Peeling Back the Layers on Phenyldichlorosilane
Historical Threads: A Look Back at How We Got Here
Phenyldichlorosilane has roots tangled deep in the surge of chemical discovery that followed the world wars. Silicon chemistry turned a corner when researchers eyed organosilicon compounds as tools for both industry and academia. Back in the mid-twentieth century, scientists in Europe and America started dabbling with arylsilanes, seeking materials that could flex in electronics, coatings, and even pharmaceuticals. Phenyldichlorosilane came out of these efforts. It represented not just a curiosity, but a real step forward in connecting carbon and silicon—a bridge that, for years, nobody had tried crossing at scale. The early pioneers juggled limited equipment and knowledge, pushing through tough hazards to wrangle this molecule, figuring out its quirks and weaknesses along the way. Those days showed how far a stubborn curiosity could drag a new branch of chemistry, and laid the groundwork for today’s high-purity standards and precision-driven synthesis.
What Sets Phenyldichlorosilane Apart
As a compound, phenyldichlorosilane walks a line between simpleness and specialized function. It combines a phenyl group and two chlorines hooked onto a single silicon atom. The liquid gives off a sharp, unmistakable odor, clear as water but with menace underneath—a reminder of the caution it demands. It’s not flashy, not something you’ll ever spot in daily life, but for those of us knee-deep in chemicals, it’s a reliable building block. Silicone polymers, specialty coatings, and surface treatments all lean on this compound. That kind of role, sitting quietly behind so many products, says a lot about the quiet backbone that chemistry provides for the modern world.
Diving Into Properties: What Makes This Molecule Tick
Phenyldichlorosilane brings a unique set of physical and chemical properties to the table. Colorless and mobile at room temperature, it stands out as more than just a solvent, but less viscous than syrupy polysiloxanes. Its standout feature, reactivity, springs from those two chlorines tightly bound to silicon. That bond melts away with the right nucleophile, opening the door to all sorts of chemical possibilities: think of it as a kind of silicon-based LEGO piece, ready for swapping out parts. Physically, its boiling point falls somewhere north of 200°C. Proper glassware and cold traps matter here—this liquid will eat through skin or eyes, not to mention most metals if you get sloppy. Hydrolysis asks for thick gloves and sharp focus, since contact with water whips up hydrochloric acid gas and silanol products fast enough to take your breath away, literally and figuratively. There’s a reason chemists treat this compound with a blend of respect and suspicion, earned through years of trial and error.
Under the Hood: How Manufacturers Create Phenyldichlorosilane
Modern preparation methods reflect decades of fine-tuning. Early experiments used direct synthesis, hammering chlorosilanes together with benzene. Today, routes commonly start with silicon tetrachloride and benzene, or diphenylsilane and phosphorus pentachloride. Each pathway balances safety, waste, and efficiency the way a brewer balances hops. Factory floors look for high purity—no use clogging reactors or screwing up downstream reactions with side-products. Rigorous distillation and quality controls set the standard in 2024, a huge leap from the slapdash days of wartime chemistry. The prep is harsh; reactions demand strict exclusion of moisture, dry solvents, and airtight gear. Any slip-up delivers toxic byproducts or eats away at profits, motivating attention to detail and ongoing process tweaks.
How Chemists Harness and Transform Phenyldichlorosilane
What draws chemists to phenyldichlorosilane again and again is the molecule’s willingness to react and be shaped. Those two chlorines are eager to be replaced; hit them with alcohols or amines, and out comes a new Si–O– or Si–N– bond. This unlocks vast families of silanes and siloxanes. My own lab work taught me one lesson above all: make sure every joint is tight and keep a bucket of baking soda close. Chlorosilanes react with water so fast, stray drops will fill a fume hood with white smoke and stinging gas. Chemical modifications allow tailoring for everything from resin crosslinkers to water-repellent sprays. The backbone, the phenyl group, lets you nudge reactivity or add thermal stability. The field keeps evolving, with more sustainable, less hazardous reagents entering the playbook. The versatility of this compound outpaces the risks, as long as protocols run tight and respect for its dangers stays high.
Behind the Names: Synonyms and Aliases in the Lab
Ask around and people use a range of aliases for phenyldichlorosilane. Some say "phenyl dichlorosilane," trimming words to save breath in a busy lab. Chemical catalogs toss out names like dichloro(phenyl)silane, and older textbooks call it chlorophenylsilane. The molecular formula, C6H5SiCl2, rarely escapes the page, but it lurks as a quiet reference on every bottle. The jumble of names sometimes causes headaches in cross-referencing safety data, but over time, every bench chemist learns the shorthand that pops up on flasks and in protocols.
Keeping Safe: Handling Real-World Hazards
Safety with phenyldichlorosilane means dialing in habits that protect both people and facilities. The material bites hard—corrosive to skin and mucous membranes, sharply irritating to lungs, and reactive with everyday moisture. Standard practice calls for well-ventilated fume hoods, face shields, and gloves resistant to chemical attack. I learned the hard way that even a casual drip on skin can burn deep, and careless venting looses clouds that drive every coworker out of a lab. Local exhaust systems aren’t an afterthought, and good record keeping keeps track of storage, waste, and usage. Regulatory agencies push hard for labeling clarity and training. The strict rules crack down for good reason. A moment of distraction in handling, storage, or disposal can put entire teams or buildings at risk.
Who Uses Phenyldichlorosilane—and Why
Application areas reach beyond what most people expect. Silicone chemistry relies on phenyldichlorosilane as a gateway to performance coatings, elastomers, and adhesives that hold up under heat or foul weather. Electronics industries tap it for protective films and as a precursor for advanced silicon materials. Research labs drill into its reactivity, carving out new siloxane frameworks or specialized ligands for catalysts. Small companies and global corporations alike find room for it in finished products without ever seeing the raw stuff themselves. Compared to other silanes, its phenyl group changes the game for compatibility and durability—features in short supply in some high-performance industries. Every improvement in its handling and downstream chemistry expands its reach, promising new options for engineers and designers facing ever-stiffer technical demands.
Progress at the Edge: Research and Moving Forward
Development never stops, with research groups exploring new reactions, safer synthesis protocols, and environmental impacts. As chemical safety standards ratchet higher, labs experiment with greener reagents and cleaner workups. Academic teams publish on catalysis advances, tweaking conditions to coax higher yields or fewer byproducts. Machine learning recently started chewing on retrosynthetic analysis, hinting at new synthetic routes that could minimize hazardous waste or lower energy bills. Every new paper spills more data about how these arylsilanes slip into organic frameworks or enable more resilient materials. The combination of practical need and intellectual curiosity ensures phenyldichlorosilane keeps showing up at the edges of new technology.
Toxicity: Lessons Learned and Current Knowledge
Toxicity research pulls no punches. Researchers documented clear risks—skin burns, eye damage, and lung irritation on brief exposure. Chronic outcomes haven't grabbed headlines in the same way as some industrial nasties, but nobody in the business shrugs off the risk. Animal studies raised alarms, pushing regulators to demand strict workplace controls. The hydrolysis byproduct, hydrochloric acid, stacks further danger. On the human side, training and equipment do most of the heavy lifting for lowering exposure. There's no magic bullet; vigilance keeps people healthy. Industry improvements come from making sure workers have what they need: information, tools, and support to backstop safe handling. Tracking long-term effects still needs attention, and public datasets usually lag behind current practice.
Looking Beyond: Where Phenyldichlorosilane Goes Next
Future prospects for phenyldichlorosilane point toward innovation tempered by a stubborn need for safer operations. As electronics keep shrinking and new materials demand more customization, the chemical’s adaptability matters. Environmental and regulatory scrutiny grows each year, driving a shift in how chemists look at waste, reactivity, and process safety. Startups and established firms move to greener chemistry, aiming to shrink hazardous byproducts for both ethical and financial reasons. From what I’ve seen, future breakthroughs won’t drop safety or reliability; they’ll push for smarter syntheses and new uses that maximize yields and minimize harm. If history teaches anything, it’s that chemistry keeps finding ways to balance risk and reward—phenyldichlorosilane will remain a key player for anyone reaching into the heart of silicon chemistry.
What is Phenyldichlorosilane used for?
Straight Talk on a Key Chemical
Phenyldichlorosilane isn’t exactly a household name, but folks working in chemistry labs or manufacturing plants definitely know it. This compound shows up as a colorless to light yellow liquid, and the smell alone reminds me of lab days when care and safety goggles meant everything. I remember the first time I watched it react; the vapors hitting moisture—smoke and hiss—showed why respect and caution matter every second you’re near it.
Building Modern Materials
Let’s cut to the chase: people use phenyldichlorosilane to create silicone polymers and resins. These polymers end up in sealants, adhesives, and certain consumer products. From waterproofing sneakers to insulating computer chips, silicone’s everywhere. Phenyldichlorosilane breaks down and helps build those strong, flexible bonds that let silicone hold up in tough spots—cold, heat, pressure, moisture. I’ve met manufacturers who swear by silicone for electronics because it doesn’t crack or give out like rubber sometimes does. That durability doesn’t just come from nowhere. It starts with chemicals like this one.
Electronics and High-Performance Uses
Silicone’s role in the electronics world makes the story more interesting. Phenyldichlorosilane acts like a starting block for silicon-based coatings that protect circuit boards. Anyone who’s replaced a phone after minor water damage, or seen a power tool flicker out from moisture, understands the pain. Silicon coatings stand between sensitive parts and the hazards out there. Ask any technician who does repairs—when these barriers hold, devices last longer and perform better.
Custom Chemistry and Research
In academic labs, chemists like using phenyldichlorosilane as a building block. It lets them design new organosilicon compounds with unique properties—stronger bonds, better heat tolerance, or resistance to certain chemicals. During my time in university, a professor showed how tiny changes in molecular structure change an entire product. One tweak in the lab using materials like this could mean a safer medical implant, a sturdier protective film, or a longer-lasting lubricant for a high-tech bearing. These discoveries ripple out, touching industries as diverse as automotive, aerospace, and biotech.
Risks and Responsibility
Handling phenyldichlorosilane isn’t your average science experiment. Any exposure to moisture or water, and it reacts violently, releasing hydrogen chloride gas. That gas isn’t something you’d want to breathe. I’ve seen labs with entire protocols built around safely transferring and storing it. Even today, anyone working with it stays sharp on regulations and keeps emergency washes within arm’s reach.
On a bigger scale, industries have a duty to manage waste and handle transportation with care. There’s no skirting the responsibility. Spills or leaks can harm workers and surrounding communities. Following the strict safety standards from organizations such as OSHA and EPA isn’t just a rule—it protects real people.
Charting a Safer Path
To tackle those risks, companies keep training strong and invest in containment systems. Proper labeling, specialized storage, and regular equipment checks matter. Some researchers search for alternatives—less hazardous but still effective. The drive for safer chemicals grows as regulations tighten. In my own experience, ongoing education and sharing real-world incidents with coworkers made the difference. Honest talk about accidents—or near misses—built vigilance into the culture.
Phenyldichlorosilane isn’t going away soon. It plays a key role in producing materials most of us rely on daily, even if we don’t realize it. Recognizing both its value and its hazards allows smart progress—balancing innovation with health and safety.
What are the safety precautions when handling Phenyldichlorosilane?
Respecting a Reactive Chemical
Phenyldichlorosilane makes a regular appearance in labs focused on organosilicon chemistry. This compound—often a clear, colorless to yellowish liquid—doesn’t ask for trouble, but it sure delivers it if you drop your guard. I learned early on that even a splash can set off noxious clouds, so I approach it with the same focus as I would a soldering iron or a spinning blade. That lesson hit home one summer when a colleague ignored a leaky stopper and nearly treated the entire lab to a choking gas leak. Keen senses and a well-placed fume hood saved the day.
Skin, Eyes, and the Right Gear
Most chemists have stories about burns or rashes—mine started with forgetting my gloves just once. Phenyldichlorosilane reacts with water, including the sweat on your skin, forming hydrochloric acid and silanols. That leaves you with burns, irritation, or worse, so I never skip gloves. Nitrile or butyl rubber gloves work well. I reach for a splash-proof lab coat and goggles even for a quick transfer; basic glasses aren’t enough. Sometimes I slip on a face shield if I’ll be pouring or stirring, overkill until the moment a drop actually lands where it shouldn’t.
Ventilation Saves More Than Comfort
One whiff of hydrogen chloride gives you a quick reason to respect this chemical. It only takes a small spill or a forgotten open flask to fill a room with corrosive, lung-searing vapors. Fume hoods take in these vapors and ship them far away from your face, saving your airways and your mood. I never work with phenyldichlorosilane outside a hood, no matter how impatient I get. Wearing a suitable respirator serves as a backup plan, especially if the extraction isn’t perfect.
Storing and Labeling Without Shortcuts
After a few close calls with aging bottles, I always check expiration dates. Phenyldichlorosilane likes a cool, dry spot far from water sources, acids, or bases. I store it in tightly sealed glass, not metal, because any hint of moisture inside can degrade the contents and pop the lid. A flammable materials cabinet has become the go-to spot, well-marked for emergency responders and coworkers. Double-labels make sure nobody mistakes it for something less hazardous.
Spill Response and Emergency Actions
Spills love bad timing. One spilled beaker and you’ll need more than paper towels. My approach starts with evacuating the area, alerting others, and then using spill kits that neutralize acid. I don’t skimp on the cleanup, and I always check the kit first thing in the day. Contaminated clothes go straight to the emergency shower before skin damage sets in. I always know the quickest route to the eyewash, since those seconds really count.
Training, Vigilance, and Shared Knowledge
Working with dangerous chemicals shapes how I work with people, too. I get new team members trained up before ever opening these bottles near them. I push for safety meetings and walk-throughs, because the cost of one mistake outpaces the time spent prepping. By staying honest about my own close calls, I encourage others to speak up before problems grow. Keeping everyone alert helps make sure everyone goes home with nothing more than a story to tell.
What is the chemical formula and structure of Phenyldichlorosilane?
Looking at the Basics
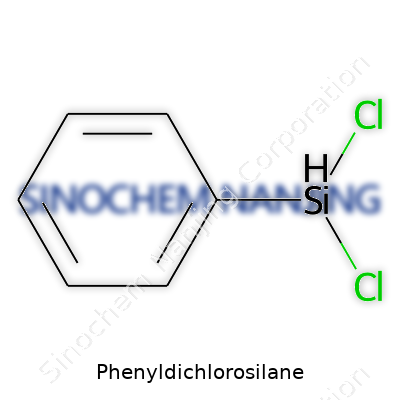
Phenyldichlorosilane goes by the chemical formula C6H5SiCl2. It pairs the toughness of silicon chemistry with the reactivity of chlorine atoms and the aromatic stability of the phenyl ring. Lay the molecule out, and you see a silicon atom at the center. It grabs two chlorine atoms and one phenyl group, along with a single hydrogen atom hanging off. The structure offers a bridge between organic and inorganic chemical worlds, which puts phenyldichlorosilane in a category of compounds called organosilanes.
Molecular Anatomy Up Close
Start with the core: Silicon, right in the middle, forms four bonds. The phenyl group, a simple circle of six carbon atoms with alternating double bonds, hooks onto silicon. Two chlorines attach nearby, and a lone hydrogen fills that last spot. If you try sketching it, you get a tetrahedral structure with the bulk of the phenyl ring on one side, two chlorines sticking out, and the hydrogen tucked in. That mix means phenyldichlorosilane carries a blend of reactivity and durability. The chlorines are easily swapped out in chemical reactions, while the Si–C bond (the link to the phenyl group) brings some solid stability.
Where Chemistry Meets Utility
Phenyldichlorosilane has been showing up in labs, especially where people need to introduce silicon into organic systems. It’s a starting point for building larger silicone polymers. By swapping those chlorines for other groups—sometimes with water, sometimes with alcohols—you can build networks or clever new molecules. It allows for making everything from heat-resistant coatings to adhesives. In my early grad school experience, I handled organosilanes to make protective coatings for electronics, and this compound was often the first bottle on the shelf before moving onto more complicated silanes.
Health and Safety Considerations
Anyone who has uncapped a bottle of phenyldichlorosilane can tell you it comes with an unmistakable, sharp odor. That’s your first warning—chlorinated organosilanes react with water in the air and release hydrochloric acid gas. That means goggles, gloves, and a good fume hood are not optional. The chemical can irritate skin, eyes, and lungs. Over the years, standard practice developed: keep it dry, and always have neutralizing agents ready in case of spills. Chemical safety groups have flagged this category of compounds for their corrosive action, too. Reliable data from NIOSH and OSHA points to the same facts: proper ventilation and quick access to water make all the difference for anyone working with it.
Industrial Footprint and Environmental Thoughts
In the bigger picture, phenyldichlorosilane helps drive innovation in specialty silicones, which are in everything from everyday caulking to sleek smartphone coatings. The chemistry industry wants new, safer ways to handle these chemicals, given their reactivity and the possibility for environmental release. Research teams continue to push for less hazardous alternatives for large-scale manufacturing, but in specialty syntheses, phenyldichlorosilane still delivers precise results you can count on.
Potential Solutions
Better handling systems, sealed reactors, and green chemistry principles—those allow industry and academia to limit exposure and waste. A promising approach involves recycling the chlorinated byproducts back into the manufacturing loop, reducing overall emissions. Experience in my own lab suggested engineers take environmental warnings seriously, with regular training and clear protocols to keep everyone safe and reduce the impact on air and water systems nearby. Keeping the focus on innovation and safety pushes this compound toward a smarter, more sustainable future.
How should Phenyldichlorosilane be stored?
Getting Real About Storage: Lessons from the Lab
I’ve spent summers sweating in underfunded research labs and watching thin glassware threaten to burst from chemicals handled wrong. Phenyldichlorosilane isn’t something people toss in a cabinet and hope for the best. It's a stubborn, reactive liquid that complicates everything if handled carelessly. Its aggressive reaction with water and its tendency to release corrosive fumes forced me to rethink my habits early in my career. Leaving a bottle loose or on a shelf near the sink led to more than one panicky evacuation drill.
Understanding the Risks
Phenyldichlorosilane lays down a challenge because it reacts vigorously with water, even with humid air. Contact leads to hydrogen chloride gas, which doesn’t just stink, it burns lungs and corrodes anything it touches. Handling this stuff with bare hands or cheap gloves might save time once, but the pain that follows teaches a lesson faster than any safety poster. Storing phenyldichlorosilane always requires respect for these dangers, or someone pays the price.
Essential Storage Practices
I grew out of shortcuts after my first near-miss. Anyone caring for phenyldichlorosilane should rely on airtight, sealed containers. Glass works well if it’s tough enough, but these containers must have ground-glass stoppers and remain free from chips. A loose seal or worn gasket invites moisture, so checking containers regularly matters. Substitute with Teflon-lined lids if the lab budget allows. I started labelling everything aggressively after watching someone mix up clear liquids during a late shift. Every bottle should carry a clear hazard label that stands out, with the cap tightly secured right after every use.
The storage location tells the difference between a quiet day and a disaster. Phenyldichlorosilane stays away from water sources and damp corners. Every time I noticed a careless shelf near a utility sink, I felt uneasy, because even a quick splash can spell trouble. Designate a storage spot in a well-ventilated, dry chemical cabinet designed for corrosives. Shelves should be lined with spill trays, since leaks lead to chaos. Metal cabinets show rust quickly, so I trust heavy-duty plastic or coated steel. Locking the cabinet keeps out curiosity from untrained hands—an absolute necessity after years around absentminded students.
Regular Maintenance and Monitoring
Over time, containers can fail and labels can fade. I once found residue on a shelf that nobody remembered spilling—proof that small leaks get overlooked until damage spreads. Chemical inventories should never gather dust; they deserve routine checks. Some labs keep moisture indicators in cabinets, so early signs of humidity don’t sneak by. Cabinets get aired and cleaned, absorbent pads swapped out, and inventory cross-checked with procurement logs. Escaped vapors or leaks should mean immediate disposal and a thorough review of safety procedures, no exceptions.
Training Makes the Difference
It only takes a quick training session to prevent a costly mistake. Newcomers to chemistry sometimes learn by doing, or by fixing disasters, but learning storage practices ahead of time saves money, health, and equipment. Sharing stories from past close calls in lab meetings helps these lessons stick. Institutional guidelines and chemical hygiene plans lay out required steps, but nothing beats a regular, real-world run-through of storage routines. Remembering a scar on a workbench or a crack in a bottle sharpens focus and reinforces careful handling.
What should I do in case of accidental exposure to Phenyldichlorosilane?
Facing the Realities of Chemical Risks
Phenyldichlorosilane, a chemical used in making silicone materials and other silicon-based compounds, proves dangerous if handled carelessly. Folks working in labs or factories making adhesives, sealants, or coatings might cross paths with this stuff. Most people would rather not think about what happens if something spills or leaks, but ignoring the risk never helped anybody. I’ve seen smart people skip simple steps and wind up with some scary trouble.
Understanding the Immediate Danger
This compound reacts fast with water, even moisture in the air. A splash or spill releases hydrochloric acid fumes and heat—enough to hurt your skin, eyes, or lungs in just a few seconds. If you’ve ever caught a whiff of acid, you remember the burning in your nose and throat. Imagine getting that on raw skin or in an eye, and you get the idea why safety teams drill folks on what to do.
Actions to Take on Accidental Exposure
Skin Contact: If any of this chemical lands on your skin, rip off those clothes right away. Sometimes people freeze up, but every second counts. Wash the exposed skin under running water—do not stop at a quick rinse. It can take fifteen minutes of steady washing to keep burns away. Skip soaps or creams unless a professional tells you otherwise.
Eye Contact: If it splashes in your eyes, head straight to the eye wash station. Hold your eyes open and flush them hard with water, at least fifteen minutes. No rubbing—that only drives it in further. Even if you think it’s all out, let a doctor have a look right away.
Inhalation: Breathing in fumes means you need fresh air, fast. Get out of the building or at least move upwind. Coughing, choking, or shortness of breath signals deeper trouble. Sometimes, people think they’re fine, then a few hours later their lungs start to close up. Medical help is not a “maybe” in this case—it’s a must.
Swallowing: Drinking or swallowing this by accident, while rare, ranks among the worst outcomes. Never induce vomiting. It only gives the acid more routes to burn tissue. Get help as soon as you can.
Why Simple Steps Make a Difference
I’ve worked at steel plants, bleach bottle factories, and research labs. Rapid response often marks the difference between a mild scare and a trip to the ER. Ignoring a spill, thinking “it’s probably fine,” only leads to regret. Every company should have Material Safety Data Sheets (MSDS) posted and easy to grab. I remember a small shop where the only posted copy hung in a closet no one unlocked—the day someone needed it, we lost precious minutes. You want safety showers and eye washes reachable within a quick walk. Even better, everyone in a workspace should run a mock drill, so panic doesn’t take over if the real thing happens.
Building Better Safety Habits
Companies can cut the odds of exposure by swapping in less hazardous chemicals where possible. Proper training stays important; people often forget what they learned last month, let alone last year. I’ve seen new and seasoned workers both skip gloves because “it’s just one quick transfer.” That habit lands you in trouble fast. Reporting near-misses also helps—too many folks keep quiet and let old mistakes repeat.
Safety at work relies on shared responsibility. Engineers, managers, and workers all have a role, from having ventilation in place to enforcing no-food-or-drink policies in the lab. Give every person the chance to review emergency plans and watch out for each other. Keeping those routines tight lets everyone go home safe at the end of the shift.
| Names | |
| Preferred IUPAC name | benzenedichlorosilane |
| Other names |
Benzenedichlorosilane Dichlorophenylsilane Phenylsilicon dichloride Phenylsilicondichloride Silane, dichlorophenyl- |
| Pronunciation | /fɛˌnɪlˌdaɪˌklɔːroʊˈsɪleɪn/ |
| Identifiers | |
| CAS Number | 80-10-4 |
| Beilstein Reference | 1462142 |
| ChEBI | CHEBI:34996 |
| ChEMBL | CHEMBL469020 |
| ChemSpider | 12317 |
| DrugBank | DB14003 |
| ECHA InfoCard | 100.006.015 |
| EC Number | 203-411-8 |
| Gmelin Reference | 78408 |
| KEGG | C14196 |
| MeSH | D018206 |
| PubChem CID | 66102 |
| RTECS number | TJ8575000 |
| UNII | 14TQ5SR66C |
| UN number | UN2185 |
| CompTox Dashboard (EPA) | `DTXSID3047822` |
| Properties | |
| Chemical formula | C6H5SiCl2 |
| Molar mass | 197.10 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent |
| Density | 1.305 g/mL at 25 °C |
| Solubility in water | Reacts |
| log P | 2.9 |
| Vapor pressure | 1 mmHg (25 °C) |
| Acidity (pKa) | 5.2 |
| Basicity (pKb) | 12.65 |
| Magnetic susceptibility (χ) | -56.0e-6 cm³/mol |
| Refractive index (nD) | 1.567 |
| Viscosity | 3 cP (20°C) |
| Dipole moment | 0.83 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 273.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -216 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -781 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331, H314, H411 |
| Precautionary statements | P210, P261, P280, P301+P330+P331, P305+P351+P338, P303+P361+P353, P304+P340, P312, P308+P313, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1-W |
| Flash point | 72 °C (closed cup) |
| Autoignition temperature | 537 °C |
| Explosive limits | Lower explosive limit: 1.5% ; Upper explosive limit: 12.7% |
| Lethal dose or concentration | LD50 oral rat 2287 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 250 mg/kg |
| NIOSH | T8019 |
| PEL (Permissible) | PEL: 5 ppm (parts per million) |
| REL (Recommended) | 3 ppm |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Trichlorophenylsilane Diphenyldichlorosilane Methylphenyldichlorosilane Triphenylchlorosilane Phenylsilane Chlorosilanes |

