Phenylmercuric Benzoate: Unpacking a Chemical with a Past and a Future
A Chemical Born from Necessity
Phenylmercuric benzoate didn’t make its entrance as a product of idle curiosity. Its story began in the early 20th century, when researchers looked for compounds that could keep microbes at bay. Preserving vaccines, eye drops, and topical medicines presented huge challenges before antibiotics and modern sterilization. The need for something effective gave rise to a family of organomercury compounds. Among these, phenylmercuric benzoate emerged because chemists found it could destroy or halt bacteria, fungi, and even some molds. For decades, its reputation as a dependable preservative echoed in the halls of both large pharmaceutical companies and smaller laboratories. People trusted it because—simply put—it worked where older, less sophisticated solutions failed.
Rubbing Shoulders with Everyday Science
Anyone who has spent time poring over ingredient labels in old pharmacopoeias may have stumbled across the name phenylmercuric benzoate. Its structure is simple enough: a phenyl group bonded to mercury, linked to a benzoate anion. It smells faintly medicinal, comes as a white to slightly yellowish powder, and it dissolves in organic solvents much more easily than in water. If you’ve handled it in lab classes, you’ll remember it well—its slightly chemical odor, the way it stuck to the spatula, the unmistakable warning signs on its bottle. In medicine, its greatest value came from being stable under a range of conditions and not breaking down easily under ordinary storage. That sort of reliability meant you could count on it to keep contaminated eye drops from going bad.
Peering into Its Chemistry
Chemists grow up learning the bright and dark sides of mercury-based compounds. The physical and chemical traits of phenylmercuric benzoate reflect that duality. It packs serious antimicrobial punch, thanks to its ability to disrupt protein and enzyme systems in microbes. Its toxicity toward microorganisms comes from its tendency to denature proteins, a trait mercury atoms lend to many of their compounds. On the technical level, scientists often measure melting points in the range of 184–187 degrees Celsius. It resists breaking down under mild heating but succumbs if pushed to higher temperatures, decomposing into various organic and inorganic fragments. This sort of resilience gave hospital workers confidence, letting them store drugs laced with phenylmercuric benzoate in less-than-perfect conditions without fear of quick spoilage.
Preparation and the Work of the Bench Chemist
Making phenylmercuric benzoate starts with making phenylmercuric acetate or other mercurial intermediates, then treating these with sodium benzoate. The process takes patience and precision; the chemist dissolves reactants separately, mixes them under controlled temperatures, filters, washes, dries, and finally obtains the white crystalline product. While the underlying chemistry may sound straightforward today, I remember my own organic chemistry professors describing the painstaking care required to purify mercury compounds. Mercury does not reward inattention with forgiveness. Even a minor spill or a wind shift in the fume hood could mean scrambled nerves, a week of environmental monitoring, or worse.
Reactions and Chemical Adjustments
Phenylmercuric benzoate stands as a remarkable example of how even small modifications to a molecule can dramatically alter biological properties. When exposed to mineral acids or alkalis, the compound can break down, releasing benzoic acid or forming more toxic mercury salts. In laboratory experiments, mixing with other organic reagents can strip away the benzoate or the phenyl group. I learned the hard way that you don’t mess around with incompatible chemicals here—safety goggles and an abundance of ventilation are must-haves, not afterthoughts. The rich chemistry of these compounds creates endless headaches for waste disposal and long-term lab cleanup, as mercury lingers stubbornly in the environment unless treated with special reagents or trapped in elaborate filtration systems.
Known By Many Names, Feared for One Reason
In the scientific and industrial world, synonyms accumulate as people in different countries or fields work with the same chemical. Phenylmercuric benzoate also answers to names like “benzoic acid, phenylmercury(II) salt” or “phenyl mercuric benzoic acid.” They sound bland, but the “mercuric” part always gives folks pause. Regulatory agencies and lab manuals call attention to the “Organomercury compound” label. No matter what you call it, its hazards remain unmistakable. Before handling it, I always did a quick mental review: gloves, lab coat, fume hood, meticulous disposal. Discipline matters, because the consequences slip by quietly at first and show up much later—not with a bang, but with long-term health effects.
Meeting Safety and Operational Rules
Running a lab that stores and works with phenylmercuric benzoate means signing up for a never-ending routine of checks and compliance. Regulations came in response to mounting evidence of mercury toxicity—a reality that played out in tragic detail in places like Minamata Bay, Japan, where methylmercury contaminated seafood and devastated entire communities. Today, strict storage in sealed containers, daily inventory, air monitoring, and specialized spill cleanup are standard operating procedures—not as boxes to tick, but as practical barriers against irreversible harm. Regulatory groups worldwide, including the European Chemicals Agency and the US EPA, treat any mercury contamination as a red flag for deeper inspection.
How It Changed Medicine and Industry
For several decades, phenylmercuric benzoate was nearly everywhere in healthcare formulations—eye ointments, contact lens solutions, vaccines, even some cosmetic creams. Doctors didn’t have many choices, especially before single-dose packaging and modern aseptics. Outside hospitals, painters and woodworkers found uses in antifungal treatments, adhesives, and certain coatings. Its broad antimicrobial spectrum made it almost indispensable. Yet all of this utility carried serious risk—repeated exposure built up mercury in bodies and spread through water systems. In the 1980s and 1990s, as mercury’s chronic toxicity became more widely recognized, the tide turned. I remember seeing the scramble as manufacturers switched to alternatives like parabens, sodium benzoate, and other less toxic preservatives.
Pushing the Boundaries in the Lab
The research community once poured tremendous effort into creating and modifying organomercury compounds, including phenylmercuric benzoate. Teams kept tweaking the molecule, trying to keep the antimicrobial benefits while softening the toxicity. More recently, the lab work shifted to studying how mercury moves through the environment and bioaccumulates in food chains. Despite the high barriers to its use, certain analytical tests and specialized syntheses still draw on its unique chemistry. I have watched researchers move into the study of molecular interactions, protein binding, and ways to trap and neutralize released mercury. Academic curiosity endures even as mainstream pharmaceutical use fades.
Grappling with Toxicity and Its Consequences
Talking about phenylmercuric benzoate without addressing toxicity would miss the crux of the matter. Researchers found out—slowly, over decades—that mercury in all forms, including organic types, concentrates in living tissue. Even low, regular exposure increases the risk of neurological symptoms, kidney problems, and developmental delays in children. Large-scale epidemiological studies, combined with disquieting animal data, pressed regulators and manufacturers to end or reduce its use where safer compounds exist. The US Food and Drug Administration and World Health Organization published recommendations years ago against organomercury preservatives in most routine applications. For the general public, the disappearance from over-the-counter products happened quietly—almost out of sight—but the positive impact on public health stands beyond dispute.
Toward a Future Without Mercury Risks
The world is moving on from phenylmercuric benzoate, but that journey isn’t complete. Some developing regions face tough choices, lacking affordable alternatives. Environmental cleanup remains a massive undertaking—building filters, neutralizing waste, and scrubbing contaminated soils and waterways to keep mercury from cycling back into food and water. The rise of green chemistry pushes researchers to design new preservatives that deliver safety without lingering harm. For now, vigilance in regulation, investment in remediation, and global cooperation chart the course forward. If there’s one lasting lesson from the career of phenylmercuric benzoate, it’s the need for humility: every chemical tool we adopt demands respect, relentless scrutiny, and the willingness to move on when the cost grows too high.
What is Phenylmercuric Benzoate used for?
What It Does in the Real World
Phenylmercuric benzoate started making waves as a preservative back in the mid-20th century. Folks working in pharmaceuticals and cosmetics favored it for one big reason: it stops microbes. Even a tiny amount of this compound can keep bacteria and fungi from taking over creams, shampoos, eye drops, or even latex paints. If you’ve ever left a tub of lotion out for months without it smelling rotten or growing fuzz, you’ve seen the power of preservatives like this.
Why Companies Chose It
Many big names across industries have turned to phenylmercuric benzoate because it offered protection where other preservatives fizzled out, especially in water-based formulas. It helped deliver stable products to market shelves and bathrooms, limiting the chance of ruined stock and customer complaints. In the lab, this compound proved itself easy to blend into different mediums, so products hit the shelves in top shape.
Health and Environmental Concerns
All that protection doesn’t come for free. Mercury isn’t friendly to humans or to the natural world. Evidence stacked up over the years showing mercury compounds can build up in the body, targeting kidneys, the brain, and nerves. Long-term or high exposures take a toll. Workers who handled raw forms or diluted solutions sometimes saw skin reactions or even subtle changes in memory and mood. Pregnant women and kids risked even more, since mercury can cross the placenta.
Waste doesn’t vanish. Much of it runs into waterways, lingers in soil, and climbs the food chain. Fish caught in polluted waters wind up with mercury in their flesh. I grew up fishing in local creeks, and health advisories weren’t rare—locals paid attention whenever a new warning landed about eating too much catfish or bass, especially for small kids.
Regulation and Shifting Practices
Governments and scientists sounded the alarm after studying health outcomes and environmental damage. The Food and Drug Administration set limits on phenylmercuric benzoate in medicines and banned it outright in some cases. The European Union took even tougher stances, removing it from most personal care and household products.
Companies scrambled for replacement preservatives. The hunt hasn’t been easy. Synthetic alternatives don’t always last as long, and natural extracts aren’t always consistent batch to batch. Some, like parabens, carry their own baggage, sparking fresh debates in science and among shoppers. What I’ve seen is that more companies started testing new recipes, adjusted packaging, and educated customers about changes. On our bathroom counter, ingredient lists started shrinking, with more products advertising they’re paraben- and mercury-free.
What the Future Holds
Cleaning up after mercury compounds takes time and effort. Scientists and industry leaders work on safer molecules and new formulas, but it takes strict oversight and independent research to make sure these don’t just swap out one problem for another. Real trust means transparency—product makers filing complete test results, tracking what happens to waste, and letting the public in on how they protect both people and nature. The world’s shifting toward more thoughtful choices, and old-school preservatives like phenylmercuric benzoate now stand as a cautionary tale about quick fixes without enough questions. Our shelves reflect those lessons, one bottle at a time.
Is Phenylmercuric Benzoate toxic or hazardous?
What Is Phenylmercuric Benzoate?
Phenylmercuric benzoate used to turn up in products like latex paint, eye drops, and some topical medicines. The point was pretty simple—kill off bacteria and fungi so products could last longer and stay safer in homes or medicine cabinets. Over time, more folks started wondering: At what cost does this preservative keep things “safe”?
The Health Risks: Mercury Is Rarely Kind
No one needs a degree in toxicology to know that mercury doesn’t play nice with our bodies. All mercurials, even in small doses, bring serious health concerns. Phenylmercuric benzoate can pass through skin and mucous membranes, working its way into the bloodstream. Once inside, it builds up, mainly in the kidneys and brain. Researchers and doctors have linked exposure to kidney damage, nerve disorders, vision changes, and, at high levels, death.
The World Health Organization, the CDC, and many health experts raise real alarms about phenylmercuric compounds. Children, pregnant people, and anyone with a weakened immune system run greater risks. Kids especially react more strongly: their nervous systems are still growing, so even what looks like a “minute” amount might cause problems later on.
Mercury in the Environment: A Cycle That Doesn’t Quit
Dispose of anything with mercury the wrong way, and there’s a ripple. Small amounts leaching from old paint, industrial waste, or pharmaceuticals eventually land in rivers and soil. Bacteria break down phenylmercuric compounds, turning them into methylmercury, the toxic form that moves up the food chain. Fish collect it, people eat the fish, and that poison settles in our tissues. The same stuff that kept a can of paint fresh for years now moves silently through our environment.
The EPA and European Union have both placed heavy restrictions or outright bans on the use of organic mercury compounds in consumer products. Nobody wants mercury back in household cleaners or medicines—public opinion has shifted, and regulators listened.
Making Better Choices: What We Can Learn
Growing up, I recall old medicine cabinets with tiny bottles of eye drops preserved with chemicals nobody really pronounced. Today, those same items use safer alternatives. That’s major progress, driven by better health science and public voices. Manufacturers switched to preservatives like benzalkonium chloride or sodium perborate. These don’t linger in your organs or drift into streams the same way mercury does.
Doctors now check product labeling more closely and teach patients to do the same. People have a right to know what goes in and on their bodies. The workplace has also shifted. Gloves, ventilation, and toxic substance controls are not optional for those handling older chemicals.
Disposal matters too. It’s not about tossing everything into the garbage. Local hazardous waste sites exist for old medicines or leftover paints. A little effort here spares downstream neighbors—and ourselves—a heap of misery.
Facing the Future
Nobody needs to take needless risks with phenylmercuric benzoate. The story of this compound shows that convenience sometimes hides costs. Regulators, doctors, and regular people now know the dangerous side of mercury-laced chemicals. Change happens when science, responsibility, and honest information lead the way.
What precautions should be taken when handling Phenylmercuric Benzoate?
Understanding the Risks
Phenylmercuric benzoate doesn’t sit on many people’s radar, but in labs, factories, and certain industries, you’ll find it. Not everyone works with chemicals daily, though anyone who does will recognize the faintly metallic tang of caution that comes with mercury compounds. I’ve seen what happens when shortcuts replace safety—sudden headaches, fatigue, and worse, regrets that linger. Exposure, even a little, stacks up risk: nervous system damage, respiratory harm, kidney trouble, even cancer. Mercury doesn’t care whether a person understood the hazard; it acts all the same.
Personal Protective Equipment: More than a Recommendation
Relying on bare hands or thin fabric never cuts it with phenylmercuric benzoate. Gloves rated for chemical resistance—nitrile or neoprene—stop this compound from seeping into the skin. Safety goggles keep splashes and tiny dust out of the eyes. Lab coats and closed shoes add a basic layer, keeping chemicals from clothes and feet. Every layer matters. In my own routines in the lab, I check gloves for pinpricks every time, because the smallest tear can undo all other precautions.
Working Space: Good Habits Prevent Accidents
Ventilation often feels like a minor detail until someone starts coughing or feels dizzy. Fume hoods or local exhaust cut down on the vapor in the room. It pays to keep any workspace clean—labelled, organized, with nothing left out in the open. Label all beakers and bottles, because memories can blur in a busy day. Clean up spills fully and right away, don’t leave it for the next person, or for “after lunch.” I tend to double-check for puddles and residues, having learned the hard way how quickly a glove can pick up traces.
Training: Knowledge Saves Time, Health, and Money
Working safely with phenylmercuric benzoate doesn’t start with the chemical, it starts with training. Regular, up-to-date information grounds every safe habit. Too many people try to muddle through with old or patchy instructions. New staff need a mentor, not just a stack of safety sheets. Any time there’s a new procedure or piece of equipment, go over the basics again. Create a culture where coworkers remind each other to tie up loose sleeves, pull on that mask, use the spill kit. No one expects a fire drill, but everyone knows what to do; the same thinking applies here.
Disposal: Mercury Never Just Vanishes
After the work ends, safe disposal keeps communities out of danger. Leftover phenylmercuric benzoate won’t simply disappear, so secure collection keeps it from groundwater or the atmosphere. Seal it in marked, compatible containers. Call in a hazardous waste service on a schedule, not just when the shelves fill up. Each waste slip gets double-checked, not for bureaucracy’s sake, but because one mixed-up batch can poison whole systems. Even cleaning rags and gloves go into designated bins, never a general trash can.
Culture Shift: Responsibility Over Routine
Over years of handling chemicals, I’ve seen careful routines keep people healthy, and carelessness create emergencies that could have been avoided. Phenylmercuric benzoate requires treating every task like it matters, even if nobody’s watching or measuring the effort. By putting safe habits and shared knowledge first, the chance of accidents drops and everyone goes home whole, every time. That’s the quiet reward, and it’s worth more than saving a few minutes or skipping a step.
How should Phenylmercuric Benzoate be stored?
Handling Mercury-Based Compounds Takes Real Care
Phenylmercuric benzoate is no everyday chemical—even chemists handle it with respect. I remember first seeing it shelved in a university storeroom: a secure, double-latched metal box, not left forgotten or stacked next to routine solvents. Anyone who works around this chemical recognizes its toxic nature. Mercury exposure can lead to long-term health issues affecting the kidneys and the brain, and the risks are real even with simple contact or inhalation. Putting safety at the center isn’t about box-ticking, but about protecting real workers and communities.
The Storage Place Sets the Tone for Safety
A lot of us underestimate the space where chemicals live. For phenylmercuric benzoate, storage means more than clearing shelf room. Store it in a tightly sealed container, glass or high-quality plastic, somewhere dry and cool. Heat and sunlight can speed up decomposition. The best setup stays simple: a locked cabinet marked clearly with hazard signs, away from acids and other reactive chemicals. Don’t crowd different types of chemicals. Some reactions kick off even in storage if the wrong chemicals tangle together.
Don’t Overlook Labeling and Monitoring
Labels tell a clear story in the lab. Leaving a bottle of phenylmercuric benzoate with smeared handwriting or missing data creates accidents waiting to happen. Each label serves not only the current worker but also the next person grabbing that container. Include full chemical names, concentration or batch information, hazard warnings, and the date received. My old lab manager stressed updated inventory logs and routine checks. This habit once helped us catch a dried cap that would have otherwise released vapor into our air.
Disposal and Leaks: Act On Problems Right Away
Find a spill, don’t walk by and wait for someone braver. Small leaks can create long-lasting problems, especially for mercury-based compounds notorious for lingering in an environment. Institute a practiced, clear routine: For example, use absorbent materials suitable for mercury spills, ventilate the area, and wear nitrile gloves and splash goggles. Regularly inspect old bottles and seals for brittleness—make sure nothing leaks onto shelves or floors. Don’t pour leftovers down the drain or toss them in the trash. Facilities must send toxic chemicals for professional disposal in compliance with local regulations.
People Training Makes a Difference
Access alone doesn’t safeguard people—regular training does. Teach everyone where these compounds are kept, what the warning signs look like, and how to respond if there’s exposure. I’ve seen labs run smooth drills with even the new interns knowing exactly which shower to use or how to call for help. A chemical like phenylmercuric benzoate shouldn’t be stored where the untrained or the curious might stumble onto it.
Solutions Wrap Around Good Practice
Chemicals aren’t only about research or industry—they test a group’s discipline and commitment to mutual safety. Using dedicated cabinets with proper labeling, inspecting containers routinely, providing chemical hygiene training, and connecting with certified waste handlers all create the right environment for dealing with high-risk substances. It’s not just about preventing accidents. It’s about respecting the power of chemistry and the wellbeing of those who put on the lab coat each day.
What is the chemical structure of Phenylmercuric Benzoate?
The Basics That Matter
Phenylmercuric benzoate has a pretty fascinating chemical composition. Structurally, it brings together a phenylmercuric group and a benzoate anion. Looking at its formula, you see C13H10HgO2. At first glance, it just looks like another compound, but the actual arrangement tells a deeper story about how chemistry gets used in practical ways.
This compound features a mercury atom nestled between a benzene ring and a benzoate group. The “phenyl” part refers to a benzene ring (C6H5–) attached directly to mercury (Hg). The other half comes from benzoic acid, where the benzoate anion (C7H5O2−) ends up bonded through the mercury to form the final salt. It’s a single mercury atom bridging these two aromatic entities, drawing out both stability and toxicity — two sides of the same coin.
Not Just a Boring Molecule
Its structure serves a real-world purpose. Growing up with a nurse in the family, I remember old medicine cabinets filled with all sorts of bottles carrying compounds with long, intimidating names. Phenylmercuric benzoate often appeared as a preservative, especially before we ever talked about “green chemistry” or mercury’s environmental toll. The molecule itself is shaped for that job. It manages to pierce the defenses of fungi and bacteria thanks to mercury’s ability to disrupt metabolic functions. The benzoate component boosts its solubility, making it blend into creams and liquids.
Chemical analysis shows that in a bottle of eye drops, you might find phenylmercuric benzoate acting as a shield. Its crystalline, stable structure means it doesn’t break down in regular storage conditions, so your medication doesn’t turn into a petri dish. I experienced the benefit myself as a kid with hay fever — those drops sat comfortably on the shelf all summer without growing any unwelcome residents.
Weighing Risks, Seeking Solutions
There’s a price for that effectiveness. With mercury at its heart, phenylmercuric benzoate carries well-known toxicity risks. The chemical structure lets it persist in places we’d rather not see it, like waterways and living tissues. Studies have repeatedly linked mercury exposure to neurological and developmental harm, reminding us that even clever chemical solutions need second thoughts.
Alternatives exist. In recent years, formulators across the pharmaceutical and cosmetic industries have searched for preservatives that avoid heavy metals altogether. Compounds like sodium benzoate or parabens now see more use, though each comes with its own set of debates. Scientists keep refining these substitutes, looking for a balance between stopping microbial growth and preserving public health. For every time one might reach for an old solution, fresh research usually reveals an option that goes easier on the world around us.
The Ongoing Conversation Around Chemistry
Understanding phenylmercuric benzoate’s structure isn’t just chemistry trivia: it’s a real part of how science and society bump up against tough questions. History shows us that the details hidden in those formulas shape policies, medical routines, and even the plants and animals down river from us. Looking at the molecule — not just the name — lays out both the promise and the price in plain terms. The dialogue about safer, smarter ingredients keeps growing, and with it, so does our responsibility to look beyond old solutions toward newer, less harmful ideas.
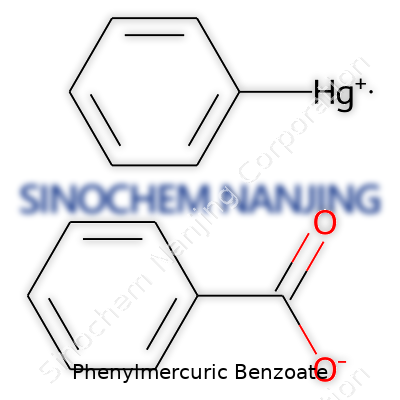
| Names | |
| Preferred IUPAC name | Oxy(diphenyl-λ^3-mercurio)benzoate |
| Other names |
Benzoic acid, phenylmercury(II) salt PMB Phenylmercury benzoate Phenylmercuric benzoate (USP) Mercury, phenyl(benzoato-O)- |
| Pronunciation | /fɪˌnaɪl.məˌkjʊə.rɪk bɛnˈzoʊ.eɪt/ |
| Identifiers | |
| CAS Number | 55-58-1 |
| Beilstein Reference | 1720742 |
| ChEBI | CHEBI:53259 |
| ChEMBL | CHEMBL2106651 |
| ChemSpider | 13757 |
| DrugBank | DB13751 |
| ECHA InfoCard | 100.011.449 |
| EC Number | 200-532-5 |
| Gmelin Reference | 16719 |
| KEGG | C18715 |
| MeSH | D010624 |
| PubChem CID | 16691 |
| RTECS number | OV9625000 |
| UNII | N9X3DL865L |
| UN number | UN3077 |
| CompTox Dashboard (EPA) | CompTox Dashboard (EPA) of product 'Phenylmercuric Benzoate' is "DTXSID8020214 |
| Properties | |
| Chemical formula | C13H10HgO2 |
| Molar mass | 354.82 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.646 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | 1.94 |
| Vapor pressure | 6.6 x 10^-7 mmHg (25°C) |
| Acidity (pKa) | pKa = 7.1 |
| Basicity (pKb) | 5.5 |
| Magnetic susceptibility (χ) | -1080.0e-6 cm³/mol |
| Refractive index (nD) | 1.610 |
| Viscosity | Viscosity: 0.948 cP (20°C) |
| Dipole moment | 1.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 401.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −92.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | Std enthalpy of combustion (ΔcH⦵298) of phenylmercuric benzoate: -9077 kJ/mol |
| Pharmacology | |
| ATC code | S01AX05 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes damage to organs; very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS06, GHS09 |
| Signal word | Danger |
| Hazard statements | H300 + H310 + H330: Fatal if swallowed, in contact with skin or if inhaled. H373: May cause damage to organs through prolonged or repeated exposure. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P260, P262, P273, P280, P308+P313, P501 |
| NFPA 704 (fire diamond) | 3-2-2-yes |
| Flash point | 104°C |
| Autoignition temperature | 275°C |
| Lethal dose or concentration | LDLO (oral, human): 2 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 44 mg/kg |
| NIOSH | WA2625000 |
| PEL (Permissible) | 0.1 mg/m³ |
| REL (Recommended) | 0.01 ppm |
| IDLH (Immediate danger) | 10 mg/m³ |
| Related compounds | |
| Related compounds |
Phenylmercuric acetate Phenylmercuric nitrate Mercuribenzoic acid |

