The Story of Thiophosgene: More Than Just a Toxic Intermediate
Historical Development of Thiophosgene
Sometimes, the story of a chemical unfolds as a series of discoveries shaped by need, urgency, and curiosity. Thiophosgene, with its sharp, unforgettable odor, arrived on the scene in the mid-1800s during a period of furious invention in European chemical laboratories. Chemists spotted its similarity to phosgene but noted right away that swapping oxygen for sulfur gave the resulting molecule unfamiliar and often troubling qualities. Its adoption leaned more toward the specialized corners of chemical synthesis than into broad industrial processes, largely because of its toxicity. By the time large-scale organic chemistry started to transform agriculture and pharmaceuticals in the 20th century, thiophosgene had found a home in research labs, helping to unlock different molecular frameworks and sparking creativity among scientists looking for ways to tweak existing reaction strategies.
Product Overview
Thiophosgene works as a tool for building complexity in organic molecules. Unlike bulk chemicals, you won’t see it rolling out of factories by the tanker-load. Its value lies in its effectiveness for bringing about difficult transformations—acting as a reagent for chlorination, for cyclization steps in making dyes, and for introducing the C=S functional group. You’ll spot it most often in small bottles, labeled with stern warnings and handled with serious caution. Researchers lean on it to bridge gaps in pathways that other chemicals can’t fill, especially in fields like pharmaceuticals, where creativity with molecular frameworks gives rise to new treatment ideas. Even graduate students who cringe at its stink respect how much ground it can cover in a synthetic sequence.
Physical and Chemical Properties
Every chemist remembers their first whiff of thiophosgene. Its deep red color makes it stand out, almost like a warning. The compound boils below 80°C and freezes around -12°C, so it doesn’t take much heat to have it floating in the air as a gas. Its molecule packs a punch, with a carbon double-bonded to sulfur and flanked by two chlorine atoms. This combination makes thiophosgene reactive—far more so than many everyday chemicals. In practice, anyone working with it keeps a close eye on containment and ventilation. Its tendency to decompose in the presence of water or basic substances gives it a shelf life that depends heavily on good storage and minimal exposure to moist air.
Technical Specifications and Labeling
Every bottle of thiophosgene carries hazard pictograms that stop you in your tracks before unscrewing the cap. Labels follow strict guidance from international safety standards. Chemists expect to see UN numbers and hazard statements like “Toxic by inhalation, in contact with skin and if swallowed.” Concentrations greater than 99% remain standard for lab and research uses to avoid unexpected results in delicate syntheses. Packaging protects not just the user but the environment, since spills or leaks can spark hazardous responses. It never turns up on a shipping receipt without thorough documentation and a clear warning to treat it as a high-risk article.
Preparation Method
Nobody prepares thiophosgene on a whim. Most supply comes from controlled chlorination of carbon disulfide. Industrial routes use chlorine gas passed over a cooled solution of carbon disulfide, cranking up yields but demanding extreme care with both the toxic starting material and the equally hazardous product. Purification includes low-temperature distillation with strict exclusion of air and moisture. Small-scale syntheses sometimes use organic solvents to keep things manageable, while automated set-ups and fume hoods act as non-negotiable barriers against accidental exposure. Every step shouts out for careful planning, from starting materials to safe venting of gases.
Chemical Reactions and Modifications
Thiophosgene thrives on reactivity. Its chlorines swap out with a wide variety of nucleophiles—amines, alcohols, or organometallic reagents—carving pathways to isothiocyanates, dithiocarbamates, and other sulfur-rich molecules. Organic chemists often rely on it to activate particular positions in molecules, creating bonds that other reagents struggle to forge. In dye chemistry, it gives rise to thioindigo derivatives. People in peptide synthesis sometimes reach for it when they want to stitch together particular backbone connections. The chemistry expands well beyond its main uses thanks to its willingness to hand over its chlorines and double bonds in reactions that drive innovation.
Synonyms and Product Names
To avoid confusion, chemists and suppliers catalog this chemical under several trusted synonyms such as trichloromethanethione, thiocarbonyl dichloride, and CTC. Each variation circles back to the same formula, helping researchers find reliable data across publications and international regulations. The variety of names shows up in databases, scientific literature, and even on import permits, so clear communication about exactly which chemical is at play keeps projects and regulatory checks moving forward.
Safety and Operational Standards
Chemists never get casual around thiophosgene. Full-face respirators, double gloves, acid-resistant aprons, and ventilated fume cabinets represent the gold standard for handling. Standards require written protocols and training before a bottle gets opened. Spills demand swift action with specialized sorbents and full evacuation of the area in larger labs. Storage stays simple and strict: away from heat, light, and especially water. Emergency procedures get drilled into memory because even a small exposure can end a workday or worse. Disposal goes straight to high-temperature incineration rather than standard chemical waste bins. Respecting these barriers preserves not just lives, but careers.
Application Area
Thiophosgene remains a favorite tool in advanced organic synthesis, especially for university labs and specialty chemical makers. Its role gets most pronounced where other reagents throw up roadblocks—especially in building complex molecular frameworks. Dye manufacturers use it to introduce sulfur into otherwise oxygen-rich colorants, shifting tones and stabilities. Drug discovery teams count on it to craft unique scaffolds that stand out from traditional carbon-based drugs, aiming for new biological activities. Teaching labs avoid it for undergraduates but keep it in the back for advanced students working under supervision, acknowledging both its power and its risks.
Research and Development
Strong interest in thiophosgene’s chemistry drives R&D efforts to find less hazardous ways to harness its power. Teams look for milder reaction conditions, safer storage, and strategies for scavenging toxic byproducts. There’s renewed attention on computer modeling of its reactivity to build more predictable, less wasteful processes. Methods like flow chemistry, where smaller amounts of reagent move through enclosed tubing, aim to cut direct handling to near zero. Research into green chemistry tools wants to move reactivity away from traditional hazardous chemicals, seeking alternatives or modifications that rival thiophosgene’s unique gifts without risking health and safety.
Toxicity Research
Toxicologists class thiophosgene as a serious respiratory risk thanks to its volatility and reactivity with proteins in the lungs. Animal studies show low lethal limits, and accidental inhalation can cause sudden, irreversible damage to mucous membranes. Reports out of academic labs tell of cautious researchers who still land in the hospital after minor leaks or mishaps. Regulations keep shifting as new data emerges, driving up requirements for engineering controls and exposure monitoring. This legacy of accidents, combined with clear animal data, has shaped not just how chemists view thiophosgene but how regulators set limits for its use and disposal.
Future Prospects
Where does thiophosgene go from here? New synthesis strategies try to channel its reactivity into safer, more contained technologies like microreactors and in situ generation. The push to “green” up chemistry puts pressure on labs and manufacturers to find replacements that don’t compromise creativity or access to key molecular targets. Safety investments have started to pay off, with more automation making direct exposure rare even in research settings. The combination of its irreplaceable reactions and tough handling means thiophosgene will stick around for specialized applications. Breakthroughs in safer alternatives will shape the future, but respect for its power remains the best safeguard, keeping progress both bold and aware of the lessons hard-won in labs over the last century.
What is thiophosgene used for?
The Character of Thiophosgene
Thiophosgene, with its sharp smell and warnings on every safety sheet, draws attention for more than just its dangers. This deep red liquid steps up as a key ingredient in the hands of chemical researchers. It isn’t found in drugstore aisles or on everyday shelves; instead, it waits in specialty labs, sealed up until someone needs its unique abilities. Having seen chemists brace themselves before uncapping a bottle, it’s clear the fuss isn’t just about hazard—thiophosgene opens doors in synthesis that often stay locked with gentler chemicals.
Building Blocks for Medicines and More
Most people never touch or see thiophosgene, yet it quietly shapes parts of the world, especially in drug development. It’s vital for making isothiocyanates, a group crucial for testing new medicines. Recent reports from publications like Journal of Medicinal Chemistry mention isothiocyanates in anti-cancer projects, and many of those reactions trace back to thiophosgene. In my graduate lab days, work with complex sulfur compounds usually meant hunting down a small vial of thiophosgene. Without it, some steps just couldn’t be finished, halting the entire molecule-building process.
Synthetic chemists rely on it to link carbon and sulfur atoms in places where simpler chemicals fumble. That’s useful for not only drug development but also for making dyes and specialty polymers that serve electronics or create colorfast clothes. Give a chemist a tough sulfur-related target, and thiophosgene lands on the workbench.
Research’s Double-Edged Sword
Risks jump out when talking about thiophosgene. Old case reports tell of gas leaks and accidental exposures, which hammered home just how cautious labs must act. Its status as a controlled substance in many countries isn’t an overreaction. Chemists wear full-face respirators and run reactions in snug glove boxes for a reason. The toxicity creates a clear boundary: only those with solid training and proper equipment should approach it. The harsh safety rules add costs and long preparation times, making it unpopular outside of highly focused research settings.
Even so, the alternatives can fall short. Green chemistry pushes for milder routes, but often those solutions don’t match thiophosgene’s power or selective touch. Efforts to find replacements keep going but have only chipped at its dominance in certain transformations. It’s not stubbornness—just the challenge of chemistry’s complexity.
How to Move Forward Safely
Part of learning chemistry means respecting chemicals like thiophosgene and keeping updated with improvements in safety. Easy-to-spot labels, better fume hoods, and digital monitoring have lowered some of the risks. Companies now invest in automated setups that seal away the most dangerous steps, protecting both the product and the people making it. Labs that take shortcuts with thiophosgene face heavy penalties, but most recognize that simple rule—keep people safe, or don’t run the reaction.
The search for gentler alternatives still deserves funding and attention. Some research groups have started designing “masked” forms of thiophosgene, releasing its reactivity only at the key moment. It’s part of a bigger goal to retain effectiveness without as much hazard. Still, as long as chemistry asks for what only thiophosgene can provide, it will stay relevant, as a careful partner in the background of new discoveries.
What are the safety precautions when handling thiophosgene?
Why Thiophosgene Commands Respect
Thiophosgene does something many chemicals don’t: it lays all the risks out on the table. This compound brings a double threat, acting both as a toxic gas and a potential chlorinating agent. Most chemists keep their hands off it unless no alternatives fit the reaction. My own work in the lab drove this point home. The pungent smell cuts through even a busy lab, and stories of symptoms after minor spills are common between chemists. The reality—skin blisters, throat burning, and delayed lung damage—makes safety gear and smart handling crucial, not optional.
The Gear That Makes the Difference
A basic lab coat or thin latex glove won’t stand up to thiophosgene. Its fumes find their way through gaps and even small cracks in protective gear. Thick nitrile gloves or, better yet, butyl rubber gloves keep hands safe. Face shields matter when you think about the potential for even a drop to splash. Reliable goggles go on before the bottle comes out of the fridge. The need for proper fit and no shortcuts with PPE isn’t just a rule—it’s experience talking after seeing what happens to those who rush.
Working Inside a Fume Hood
Every bottle of thiophosgene should stay under a working fume hood. Any attempt to work outside the hood courts disaster. The hazard isn’t only inhaling the vapor. The stuff can turn a minor leak into a shutdown situation quick, driving everyone out. I’ve watched coworkers run evacuation drills because of a broken flask. A working sash and good airflow are the only barriers that really make sense.
Storage is No Afterthought
Thiophosgene belongs away from acids and bases, never in direct sunlight, with the bottle sealed tight. Keeping it cold—below room temperature—reduces the risk of fumes building up. In labs I’ve worked in, the rule is clear: only buy what you can use soon. Old bottles become even more dangerous, and disposal becomes a headache. Chemical refrigerators labeled for such reagents keep accidents rare.
Proper Planning and Training Save Lives
Most labs have at least one training session a year because relying on memory under stress doesn’t work. Those of us who’ve handled spills know the drill: emergency shower, eye wash, and a run to the fresh air. Everyone needs clear instructions on what to do and where to go if something spills. Emergency numbers should be close, and safety data sheets for thiophosgene must stay within reach.
Disposal: No Shortcuts Here
Pouring leftovers down the drain isn’t just illegal—it threatens the whole building. Labs arrange for chemical waste pick-up, labeling every container and never mixing thiophosgene with other waste. If accidents do happen, neutralizing residue safely using sodium bicarbonate or a commercial neutralizer is non-negotiable.
Real Solutions Start with a Safety Culture
Accountability comes from every chemist taking these threats seriously, helping catch mistakes before they multiply. This means double-checks, slowing down, and asking for help. It took years for me to build these habits, but no shortcut replaces knowing that a little more time can keep everyone safe.
What is the chemical formula and structure of thiophosgene?
Understanding Thiophosgene: A Simple but Potent Compound
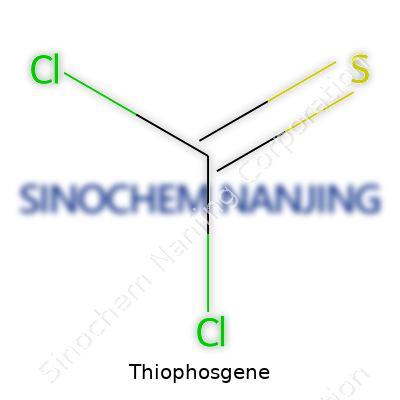
Thiophosgene, known in science for playing a tricky game in labs, carries the chemical formula CSCl2. Those six symbols matter. Each letter stands for something that deserves respect—carbon, sulfur, and chlorine coming together. To get why thiophosgene is important, you don’t need a lab coat, just a willingness to see that solutions in chemistry often start with the basics.
Getting Closer to Its Structure
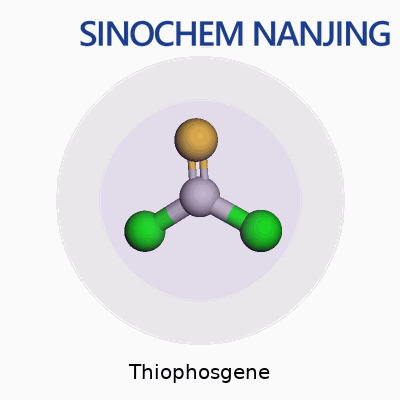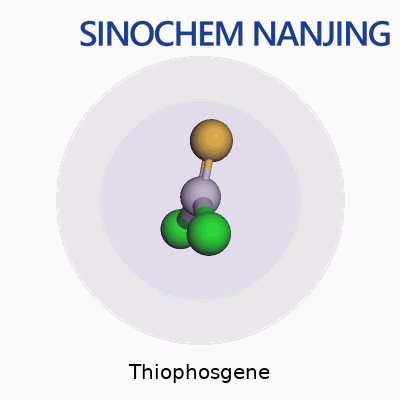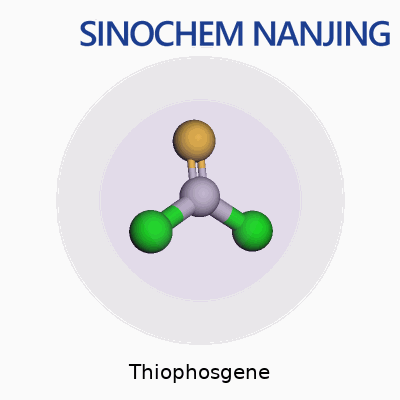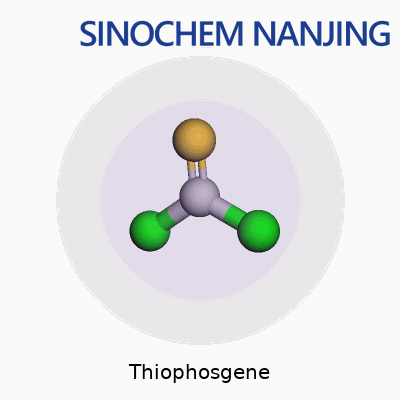
Thiophosgene doesn’t hide its structure: just a carbon in the center, double-bonded to a sulfur atom, and linked to two chlorine atoms. If you ever drew a stick figure as a kid—one head in the middle and arms outstretched on both sides—picture this layout with chemical bonds. The carbon puts one hand on sulfur and the other two on separate chlorines. It’s not fancy, but this arrangement gives the molecule a lot of personality.
This structure isn’t just chemistry trivia. The linear form, with sulfur tied tightly to carbon, gives thiophosgene its bite. Chemists will tell you the strength of these bonds brings both good uses and real hazards. Thiophosgene can swap its chlorine atoms out for something else during a reaction—making it a team player in creating all sorts of complex organic molecules. A clear structure, plain to see, provides opportunities and risks alike for those who work with it.
Why Thiophosgene Matters Beyond Its Formula
Many folks, including myself, bumped into thiophosgene in textbooks before ever laying actual eyes on it. The name tends to ring a bell for organic chemists. Its formula, CSCl2, echoes phosgene (COCl2), which history knows all too well from early warfare and as a tool in plastics production. Swap one oxygen out for sulfur—suddenly, you’ve got a different story in your hands.
Thiophosgene's reactivity made it a backbone in synthetic chemistry. Want to slip a sulfur atom into a molecule? Need a convenient way to forge isothiocyanates for drug research? Here’s your reagent. That utility puts it on the shelves of research labs worldwide, though always behind locked doors and under fume hoods.
Moving away from academic uses for a second, thiophosgene also carries risks. With toxicity matching some of the harsher chemicals in the lab, safe handling moves from suggestion to requirement. Folks working with it keep emergency routines sharp, always wary that a small mistake invites real harm.
Risks, Responsibility, and Safer Solutions
Nobody should handle thiophosgene without serious preparation. Its structure—though straightforward—leads to a volatile nature, inviting trouble through inhalation or skin contact. Safety standards aren’t just formalities here; they come from generations of hard lessons. Fume hoods, proper gloves, and thorough training shape the only way to work with this stuff.
Modern chemistry still looks for ways to mimic thiophosgene’s chemical tricks without the same hazards. Green chemistry, a field I’ve followed for years, now offers alternate reagents that nudge reactions in the right direction with less risk. As demand grows for safer labs and less toxic waste, solutions draw from both the legacy of compounds such as thiophosgene and the drive for innovation. In the meantime, those who open a bottle labeled CSCl2 know well what kind of respect it demands.
How should thiophosgene be stored?
Understanding the Threat
Anyone who’s handled thiophosgene even once realizes the substance isn’t something you’d want to take chances with. I remember my first experience in a synthetic lab; the smell alone could make a lasting impression. Thiophosgene has a reputation—one that goes beyond stories in textbooks. Its volatility, toxicity, and potential to break down in contact with air and moisture call for respect and discipline.
Physical and Chemical Risks
Let’s break it down: thiophosgene, a red liquid, boils at less than 80°C. That means it evaporates easily at room temperature. Inhalation or skin contact poses immediate danger to health. Unstable even on the shelf, the compound will react quickly with water to release HCl and ugly gases. These aren’t characteristics to ignore or underestimate. Sources like the CDC and NIOSH warn about both acute inhalation and chronic effects, which remain persistent risks in any technique-heavy chemistry lab.
Storage: Practical Habits from Real Lab Benches
Safe storage starts with environmental control. Based on my time in university and industry labs, no one keeps thiophosgene in the general chemical storage. Instead, it always goes in a corrosion-resistant, tightly sealed glass or PTFE container. Screw caps must close tightly to keep water vapor out; exposure to moist air can degrade the material and threaten shelf life, not to mention safety on opening. I’ve seen clouded glass bottles and pitted aluminium caps—the results of ignoring such details until it’s too late.
Cold temperatures slow down decomposition. Don’t store it in the deep freezer, but a dedicated flammable materials fridge checked often for temperature stability reduces pressure build-up, degradation, and vaporization. The worst mistake I’ve seen happens when someone sticks thiophosgene into a regular fridge, right next to biological samples or benign chemicals—a disaster waiting to happen.
Preventing Exposure
Most seasoned chemists use a secondary container, often a sealed metal can or purpose-built container, to contain leaks or breakage. Spill trays lined with absorbent material sit under heavy-use chemicals for a reason. Lab managers emphasize double-labeling—one on the bottle, one on the outer secondary container—to keep emergencies from spiraling out of hand.
Don’t store large volumes unless absolutely needed. Spreading out risks by keeping the minimum required amount, away from oxidizers and bases, reflects a lesson learned from watching an entire shelf’s contents react badly during a minor fire inspection incident.
Regulations and Documentation
Government agencies take this seriously. OSHA, EPA, and European REACH guidelines spell out quantity limits, approved storage technologies, and environmental systems for handling toxic chemicals. At my own workplaces, regular audits and inventory control forced everyone to review expiry dates on containers. People who keep detailed logs and regular hazard reviews cut accident rates dramatically, as numerous lab incident reports show.
Safer Workplaces through Habitual Care
Training and signage matter just as much as hardware. Even one slip-up—picking the wrong gloves or working outside a fume hood—can bring big consequences. Everyone from students to senior scientists benefits from refreshers on handling unexpected leaks or exposure, and stories from real accidents reinforce the point better than any manual.
Thiophosgene storage isn’t just about compliance or ticking off boxes. Serious attention and disciplined habits can mean the difference between routine research and a trip to the hospital. Culture makes the key difference—careful labeling, proper containers, thoughtful physical separation, and making sure every step gets reviewed regularly.
What hazards are associated with thiophosgene exposure?
Straight Talk About Thiophosgene Hazards
Plenty of chemicals floating around in manufacturing or research labs feel intimidating. Thiophosgene lands high on that list, shaken by both its sharp, choking odor and the stories people share about accidental exposure. Whenever you start working with thiophosgene, reality sets in quickly: this is not the kind of substance to underestimate.
What Accidental Exposure Looks Like
A small leak. A quick splash. Maybe it clouds the air for just enough seconds to sting your throat. Thiophosgene's danger comes mainly from inhaling its vapor, which irritates the eyes, nose, and lungs. That burning feeling in your chest warns you right away, but sometimes, the worst symptoms creep up hours later. Back in my student days, a colleague got exposed during a rushed experiment. There were coughs, watery eyes, and headaches. He brushed it off, but by evening he ended up in the emergency room struggling to breathe. Turns out, even brief, unprotected contact with thiophosgene vapor can cause delayed lung injury, including life-threatening pulmonary edema.
The Risks Don’t Stop at Breathing
Getting thiophosgene on skin brings a whole different set of problems. The liquid burns, and even diluted splashes can leave painful welts or ulcers. Add to that, eye exposure leads to severe irritation and, in bad cases, loss of vision. These are not distant hazards. In any lab where chemicals pass through fume hoods or transfer lines, all it takes is a loose connection or a hurried pour to cause a spill.
Not Just a Workplace Hazard
The risks extend beyond the person handling the chemical. Thiophosgene vapor lingers. Without proper ventilation, it sticks around to affect other people working in the same space. It reacts with water in the air, producing toxic gases such as hydrogen chloride and carbonyl sulfide, multiplying the hazard. Accidental releases don’t just harm individuals—they can clear out entire facilities until professionals declare them safe.
Why These Concerns Deserve Attention
Research and workplace injury reports paint a clear picture: acute thiophosgene exposure often leads to hospitalization, with long-term lung issues or chemical burns that take weeks to heal. Fatalities have occurred when people did not have access to protective gear or underestimated the chemical’s power. The U.S. National Institute for Occupational Safety and Health (NIOSH) sets extremely low permissible exposure limits for thiophosgene, and for good reason. The margin between a safe dose and a toxic one is razor-thin.
Practical Solutions That Make a Real Difference
Nobody expects to spill a hazardous chemical, yet mistakes happen. Effective solutions rest in layered protection. Fume hoods, full chemical goggles, proper gloves, and laboratory coats form the basics. Emergency showers and eyewash stations must be easy to reach. Workers need regular safety training to recognize symptoms, handle spills, and respond to an unexpected release. Having a buddy system can make a huge difference—another set of eyes helps spot leaks or unsafe practices in real time.
Reporting near-misses and learning from them strengthens group safety, too. Companies willing to invest in robust ventilation, leak detectors, and real-time air quality monitoring reduce the risk for everyone. Substitute less toxic chemicals whenever possible, but when thiophosgene absolutely can’t be replaced, strict rules need to become routine, not just recommendations.
Looking Out for Each Other Matters
Most people who’ve seen thiophosgene accidents become the biggest advocates for safety. The stories stick, shaping the culture of any team handling dangerous chemicals. Taking thiophosgene hazards seriously isn’t just about compliance—it’s about sending everyone home healthy each day. That’s what builds respect and trust in the lab, and keeps science moving forward without sacrificing anyone’s well-being.
| Names | |
| Preferred IUPAC name | Trichloromethanethione |
| Other names |
Thiocarbonyl dichloride Thioketene dichloride Thioxomethylene dichloride Carbonic dichloride sulfide Dichlorothiocarbonyl Carbonothioyl dichloride |
| Pronunciation | /ˌθaɪ.oʊˈfɒs.dʒiːn/ |
| Identifiers | |
| CAS Number | 463-71-8 |
| 3D model (JSmol) | `/graphics/jsmol/jmol.php?model=thiophosgene` |
| Beilstein Reference | BJ5175000 |
| ChEBI | CHEBI:38200 |
| ChEMBL | CHEMBL38379 |
| ChemSpider | 21402 |
| DrugBank | DB06733 |
| ECHA InfoCard | 100.004.861 |
| EC Number | 207-611-0 |
| Gmelin Reference | Gmelin1041 |
| KEGG | C14325 |
| MeSH | D013849 |
| PubChem CID | 7909 |
| RTECS number | XN8575000 |
| UNII | F439U620FS |
| UN number | UN1709 |
| CompTox Dashboard (EPA) | DJYRVLAGFYHUJI-UHFFFAOYSA-N |
| Properties | |
| Chemical formula | CSCl2 |
| Molar mass | 99.00 g/mol |
| Appearance | Red liquid |
| Odor | Disagreeable, suffocating |
| Density | 1.64 g/mL at 25 °C |
| Solubility in water | Decomposes |
| log P | 1.85 |
| Vapor pressure | 41 mmHg (20 °C) |
| Acidity (pKa) | -0.7 |
| Basicity (pKb) | -5.17 |
| Magnetic susceptibility (χ) | χ = -42.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.595 |
| Viscosity | 0.55 mPa·s (20 °C) |
| Dipole moment | 1.76 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 48.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -214.6 kJ/mol |
| Hazards | |
| Main hazards | Toxic if inhaled, swallowed, or in contact with skin; causes severe burns and eye damage; may cause respiratory irritation. |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H311: Toxic in contact with skin. H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P260, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 4-2-2-T |
| Flash point | 25 °F (-3.9 °C) (Closed cup) |
| Autoignition temperature | 180°C |
| Explosive limits | Explosive limits: 7.1–26% |
| Lethal dose or concentration | LD50 oral rat 56 mg/kg |
| LD50 (median dose) | LD50 (median dose): 8 mg/kg (oral, rat) |
| NIOSH | TH1200000 |
| PEL (Permissible) | 1 ppm |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | 2 ppm |
| Related compounds | |
| Related compounds |
Phosgene Trichloromethanesulfenyl chloride Ethanethiol Carbon disulfide |

