Phenylmercuric Nitrate: A Deep Dive from Labs to Real-World Uses
Historical Development
Chemistry across the twentieth century moved fast, and phenylmercuric nitrate rode that wave as well as any other compound. Its earliest forms arrived around the late 1800s, when combining organic molecules with metals lit up possibilities for new drugs and materials. The need for solid, stable antiseptics during the early twentieth century, especially with hospitals stretched thin by infection control woes, kicked research into gear. By the 1920s, phenylmercuric salts turned up on medical shelves, thanks in large part to the practical results these compounds delivered—broad spectrum antimicrobial activity, stability in creams and solutions, and reliability under harsh storage conditions. Scientists kept tinkering, refining methods for large-scale synthesis by the 1950s, at which point phenylmercuric nitrate took root in ophthalmology, topical creams, and industrial labs. As safety awareness grew, the chemical’s dual status—prized tool and tricky toxin—sparked waves of regulatory scrutiny and research, shaping how the compound gets handled even today.
Product Overview
Phenylmercuric nitrate isn’t found as some rare curiosity on a dusty shelf. For decades, manufacturers sold it as a white to off-white powder. Pharmacies stocked bottles labeled with strict handling warnings. Chemists knew its range; not just a laboratory chemical, it lived in eyedrops, ointments, and even paints, appreciated for strong preservation and anti-fungal powers. These advantages put it in crosshairs once regulatory landscapes started shifting, as concerns about mercury in health products led to hard questions and dropped approvals. In practice, you had to respect the balance: efficacy on one side, hazard on the other.
Physical & Chemical Properties
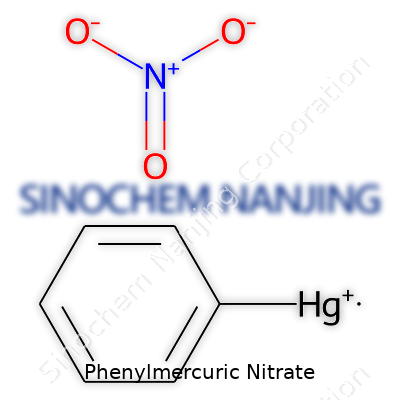
In practical terms, phenylmercuric nitrate gives off no smell, burns at high heat, and dissolves best in water or alcohol. Its white crystalline texture is dense—mercury adds a lot of heft. The formula, C6H5HgNO3, features a phenyl ring bonded to mercury, which acts as a carrier for both organic and inorganic traits. In real use, its decomposition leaves behind mercury oxide and phenol, both hazardous in wrong hands or under poor ventilation. It reacts poorly with acids or reducing agents, making clear why chemical storage demands careful control.
Technical Specifications & Labeling
Any bottle or jar of phenylmercuric nitrate wears a label with more warnings than most household cleaners: “Toxic by inhalation, ingestion, or skin contact,” “Environmental Hazard,” and full hazard pictograms. Chemical suppliers stamp out certificates of analysis with details like melting point (around 110°C), moisture threshold, purity often above 99%, and exact mercury content. Batch numbers allow traceability, a must for anyone using this in regulated lab settings or pharmaceutical processing. Specialized secondary containment and robust steel shelving reflect the seriousness required for proper storage.
Preparation Method
Making phenylmercuric nitrate always begins with fresh phenylmercuric acetate or phenylmercuric chloride, made beforehand using benzene and basic mercury salts. Through a swap reaction with nitric acid, technicians yield the nitrate, filter, and recrystallize it to remove impurities. This multi-step process happens under powerful ventilation, with full protective gear standard. Any scrap, wash water, or residue counts as hazardous waste, following national disposal laws that often resemble hazardous material routes. On the synthesis side, strict controls on temperature and mixing prevent runaway reactions.
Chemical Reactions & Modifications
Phenylmercuric nitrate doesn’t just sit inert; it lends itself to organometallic reactions, especially halogen exchanges and nitration steps. In the pharmaceutical sector, chemists sometimes adjust its backbone, adding or swapping functional groups to shape properties like solubility for special applications. Heating leads to dangerous decomposition, which kicked off plenty of debate about storage and shelf-life. In labs, users find it handy as a catalyst or as a reference in testing mercury content for analytical chemistry. The compound’s reactivity with sulfide-bearing chemicals underscores its strict incompatibility with many standard reagents.
Synonyms & Product Names
Phenylmercuric nitrate shows up under dozens of catalogue names, like mercury, phenyl nitrate or PMN, and at times in old medical paperwork under Phenylmercurinitrate, Mercurophen, or Nitrophenylmercuric. Vendors catalog it with different spellings and abbreviations, so researchers cross-check synonyms before ordering or scanning old toxicity studies.
Safety & Operational Standards
Working with phenylmercuric nitrate means planning for exposure control first, not getting seduced by its utility. Labs and factories writing safety protocols go well beyond dust masks, mandating enclosed glove boxes, local exhaust hoods, and mercury monitors. Cleanup kits, spillage procedures, and staff training focus on immediate decontamination and zero-tolerance for spills. Guidelines—driven by OSHA, REACH, and EPA—set exposure caps in micrograms per cubic meter, and annual health checks track even trace worker exposure. Pharmacies once compounding topical creams with phenylmercuric nitrate—especially for the eyes or wounds—now face regulatory bans and educate about safer alternatives.
Application Area
Pharmaceuticals once treated phenylmercuric nitrate as a staple in eye drops, nasal sprays, and anti-microbial creams thanks to its sturdy germ-killing power. Outside medicine, it gave paints mildew resistance and protected latex from spoilage. In research, it became a tool for studying organic mercury chemistry, providing a reference standard for spectroscopic and toxicological studies. The modern landscape, with its strict mercury restrictions, shrank these uses sharply. Today, you spot phenylmercuric nitrate mainly in tightly regulated industrial or academic labs, not on consumer shelves. Some niche medical preparations in countries with different regulatory frameworks may still use it, but most developed countries consider it a relic of older medicine cabinets.
Research & Development
Through the 1960s and 1970s, pharmaceutical and chemical companies invested heavily in ways to tame phenylmercuric nitrate—searching out analogues with lower human toxicity but the same antimicrobial punch. Studies in microbiology showed it worked well against bacteria and fungi, yet toxicity shadowed every potential breakthrough. Modern research pivots to environmental tracking, unveiling just how persistent this compound becomes in soil, water, and even the food chain. Analytical chemists tune instruments to track parts per billion, sparking innovations in sensing and rapid detection. Despite the decline in direct human use, research into remediation, waste treatment, and molecular modification keeps phenylmercuric nitrate in the scientific conversation, urging more responsible mercury stewardship.
Toxicity Research
Long-term studies connect phenylmercuric nitrate to kidney and nervous system damage, bioaccumulation, and teratogenic effects. Early reports underestimated the risks—even topical exposure led to systemic absorption, as confirmed by blood mercury levels in lab staff and patients. Now, toxicity mechanisms are better mapped: mercury binds to sulfhydryl groups in proteins, blocking essential enzyme activity. Mercury’s persistence in the environment, especially from wastewater or improper disposal, haunts regulators and drives strict vigilance. Animal studies, alongside population reports, helped phase out this preservative in drugs for infants and pregnant women. The upshot remains clear—every step, from synthesis to disposal, requires controlled handling and robust medical surveillance.
Future Prospects
Phenylmercuric nitrate symbolizes the evolutionary arc of chemistry, from essential tool to legacy risk. As alternatives emerge for medical and industrial preservation—quaternary ammonium compounds, silver-based agents, and advanced synthetic antimicrobials—the market for phenylmercuric nitrate dries up. Specialists still see it pop up in analytical applications and as reference material. Environmental technology might carve out a comeback, with research pounding away at new mercury capture techniques or ways to remediate old contamination. The future—if it still involves phenylmercuric nitrate at all—rests not on new uses but on responsible retirement, safe disposal, and the lessons scientists draw about balancing utility and hazard in chemical innovation.
What is Phenylmercuric Nitrate used for?
The Story Behind phenylmercuric nitrate
Dealing with substances that carry a warning label tends to make people uncomfortable, and “phenylmercuric nitrate” triggers that response. Most folks don’t run into it at the grocery store. Yet, this chemical found its way into a host of medical and industrial products over the decades. I first stumbled into it during college research, flipping through an old pharmacology guide. My professor—proudly old school—explained how this compound kept eye drops safe back when preservatives like benzalkonium chloride weren’t everywhere.
Medical Uses and Risks
For a long stretch, phenylmercuric nitrate showed up in eye drops and nasal sprays. Its value boiled down to stopping bacteria and mold. Mercury has a nasty reputation now, but in the early- and mid-1900s, companies looked the other way because infections were the bigger threat. There’s no sugar-coating that mercury accumulates in the body and causes health problems. In relaxed regulations of decades past, the benefits seemed to outweigh the risks, at least on paper.
Regulators in the United States and abroad eventually changed the story for phenylmercuric nitrate. Scientists recognized the dangers. Chronic mercury exposure links to nervous system damage and kidney problems. In 1998, the FDA called for drug manufacturers to swap it out, especially in products that stick around in the body. Eye drops for infants or folks with damaged corneas made the situation worse because the preservative lingered, potentially harming vulnerable groups. Today, new ophthalmic products in the US avoid it altogether. Some countries still allow trace amounts, but the writing is on the wall: safer chemicals now take its place.
Behind the Chemistry: Other Uses
Step outside medicine, and phenylmercuric nitrate pops up in odd corners of industry. Old paint formulas used it as a fungicide. Certain adhesives and caulks used to rely on it for mold control. My landlord, a retired contractor, still shakes his head at how many people painted their kitchens with toxins in the ‘60s and ‘70s, thinking only about mildew on their windowsills.
Environmental groups tracked residue in landfills and water runoff. As municipal water labs improved testing, scientists started to find traces of phenylmercuric nitrate—and other mercury preservatives—spreading farther than most people ever guessed. Once the scope became clear, countries tightened regulations. Factories now label waste much more strictly, and safer treatments for fungus and bacteria edge out the old standbys.
Seeking Solutions
The story isn’t about demonizing past choices. People were making do with the tools they had. Even now, finding the right balance between safety and effectiveness can challenge manufacturers. Companies switching away from mercury tend to reach for parabens, benzalkonium chloride, or newer preservatives. EWG and medical journals compare toxicity data, hunting for anything with less risk after long-term exposure.
Public health education needs to keep up. Students in biology and chemistry classes should dig into why older solutions faded out and what new options promise. Doctors and pharmacists talk to patients about preservatives in products, encouraging folks with allergies or health issues to ask questions. A more informed public can catch outdated products still hanging around.
Modern technology keeps offering alternatives, both in labs and in how information gets shared. Regular testing, regulations with teeth, and transparent ingredients lists stand out as the simplest ways to avoid repeating the old mistakes. With safer substitutes, better awareness, and open debate, there’s hope for a healthier balance between preserving products and protecting people from harm.
Is Phenylmercuric Nitrate safe for human use?
Understanding the Risks
Phenylmercuric nitrate used to serve an important job in eye drops and ointments. Over time, people started paying more attention to what’s actually inside these medications. Turns out, this compound contains mercury, which brings a whole set of concerns.
Doctors, pharmacists, and researchers have raised repeated alarms about exposure to mercury, even in small amounts. Studies show that even a tiny bit of mercury absorbed through the skin can gradually build up in the body. I’ve seen how the medical community stresses extra caution, especially in products that touch the eyes or skin where absorption rates are higher. Poisoning isn’t a word most folks like to hear when dealing with over-the-counter treatments or prescription medications.
Why Mercury-Free Matters
Digging through medical research, it’s not hard to spot a pattern—a number of reports link phenylmercuric nitrate with allergic reactions, skin irritation, and risk of mercury toxicity. The eyes are especially sensitive, and I would hate to take a gamble with vision for the sake of preserving eye drops a little longer. The US Food and Drug Administration pulled its approval for several mercury-based preservatives in the 1990s, mainly because safer alternatives exist. The European Medicines Agency went down a similar road.
My own take, shaped by years spent covering stories in healthcare, is to avoid compounds that carry these kinds of long-term risks. Mercury gets trapped in the body over time. Exposure can affect the kidneys, nerves, and even thinking and memory. Pregnant women and children are at the greatest risk, which hits home for many parents looking for trustworthy wound or eye care.
What Safer Preservatives Bring to the Table
The good news? Developers and pharmacists have cooked up a whole range of modern preservatives that don’t carry the same risks. Benzalkonium chloride, sodium perborate, and stabilized oxychloro complex are just a few examples. These have clear safety records and offer plenty of protection against bacteria. Based on peer-reviewed evidence, these alternatives don’t stick around in the body or pose the same risk to sensitive organs.
Walking on the side of caution serves everyone better, especially when effective, lower-risk options exist. Every time I talk with healthcare professionals about safe medications, they bring up the burden of proof: if something safer exists, why use what’s more hazardous?
Steps Toward Safer Choices
Anyone using medicated creams, drops, or eye products can check ingredients with a quick glance or a call to their pharmacist. If prescribers still suggest older formulations, asking for an alternative never hurts. Medical providers have an obligation to stay updated on ingredient safety. Most do, and that’s reassuring.
No one wants to trade convenience for risk if it can be avoided. Most reputable brands have already switched away from mercury-based preservatives. Folks with older medications tucked away in a drawer can check expiration dates and ingredients, then ask their doctor if it’s time for an upgrade.
Looking at the Bigger Picture
Medical science tends to move on as more evidence comes to light. Phenylmercuric nitrate no longer makes sense in today’s shelves—safer options are available, and nobody benefits from unnecessary exposure. Trust builds over time when companies and doctors listen to safety data and adjust their offerings. That’s how confidence in medicine grows.
What are the potential side effects of Phenylmercuric Nitrate?
Understanding the Real Consequences
Phenylmercuric nitrate found its way into healthcare as an antiseptic and preservative, especially in eye drops and topical ointments. At first glance, this might sound promising. After all, products get more shelf life and better protection against bacteria. Turn the bottle around, though, and there’s mercury. That’s never a comforting word. Years of medical experience show that anything containing mercury deserves respect and caution—side effects often slip in quietly and stick around much longer than expected.
The Body’s Unwelcome Response
Exposure, even at low levels, can upset almost every organ system. In eye products, phenylmercuric nitrate soothes infection but leaves a risk of allergic reactions, irritation, and deeper tissue damage. One patient came in with what seemed like simple redness after using prescription eye drops. It didn’t clear up as usual. The skin around her eyes swelled, and she started to notice blurry vision. Her doctor traced the cause back to phenylmercuric nitrate—a clear reminder that small doses carry serious weight.
The most troubling impact shows up with repeated, longer exposures. Mercury accumulates. In the world of public health, this sparks worry about mercury poisoning, bringing neurological symptoms to the surface: memory problems, tremors, numb fingers, trouble with coordination. The kidneys, tasked with cleaning the blood, pick up the slack but get strained in the process. Studies on healthcare workers, lab techs, and patients show that inhalation or skin contact, even with so-called “trace” amounts, adds up over time.
Health Risks Outweigh the Benefit
The FDA no longer allows phenylmercuric nitrate in new medications. Medical journals point out its link to skin irritation, eczema-like rashes, and sometimes systemic toxicity—especially in infants. Babies and young children don’t process mercury as efficiently as adults. Elderly users or anyone with kidney trouble face added risk. Some hospital records tell stories of toddlers who developed rash and swelling after a few rounds of eye drops—side effects disguised as regular “pink eye.”
Environmental health studies show another angle. Mercury leaches into water. Residents in areas near waste disposal or pharmaceutical manufacturing have higher levels of mercury with similar patterns of headache, fatigue, and mood swings. Health officials keep a tight watch, but once mercury’s in the ecosystem, there’s no turning back.
How Can We Do Better?
Banning phenylmercuric nitrate in most countries was a step in the right direction, yet imported goods still pose a risk. Pharmacies caught with old stock sometimes unwittingly expose patients. It helps to double-check ingredient lists and ask a doctor or pharmacist before using any ointment with unfamiliar chemicals. Patients with a history of allergies or reactions to preservatives should stay on the alert.
In hospitals and clinics, switching to safer preservatives costs a bit more, but clearing mercury from the supply chain saves money and lives over time. Reporting side effects—no matter how small—helps authorities spot trouble before it spreads.
Final Thoughts
Mercury has no place in modern medicine. Respect for patients means erring on the side of caution, listening to experience, and reading the fine print. Healthy skepticism and solid science both say the same thing: some risks aren’t worth taking.
How should Phenylmercuric Nitrate be stored?
Looking at the Risks Up Close
Phenylmercuric nitrate isn’t the face of your local chemistry poster—far from it. Over the years, working around labs and talking with industrial folks, I’ve noticed these mercury compounds earn careful attention, and for good reason. This white or colorless crystalline powder hits the body hard if you mess up storage or handling. Even a little slip invites direct effects on skin, kidneys, and nervous system. It doesn’t take much. Mercury compounds in general don’t forgive carelessness.
Keeping People Safe Starts With Storage Choices
Smart storage choices cut down on the chance of accidents and long-term exposure. I’ve seen large institutions lock these containers in cabinets with dedicated warning labels and extra ventilation. That approach works. Room temperature storage usually works, but keeping it away from heat sources or sunlight keeps degradation at bay. Moisture spells trouble for phenylmercuric nitrate, so dry air makes a practical difference—think of silica gel packs in storage cabinets.
Container Materials Matter
Glass and high-quality plastics handle phenylmercuric nitrate best. Metal containers signal a problem—mercury and its compounds react with certain metals, and this increases the odds of unwanted reactions. Store the material in tightly sealed containers to prevent vapor leaks or contamination from the outside.
Segregate Like You Mean It
Experience teaches that one poorly segregated compound can ruin your week—or your health. Strong acids, alkalis, and ammonia shouldn’t share space with phenylmercuric nitrate. Mixing storage heightens risk of release or even explosions. I always recommend double-checking chemical compatibility charts, which are available in most reputable lab safety guides.
Labeling—Clear and Bold
Accurate labeling with clear hazard warnings helps everyone who walks through a storage room. Don’t count on everyone knowing what sits inside. I once saw a secondary bottle missing a proper mercury warning sticker; that kind of oversight is an accident waiting to happen. Include the name, hazard symbols, and date received or opened.
Regulations and Real-world Shortages
Laws about toxic mercury compounds range from strict tracking in hospitals, to full-on bans in some countries. Institutions that still use phenylmercuric nitrate stay up-to-date on local storage, disposal, and reporting rules. In places where regulations shift fast, outdated practices stick around. I believe ongoing training helps people keep pace—even a short annual review goes a long way.
Safe Storage Builds Lasting Trust
Chemical safety goes beyond compliance. Proper storage wins trust across teams. It shows everyone—supervisors, researchers, custodians—that risk isn’t getting swept under the rug. Keeping phenylmercuric nitrate behind lock and key, with good training and honest respect for its danger, goes much further than a checklist. Real safety habits stick, while shortcuts catch up sooner or later. If I learned anything from fieldwork, it’s to invest in those habits.
What precautions should be taken when handling Phenylmercuric Nitrate?
The Risks Lurking in the Bottle
Phenylmercuric nitrate ranks high among the lab chemicals that call for extra respect. This compound, found in some medicines and industrial products, releases mercury—a heavy metal with a reputation nobody wants on their hands or, worse, in their body. Exposure can cause headaches, tremors, nerve problems, and kidney damage. Even at low levels, mercury sticks around, building up in tissues and making small mistakes costly in the long run.
Smart Ways to Handle and Store
Anyone who’s spent time in a lab knows the rule: gloves before touching. With phenylmercuric nitrate, that means donning nitrile or neoprene gloves, not just latex. Skin acts like a sponge for certain chemicals; skipping proper protection can mean absorbing mercury without realizing. A splash to the eye isn’t just painful—it’s dangerous. Lab goggles with side shields cut down the risk, and for folks working with powder or concentrated forms, a face shield adds another layer.
Good ventilation does more than stop unpleasant smells. Using a fume hood pulls invisible vapors away from your face, so you’re not breathing in toxic mercury compounds. Bottles get stored tightly sealed, away from acids and heat. Labels ought to shout warnings, not whisper, to keep everyone aware.
Personal Experience: Mistakes Leave Marks
During grad school, a forgotten bottle of phenylmercuric nitrate turned up in a dusty chemical closet. Complacency led someone to open it without the right gloves or a fume hood. A few days later, headaches started, and blood tests flagged mercury exposure. This served as a wake-up call: small lapses in judgment with mercury leave lasting reminders. Easy to avoid, hard to undo.
Cleanup Isn’t Just About Mopping Up Spills
Spills demand swift, exact steps. Most labs keep a mercury spill kit on hand—sulfur powder, sponges, and zipper bags to trap every bit. Regular towels won’t do. A sloppy response spreads contamination, raising the risk for everyone who works there afterward. Every tool, beaker, and glove that met the nitrate must go in a hazardous waste bag, not the regular trash or sink drain. Improper disposal turns the building into a risk zone, not just the bench.
Looking Forward: Protecting People, Not Just Lab Gear
Better training helps more than fancy equipment. Labs where everybody—from students to seasoned staff—gets straightforward, scenario-based safety lessons see fewer incidents. Supervisors hold the line by checking safety gear, enforcing rules, and encouraging staff to speak up about bad practices. Some institutions push for less toxic preservatives in pharmaceuticals and paints, phasing out mercury-based ingredients entirely. This protects not just those in the lab, but people downstream in waste facilities and beyond.
Pushing for Safer Habits
Every time we glove up, check those labels, and store chemicals right, we invest in a safer lab culture. Cutting corners with phenylmercuric nitrate bites back, and the cost isn’t just regulatory—it’s personal health. Small habits, shared knowledge, and up-to-date training keep science on track and people out of harm’s way. That’s worth much more than the cost of a box of gloves or a proper waste bag.
| Names | |
| Preferred IUPAC name | oxido-(phenyl)mercury |
| Other names |
Mercurochrome nitrate Mercuric nitrate, phenyl- Phenylmercuric nitrate(V) Phenyl mercury nitrate |
| Pronunciation | /ˌfiː.nɪl.məˈkjʊə.rɪk ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 100-97-0 |
| Beilstein Reference | 14607 |
| ChEBI | CHEBI:53502 |
| ChEMBL | CHEMBL17461 |
| ChemSpider | 54640 |
| DrugBank | DB01474 |
| ECHA InfoCard | ECHA InfoCard: 100.013.758 |
| EC Number | 231-213-3 |
| Gmelin Reference | Gm. 2252 |
| KEGG | C01425 |
| MeSH | D010625 |
| PubChem CID | 16684482 |
| RTECS number | OV4550000 |
| UNII | 9ZM1TM842A |
| UN number | UN1673 |
| Properties | |
| Chemical formula | C6H5HgNO3 |
| Molar mass | 357.29 g/mol |
| Appearance | White or yellowish-white crystalline powder |
| Odor | odorless |
| Density | 3.98 g/cm³ |
| Solubility in water | Soluble |
| log P | -0.04 |
| Vapor pressure | 1 mm Hg (20 °C) |
| Acidity (pKa) | |
| Basicity (pKb) | 7.58 |
| Magnetic susceptibility (χ) | -97.0e-6 cm³/mol |
| Refractive index (nD) | 1.613 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.88 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 274.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -157.5 kJ/mol |
| Pharmacology | |
| ATC code | S01AX05 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; causes burns; may cause sensitization by skin contact; environmental hazard. |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H300 + H330: Fatal if swallowed or if inhaled. |
| Precautionary statements | H260, H300, H310, H330, H373, H410, P210, P222, P223, P231, P233, P260, P262, P271, P273, P280, P284, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P313, P310, P320, P321, P330, P361, P363, P391, P403, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-OX |
| Autoignition temperature | > 250°C |
| Lethal dose or concentration | LD50 oral rat 26 mg/kg |
| LD50 (median dose) | LD50 (median dose): 29 mg/kg (oral, rat) |
| NIOSH | MI140 |
| PEL (Permissible) | 0.1 mg/m³ |
| REL (Recommended) | 0.01 mg/m3 |
| IDLH (Immediate danger) | 10 mg/m3 |
| Related compounds | |
| Related compounds |
Phenylmercuric acetate Mercuric nitrate |

