Looking Beyond the Flask: A Commentary on Phenylphosphonothioic Dichloride
Historical Context
Phenylphosphonothioic dichloride rings a bell most loudly for those who have passed through the doors of an organophosphorus chemistry lab. Synthetic phosphorus compounds carved out their share of organic chemistry during the 20th century, riding advances in both industrial and agricultural sciences. By the 1950s and 1960s, as nations leaned heavily on farm productivity and plastics innovation, laboratories worldwide turned to organophosphorus reagents to solve knotty problems in crop protection, flame retardancy, and material modification. Within this family, phenylphosphonothioic dichloride joined the roster not just as a raw material, but soon as a tool that could deliver sulfur into molecular skeletons, helping chemists reach structures that had once demanded several laborious steps.
Product Overview
This compound rarely sits on the shelf for those outside the synthesis bench. Coming as a colorless-to-pale-yellow oily liquid, phenylphosphonothioic dichloride brings a biting, almost acerbic odor that often lingers longer than you’d like. Its role as a versatile intermediate springs from its highly reactive dichloride groups and that phosphonothioic backbone, which finds resonance in both agriculture and material chemistry circles. It's not a household name, but it's a familiar face to those crafting flame retardants, plasticizers, and occasionally bioactive phosphorus-containing molecules.
Physical & Chemical Properties
Phenylphosphonothioic dichloride doesn’t hide from its reputation as a reactive, moisture-sensitive player. Volatility at room temperature, hydrolysis on contact with water, and reactivity toward nucleophiles set the tone in any operation. Its density outpaces water, and direct contact means trouble for skin, eyes, or lungs. Storage usually means sealed glass, dry spaces, and constant vigilance to leaks and vapors. It burns chlorine and sulfur into the nose — a reminder of its stubbornly non-benign nature.
Technical Specifications & Labeling
Any reputable laboratory supply brings this chemical clearly labeled, under a raft of warnings about its corrosive, moisture-reactive personality. Chemists know to watch out for the compound’s CAS number (also often found as 2524-32-1) and to demand high purity to avoid side reactions, especially during scale-up. Regulatory standards flag this molecule due to its potential for misuse and its acute health hazards; labels on bottles scream with pictograms for toxicity, corrosivity, and environmental hazard, all backed by official statements regarding storage and disposal.
Preparation in Practice
Traditional lab syntheses for phenylphosphonothioic dichloride usually follow careful chlorination of phenylphosphonothioic acid or derivatives. Chlorinating agents show up in glassware: thionyl chloride or phosphorus pentachloride are standard fare. The reaction fumes require robust ventilation, and the work-up keeps you on edge. Yields run acceptably high under dry, low-temperature conditions, but the fine margin between textbook success and hazardous mess drives home why even seasoned technicians prepare only the amount they need for immediate use.
Reactions, Modifications, and Real-World Chemistry
Most organic chemists remember their first reaction with phenylphosphonothioic dichloride. The reagent transforms in the presence of various nucleophilic partners: alcohols, amines, and phenols readily substitute the two chlorine atoms, paving the way for much more complex phosphorus-sulfur chemistry down the line. These reactive chlorides let one build custom ligands, forge fire retardants, or bolt functional groups onto pharmaceuticals. In practice, every reaction requires precise control of stoichiometry and temperature; side products can complicate purification, and traces of water can turn surfaces tacky and glassware cloudy. Researchers often modify this backbone for application-specific roles; the world of flame retardancy leans heavily on such modifications, driving performance in treated textiles and polymers.
Synonyms & Alternate Product Names
Lab veterans might hear phenylphosphonothioic dichloride referred to by names such as O,O-dichlorophenylphosphonothioate or dichlorophenylthiophosphonate. Each reflects a slightly different perspective on structure; in practice, the bottle on the shelf carries multiple synonyms, hinting at a history of varied nomenclature and cataloging.
Safety, Operational Standards & Practical Vigilance
Lab safety kicks in long before you break the seal on a container of phenylphosphonothioic dichloride. Gloves and splash goggles only offer the first defensive layer; users favor chemical-resistant lab coats and full-face shields, especially with larger batches. Even small spills demand quick attention, as vapors and splashes corrode surfaces and burn skin. Fume hoods are standard, but I’ve seen veterans equip their ventilated spaces with backup carbon masks, just in case the exhaust falters. Waste requires careful neutralization and appropriate containment as regulations governing hazardous phosphorus waste grow stricter each year. Regular safety training, thorough pre-approval of protocols, and vigilant supervision keep most close calls from turning serious — but even with all that, too many old labs carry battle stories attesting to what happens when anyone gets complacent.
How Applications Shape Demand and Research
In the world outside academic chemistry, phenylphosphonothioic dichloride rarely takes starring roles, yet its fingerprints show up in processes from polymer functionalization to flame retardant synthesis. The agriculture sector at one time sought after organophosphorus compounds for pesticide work, only to draw back as toxicity concerns mounted. Modern regulations shape much of the application domain, throttling back on loose ends and pressing chemists to look for safer, less persistent alternatives. In industrial plastics, the drive to engineer performance flame retardants with higher thermal stability keeps this compound and its derivatives in play, while new generations of phosphorus-containing ligands and intermediates rely on its chemical backbone.
Research, Toxicity, and Responsible Development
Toxicology studies rarely flatter organophosphorus chemicals, and phenylphosphonothioic dichloride proves no exception. Strong irritant effects strike skin, eyes, and respiratory tract; acute poisoning risks demand respect and careful handling. Animal toxicity tests track organ damage; chronic exposure data remains sparse, though short-term exposure thresholds force researchers to minimize personal risk. Regulatory agencies treat the material with a strong dose of skepticism, and most university or company labs roll out detailed risk assessment protocols and environmental controls, enforced by law and experience alike. The wave of green chemistry pushes against reagents with high toxicity and persistent environmental profiles, nudging research toward improved containment, reduced residual waste, or altogether different synthetic paths.
Looking Forward: Better Science, Safer Workspaces
The future for phenylphosphonothioic dichloride walks the tightrope between chemical utility and regulatory burden. Academic teams hunt up milder, safer reagents, questioning the legacy routes that made this molecule so widely available. Scale-up for industrial use faces more hoops with every revision of hazardous substance lists. I see grassroots changes in how young chemists approach synthesis: less bravado, more risk calculation, and increasing collaboration with environmental scientists. If phenylphosphonothioic dichloride rises to new prominence, it’ll be due to thoughtful innovation — not just because it’s always been there on the shelf, waiting for the next batch reaction.
What is Phenylphosphonothioic Dichloride used for?
What This Chemical Does
Phenylphosphonothioic dichloride sounds like something straight out of a chemistry textbook, but it's no stranger in industrial and agricultural circles. This chemical shows up where folks look to synthesize organophosphorus compounds. You find these in everything from pesticides to certain flame retardants. Factories rely on it to build other chemicals piece by piece, linking up molecules in ways basic raw materials can’t manage. I learned about it during a summer job at a specialty chemicals plant, where its sharp, stinging odor stuck with me longer than most lectures ever could.
Where It Ends Up
Most of the time, phenylphosphonothioic dichloride acts as a middleman—in chemical lingo, a “reagent” or an “intermediate.” For example, manufacturers take this compound and react it with other stuff to make pesticides that protect crops from disease and insects. Sometimes, it’s used in making flame retardants for electronics and insulation. There’s not a lot of glamour in talking about molecules, but the difference between an overheated phone and a safe one can start in a chemical plant.
People outside the chemical industry rarely see or hear about phenylphosphonothioic dichloride directly, and for good reason: it’s corrosive, hazardous, and tough on the environment if handled carelessly. Skin burns and lung irritation are no joke. Workers wear gloves, masks, and full-body suits, and companies spend time and money to contain spills. The risks aren’t just about individual exposure; nearby communities can get nervous, especially if there’s a history of leaks.
Why Watchdog Groups Care
Concerns pop up because many chemicals with “chloro” or “thio” in their names have a checkered safety history. A number of these compounds have shown up as nerve agents or chemical weapons in the past. Regulations keep a close eye on phenylphosphonothioic dichloride. The United States Environmental Protection Agency and its EU counterparts require companies to report how much they make, use, and store. Cross-country shipments raise red flags for custom inspectors.
Handling toxic stuff is never free from risk. I once saw a drill go wrong while emptying a reactor in the plant—the emergency response that followed showed how crucial proper training and good equipment can be. Every extra layer of safety stops an accident from turning into tragedy.
Making Industry Safer and Smarter
Safer alternatives do exist in some applications. Green chemistry programs push for less aggressive compounds or designs where workers never touch the nastiest ingredients. Teachers and trainees push the basics: label everything, don’t get complacent, never skip the vent hood. Engineers look for ways to automate transfers and doses so fewer people get exposed. Research never stops, and some labs try to develop newer pesticides and flame retardants using friendlier starting materials.
Waste comes as a cost both to people and planet. Good practice means capturing and neutralizing chemical by-products before they leave the factory. Scrubbers, filters, and better containment systems limit how much leaks into air or water. A community has a right to know what’s in their backyard, and factories owe honest answers about what they store and how it’s managed.
Taking Responsibility
People who work with hazardous intermediates like phenylphosphonothioic dichloride carry a special responsibility. They protect not only their own health, but also their coworkers and neighbors. Regulators set rules, but personal vigilance makes the biggest difference. Being open about risks and always looking for better ways to work goes a long way, whether in a small batch shop or a global chemical powerhouse.
What are the safety precautions when handling Phenylphosphonothioic Dichloride?
Why Respect for Chemicals Matters
Years ago, in my first real lab job, I was surprised by how much fuss surrounded the easy stuff—gloves, goggles, labels. Nobody cut corners, and for good reason. Phenylphosphonothioic dichloride commands just that sort of respect. It doesn’t care about shortcuts, and ignoring safety turns a simple experiment into a medical emergency.
Direct Contact Spells Trouble
One splash, one cloud of vapors can ruin your day. Phenylphosphonothioic dichloride reacts with water, releasing corrosive and toxic gases, including hydrochloric acid. Eyes and skin burn quickly on contact. Even ordinary disposable gloves melt away. This chemical demands proper personal protective equipment—think thick nitrile gloves, chemical-resistant splash goggles that seal, and a sturdy lab coat. I’ve seen cheap gloves eaten away during demonstrations, so cut-rate gear does not cut it.
Ventilation and Air Quality
Fume hoods save lives. Despite the inconvenience and noise, working in one slashes risks dramatically. Phenylphosphonothioic dichloride vapors threaten lungs and airways—one good whiff leaves a lingering burn in the back of the throat. The fume hood pulls vapors away before lungs catch them. Only a functioning, regularly checked hood offers real protection. Depending on building age and budget, that can require some advocacy from staff who work with these chemicals every day.
Containment and Storage
No open containers, no casual refilling. Phenylphosphonothioic dichloride demands tough, sealed bottles—no glass with old chips, no containers you grab from recycling. Store it away from water sources and base chemicals because accidental mixing triggers reactions you can’t always stop. Lock it up, label it clearly, and keep the storage records up to date. Even interns need to be able to find the safety cabinet in the dark.
Spill Response: Ready Before the Worst
A friend once spilled just a little of this stuff down a bench leg. The panic lasted longer than the spill. Emergency training and spill kits matter much more than most realize. Acid-neutralizing agents, absorbent pads, and a quick hit on the eyewash station turned a dangerous accident into a lesson instead of a disaster. Printed instructions next to the kits make a difference when hands shake and instincts start to falter.
Data and Documentation
It’s easy to roll eyes at the paperwork. But exposure records, training logs, up-to-date Safety Data Sheets—these matter when things go wrong. OSHA and local regulations may feel frustrating, but after an incident every record keeps the bigger disaster away. Failure to keep track puts future staff in harm’s way.
Real Solutions Build Good Habits
The list feels long, but cutting any corner with phenylphosphonothioic dichloride makes life easier for exactly no one. Practice routine, drill emergencies, and challenge colleagues who let things slide. An approach rooted in lived experience, clear communication, and everyday vigilance keeps disasters off the front page. Every glove, every hood, every label matters because the risk turns real in an instant.
What is the chemical formula and structure of Phenylphosphonothioic Dichloride?
Understanding Phenylphosphonothioic Dichloride
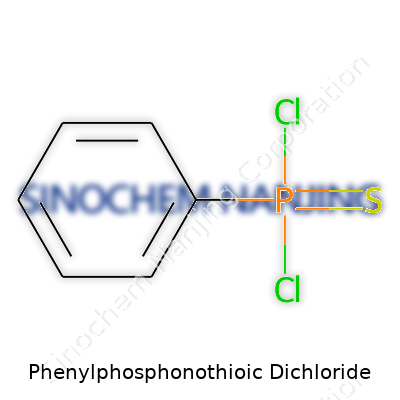
Growing up near an old industrial park, I spent long afternoons watching tank trucks roll in, bringing chemists a wild medley of specialty chemicals. Phenylphosphonothioic dichloride stood out because of its reputation—common in labs, but tricky enough to keep locked away from high school science classes. The formula reads as C6H5PSCl2, and for many, that jumble of letters and numbers hides a world of reactivity and hazard.
Breaking Down the Formula
One phenyl group (C6H5) anchors the molecule. The core features a phosphorus atom linked to a sulfur atom by a double bond (P=S). Two chlorine atoms complete the set, each attached directly to the phosphorus, making the full name accurate: phenyl + phosphono + thioic + dichloride.
Now, the structure looks something like this: the phenyl ring attaches to phosphorus, which bonds to sulfur with a strong double connection, and stretches two chlorines off to the side. Real chemists sketch it so that the phosphorus sits center-stage, with lines shooting out to a benzene ring, two Cl’s, and a S double bond. In 2D form:
- Phenyl group (C6H5) on phosphorus
- P=S bond
- Two P–Cl bonds
This formula and arrangement matter, especially for people handling synthesis or responding to chemical spills. Chlorides bristle with reactivity, and the phosphorus-sulfur linkage opens doors to unique transformations in organic chemistry. The structure lets it react sharply with water, releasing corrosive hydrogen chloride. If someone in a lab cracks open a container without gloves and goggles, the risks get real, fast.
Why Structure Matters So Much
In chemical manufacturing, small changes in core structure alter the whole game. Swap the sulfur for an oxygen, and suddenly you end up with phenylphosphonic dichloride — a very different animal. Phenylphosphonothioic dichloride fits precisely where chemists need its unique sulfur-phosphorus pairing. For example, it steps into the ring during synthesis of pesticides, flame retardants, and specialized organophosphorus compounds.
One misstep handling the molecule can introduce HCl fumes; ask anyone who’s worked in a poorly ventilated hood about the sting in their eyes. For years, safety in smaller businesses got overlooked. Strict documentation and personal stories changed that, pushing the industry to invest in high-quality gear and constant training. It’s not just the reactive chlorides; anything bonded to phosphorus and sulfur-leans toxic, often affecting the nervous system with repeated exposure. The concrete reality? A well-marked bottle, a solid hood, and gloves that don’t spring leaks.
Inside the structure, you see phosphorus’s tendency to bond in unique ways, which makes it valuable in synthesis but brings headaches if spilled into the environment. Regulations in the EU, US, and China reflect lessons learned: tightly tracking use, advocating quick cleanup protocols, and researching less hazardous substitutes.
Building Strong Safety Habits
Anyone spending time in a lab with this stuff learns to double-check their chemical labels—and their health and safety protocols. I know colleagues who keep detailed logs, snap photos of labeling, and never transfer the liquid without a buddy present. Over the last decade, strengthened reporting, stricter handling guidelines, and community pushback have helped make chemistry safer, but everything starts with that central formula: C6H5PSCl2. Understanding it isn’t just about scoring high on a quiz—it keeps people healthy and workplaces open, and it reminds us that in chemistry, nothing beats respect for what a single molecule can do.
How should Phenylphosphonothioic Dichloride be stored?
Why Handling Matters
Phenylphosphonothioic dichloride isn’t on most people’s weekly shopping list, but in the chemical industry, this substance plays a role in making certain herbicides, plasticizers, and flame-retardants. It carries risks, too: strong fumes, a sharp odor, and the sort of reactivity that doesn’t mix well with carelessness. Stories pop up in trade publications about near-misses simply because someone underestimated what it can do. Mishandling has real consequences, from frightening chemical reactions to lasting damage in storage facilities and labs.
The Right Approach to Storage
Solid walls and a sturdy floor matter, but storing this chemical demands something extra. Moisture triggers it, so sealed containers sealed tight are the first step. Humid air or a stray drop of water can create fumes and heat—neither of which anybody wants in their storage area. Every year, fires and injuries around the world underline the need for vigilance.
Shelving sits at waist-height, never on top of high cabinets. Dropping the container from any height brings risk. I’ve seen a cracked vial turn one spotless storeroom into a hazmat zone in under a minute. Use glass or specialty plastic containers built to handle aggressive chemicals—metal lids, especially those with aluminum, react and corrode.
Separation Makes A Big Difference
Phenylphosphonothioic dichloride clashes with more than just water. Strong bases, alcohols, and oxidizers don’t belong anywhere nearby; storing them in the same room increases the chance of cross-contamination. I remember a lab in college where a newbie housed alcohol right next to a bottle of this material. Nobody slept well that week until safety officers cleared the danger.
This substance prefers its own space, ideally in a chemical storage cabinet with exhaust ventilation. Airflow helps carry away any accidental vapors before they collect and do harm. Strong shelves, spill containment trays, and warning labels at eye level form the backbone of a responsible setup.
PPE and Emergency Tools
Entry into the storage room without goggles, gloves, and a long-sleeved coat tempts fate. It only takes a small splash to cause burns or breathing problems. Nearby, a working eyewash station and shower stay ready for one of those “just in case” moments everyone dreads but plans for. I’ve been through enough safety drills to know that a few extra seconds can change an accident’s outcome.
Temperature and Spill Control
Room temperature generally keeps this substance stable, avoiding both freezing and overheating. Direct sunlight can break down containers or warm liquids too quickly, so dark, cool closets beat open windows and hot corners every time. In case a leak does happen, mineral oil or sand can help control the spill until experts arrive, since water only makes things worse.
Good Records Make for Fewer Surprises
Clear labeling, locked cabinets, and an up-to-date inventory stop unwanted surprises. Every entry and exit matters—if a container goes missing, a quick response can mean the difference between routine paperwork and a company-wide scare. Regular checks reveal weak seals or signs of corrosion before they spark an incident.
In my own work, constant vigilance turned potential accidents into harmless stories told around lunchrooms. Respecting substances like phenylphosphonothioic dichloride doesn’t spring from fear; it comes from seeing what carelessness really costs. Safety, in the end, always pays back more than it asks.
What are the physical and chemical properties of Phenylphosphonothioic Dichloride?
How This Chemical Looks and Acts
The name “Phenylphosphonothioic Dichloride” may trip up your tongue, but the stuff itself shows up as a clear or pale yellow liquid. It gives off fumes that can catch your nose pretty quickly; keep it in a sealed container, and you’ll notice a sharp, pungent smell if you open it. The structure shows a phosphorus atom with a phenyl ring, a sulfur atom, and two chlorine atoms—so it’s got some bite.
It doesn’t dissolve much in water, but in organic solvents like toluene or chloroform, it blends in easily. That poor water solubility means spills don’t always wash away with a simple splash—cleanup needs care. It reacts strongly to moisture and won’t just stay put in humid air. As it sits out, it starts to break down, and the result isn’t pleasant: hydrogen chloride and some sulfur-based byproducts float off, turning a spill into a real mess.
Reactivity and Storage Hazards
This isn’t the kind of thing you want hanging around a crowded workspace. Once it hits water—even the moisture in the air—hydrolysis fires up, leading to the release of gases that irritate the eyes, nose, and throat. The fumes can sting the skin or lungs on contact. Splash some on a metal tool, and you’ll probably notice quick corrosion. If it runs across bases or alcohols, a strong, sometimes violent reaction kicks in, so it demands tough storage conditions: cool, dry, and sealed away from common chemicals.
Real-World Use and Why Handling Matters
Chemists in labs and factories use phenylphosphonothioic dichloride to whip up pesticides and flame retardants. In certain organic syntheses, it reacts fast and gets things done—but if safety gear slips, burns show up in minutes. In my own time working around lab chemicals, small mistakes with similar agents turned into lessons: gloves, goggles, good ventilation, and a ready wash station make the difference between an ordinary day and regret.
This chemical also stands out as an intermediate. It serves as a building block with selective reactivity—one reason factories value it for making custom molecules. Sometimes, it takes part in processes where its chlorine atoms swap out for other groups, letting chemists tailor things for specific end uses. But those same properties mean anyone nearby has to respect the risks. The history of workplace accidents shows what happens once planning slips or equipment breaks down: accidental exposure can lead to severe burns, respiratory distress, and long-term scarring.
What We Can Do Better Regarding Safety
Strict containment measures change the story. Tanks and lines built to stand up to corrosion, automatic leak alarms, and air filtration can all reduce risk. Training matters just as much—anyone handling this needs clear, real-world advice, not just a safety sheet taped to the wall. Emergency showers and breathing masks aren’t just extra—they’re baseline protection.
Regulators and chemical manufacturers stress tracking and tracing supplies, since misuse or accidental release causes serious harm. By following best practices and inspecting equipment regularly, companies can keep accidents to a minimum.
Final Thoughts
Phenylphosphonothioic dichloride gets the job done in industry, but the demanding storage and handling remind us: chemistry isn’t just about the science, but taking care of the people working with it every day.
| Names | |
| Preferred IUPAC name | Phenylsulfanylidynephosphane dichloride |
| Other names |
Thiophosphoryl chloride Thiophosphonic dichloride Phosphonothioic dichloride Phenylthiophosphonic dichloride O,O-Dichlorothiophenylphosphonate |
| Pronunciation | /fəˌnaɪlˌfɒs.fəˌnəʊˈθaɪ.ɪk daɪˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | [2524-32-1] |
| Beilstein Reference | 2060884 |
| ChEBI | CHEBI:51790 |
| ChEMBL | CHEMBL4292071 |
| ChemSpider | 22216 |
| DrugBank | DB14599 |
| ECHA InfoCard | InChI=1S/C6H5Cl2PS/c7-10(8,9)6-4-2-1-3-5-6/h1-5H |
| EC Number | 208-118-1 |
| Gmelin Reference | 60714 |
| KEGG | C19324 |
| MeSH | D010668 |
| PubChem CID | 69986 |
| RTECS number | SZ9625000 |
| UNII | 0LRL0U0A9W |
| UN number | UN2834 |
| CompTox Dashboard (EPA) | `DTXSID3035687` |
| Properties | |
| Chemical formula | C6H5PSCl2 |
| Molar mass | 231.06 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | pungent |
| Density | 1.598 g/cm3 |
| Solubility in water | Reacts |
| log P | 1.9 |
| Vapor pressure | 0.4 mmHg (20 °C) |
| Acidity (pKa) | 2.1 |
| Basicity (pKb) | -4.5 |
| Magnetic susceptibility (χ) | -64.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.616 |
| Viscosity | 1.4 mPa·s (20 °C) |
| Dipole moment | 3.13 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 352.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -116.8 kJ/mol |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes severe skin burns and eye damage; may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P261, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | 230 °F (110 °C) - closed cup |
| Lethal dose or concentration | LD50 oral rat 140 mg/kg |
| LD50 (median dose) | LD50 (median dose): 100 mg/kg (rat, oral) |
| NIOSH | SY8575000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.05 mg/m3 |
| IDLH (Immediate danger) | IDLH: 1 ppm |
| Related compounds | |
| Related compounds |
Phenylphosphonous dichloride Phenylphosphonic dichloride Phenylphosphonic acid Phenylphosphinothioic chloride Thiophosphoryl chloride |

