Phenyltrichlorosilane: Navigating a Chemical Cornerstone in Modern Science
Historical Development
Phenyltrichlorosilane has roots stretching back to the early days of organosilicon chemistry, which picked up steam in the 1940s and 1950s. Chemists realized they could take the properties of both silicon and carbon and combine them in ways that natural minerals and polymers couldn’t match. Phenyltrichlorosilane quickly became one of the workhorses in this new world. Scientists experimented with it to build more complex silicon-based molecules, especially as the electronics and coatings industries demanded better materials. Each decade, research brought another layer of understanding, moving from simple syntheses to targeted designs. For those at the bench or on the plant floor, seeing how one compound like this could influence advances in everything from waterproofing agents to microchips gives a strong sense of how chemical history shapes modern technology.
Product Overview
This substance stands out for its utility as a building block. In industrial settings, it gets used to prepare both specialty and commodity silicones. Its formula—C6H5SiCl3—might look unremarkable, but that combination of a phenyl group and three chlorines on a silicon atom opens doors to countless chemical conversions. I’ve seen research labs reach for phenyltrichlorosilane as a starting point when they're after something more exotic, like phenyl-modified siloxanes for advanced rubber blends or specialty coatings. Its availability and reactivity make it popular, but the excitement comes from what it turns into, not just what’s in the drum.
Physical & Chemical Properties
Anyone working with phenyltrichlorosilane notices right away that it’s a colorless to pale yellow liquid with a sharp, distinctive smell. It tends to hydrolyze rapidly when exposed to air, pulling moisture to create hydrochloric acid and white fumes—an immediate reminder to respect proper ventilation and personal protection. The molecule’s boiling point sits around 201-203°C, giving it enough thermal stability for most laboratory work, but it stays volatile enough to require careful storage. It dissolves in organic solvents like ether or toluene, which suits organic chemists trying to dissolve or react it under controlled conditions. The three chlorine atoms not only flag it as a reactive compound but make it a tough customer for those handling waste disposal and emissions control—real-life concerns for those of us who’ve ever dealt with cleanup after a spill.
Technical Specifications & Labeling
Regulators and manufacturers follow clear guidelines for labeling phenyltrichlorosilane, not out of bureaucratic comfort but because errors carry consequences. Containers must show its chemical identity, hazard pictograms, and key safety warnings, particularly regarding toxicity and reactivity with moisture. Most packaging—whether for research or industrial stock—uses materials that resist corrosion and keep out water. I’ve learned firsthand that even small labeling lapses introduce confusion and risk. In practice, consistent technical specs build trust between supplier and user; reliable purity assures both innovation and safety across every downstream application.
Preparation Method
Chemical plants typically produce phenyltrichlorosilane by reacting chlorosilanes with benzene under catalyzed conditions, exploiting the affinity of silicon for aryl groups. The most common route involves the direct chlorination of phenylsilane or through Grignard reactions, which connect phenyl magnesium halide to silicon tetrachloride. Engineers favor methods that compress time and reduce waste, though every setup comes with its trade-offs. When you see a team fine-tune running conditions, you grasp quickly that economic yield and by-product control shape not just manufacturing costs but also environmental footprint. This is the kind of detail that separates academic curiosity from daily operational reality.
Chemical Reactions & Modifications
Phenyltrichlorosilane earns its place in a chemist’s toolkit because of those reactive chlorine atoms. It acts as a crosslinker for silicone polymers, driving network formation that gives rubbers their elasticity or resins their high-temperature performance. When exposed to alcohols, it undergoes controlled hydrolysis to create siloxane bonds—this forms the cornerstone of most flexible, water-repelling coatings and adhesives. I’ve seen teams modify its phenyl or silicon center to further tune properties, such as incorporating fluoroalkyl groups for ultra-repellent surfaces, or using it in tandem with other organochlorosilanes to balance flexibility and durability. Each reaction captures a balance of simplicity and creativity, blending reliable old chemistry with modern performance demands.
Synonyms & Product Names
Across labs and supplier catalogs, phenyltrichlorosilane also goes by alternatives like trichlorophenylsilane or phenylsilicon trichloride. While the differences seem slight, clarity in nomenclature prevents mix-ups, especially since many organosilanes share similar formulas but differ by just a group or two—those details matter both for procurement and safe use.
Safety & Operational Standards
Dealing with phenyltrichlorosilane means respecting both chemical hazards and regulatory frameworks. Inhaling its vapors or contacting its liquid forms risks burns and lung damage. Facilities turn to best available engineering controls—ventilation, protective gear, emergency showers—not as box-ticking but as lessons learned through tough experience. Good training ensures staff know first aid for exposures and can handle decontamination or leaks. Years ago, an unexpected splash during a transfer taught me the difference between reading a safety data sheet and living one; fast action and solid teamwork kept minor mishaps from becoming major incidents. Regulatory agencies scrutinize documentation, storage, transport, and waste with good reason: slip-ups reach well beyond the lab bench, potentially affecting air and water quality.
Application Area
Phenyltrichlorosilane’s legacy runs through industries that shape daily life, even if most people never see its name on a label. Electronics manufacturers use its derivatives for electronic encapsulation and surface treatments that defend devices against moisture. Paint and coatings scientists employ it when they want weather-resistance and durability. Medical device researchers experiment with it for modifying silicone implants or lubricious tubing, weighing risks and rewards compared to other materials. And, in the construction world, waterproofing agents often start from its chemistry. In my own work, I’ve seen it unlock pathways that let teams develop safer, stronger, and smarter products, but also push us to balance performance gains against environmental responsibility.
Research & Development
Research labs focus on pushing the limits of what phenyltrichlorosilane can do, aiming for more sustainable and efficient processes or exploring new applications like functional coatings that interact with light or resist fouling from biological growth. Teams look at how tweaking its structure enables new catalysts or more specialized elastomers. Journals fill with articles about balancing reactivity with selectivity or marrying silicon chemistry to green principles. For those who value science, seeing such active R&D tells you this chemistry hasn’t reached its peak but continues evolving with society’s needs.
Toxicity Research
Scientists continue to probe the health effects of phenyltrichlorosilane, both in the short term—burns, respiratory distress—and in the long run, where repeated low-level exposures demand careful study. Animal testing and mechanistic research guide regulations, driving controls that aim to minimize accidental releases and occupational exposure. The presence of hydrochloric acid as a hydrolysis product raises both acute and chronic concerns, pushing companies and labs to invest in mitigation and medical monitoring. Communication about these risks means more than ticking boxes: it keeps accidents rare and trust high.
Future Prospects
The future for phenyltrichlorosilane depends on balancing innovation with stewardship. Chemists chase new functional materials for clean energy, biocompatible implants, and smart surfaces. At the same time, community awareness about hazardous substances puts pressure on manufacturers to use greener synthesis routes and safer handling protocols. Cleaner reactions—maybe with less hazardous by-products—or better recycling methods could make the most of this chemistry without risking health or the environment. From what I’ve seen over decades, every breakthrough sparks fresh questions about responsibility, and phenyltrichlorosilane’s story is far from ending as science and society both continue to change.
What is Phenyltrichlorosilane used for?
What Makes Phenyltrichlorosilane Stand Out?
Phenyltrichlorosilane sounds like one of those chemicals you’d only see in the back of a dusty textbook, but in reality, plenty of people cross paths with its results every day. In the world of manufacturing and material science, it carves out an important role. When chemists and engineers talk about silanes, they usually mean compounds built around silicon and hydrogen. Stick a phenyl group and some chlorine on, as with phenyltrichlorosilane, and a new set of uses opens up—especially for anyone dealing with surfaces and coatings that need a specific touch.
Why It Pops Up in Industry
Factories that make silicone resins often use phenyltrichlorosilane, since it helps set up the three-dimensional web needed to make those high-performance materials. Without something that links the pieces together, silicone might not offer the same resilience, heat resistance, or electrical properties that shape our daily routines. Take the coatings inside certain electronics or the molds in foundries—those coatings owe part of their stability to how phenyltrichlorosilane changes the surface chemistry.
The story goes beyond just major industry. Labs use this stuff to modify glass surfaces and add specific, often water-repellent layers. Imagine a microscope slide where nothing should stick except a targeted chemical reaction. It’s tougher to design selective, reliable, repeatable experiments without the help of compounds like phenyltrichlorosilane.
Why Anyone Should Care
It’s easy to overlook building blocks like phenyltrichlorosilane since so many products start with tiny molecules before blending, heating, or transforming them into something brand-new. But silicon-based compounds changed how many industries approach durability and environmental resistance. If you own a computer, some of the insulating materials inside may owe their properties to just this compound. Car windows and optical fibers rely on smart chemical reactions with this type of silane, lending them scratch resistance or extra clarity.
Working with chemicals like this takes expertise and trust in the supply chain. Mishandling it could lead to harsh byproducts, including hydrogen chloride gas, which brings plenty of health and environmental risks. That's why most of the world treats phenyltrichlorosilane under the watchful eye of strict regulations.
Solutions: Safer Production, Smarter Uses
Plenty of factories have started to focus on recycling chlorosilane byproducts and finding greener reaction pathways. Investment in closed-loop systems, where gases and waste are captured and reused, cuts down on emissions. Academic labs and smaller manufacturers learn from these big players, installing improved ventilation and better detectors in storage areas.
Regulation keeps pace with risk. Agencies demand detailed tracking and emergency planning to prevent spills or accidents. By training workers—not just at the top but on the floor—companies support a safer environment. Collaborative research also brings new silanes that lower risk without losing performance, drawing on lessons from years of industrial experience.
From glass slides in a DNA lab to the shell of an electric motor, phenyltrichlorosilane often plays a supporting role, but none of those results turn out the same without it.
What are the storage and handling requirements for Phenyltrichlorosilane?
Why Phenyltrichlorosilane Deserves Serious Attention
Every person who’s worked with Phenyltrichlorosilane knows this compound does not play games. It shows up as a clear, colorless liquid with a sharp, pungent odor. Once exposed to moisture, a cloud of hydrogen chloride gas can form, which irritates both skin and lungs. Keeping this chemical contained and under control has always been a part of daily safety practice, not just a checklist formality.
Choosing the Right Storage Location
A dry, cool spot where water never sneaks in makes all the difference. Even a small leak or drip leads straight to trouble, and that cloud of acid vapor appears before you realize it. Concrete or steel shelves need a tough, sealed coating because Phenyltrichlorosilane etches right through ordinary surfaces. Good air circulation cuts down fume accumulation, but a spark-free environment is just as important. The fumes can corrode electrical components and wires, which turn small mistakes into bigger issues.
What Kind of Containers Hold Up?
Stainless steel or tightly sealed amber glass bottles shield Phenyltrichlorosilane from light and moisture. Those screw caps should come with special PTFE or Teflon linings because this compound chews through ordinary rubber in record time. Double containment works best in shared lab spaces: secondary trays made from polypropylene or other resistant plastic catch spills, and color-coded hazard labels remind everyone that even a small amount brings hazards.
Everyday Handling—Details that Matter
Direct skin contact burns fast. Full face shields, neoprene gloves, and lab coats cover the bases, and most of us keep a set shower and eyewash station within five steps. Any transfer or sampling means working in a chemical fume hood; open-air pouring is a shortcut that makes accidents far more likely. Spills quickly react with any dampness, so cleaning material needs to neutralize acids—never just towels or paper.
Remember the Reactions: What Can Go Wrong?
Rain, leaky pipes, or high humidity push Phenyltrichlorosilane to its limits. Droplets on containers start the hydrolysis chain, creating sticky residues and releasing toxic gases. More than one experienced chemist remembers how a rushed move or a missing gasket punched holes in a safety record. Fire risk shouldn’t get overlooked, since those vapors catch flame from static, heat, or open circuits.
Training, Signs, and Emergency Kits
Onboarding for new team members requires hands-on walkthroughs. Written instructions next to storage areas, plus spill kits packed with calcium carbonate or soda ash for neutralization, matter far more than fancy floor signs. Every worker should report damaged bottles and skips no step in daily inspections. Team drills for a major leak or accidental splash reinforce habits, and nobody expecting an emergency handles it with the right speed.
Staying Safe Means Respecting Every Step
Phenyltrichlorosilane never excuses shortcuts, no matter the rush or workload. Transparent policy, clear hazard markings, and regular maintenance carry more weight than any single safety device. A safe operation relies on discipline, not luck. Lessons learned from mishaps stick with a person, and the best labs share tips and strong routines instead of hoping things turn out all right.
What are the hazards and safety precautions associated with Phenyltrichlorosilane?
The Real Hazards of Handling Phenyltrichlorosilane
I’ve worked in environments where chemicals like phenyltrichlorosilane turn a regular day into a high-stakes affair. This compound, often found in labs and manufacturing, doesn’t mess around. A splash on the skin or eyes leaves a mark you won’t forget, and a whiff of its vapors makes you respect your respirator.
Phenyltrichlorosilane reacts fast and violently with water—even the humidity in the air is enough to set it off. That reaction produces hydrochloric acid and white smoke. During college, I watched a minor spill chew through a countertop, releasing fumes that sent everyone scrambling for the door. Hydrochloric acid is aggressive; it doesn’t just burn on contact, it hangs in the air and attacks the lining of your nose and throat.
Direct contact causes chemical burns, redness, and blistering. Eyes take it worst—exposure can lead to permanent damage or blindness. Breathing in fumes stings the nose and throat, makes the chest tight, and causes coughing fits. Regular exposure boosts the risk of chronic respiratory problems. With a low boiling point, it evaporates quicker than you’d expect, raising the odds of inhalation if the ventilation stumbles.
Staying Ahead with Smart Safety Steps
Over the years, safety habits around chemicals like this have kept my team and me out of the ER. Personal protection is non-negotiable. We suit up in gloves made from rubber that can handle acids, safety goggles, and face shields—standard glasses won’t cut it. Full-length lab coats or chemical-resistant suits shield the skin from accidental splashes.
Respiratory protection safeguards against inhaling this stuff. Properly fitted respirators, either full face or with acid gas cartridges, do more than offer peace of mind—they keep lungs working. Good ventilation is essential. I’ve seen labs with fume hoods so loud you have to shout, but nobody complains. It’s far better than coughing up your morning coffee after one bad breath of vapor.
Storage calls for close attention. Phenyltrichlorosilane doesn’t belong near water or sources of humidity. Sealed containers with dry inert gas like nitrogen keep it in check. I always double-check labels to avoid mix-ups, and we store it away from acids, bases, and oxidizers that might cause trouble. Spills call for immediate action—spill kits built for acids and a clear exit plan keep a serious incident from spiraling. Quick steps and training make the difference between an easy cleanup and an emergency room visit.
Disposal means dealing with hazardous waste rules head-on. Dumping it down the drain never happens. Licensed contractors collect and neutralize leftovers. It costs more, but it’s nothing compared to the cost of a chemical injury or environmental citation.
Building a Stronger Safety Culture
Clear, regular training helps. Everybody working with phenyltrichlorosilane learns not only the risks, but real-life stories behind near misses and accidents. I’ve seen new lab members ask questions without fear because the culture puts safety first. Experienced colleagues step in with practical advice about PPE, emergency showers, and safe transfer techniques. Open communication turns dangerous work into manageable work.
Phenyltrichlorosilane isn’t going anywhere in industry or labs. Respect, not fear, guides safe handling. Clear rules, solid equipment, and people who look out for each other keep its hazards from becoming tragic headlines.
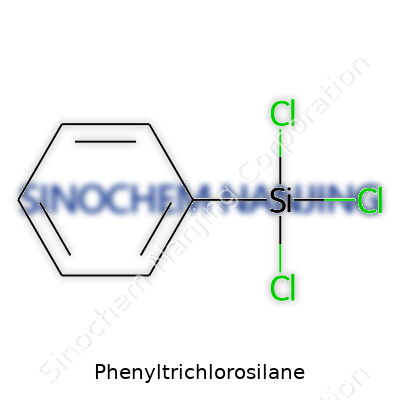
What is the chemical formula and structure of Phenyltrichlorosilane?
Chemical Formula and Structure
Phenyltrichlorosilane stands out in the chemistry world for both its formula and its structure. The formula is C6H5SiCl3. What this really means: one benzene ring hooked up to a silicon atom, which in turn grips three chlorine atoms. I’ve worked in labs where this compound makes a regular appearance, thanks to its reactive nature.
To sketch it out in your mind: imagine a six-carbon hexagon—the classic benzene ring—tagged onto a silicon atom. Instead of a silicon atom surrounded by four chlorines, here only three positions have chlorines, and the fourth spot gets the whole phenyl (benzene) group. Chemists call this structure an organosilicon halide for a reason. That silicon doesn’t just play a passive role. Its link to the aromatic ring changes how it reacts, opening doors to all sorts of functional chemistry you won’t get with plain trichlorosilane.
Why That Structure Matters
The combination of the phenyl group and the three chlorines gives it a versatility in synthesis. I’ve seen it used as a foundation for making siloxanes, resins, and coatings. Silicon sticking to the aromatic ring creates a bridge between classic organic chemistry and the world of materials science. The chlorines are reactive, so the molecule hooks up easily to other compounds—especially in controlled, anhydrous conditions.
Without moisture control, you’ll see white smoke whistling out of the flask. That’s because those chlorines come off the silicon pretty eagerly when water is around, releasing hydrochloric acid and forming silica-rich solids. Anyone who’s tried handling this stuff in a humid room won’t forget the experience. I learned early that only dry glassware cuts it for these reactions.
Role in Industry
Phenyltrichlorosilane crops up often in the production of silicones. The starting point for many high-performance materials, it helps build up siloxane chains that bring flexibility, thermal stability, and water-repellent qualities into products. Take electronics. Manufacturers rely on these traits for protective coatings that stand up to heat and don’t absorb water. In adhesives and sealants, that link between phenyl and silicon boosts resistance to chemicals where regular organosilicon compounds might fail.
From an environmental and safety perspective, the key is the chlorine content. Improper handling risks both human health and environmental release of hydrochloric acid. Strong workplace controls, well-maintained ventilation, and respect for proper personal protective equipment keep people and setups safe. Education in chemical handling goes a long way—my own workplace training made all the difference, especially for new chemists getting familiar with volatile chlorosilanes.
Looking at the Future
The demand for specialty silicone materials won’t slow down any time soon. The foundational chemistry behind phenyltrichlorosilane holds value, but greener synthesis routes have to keep pace. There’s room for new catalysts and process improvements to cut waste and energy use. In my experience, collaboration between chemists, engineers, and safety officers energizes safer production and handling.
Phenyltrichlorosilane brings together basic science and hands-on application. Its chemical formula isn’t just a string of letters; it’s an invitation to solve real-world problems and to advance both technology and safety.
How should Phenyltrichlorosilane be disposed of?
Why Disposal Choices Matter
Phenyltrichlorosilane doesn’t look all that different from any other industrial chemical until you start working with it. I remember a lab job early in my career — that potent, sharp smell stayed on my mind. The danger doesn’t end after the last experiment wraps up. It reacts with water, releasing hydrochloric acid, and that brings trouble for skin, eyes, and anything made of metal. A careless pour down the drain can spark a bigger mess than just a plumbing bill.
According to the Environmental Protection Agency, mishandling this chemical puts both health and groundwater at risk. Even small spills sneak their way into soil or streams, building up over time. I’ve seen stressed-out janitors and maintenance folks dealing with the aftermath of poor handling. This is why disposal isn’t just housekeeping. Done wrong, and the cost snowballs for the whole community.
What Actually Works
Dumping hazardous chemicals in regular trash or pouring them into a sink never cuts it. They're built for hazard, not household pipes. Professionals use a process that neutralizes the danger. In my own lab, we kept every drop in a tightly-sealed, labeled container. Once filled, a hazardous waste contractor picked it up. These folks don’t mess around — their trucks show up with the paperwork and the right gear, and they log where everything goes.
Treatment facilities break down the compound in controlled settings. Under careful supervision, experts hydrolyze Phenyltrichlorosilane. They use water under contained, vented systems, which controls the toxic gas as it forms. Facilities capture both the hydrochloric acid and the leftover silicon waste — both get processed or neutralized. This chain of custody keeps the toxin out of waterways and off job sites.
Rules Keep Cities Safer
Laws don’t just fill paper. OSHA and the EPA demand proof: tracking, documentation, transport by licensed pros. Big fines land on companies that skirt these rules. Years ago, a colleague tried to skip protocol to save time. The company paid thousands, and local water was tested for months. Regulations force us to slow down and do things right.
Another part of the equation comes from local authorities. Many cities offer hazardous chemical collection days, but they still warn against showing up with industrial waste. It’s about capacity and safety. Residents should always ask before assuming a community drop-off is enough.
What We All Can Do
Companies could treat workers better with simple, clear training. Too often, new hires get left with a thick binder and little real-world context. The best bosses walk you through disposal steps in person. Every workplace should post emergency numbers and keep spill kits handy. Labels shouldn’t fade. Collect containers regularly, never waiting until they’re overflowing.
On a personal note, curiosity and double-checking mean fewer emergencies and less stress. Reading the SDS (safety data sheet) is routine, but treating those instructions as gospel builds a safer world for everyone downstream.
Looking Forward
Better methods will come as technology grows. Right now, careful disposal and honest documentation serve our neighborhoods and the planet. The stuff made in the lab shouldn’t haunt the tap water or the air outside.
| Names | |
| Preferred IUPAC name | Trichloro(phenyl)silane |
| Other names |
Trichlorophenylsilane Phenylsilicon trichloride Silane, trichlorophenyl- Phenyltrichlorsilan |
| Pronunciation | /ˌfiː.nɪlˌtraɪˌklɔː.rəˈsɪˌleɪn/ |
| Identifiers | |
| CAS Number | 98-13-5 |
| Beilstein Reference | 3856963 |
| ChEBI | CHEBI:51989 |
| ChEMBL | CHEMBL156141 |
| ChemSpider | 21849 |
| DrugBank | DB14253 |
| ECHA InfoCard | 100.006.209 |
| EC Number | 213-935-1 |
| Gmelin Reference | 63616 |
| KEGG | C19212 |
| MeSH | Organosilicon Compounds |
| PubChem CID | 66202 |
| RTECS number | WX2975000 |
| UNII | UJ8LDY7C8Y |
| UN number | UN1810 |
| CompTox Dashboard (EPA) | DTXSID6020140 |
| Properties | |
| Chemical formula | C6H5SiCl3 |
| Molar mass | 263.57 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent |
| Density | 1.387 g/mL at 25 °C |
| Solubility in water | Reacts |
| log P | 2.9 |
| Vapor pressure | 1 mmHg (25°C) |
| Acidity (pKa) | -3.7 |
| Magnetic susceptibility (χ) | -72.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.553 |
| Viscosity | 1.44 cP (25°C) |
| Dipole moment | 1.30 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 324.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -420.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1711 kJ/mol |
| Pharmacology | |
| ATC code | |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331, H314, H335 |
| Precautionary statements | P260, P261, P264, P271, P280, P301+P330+P331, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P310, P312, P321, P330, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-W |
| Flash point | Flash point: "≥ 110 °C |
| Autoignition temperature | 232 °C |
| Lethal dose or concentration | LD50 (oral, rat): 2300 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 3600 mg/kg |
| NIOSH | B255 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Phenyltrichlorosilane is "1 ppm (5 mg/m3) |
| REL (Recommended) | REL (Recommended Exposure Limit) of Phenyltrichlorosilane is "1 ppm (5 mg/m3)". |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Triphenylmethyl chloride Benzyltrichlorosilane |

