complex molecules, especially where phosphorus must join or modify organic frameworks. Each time, the acid adapts, not by hogging attention, but by quietly solving specific chemical challenges.
Research and Development
Scientists keep pushing the boundaries of what phosphonic acid can do. Researchers test its reactivity, aiming for more efficient crop protection agents, cleaner water treatments, or novel catalysts. Academic teams examine its molecular flexibility, searching for derivatives with selective biological activity or less environmental persistence. Some labs develop new synthetic routes that reduce waste, cut down emissions, or simplify purification, tying sustainability into future supply chains. The constant search for greener, safer chemicals puts phosphonic acid under the microscope, both literally and figuratively, as developers try to tweak its structure for specialized uses. Collaborations between universities and industry look promising, with patents climbing for new phosphonate-based drugs or antimicrobial agents. With each new use case, data collection and peer review stay central, both to safeguard people and to support evidence-based expansion into new fields.
Toxicity Research
No commentary on phosphonic acid feels complete without talking about the shadow it casts in terms of toxicity. Acute exposure brings the typical risks of a strong acid: skin blisters, eye irritation, and respiratory struggles if dust gets inhaled. Long-term studies show that, compared to many industrial chemicals, phosphonic acid features moderate toxicity; the real danger lies in concentration and chronic environmental exposure. Early animal studies suggest slow metabolism and limited accumulation in higher organisms, but the acid’s derivatives paint a more complicated picture, especially in agricultural run-off scenarios. Regulators set strict occupational limits and demand clear documentation of releases, while researchers continue feeding fresh toxicity data into chemical hazard databases. Societal pressure for cleaner industry means companies and scientists must keep sharing results—no hiding behind industry jargon or incomplete studies.
Future Prospects
Looking forward, phosphonic acid’s story remains unwritten on several fronts. Demand for sustainable agriculture, efficient water purification, and advanced pharmaceuticals could lift its profile. Researchers searching for phosphorus-based alternatives to harsher chemicals lean into its versatility and predictable reactivity. As global regulations tighten around chemical storage, discharge, and manufacturing waste, producers must invest in cleaner synthesis and better containment. New applications in battery technology or nanomedicine may emerge as chemists probe deeper into structure-activity relationships. Everyone in the supply chain, from lab techs to policymakers, needs to take a hard look at both benefits and risks, pushing for transparency and rapid adoption of safer alternatives where risks show up. The future promises fresh debates and breakthroughs, always anchored by the lessons learned from this quietly essential molecule.
What is phosphonic acid used for?
A Look at Where Phosphonic Acid Shows Up
Phosphonic acid pops up in places you might not expect. For years, farmers have counted on it as part of their routine. It helps plants fight off diseases, especially those caused by fungi. Grape growers spray it to hold back downy mildew, which without treatment destroys crops and hurts food supply. Crops like potatoes and cucumbers also benefit, with yields staying healthy. Its ability to support plants without leaving lasting residues in the soil means it works well for those wanting to keep land productive over the long haul.
Keeping Water Clean
Water treatment companies use phosphonic acid to stop pipes from clogging with scale. Hard water mixes minerals that settle inside pipes and equipment, cutting down efficiency and life span. Adding small amounts of phosphonic acid helps prevent these buildups; this makes it easier to maintain clean systems for both homes and factories. In personal experience, fewer plumbing repairs and cleaner water heaters add up quickly both in convenience and savings. You cannot overstate the frustration of dealing with scale in household appliances, either.
Help for Industry
Factories and manufacturing plants often work with metals that need to resist rust. Phosphonic acid helps here too. Metal surfaces washed with it take on a thin layer that blocks corrosion, adding years to their service life. I’ve seen how much money companies spend repairing rusted equipment or dealing with failures that interrupt work. A simple treatment can mean fewer breakdowns and lower costs in the long run.
Household Products
Many cleaning items in cupboards contain phosphonic acid—or ingredients related to it. Dishwashing detergents, stain removers, and limescale treatments rely on its ability to bind minerals and lift away stubborn residue. This chemical isn’t just a behind-the-scenes helper for farms and factories. It makes everyday chores at home less of a hassle, from scrubbing sinks to removing spots from glasses. Safe and smart use on the household level frees up extra time and cuts frustration, especially with hard water in cities.
Environmental Considerations
More products containing phosphonic acid show up each year, but this brings up questions about safety and waste. If it runs off fields or gets flushed from homes and factories, it reaches rivers and lakes, potentially affecting plants and animals. Some countries keep a close eye on its use, setting clear limits and testing water regularly. Watching out for environmental health keeps food, water, and communities safe.
Looking Ahead: Smarter Choices
Solutions involve smarter application. Precision spraying, better wastewater treatment, and regular soil tests reduce risks while supporting the benefits for crops and infrastructure. Farmers and engineers can share tips and data for safer, more efficient use. Governments can offer better training and rules shaped by ongoing research. Small changes, like switching to more accurate dosing or improved filtration, ripple out to help everyone.
Learning about phosphonic acid means recognizing its everyday presence and the role it plays in protecting food, water, and materials. Paying attention to how we use it and deal with the leftovers matters most if we want to keep taking advantage of its strengths without creating problems down the line.
Is phosphonic acid safe to handle?
Understanding the Substance
Phosphonic acid doesn’t grab headlines, but in industries like agriculture, cleaning, and water treatment, workers use it all the time. Chemically, it’s related to phosphoric acid, sitting in the same family as compounds that play big roles making fertilizers, cleaners, and rust inhibitors. Some people hear “acid” and think about bubbling test tubes or lab accidents in movies. In real life, it’s not so dramatic—until something slips up.
Risks You Can’t Ignore
Forget the stereotypes—phosphonic acid is not something anybody wants on their skin (or worse, in their eyes). The stuff corrodes tissues and causes burns fast. Reports collected by the National Institute for Occupational Safety and Health show even short exposures may trigger redness, swelling, and long-lasting irritation. Small spills on unprotected skin can do real damage. Inhaling vapors or mists also causes coughing, throat pain, or lung distress. The Centers for Disease Control and Prevention singles out strong acids in its chemical hazard database, and phosphonic acid lands on that list.
Why Handling Safely Matters
Coming from years working around chemical warehouses, I saw the difference between respecting these products and cutting corners. My very first week unloading drums, an older worker cracked a lid open too fast and splashed dilute acid across his gloves. He was lucky, but those gloves melted through at the fingertips. Gloves, face shields, and aprons hold up for a few moments against a spill, but soaking through happens faster than most folks expect. One minute of carelessness leads to weeks off work and sometimes permanent scars. These accidents become stories told in staff rooms, reminders to the new crew just how fast something can go wrong.
Safe Handling: Steps to Make Routine
Personal protective equipment (PPE) is not a suggestion from a dusty safety poster but a barrier that saves skin and eyesight. Splash goggles, chemical-resistant gloves, aprons, and face shields remain non-negotiable for anyone pouring, mixing, or moving phosphonic acid. Proper ventilation helps catch stray fumes before they build up. Emergency eye-wash stations and safety showers must always be close to where work takes place, not just installed for compliance points.
Labeling matters just as much. Workers sometimes grab the wrong container when labels fade or peel off, especially in a busy shift. That mistake could lead to splashing something stronger than they expected. The Occupational Safety and Health Administration points out that clear labeling and up-to-date safety data sheets stay essential for anyone managing acids.
A Culture of Care—Not Just Compliance
Safety routines work best when the people using chemicals speak up about odd smells or cracked hoses before accidents happen. Trainings that give real stories—not just rules—help new workers see the point of all those gloves and goggles. Managers who spend time on the line lead by example, swapping out worn gear or reporting faulty lids themselves.
Investing in good training and equipment saves companies money long term. Lawsuits, medical bills, and replacement costs stack up much higher than ordering new gloves. Fines from regulators remind even the friendliest workplaces about the real cost of looking the other way. Using phosphonic acid safely boils down to respect—for the workers, the chemical, and everyone who depends on a job done right.
What is the chemical formula of phosphonic acid?
The Chemical Profile
Phosphonic acid carries the formula H3PO3. This isn’t just another string of letters and numbers in a chemistry textbook. Phosphorus sits at the core of many essential life processes, and in the form of phosphonic acid, it does much more than most folks realize. If you’ve ever wondered what holds together some of the pesticides that keep crops healthy or why certain water treatment solutions remain effective, you’re looking at a compound that often flies under the radar: H3PO3.
Everyday Impact and Application
A fair share of my understanding grew after working alongside a water treatment technician. He pointed out that phosphonic acid derivatives combat mineral scale and corrosion in water systems. He explained the frustration of dealing with calcium and magnesium deposits—those stubborn white streaks inside kettles and pipes. The use of chemicals based on H3PO3 helped the system run longer, with fewer breakdowns. If you’ve ever benefited from clear tap water or shiny dishware coming out of a dishwasher, you have these types of compounds to thank.
Farmers also use phosphonate-based pesticides to protect plants from disease. Research by the Food and Agriculture Organization highlights that crop yields suffer when downy mildew and other diseases attack, which directly threatens food supply. By building protection into their crops, farmers keep markets stocked. It’s important for households and economies relying on steady food prices and availability.
Safety and Environmental Concerns
People often overlook the risks tied to phosphorus compounds. Overuse in agriculture can disrupt natural water systems. Excess phosphorus in lakes and rivers triggers algal blooms, choking out fish and threatening drinking water supplies. A 2022 study in Environmental Science & Technology traced these links between phosphorus runoff and ecosystem collapse. Watching a beloved fishing lake turn green and smell rotten drives home how close to home these issues can hit.
Manufacturing and handling phosphonic acid call for clear safety measures. Regulatory bodies such as the Occupational Safety and Health Administration (OSHA) insist on using gloves and eye protection to avoid irritation. Responsible industry practices, including strict labeling and storage, help workers and the environment. My neighbor once shared a story about a chemical spill at a small plant; the emergency response team knew their stuff, but cleanup still lasted days and cost thousands. These stories remind us why strong protocols and quick responses matter.
Pathways to Smarter Use
Sustainable agriculture and industry depend on better phosphorus management. Adopting precision farming practices cuts down non-essential phosphorus use. Crops get what they need without dams and rivers carrying away the excess. Water treatment companies can design closed-loop systems, recycling phosphonates before they cause problems downstream.
Education also holds big potential. Homeowners and small business operators need plain language guides on safe handling and disposal. If more people pay attention to what flows down their drains, it can dramatically shift the burden off municipal systems and natural ecosystems. Public investment in research pays off, too—new forms of phosphorus recovery from waste can keep the cycle going with fewer losses.
Understanding H3PO3 means facing both its usefulness and its risks—something that sticks with you once you’ve seen both thriving crops and clogged rivers up close.
How should phosphonic acid be stored?
A Practical Take on Safe Handling
Storing phosphonic acid takes more than dropping it in a random corner of the warehouse. Its nature as a corrosive chemical demands attention. Many have experienced the sting of working around materials that, if handled the wrong way, turn a small oversight into a big problem. Phosphonic acid, clear and almost unassuming, doesn’t give visual warnings. It reacts with skin, eyes, and everyday surfaces, breaking down common materials over time.
Physical Storage: Location Matters
Years working around chemical storage gave me a sense for secure spaces. The most reliable option for phosphonic acid always starts with a cool, dry room where sunlight can’t peek in. Temperatures need to remain steady: avoid any place prone to freezing or getting very hot. Insulation against temperature swings pays off. High heat or freezing temperatures can change the properties of the acid, or worse, damage the container and cause leaks.
At one warehouse I worked in, a poorly insulated shed left bottles too close to an outer wall. Humidity seeped in, lids corroded, and acid dripped onto metal racks. That earned a lasting reminder—secure, sealed containers should line the shelves, preferably of high-density polyethylene or glass. Metal storage falls short here, as the acid gradually eats away at many metals. Stainless steel resists corrosion better, but plastic containers usually beat it for practical, long-haul storage.
Labeling and Segregation
Clear labeling supports safety. A sticker with bold writing and an easy-to-see hazard symbol tells everyone at a glance what sits inside. Don’t trust memory or half-torn masking tape. Toxic substances lined up together cause headaches for everyone, so keep phosphonic acid on a shelf away from bases, cyanides, and oxidizing agents. Chemical mixing triggers reactions, and anyone who’s seen a small spill grow into a frantic evacuation will set down firm rules about separation.
Emergency Tools Close at Hand
Anyone dealing with phosphonic acid, whether at a farm, manufacturing plant, or lab, learns the value of easy-access safety stations. This means eyewash units, drench showers, and, crucially, well-marked exits. I once watched a colleague splash acid on his forearm; quick steps to an eyewash station made all the difference. A good routine involves checking these stations and keeping spill kits close. These aren’t just compliance measures. In an emergency, seconds matter.
Regular Checks and Training
Complacency leads to trouble. Store managers and warehouse staff should check containers every week. Look for leaks, cracks, rust stains, or anything unusual. Swapping out tired labels and replacing damaged bottles is part of the job. Training matters too—every team member needs to know handling, first aid, and emergency response steps. I’ve watched new hires freeze when faced with an unfamiliar hazard symbol, so hands-on demonstrations always beat paper checklists.
Why Storage Details Build Trust
Beyond physical safety, proper storage builds trust between employers and their teams. People work more confidently when they know risks are managed. Customers, regulators, and communities recognize good chemical management as a sign of a responsible operation. Public records back this up. The Environmental Protection Agency regularly lists poor storage as a top reason for safety citations and costly cleanups. Simple steps—better containers, and cleaner, dry shelves—help prevent these problems from landing on the nightly news.
What are the main applications of phosphonic acid in industry?
Water Treatment and Scaling Prevention
Factories, power plants, and municipal water facilities run into a constant headache: scale. Lime, rust, and other gunk build up in pipes and boilers, choking off flow and wasting energy. Phosphonic acid steps in as a sort of bodyguard, binding unwanted minerals like calcium so they stay dissolved. This stops scale before it even gets started. I've heard engineers praise the way a pinch of phosphonic acid in cooling towers keeps things running smoothly for months instead of weeks. This matters for everyday life too. Nobody likes boil-water advisories or higher energy bills caused by clogged infrastructure. Reducing downtime means cities and companies can serve more people without constantly fixing or replacing pipes.
Detergents and Cleaning Products
Many people don't realize what goes on under the sink or behind the laundry room walls. Those sparkling tiles and spotless shirts get that way because of the chemistry we don't see. Phosphonic acid delivers strong cleaning power, but it isn’t harsh on surfaces. In dishwashing detergents and laundry powders, it softens hard water, stops stains from resettling, and even helps keep glassware clear. For folks dealing with stubborn grime — think restaurants or hospitals — cleaners with a backbone of phosphonic acid handle tough jobs without excessive scrubbing or the need for aggressive, toxic solvents.
Textile Processing
Anyone who’s worked in textiles knows fabric quality depends on more than the cloth. Colors fade, fibers roughen, and production lines can jam up if the water isn’t just right. Phosphonic acid acts as a stabilizer in dye baths, keeping colors vibrant and helping the dye spread evenly on every strand. It also protects fibers during scouring and bleaching, so finished products feel softer and last longer. Garment factories chasing higher quality have come to rely on this compound, especially as consumers reject “fast fashion” that falls apart after a handful of washes.
Crop Protection and Fertilization
Modern farming doesn’t look much like the storybook fields of decades ago. Fields today must produce more while using fewer resources. Phosphonic acid supports plant health in two big ways. It helps crops absorb nutrients from the soil, acting almost like a booster shot for roots. It also lends a hand fending off harmful fungi and diseases, making it valuable for orchard owners, vineyard managers, and greenhouse operators. This approach reduces reliance on harsh pesticides, giving both conventional and organic growers a tool for healthier harvests with less chemical baggage.
Corrosion Inhibition in Industry
Steel tanks, pipelines, and machinery face constant assault from moisture, oxygen, and chemicals. Rust eats billions of dollars from industrial budgets every year. I’ve seen engineers protect investments with coatings and additives loaded with phosphonic acid. These molecules form a protective layer on metals, slowing corrosion and extending the lifespan of vital parts. In sectors like oil and gas or automotive manufacturing, this adds years of use, keeps maintenance costs low, and helps prevent dangerous failures.
Conclusion
Phosphonic acid isn’t the flashiest ingredient in any formula, but the results speak for themselves across dozens of industries. From softer clothes and cleaner water to healthier harvests and tougher machinery, it’s doing important work in places most of us never see. That’s worth noticing, even if you don’t spot its name on the label.
| Names | |
| Preferred IUPAC name | Phosphonic acid |
| Other names |
Phosphorous acid Phosphonic acid Orthophosphorous acid Dihydroxyphosphine oxide |
| Pronunciation | /fɒsˈfɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 13598-36-2 |
| Beilstein Reference | 1852536 |
| ChEBI | CHEBI:44970 |
| ChEMBL | CHEMBL504 |
| ChemSpider | 58312 |
| DrugBank | DB09460 |
| ECHA InfoCard | 13a4bf88-07df-4090-9256-c8c7d7c35863 |
| EC Number | 015-157-00-0 |
| Gmelin Reference | 63525 |
| KEGG | C01104 |
| MeSH | D010766 |
| PubChem CID | 24457 |
| RTECS number | TB6300000 |
| UNII | 9Q3KT2BG4V |
| UN number | UN1805 |
| CompTox Dashboard (EPA) | DTXSID8020129 |
| Properties | |
| Chemical formula | H₃PO₃ |
| Molar mass | 81.963 g/mol |
| Appearance | Colorless crystals or liquid |
| Odor | Odorless |
| Density | 1.65 g/cm³ |
| Solubility in water | soluble |
| log P | -2.0 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 1.7, 7.2 |
| Basicity (pKb) | 1.3 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.410 |
| Viscosity | Viscous liquid |
| Dipole moment | 1.45 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 77.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1281.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1277.2 kJ/mol |
| Pharmacology | |
| ATC code | A01AD11 |
| Hazards | |
| Main hazards | Corrosive, causes severe skin burns and eye damage, harmful if swallowed or inhaled. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H290, H314 |
| Precautionary statements | P264, P280, P301+P312, P305+P351+P338, P337+P313, P330, P501 |
| NFPA 704 (fire diamond) | Health: 3, Flammability: 0, Reactivity: 2, Special: - |
| Autoignition temperature | 280 °C (536 °F; 553 K) |
| Lethal dose or concentration | LD₅₀ (Oral, Rat): 1530 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1,890 mg/kg (rat, oral) |
| NIOSH | WA6475000 |
| PEL (Permissible) | PEL: 1 mg/m³ |
| REL (Recommended) | ≥99% |
| IDLH (Immediate danger) | IDLH: 1000 mg/m3 |
| Related compounds | |
| Related compounds |
Phosphoric acid Phosphorous acid Hypophosphorous acid Phosphonate Phosphate |

