Phenylmercuric Sulfanilamide: A Close Look at Progress, Pitfalls, and Future Paths
Unpacking the Past: The History Behind Phenylmercuric Sulfanilamide
Phenylmercuric sulfanilamide entered the world stage at a time when the search for potent antimicrobial agents swept through both chemistry and medicine. Its creation mirrored early 20th-century efforts to blend organic and inorganic chemistry, banking on both mercury's toxic punch and sulfanilamide's microbial-fighting spirit. While it never became a household name on the scale of penicillin, researchers gave it a shot in many preservation, industrial, and therapeutic applications. People looked to mercury derivatives for their ability to knock out tough bacteria and fungi, before the tide turned as scientific evidence stacked up regarding toxicity. Documenting this evolution matters, since today’s assessments of chemical use cannot be separated from yesterday’s enthusiasm for quick, sweeping solutions—and the cautionary tales that came from them.
Phenylmercuric Sulfanilamide Up Close: Properties and Profile
This compound combines a phenylmercuric group with a sulfanilamide. In practice, that points toward a white to off-white crystalline powder, only slightly soluble in water, more so in organic solvents. The inclusion of sulfur and mercury atoms gives rise to strong intermolecular bonds. Its distinctive profile, including melting point in the moderate range typical for organomercury compounds, gave it flexibility for formulation, whether in preservatives, germicides, or select pharmaceutical preparations. In the real world, users watch for stability in light and air; here, phenylmercuric sulfanilamide stands out for its relative resilience up until strong acids or bases show up, which can break down its structure. That kind of background knowledge proves crucial whenever handling or reformulating the compound for research or industrial use.
Naming and Identity: Avoiding Confusion
Chemical names tend to get unwieldy, so researchers and industry players developed a host of aliases for phenylmercuric sulfanilamide—sometimes listing it as phenylmercuric para-aminobenzenesulfonamide, or simply as PM-SA in lab shorthand. Keeping track of these names matters for very practical reasons. Cross-referencing studies or regulations becomes a nightmare if synonyms are used loosely. In nearly every story I’ve heard from lab workers, confusion over product identity leads to wasted resources, administrative headaches, and, at worst, safety risks that could have easily been avoided.
Preparation and Tinkering: How Synthesis Shapes Use
Preparation methods often start with reacting phenylmercuric acetate or nitrate with sulfanilamide derivatives in carefully controlled pH and temperature settings. Small differences in reaction times, purification steps, or intermediate compounds can influence both the yield and the impurity profile. Having spent stretches of time working in chemical labs, I’ve noticed that shortcuts or scaling up without deep dives into the reaction details directly affect how the final material performs—and whether costly waste can be avoided. On the technical front, modern labs depend much less on mercury chemistry, but in specialty synthesis, control over these parameters remains a make-or-break difference.
Frontiers of Use: Application Areas and Scientific Curiosity
Phenylmercuric sulfanilamide once found broad utility as an antimicrobial agent in creams, ointments, eye drops, and topical wound treatments. For a while, it also had a place in preservatives, as mercury’s broad-spectrum kill rate was tough to rival. Regulatory winds have since blown cold, yet researchers revisit its structure in medicinal chemistry, either to understand mercury’s biological interactions or to develop safer analogs inspired by its mode of action. Genuine innovations often grow out of painstaking study of “old” scaffolds. In my circles, I’ve seen academic teams draw lessons from the detailed interactions of mercury compounds to strategize how persistent agents can be replaced without losing antimicrobial punch.
Safety Isn’t a Footnote: Handling and Health
Safety always rides up front whenever mercury enters the scene, and phenylmercuric sulfanilamide is no exception. Even in the mid-20th century, before today’s hazard labeling took hold, practitioners reported skin rashes and renal disturbances linked to repeated contact. As data poured in, chronic low-level exposure raised alarm bells about cumulative neurotoxic and nephrotoxic risks. Proper protective gear—gloves, goggles, fume hoods—became as essential as knowing the CAS number. I remember a tenured safety officer repeating that “no one gets points for ignoring MSDS sheets,” and the mantra holds up whenever working with organomercury compounds. Legislation like the Minamata Convention placed lasting restrictions on the entire class, reinforcing the need for alternatives whenever possible.
Hazards in the Lab and Beyond: Toxicity Realities
Lab experiments tell only half the story. Out in the real world, mercury doesn’t just disappear once a product is discarded or washed away. Reports detail environmental accumulations, especially in aquatic systems, where organic mercury morphs into methylmercury, building in fish, and up the food chain, even reaching dinner plates and baby formulas halfway around the planet. Data published over decades link organomercury exposure with learning and developmental challenges, particularly in children. This carries heavy weight in any discussion about future allowable uses. Outdated disposal practices, such as sending residues down the sink, led to tragic consequences in both wildlife and small rural communities that lacked the resources to clean up after old chemical works. Strong labeling, clear usage limitations, and robust waste management protocols reflect the lived realities of scientists, families, and downstream users.
Redrawing the Risk Map: Technical and Legal Barriers
No company wants the reputation hit that comes from a mercury release, so over time, various national and international bodies set technical and labeling standards for compounds like phenylmercuric sulfanilamide. Documentation now emphasizes batch purity, storage temperature boundaries, restricted applications, and mandatory spill response strategies. Labs and facilities that keep working with these compounds stand under the gaze of regulators, environmental activists, and local communities. The lesson here? Safe handling is a group effort; it only works if everyone from procurement staff to lab techs joins in on training and vigilance. From my own experience, an honest audit of actual chemical use and disposal habits reveals gaps far quicker than any annual safety review or distant compliance office memo.
Paths Forward: Research, Replacements, and Ethical Choices
Modern R&D on phenylmercuric sulfanilamide splits in two. On one hand, basic researchers keep studying organomercury interactions with bacterial structures to inspire next-generation antimicrobials that eliminate pathogens while leaving humans unharmed. Other scientists invest their time in environmental chemistry, hunting for cheap, scalable ways to break down and neutralize mercury residues. There’s a lot to learn from past successes and failures—both in how quickly chemicals strutted onto pharmacy shelves and how stubborn some of their negative legacies remain. Sustainable chemistry advocates now look for “benign by design” rules, setting out from the drawing board with a hard line against persistence and bioaccumulation.
Beyond the Present: What Might Come Next
Looking ahead, the main challenge involves balancing antimicrobial power with environmental and human safety. Phenylmercuric sulfanilamide offers chemists a cautionary template—a potent weapon, sure, but one that introduced cascading hazards. Tomorrow’s products need to sidestep those pitfalls. This calls for coordinated efforts among scientists, engineers, clinicians, and everyday people forced to face the fallout from yesterday’s choices. Progress shows up slowly, through better chemical education, cross-disciplinary studies, and policy frameworks that address both technical performance and the realities of long-term use. If researchers, regulators, and users stay honest about limits and costs, we may yet turn lessons from the tricky history of organomercury chemistry into working blueprints for safer, smarter, and more responsible innovations.
What is Phenylmercuric Sulfanilamide used for?
Understanding Phenylmercuric Sulfanilamide
Phenylmercuric sulfanilamide slips into the conversation when talking about chemicals that try to control bacterial growth. This compound, a combination of organic mercury and sulfanilamide groups, shows up in certain pharmaceutical and cosmetic products. The world once saw lots of preservatives loaded with mercury-containing chemicals because these showed remarkable strength against bacteria and fungi. In practice, phenylmercuric sulfanilamide made its way into ointments, creams, and even eye drops for its ability to keep things clean over time.
Why Manufacturers Turned to It
What stands out with mercury-based compounds like this one is their strong antimicrobial effect. Back before the risks of mercury exposure became clear, companies leaned into these preservatives to prolong the shelf life of medicines and personal care products. People wanted products that stayed safe from bacteria—nobody likes the thought of an eye infection from a contaminated dropper. At the time, few alternatives matched the power of mercurials for warding off unwanted microbes in water-based mixtures.
I came across older pharmacy guides listing phenylmercuric sulfanilamide as part of many topical creams. The thinking was simple: fight infection and spoilage. It was not unusual to see small amounts blended into ointments for burns or wounds. You might even spot it listed in some over-the-counter antiseptic sprays and nasal preparations produced before the 1980s.
Health Risks Get Recognized
Better toxicology research opened everyone’s eyes to mercury’s dangers in the late 20th century. Both organic and inorganic mercury accumulate inside the body over time, especially with regular exposure. Health authorities found mounting evidence linking mercury compounds to neurological issues, kidney problems, and allergic reactions. The skin absorbs these compounds, and the eyes prove even more sensitive to their effects.
The U.S. Food and Drug Administration responded to growing evidence by tightening rules for mercury in medicine. Many developed countries now reject Mercury-based preservatives for most personal care products. In my time working alongside pharmacists, I watched companies pull these ingredients from their formulas, switching to safer antimicrobial agents like parabens or benzalkonium chloride. Some older stock still turns up on dusty drugstore shelves or in legacy collections, but you won’t spot them in most mainstream products anymore.
Global Moves Away From Mercury
The push to safeguard public health goes beyond product labeling. International agreements like the Minamata Convention urge countries to phase out unnecessary mercury uses, reflecting hard lessons learned about pollution and toxicity. Governments now focus on education, safe disposal, and stricter regulation. Mercury poisoning, once associated mainly with industrial disasters, now serves as a lesson about underestimating everyday exposures.
Searching for Modern Solutions
People expect safe, long-lasting medicines and cosmetics. With bans in place, scientists keep searching for alternatives that do the job without risking user health. If a preservative turns out to cause more trouble than it prevents, smart policy and innovation set the industry straight. Beyond checking ingredient lists, doctors and pharmacists help patients navigate product choices—especially those who react badly to common preservatives. Patch testing and better packaging contribute as well.
If you’re digging through old medicine cabinets or inherit supplies, it’s worth checking labels. Any bottle listing “mercuric” compounds belongs in a hazardous waste bin, not your daily care routine. Every step away from potentially harmful chemicals gives peace of mind, whether you’re treating a scratch or just using a new face cream before bed.
Is Phenylmercuric Sulfanilamide safe for human use?
Trust Earned Through Straight Facts
Whenever doctors or pharmacists come across unfamiliar chemicals, safety should come before convenience. Phenylmercuric sulfanilamide looks like just another mouthful of a chemical name, but hiding behind its syllables is a story about safety, trust, and science catching up with old habits. For decades, this substance ended up as a preservative in creams, ointments, and topical medicines. Its job? Stop bacteria and fungi from turning medicine into mush. The intent was good. But good intentions don’t always mean good outcomes, especially where mercury is involved.
Mercury Compounds: No Such Thing as a Harmless Dose
Take it from researchers who dig into toxicology—mercury compounds can cause damage even when they’re used in small amounts. Our bodies have no systems built to safely get rid of mercury, so it sticks around. This isn’t paranoia. The U.S. Food and Drug Administration (FDA) banned most mercury-based ingredients from over-the-counter drug products starting in the 1990s. The European Union phased out phenylmercuric compounds for similar reasons, stating high risks of toxicity compared to minimal benefits. Regulatory bodies rarely agree on everything, but mercury gets few defenders nowadays.
What Happens When People Use It?
Experience counts for something, and stories from patients stand out here. Reports include burning, rashes, and even more severe allergic responses. Researchers tracked those reactions back to both direct skin exposure and systemic buildup over time. The mercury in phenylmercuric sulfanilamide doesn’t just vanish. In the worst cases, it can lead to kidney damage, nervous system changes, and developmental issues for fetuses. Medical literature shows infants and pregnant women are especially vulnerable to these effects. No one welcomes risk without a real, proven reward.
Science Can Do Better
Plenty of safer preservatives work well in creams and ointments. Parabens, sorbic acid, and modern antimicrobial agents keep products stable with far less risk. Some people worry about parabens, too, but regulatory agencies such as the European Medicines Agency and the FDA still see them as much less risky than anything containing mercury. That choice between mercury—an undisputed neurotoxin—and a well-studied alternative should be easy.
What People Can Do About It
Ask questions about what goes into medicine and personal care products. Don’t settle when the label lists ingredients you don’t trust. Pharmacists owe it to patients to check up on the latest guidance—no more coasting on outdated information from dusty pharmaceutical guides. Product recalls and updated recommendations happen for a reason. That isn’t fearmongering; it’s staying safe. If something on the label looks like a chemical tongue-twister with “mercuric” inside, leave it on the shelf. Health comes from informed choices, not nostalgia for old formulations.
Choosing Health Over Habit
People trust doctors and companies with their health, expecting that no ingredient sneaks into a formula unless it truly belongs. Phenylmercuric sulfanilamide doesn’t belong in medicines anymore. Real safety means replacing it with cleaner, safer options. Let’s keep learning from the past—nobody benefits from ignoring history or pretending modern science hasn’t moved on.
What are the side effects of Phenylmercuric Sulfanilamide?
Living With a Name Hard to Pronounce
Phenylmercuric sulfanilamide rarely pops up in average conversation. Unpacking its risks means tracing back to older formulations in medical history, since this compound once found a home in creams, eye drops, and powders for its antimicrobial punch. Mercury comes baked into its core. The very thing that gave it some medical usefulness has also set off alarm bells for anyone worried about safety.
What the Body Faces
Once this chemical gets into the body—on purpose or by accident—problems start to line up. There’s the basic skin irritation: itching, burning, peeling, and sometimes blisters right where people applied a cream. Redness can pop up quickly. With repeated use, the burning gets worse. Scratching makes the situation spiral. I saw a neighbor’s child get a rash from an old antiseptic powder and it never quite cleared until the stuff was tossed.
Eyes react sharply too. Mercury-based agents in eye drops often trigger stinging that doesn’t quit. I’ve read about swelling and watery eyes that outlast the medicine. Using mercury preservatives can even break down the protective layer of the eye, turning a cure into a new problem. In severe cases, people complain about blurry vision or increased sensitivity to light. When I volunteered at a local health clinic, there were strict rules about what could be prescribed for anything near the eyes—mercury compounds were not even up for discussion.
Mercury’s Hidden Threat
Here’s the bigger danger: mercury builds up. Even small amounts, over time, can poison tissues. Early toxicity signs often slip by unnoticed. People might feel a metallic taste, get headaches, find themselves unusually tired, or start to lose weight for no obvious reason. Loss of appetite seems to hit most users first. Longer exposure means tremors, mood swings, or, worst of all, kidney trouble. Medical evidence on mercury tells the same story: the nervous system takes the hardest hit, leaving real damage behind if the exposure pile up.
Reproductive and Environmental Impact
The concern doesn’t stop with skin and nerves. Mercury agents—including phenylmercuric sulfanilamide—do cross into breast milk and through the placenta, opening the door to risks for babies and pregnancies. Studies show that prenatal exposure to mercury links to developmental delays. I know pediatricians who won’t let anything even close to mercury get used around expecting mothers or young children. It just isn’t worth the gamble.
Mercury compounds don’t break down well after entering the water. Throwing old tubes of cream in the trash can send mercury out into the soil and groundwater. This environmental hazard lingers far after the packaging starts to crack, and it contaminates food sources, especially fish. The World Health Organization and EPA both flag mercury preservatives for just this reason.
Better Safe Than Sorry
Modern medicine has safer alternatives. Manufacturers pulled most mercury-based antibiotics and preservatives off pharmacy shelves decades ago. Still, old tubes and bottles can hide in medicine cabinets or pop up at thrift stores. Tossing these products at hazardous waste sites makes a difference. Education goes a long way; I always remind family and friends that medicine should evolve with time, and old isn’t always gold—especially where mercury’s involved.
The strongest advice from doctors and scientists says, read the label and steer clear of anything with names ending in "mercuric." Mercury damage doesn’t mend overnight, and the risk just isn't worth it for a little extra shelf life or infection control.
How should Phenylmercuric Sulfanilamide be stored?
Storing a Powerful Compound Safely
Phenylmercuric sulfanilamide might not pop up in every household, but it holds a spot in laboratories and some medical contexts because of its antimicrobial punch. Its use traces back decades, but its very nature—combining mercury with sulfanilamide—raises red flags for safety. This isn’t just some ordinary chemical. From my years in science labs, handling even small amounts taught me one thing: don’t cut corners on storage.
Recognizing Its Risks and Sensitivities
The compound brings together two dangerous properties: the toxic bite of mercury and the reactive tendencies of sulfanilamides. You can’t treat this like you would a bottle of acetone or even concentrated acids. Law and common sense both demand respect here. Inhaling the dust or vapors, touching it with bare hands, or letting it contaminate common workspaces carries real health risks. Mercury compounds in particular show up in environmental incidents, and even tiny spills put coworkers or local wildlife at risk.
Protection Starts with Environment
A dry, cool, and stable spot works best. Heat or direct sunlight kicks chemical reactions into gear or speeds up degradation, and this leads straight to unsafe conditions. Temperature swings crack glass containers, and that’s a nightmare scenario. Fluctuating humidity can corrode metal lids, seeping contaminants into the air. A windowless chemical storage room or ventilated cabinet does well. In research jobs, I saw that a sign clearly listing “TOXIC - MERCURY COMPOUND” on the door kept everyone alert and saved a few near-misses.
Keeping Containers Tight and Secure
Sealed glass or specially lined plastic holds up best. Phenylmercuric sulfanilamide won’t play nice with rusty caps or unprotected metal. One drop leaking into the wrong shelf slot becomes an emergency. Make sure containers can’t tip or break in a shake or a bump—sturdy shelving with easy access wins every time. Segregating mercury compounds from acids and bases isn’t just a bureaucratic rule; mixing spills create hazardous gases or unexpected explosions.
Labels That Leave No Doubt
Every label needs to do more than check a regulatory box. Handwritten scrawls fade and confuse. Strong, waterproof labels screaming “MERCURY - DO NOT TOUCH” have saved more than one new technician from a close call. Dates and concentrations help at audit time. I’ve learned that opening a mystery jar ends up in lengthy incident reports and ruined gloves.
Managing Waste and Cleanup
After experiments, leftover phenylmercuric sulfanilamide must not linger on benches or go down the drain. Closed, labeled hazardous waste bottles that stay away from common trash bins remove headaches and injuries. Partnering with specialized waste disposal companies means harmful byproducts avoid landfill disasters. Chemical spill kits—gloves, absorbent pads, mercury-specific vacuums—should stay stocked and checked. The best cleanup happens the moment something spills, not a day later.
Building Good Habits
Training must reach everyone in the lab, not just the head chemist or longtime employees. Labs run safer when everyone buys in. In graduate work, buddy systems for handling toxic chemicals caught mistakes early and built real confidence. Supervisors walking the talk—labeling, double-checking seals, and running practice drills—bring results no classroom slideshow can match.
Are there any alternatives to Phenylmercuric Sulfanilamide?
Why People Rethink Mercury Compounds
Every chemist who has spent time in a lab knows that mercury deserves a wary respect. Phenylmercuric sulfanilamide shows up in some medical and cosmetic formulas due to its ability to wipe out bacteria and fungi. Still, the mounting evidence has forced folks to look for better options. Mercury doesn’t just slip out the back door after use. It sticks around in the environment. It works its way up the food chain and eventually turns up in people. Health experts link long-term mercury exposure to kidney damage, cognitive issues, and developmental setbacks in kids. Meanwhile, more consumers read labels and press for cleaner options. The need for practical, effective, and less risky substitutes stands out more than ever.
Alternatives People Trust More
Parabens once grabbed plenty of that shelf space previously reserved for mercury preservatives. You can thank their track record for staving off mold and yeast in creams and gels. That glow has faded in recent years. More studies point out how parabens mimic hormones, which brings a whole new can of worries.
Enter the next wave: Phenoxyethanol picked up steam, landing in many lotions and ointments. Scientists back its safety in lower concentrations. Studies report fewer side effects unless you really pile it on. Personal experience with phenoxyethanol has shown it’s easier on the skin than many older compounds, and it doesn’t pack up environmental baggage.
You will also run into organic acids, like sorbic acid and benzoic acid. These tend to show up more in food and mild personal care products. They break down pretty quickly and don’t stick around the way mercury compounds do. For mild formulas, these acids hold up, though they don’t tackle every pathogen.
Making New Preservative Choices Work
Every substitution trade-off comes with its challenges. Fewer people react to phenoxyethanol, but using it in the wrong blend or at too high a level irritates skin. Parabens last a long time but raise hormone concerns. Benzoic and sorbic acids work best at a low pH, so the rest of the formula needs to match.
Plant-based preservatives have gotten more attention. Brands looking to build a natural image sometimes choose grapefruit seed extract or tea tree oil. The problem is, these hits and misses pile up; plant extracts break down under high heat or light, and sometimes molds slip past. In tests, synthetic preservatives keep products safer for longer stretches.
The Reality of Finding the Right Substitute
Any chemist swapping out phenylmercuric sulfanilamide will tell you: Test, test, and test again. Success means screening possible substitutes for both safety and actual performance in real-world storage and use. Regulators worldwide press for lower-toxicity choices, nudging companies to keep searching for smarter chemistry.
Switching out mercury-based formulas rarely happens overnight. Small producers struggle to afford broad testing. Consumer education takes time, and skepticism always has a seat at the table. Even so, with enough industry cooperation and a hard look at health impacts, the future promises safer blends that everyone — from the factory to the home — can handle with less worry.
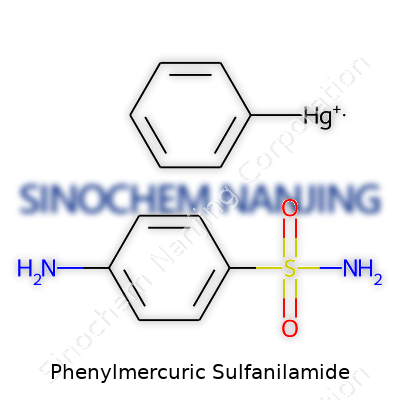
| Names | |
| Preferred IUPAC name | Phenylsulfanylaminomercury |
| Other names |
Mercurochrome sulfanilamide Sulfanilamide mercury Phenylmercury-p-sulfanilamide |
| Pronunciation | /ˌfiː.nɪl.məˌkjʊər.ɪk sʌlˌfæ.nɪlˈæm.aɪd/ |
| Identifiers | |
| CAS Number | 100-56-1 |
| Beilstein Reference | 2348606 |
| ChEBI | CHEBI:75010 |
| ChEMBL | CHEMBL2106728 |
| ChemSpider | 20632845 |
| DrugBank | DB01361 |
| ECHA InfoCard | 03a09b7a-0841-47d1-b424-aa838dc4277f |
| EC Number | 212-099-7 |
| Gmelin Reference | 2119985 |
| KEGG | C18746 |
| MeSH | D010631 |
| PubChem CID | 21836505 |
| RTECS number | OV9275000 |
| UNII | DE9M0J32R7 |
| UN number | UN2025 |
| Properties | |
| Chemical formula | C12H12HgN2O2S |
| Molar mass | 557.93 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 0.5 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.1 |
| Vapor pressure | Negligible |
| Acidity (pKa) | pKa = 10.4 |
| Basicity (pKb) | 10.47 |
| Magnetic susceptibility (χ) | -1021.0 × 10^-6 cm³/mol |
| Refractive index (nD) | 1.67 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.21 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | Unknown |
| Pharmacology | |
| ATC code | D08AJ05 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or absorbed through skin; causes damage to kidneys, liver, and central nervous system; may cause allergic skin reactions; very toxic to aquatic life. |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H300 + H330: Fatal if swallowed or inhaled. H310: Fatal in contact with skin. H373: May cause damage to organs through prolonged or repeated exposure. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P210, P260, P262, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P313, P314, P330, P391, P501 |
| NFPA 704 (fire diamond) | 2-2-3-W |
| Lethal dose or concentration | Lethal dose or concentration: **LD50 (oral, rat) 740 mg/kg** |
| LD50 (median dose) | LD50 (median dose): 164 mg/kg (rat, oral) |
| NIOSH | SY8225000 |
| PEL (Permissible) | 0.1 mg/m³ |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | IDLH: 10 mg/m3 |
| Related compounds | |
| Related compounds |
Phenylmercuric nitrate Phenylmercuric acetate Mercuric chloride |

