Phenylphosphonic Dichloride: Understanding Its Legacy and Pushing Its Boundaries
Historical Development
Walking through the legacy of organophosphorus chemistry, phenylphosphonic dichloride holds a reputation earned through decades of clever laboratory work. In the mid-20th century, chemists hunted for adaptable phosphorus compounds that could bridge the needs of industrial synthesis and academic study. Phenylphosphonic dichloride, bearing the formula C6H5POCl2, emerged from this search due to its straightforward formation from phosphorus trichloride and benzene derivatives. My own introduction to this substance came during a university synthesis course, where the tang of chlorinated compounds and wary respect for their reactivity etched memories in a way no textbook could manage. Then as now, its preparation reflects the intersection of industrial ambition and the controlled precision of synthetic organic chemistry.
Product Overview
This organophosphorus compound steps into the scene as a versatile intermediate, often overshadowed by its more familiar cousins like triphenylphosphine or phosphoric acid. Its structure features a phosphorus atom doubly connected to chlorine and a benzene ring, a setup that supports more than a few possibilities for transformation. On the shelf, it appears as a colorless to pale yellow liquid, which belies the intensity of chemical activity it can unleash in the right hands. Synthetic chemists favor it for its capacity to introduce phosphorus into more complex molecules, especially during the preparation of ligands, flame retardants, and plasticizers. This is not a material one tosses about lightly; it demands a healthy respect arising from both its utility and potential for harm.
Physical & Chemical Properties
Phenylphosphonic dichloride displays properties you’d expect from a reactive organochloride. It generally sits liquid at room temperature, with a boiling point over 260 degrees Celsius—stubbornly persistent, except in the company of hydrolytic agents. Water triggers a rapid and often violent reaction, tearing chlorine away and yielding phenylphosphonic acid and hydrogen chloride gas. In real lab situations, steam or muggy air can set off hazardous fumes. Its density usually comes in just over 1.3 g/cm³, and it carries a distinctive, pungent odor. In practice, this means storage demands vigilance: sealed glass containers, moisture-excluding atmospheres, and careful control of workspaces where it’s used. These characteristics mean even routine handling must feel deliberate and systematic.
Technical Specifications & Labeling
Labeling does more than list a name or hazard symbol here. Every bottle of phenylphosphonic dichloride needs hazard warnings for corrosivity, toxic inhalation, and environmental damage. Chemists cannot dodge the GHS skull and crossbones or the clasp of the black exclamation mark. Specifications on purity loom large because trace water or unreacted starting material can spell disaster in sensitive syntheses. In the industry, purity thresholds need to hit 98% or higher for most advanced uses, with moisture content often strung tight to less than a fraction of a percent. Accurate labeling gives chemists a fighting chance to predict and contain the energy this molecule holds.
Preparation Method
Making phenylphosphonic dichloride begins with the measured reaction of benzene or chlorobenzene with phosphorus trichloride and sometimes oxygen. The route takes advantage of electrophilic substitution, placing the phosphorus onto the benzene backbone while keeping side reactions in check. Industrial syntheses scale this up under steady-flow conditions, keeping strict control over temperature and atmosphere—no small feat, given the volatility of both phosphorus trichloride and hydrogen chloride. For a synthetic chemist in a teaching lab, scraping together small amounts means slow addition, glassware protected by drying tubes, and a readiness for nasty vapors. When done right, the product distills away, a testament to both tradition and precise chemical know-how.
Chemical Reactions & Modifications
Phenylphosphonic dichloride tends to attract the attention of anyone needing to tinker with phosphorus-based scaffolding. It reacts briskly with nucleophiles: amines, alcohols, and water. Alcohols turn it into esters, popular for flame retardant research and as building blocks for more exotic ligands. Amines lead to a host of phosphonamidates, some of which are critical in medicinal chemistry. In my own bench days, a small addition of this compound to a cooled solution could turn a quiet afternoon into a scramble for the fume hood. Hydrolysis occurs in a blink, but with skillful handling, selective substitutions become feasible, letting chemists layer complex functionality onto the phenylphosphonate framework.
Synonyms & Product Names
You’ll spot phenylphosphonic dichloride under several aliases. In some catalogs, it appears as dichlorophenylphosphine oxide, or simply PPDC, though most stick to the longer, systematic name for clarity in communication. Old texts sometimes call it phenylphosphonyl dichloride. No matter the name on the bottle, the hazards and possibilities remain the same.
Safety & Operational Standards
Working with phenylphosphonic dichloride means drawing on every thread of safety knowledge. Gloves and goggles won’t cut it on their own; the fume hood makes a non-negotiable line of defense, and SCBA-level precautions come into play at industrial scale. Spills can corrode metal and skin alike, and inhalation of vapors carries the risk of pulmonary damage. The best practices involve sealed transfers, inert atmosphere glove-boxes for sensitive steps, and waste protocols that neutralize and absorb both hydrochloric acid and residual organophosphorus compounds. Lab veterans share stories of overlooked droplets etching glass or the acrid tang of escaping gas, reminders that routine shortcuts invite serious harm. The rules here carry real consequences, and the best chemists treat them as lifelines, not just checkboxes.
Application Area
Phenylphosphonic dichloride forms the backbone for a staggering array of products. Companies depend on it for crafting flame retardants that end up in electronics and polymers, pushing up fire resistance without adding bulk. It plays a role in surfactant chemistry, especially for detergents tough enough to handle greasy, hydrophobic messes. Some researchers tweak the core molecule, using it as a springboard for pharmaceuticals that rely on phosphorus for enzyme targeting. In material science, it helps anchor organic groups onto metal surfaces—one route for tuning surface properties on nanoparticles or metal-organic frameworks. Here, versatility often means necessity, as very few other compounds allow for the reliable and scalable attachment of both phenyl and phosphorus centers.
Research & Development
Recent years have seen an explosion of interest in greener synthesis, and the world of organophosphorus chemistry has not stood still. Researchers keep pressing for new methods that dial down hazardous byproducts, searching for base-metal catalysis to replace antiquated chlorination routes. Teams in academia push for solventless processes or swap out benzene for safer arenes, seeking the holy grail of reduced toxicity without sacrificing yield or selectivity. Instrumental analysis now tracks trace impurities more closely, informed by both regulatory demands and the higher stakes of pharmaceutical applications. The drive to reimagine processes using phenylphosphonic dichloride fits into a broader movement away from legacy hazards, showing real progress in lab-scale invention and production-level optimization.
Toxicity Research
This compound's dangers are not abstract. Animal studies document corrosive effects on mucosa and skin, and inhalation exposure punches straight at respiratory tissues. Chronic exposure links to organ damage, mutagenicity questions, and risks for workers not shielded by proper protocols. Regulatory agencies demand careful monitoring in both air and liquid waste streams. From my own perspective, awareness of these risks sharpened my discipline at the bench, prompting a renewed focus on spill containment, rapid neutralization, and timely medical follow-up if accidental contact occurred. The stakes are doubly high in settings with incomplete training or absent safety culture, and reports from poorly regulated markets paint a stark picture—lax handling translates quickly into real harm.
Future Prospects
Pressure mounts for every hazardous reagent to earn its place in modern chemistry. For phenylphosphonic dichloride, the future depends on steps toward safer manufacturing, greener reaction pathways, and ultimately on unlocking new applications where its reactivity carves out unique performance gains. The ongoing transition to halogen-free flame retardants and new surface modification techniques could nudge its use upward, assuming researchers find willing partners in regulatory agencies and materials companies. If the community keeps prioritizing toxicity research, adopts best practices for exposure control, and advances greener synthesis, phenylphosphonic dichloride could ride the wave of next-generation chemical development rather than be swept aside by safer alternatives.
What is Phenylphosphonic Dichloride used for?
Behind the Formula: Why This Chemical Matters
Phenylphosphonic dichloride barely makes the headlines, but it supports important work in both industry and research labs. Anyone who’s spent time around chemical synthesis recognizes its sour, stinging odor and its corrosive bite. Most folks hardly ever deal with it outside a controlled setting, and that’s for a reason. Still, its role in making flame retardants, plastic additives, agricultural chemicals, and specialty materials deserves a closer look.
Building Better Polymers
In my days helping out at a polymer research lab, we used phenylphosphonic dichloride as a building block for specialty plastics. It reacts well with alcohols and amines, which lets chemists create molecules that stand up to heat and stress. Factories that turn out circuit boards or high-temperature coatings count on this compound. The finished products don’t just last longer; they offer protection in electrical systems and keep things safer everywhere from airplanes to kitchens.
Flame Retardants and Fire Safety
House fires don’t spare much, and as materials scientists worked to keep buildings and textiles safer, they leaned on phosphorus-based flame retardants. Phenylphosphonic dichloride lets chemists tie phosphorus atoms into complex molecules that can slow or even block burning. I’ve seen the difference it makes—treated foam and fabrics don’t flare up as quickly, and seconds count when escaping a burning room. It's not magic. It’s chemistry protecting people and property.
Crop Protection: Supporting Food Security
Farmers fight pests and blights year after year. Crop protection science calls for powerful, reliable chemistry. Whether it’s herbicides, fungicides, or growth regulators, experienced agricultural chemists often turn to derivatives of phenylphosphonic dichloride. These molecules disrupt pest processes or help plants resist tough conditions. This side of the story matters because global food demand keeps rising. Better chemistry gives farmers tools to feed more people without clearing more land.
Safety and Environmental Concerns
Working with phenylphosphonic dichloride means strict protocols. It’s reactive and dangerous if handled carelessly—it burns on contact and fumes aggressively, so a good fume hood and gloves come standard. Regulatory oversight keeps people safer both on the job and in communities nearby. Some countries press for lower emissions and stricter disposal rules to prevent water or soil contamination. This approach makes sense, since mishandling can cause long-term health problems for workers and neighbors.
Responsible Innovation
Chemists continue looking for greener routes—better waste management, less hazardous alternatives, and new recipes that deliver performance without extra toxins. Renewable feedstocks or cleaner reactions might someday reduce the need for phenylphosphonic dichloride. Until then, careful stewardship means workers, end-users, and the environment all get proper attention.
Trust and Transparency in Industry
In any high-risk industry, people want transparency. I’ve seen communities demand open communication from plants using tricky chemicals like phenylphosphonic dichloride. Companies and regulators have to listen—public trust takes years to build, but only a single disaster to lose. Sharing safety data, publishing accident reports, and opening up about chemical handling practices helps. Those who work in chemical safety understand: if you cut corners, the price gets paid in lives or lawsuits.
What are the safety precautions when handling Phenylphosphonic Dichloride?
Understanding What You’re Handling
Phenylphosphonic dichloride packs a punch. It’s a sharp-smelling, highly reactive liquid that manufacturers use in making flame retardants, plasticizers, and other specialty chemicals. Anyone working with this stuff knows it’s not just another bottle on the shelf. The dangers run deep: skin burns, painful eye injuries, lung damage, and, if inhaled for too long, something far worse.
Why Strong Safety Rules Matter
You learn quickly that careless handling can ruin your day or land you in the hospital. Education and training should come before your first contact with this chemical. Don’t trust luck or cut corners. Companies must provide regular training updates since guidelines and recommended gear change, and even seasoned workers sometimes forget the basics.
What You Wear Really Counts
Put on chemical-resistant gloves, eye protection, and splash-proof suits—no excuses. Even one drop can cause a nasty chemical burn. If it splashes on skin or eyes, speed matters. Wash off with lots of water and seek help. Respirator masks make a big difference in workplaces with poor ventilation because fumes and vapors irritate airways fast. In some labs, I saw colleagues skip protection for "just a second" and pay the price with rashes or rough breathing for the rest of the week.
Controlling Spills and Storage
A tight, leak-proof container is a must. Keep it away from water since this chemical reacts violently with moisture, releasing hydrogen chloride gas. That gas can damage lungs and corrode equipment. From experience, a splash or spill can turn a quiet day into an emergency, so easy-to-access spill kits help. Sand and inert absorbents can save time during cleanup. Never store this chemical near bases, oxidizers, or anything that can ignite.
Ventilation Is Not Just Box-Checking
Good airflow in your lab or plant matters more than folks realize. Open windows won’t cut it; fume hoods or designated exhaust systems must pull vapors away from you. One afternoon in a school lab showed how quickly fumes could spread—even a tiny release can ruin indoor air, leading to headaches or worse. Keeping the place clean and organized helps spot leaks before problems grow.
Routes for Safer Workplaces
Supervisors have to lead by example, always wearing the right gear and encouraging questions. Workers without enough experience need to shadow those who’ve already handled the chemical safely. It's not just about following a checklist. Having regular emergency drills helps everyone know exactly what to do during an accident—waiting to act breeds confusion.
Label containers well, carry safety data sheets nearby, and keep emergency contact numbers within sight. Using these simple steps has protected colleagues, and sometimes, it's the only thing standing between a close call and a disaster. Routine checks, transparent communication, proper waste disposal, and practicing what you teach all add up to a culture where everyone trusts each other with their safety. That’s not naive optimism—it’s what keeps people alive.
How should Phenylphosphonic Dichloride be stored?
Understanding the Risks
Phenylphosphonic dichloride isn’t something you want near your lunch or anywhere children roam. I’ve worked around chemicals most of my career, and this stuff stands out for its nastiness. A small spill releases toxic fumes that fix the nose hair burning feeling in your memory. It reacts strongly with water, kicks off clouds of hydrogen chloride, and even a few drops on your clothes can end with a ruined pair of jeans or worse—serious burns.
The Right Storage Space
You need a spot built for danger. A locked chemical storage cabinet, made from corrosion-resistant steel, keeps temptation and accidents away. Choose a cool, dry room—heat speeds up chemical breakdown and moisture leads straight to a toxic mess. Never store it in glass, since it can corrode the stopper; high-density polyethylene bottles or Teflon-lined containers stop the slow eating away that happens over time. I’ve seen folks get tempted to “make do” with whatever’s handy. That shortcut can end with a trip to the hospital or a hazardous cleanup bill.
Segregation: No Roommates Here
In my first year in the lab, our supervisor had a rule—never store chlorinated reagents with water or alcohol. Mixing rivals means one crack in the shelf and you could start a reaction that throws gases nobody wants to breathe. Keep phenylphosphonic dichloride on its own shelf or in a dedicated cabinet, not next to anything containing water, bases, or strong oxidizers. One wrong combination and you won’t get a second chance.
Regular Checks and Safety Gear
Storage isn’t “set it and forget it.” Check bottles for leaks or cloudiness every month. Labels fade, so refresh them. I still remember the day someone tossed what they thought was empty packaging, only for a cleaner to open it and end up with severe burns. PPE like goggles, gloves, and an apron with a chemical splash resistance rating belong in arms’ reach—you wouldn’t work on a car without tools, so don’t handle chemicals without protection.
Training Beats Luck Every Time
No substitute exists for education. Give real training, not just a stack of papers to sign. Walk people through spill kits, eyewash use, and emergency ventilation. Speak from experience—the lab down the hall once left a bottle at the wrong temperature and ended up calling the fire service before lunch. Mistakes often start with skipped training steps.
Solutions That Last
Invest in purpose-built storage. Use digital logs to track checks and splits. Don’t forget the sensors—hydrogen chloride monitors will warn you before a noseful tells you the same thing. Local exhaust ventilation, either as a fume hood or vented cabinet, blocks the worst hazards. A written response plan saves time when things go sideways, which always happens sooner or later.
Complacency breeds danger with chemicals before you even notice. Stick to proven steps, push for ongoing safety education, and make sure the budget for quality storage never runs dry. Phenylphosphonic dichloride rewards careful habits with safety—and punishes shortcuts. Choose safety. Your coworkers and community rely on it.
What is the chemical formula of Phenylphosphonic Dichloride?
Understanding Phenylphosphonic Dichloride
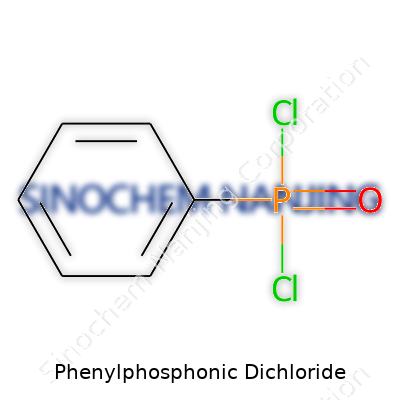
Phenylphosphonic dichloride has the formula C6H5P(O)Cl2. It brings together a phenyl group, a phosphorus atom, and two chlorine atoms, with one double-bonded oxygen attached to the phosphorus. I remember the first time I drew it out in a cramped university lab; balancing the structure wasn’t as simple as other phosphorus compounds. This detail sticks with me because it highlights the importance of each atom’s role when working with chemicals that shift the direction of a reaction pretty fast.
Why Chemical Structure Is Critical
Chemical industries depend on precise formulas to predict outcomes in reactions and avoid costly mistakes. Phenylphosphonic dichloride acts as a building block in creating flame-retardants, plasticizers, and some pharmaceuticals. Getting this formula wrong doesn’t just mean a theoretical error. The wrong atoms could trigger an unwanted side-reaction, lead to a hazardous situation, or make production batches unusable. For years, a friend of mine worked in a facility that handled organophosphorus compounds. They had protocol meetings nearly every week, and every single time, the formula—C6H5P(O)Cl2—got reviewed. Every chemist there knew that confusion between a sulfonyl and a phosphoryl group could ruin output and potentially risk lives.
Real-World Hazards
A misread label or typo on a chemical formula sometimes leads to exposure to toxic substances, especially where chlorine atoms come into play. Phenylphosphonic dichloride produces corrosive fumes if it hits water, so understanding what’s in the bottle keeps everyone in the lab safe. I recall one incident where a new technician misread the bottle and attempted dilution in a regular sink. The chemical splashed, reacting harshly with moisture, and triggered an emergency wash that afternoon. If more training had occurred on identifying and handling this compound by both its structure and hazards, the stress, risk, and clean-up might have been avoided.
More Than Just Letters and Numbers
For most people outside the lab, chemical names and formulas seem like a jumble. In practice, getting each letter and number right is as important as following a recipe for bread, except the stakes are much higher. Suppliers label each drum or flask with exacting detail because manufacturers downstream depend on it for synthesis. Errors slow production, waste material, and even spark regulatory trouble.
Ways Forward: Building Safer, Smarter Labs
Training on structural formulas should start early—and not just as a dusty classroom routine. Regular drills, digital flashcards, and supervised handling sessions helped me and countless others build muscle memory. Visual aids go a long way, too. In one well-organized lab, every major reactant and end product appeared on laminated info cards with color codes and easy-to-spot drawings. For new hires, this made all the difference. Hiring managers can set up orientation periods to reinforce these identities, and software that scans and double-checks order forms adds a backup layer for everyone rushing through busy days.
Staying Sharp Matters
Attention to chemical formulas like C6H5P(O)Cl2 can prevent mishaps and keep processes on track. This isn’t just pencil-pushing; it protects people, companies, and the environment. Anyone who’s faced beakers and towering stacks of glassware for days on end will agree—accuracy isn’t optional in this work.
Is Phenylphosphonic Dichloride hazardous to health or the environment?
What Really Happens with Exposure
People working around phenylphosphonic dichloride understand that health risks go beyond the label on the drum. A single whiff in the lab can send someone into a coughing fit, and a splash on the hand can sting for hours. Eyes water, skin reddens, throats burn. This isn’t just one person's story; toxicology records back this up. Direct exposure can cause chemical burns on skin or eyes, persistent breathing trouble, and, in heavy enough doses, damage deeper inside the body. There’s a reason for strict handling rules. I remember a researcher in our group who removed gloves too early. Even after a quick rinse, the irritation stuck around longer than any of us expected.
Years of data from the National Institute for Occupational Safety and Health show that accidental inhalation lands workers in clinics with respiratory issues, sometimes for weeks. Phenylphosphonic dichloride doesn’t just stop at an angry red mark—it can deepen the injury, sometimes without much warning. Toxicologists raise alarms about cumulative effects as well. This puts pressure on everyone in the chain, whether you’re producing the compound or processing downstream chemicals.
What Happens Outside the Lab
People rarely talk about what happens after a spill or leak. Phenylphosphonic dichloride reacts strongly with water, releasing hydrochloric acid and other nasty byproducts. These reactions have real consequences outdoors. Even with small leaks, the vapors can move fast. They can travel beyond fences, affecting not just workers but anybody nearby. A neighbor downwind from a chemical plant shared how, on a foggy morning, she got caught in a chemical cloud—just walking her dog. Numb lips, burning eyes, cough that stuck through lunch. Months later, that memory hasn't cleared up for her.
Researchers note that the environmental impact lingers. Local waterways take the brunt. Hydrolysis sends acidic byproducts downstream, where fish and small aquatic life face trouble. Community monitoring near factories in China and Europe found drops in aquatic health after nearby chemical leaks. Microbes involved in breaking down organic waste slow their work, so pollution sticks around longer. The drop in water pH stresses ecosystems, killing off insects and some plants outright.
What Can Actually Make a Difference
Prevention starts with airtight containers and real-time leak detection—something basic that gets overlooked until sirens sound. Plants that focus on automated shutoff systems report almost zero accidental releases. Staff training needs to go beyond videos or manuals. Full-scale emergency drills work. The teams that practice every month spot trouble before it gets out of hand.
In the bigger picture, factories that switch to closed systems with recovery units catch most leaks before they reach the air or soil. They turn potential waste back into useful feedstock, so there’s less pollution and less raw material bought. Environmental agencies could double down on monitoring downstream waterways, picking up early warning signs with regular sampling. Giving nearby residents fast alerts makes a difference, too. A text message about conditions helps someone dodge an invisible cloud during a morning walk. Simple but effective.
The track record on phenylphosphonic dichloride shows that meaningful improvement comes from better equipment, up-to-date training, and stronger local oversight. Health and ecology both face real threats, but smarter action can shrink those risks.
| Names | |
| Preferred IUPAC name | Phenylphosphonodichlorid |
| Other names |
Dichlorophenylphosphine oxide Phosphonic dichloride, phenyl- Phenylphosphonous dichloride Benzeneposphonyl dichloride |
| Pronunciation | /ˌfiː.naɪl.fɒsˈfɒn.ɪk daɪˈklɔː.raɪd/ |
| Identifiers | |
| CAS Number | 824-72-6 |
| Beilstein Reference | 1462254 |
| ChEBI | CHEBI:38770 |
| ChEMBL | CHEMBL15977 |
| ChemSpider | 20541 |
| DrugBank | DB11156 |
| ECHA InfoCard | 100.004.995 |
| EC Number | 208-194-3 |
| Gmelin Reference | 81883 |
| KEGG | C19206 |
| MeSH | D010629 |
| PubChem CID | 85721 |
| RTECS number | TH3850000 |
| UNII | F8L714J1FH |
| UN number | UN1817 |
| Properties | |
| Chemical formula | C6H5POCl2 |
| Molar mass | 266.98 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent |
| Density | 1.348 g/mL at 25 °C (lit.) |
| Solubility in water | Reacts |
| log P | 1.9 |
| Vapor pressure | 0.5 mmHg (20 °C) |
| Acidity (pKa) | 1.10 |
| Basicity (pKb) | BASICITY (PKB): 10.10 |
| Magnetic susceptibility (χ) | -55.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.578 |
| Viscosity | 1.339 cP (25°C) |
| Dipole moment | 1.84 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 352.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -444.7 kJ/mol |
| Hazards | |
| Main hazards | Corrosive, causes severe skin burns and eye damage, toxic if inhaled, reacts violently with water. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1-W |
| Flash point | 77 °C |
| Autoignition temperature | 250 °C |
| Explosive limits | Lower 1.2%, Upper 11% |
| Lethal dose or concentration | LD50 oral rat 630 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat: 1400 mg/kg |
| NIOSH | WX8225000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Phenylphosphonic Dichloride: Not established |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | IDLH: 5 ppm |
| Related compounds | |
| Related compounds |
Phosphorus trichloride Triphenylphosphine Phenylphosphonic acid Phenylphosphine oxide Dichlorophenylphosphine Diphenylphosphinic acid |

