2-Aminophenylarsonic Acid: Beyond the Formula
Looking Back: A Chemical’s Place in History
Few compounds spark as much debate in scientific and agricultural circles as 2-Aminophenylarsonic acid. Chemists first explored organoarsenic compounds like this one in the late nineteenth and early twentieth centuries, motivated by the promise of new treatments for diseases and better crop yields. The link between arsenic and toxicity was already recognized, but the ability to harness arsenic’s power in controlled ways led to a surge in research. 2-Aminophenylarsonic acid became best known for its inclusion in animal feed additives, especially in poultry production, from the mid-1900s onward, at a time when farming raced to expand output. For many years this substance, often called “roxarsone,” helped control parasites and improve feed efficiency. Public health concerns eventually shifted the conversation, as more rigorous research unearthed possible environmental and toxicological risks. Still, the compound’s history underlines a pattern seen across medicine and agriculture: hope, adoption, scrutiny, need for reassessment.
What the Compound Brings to the Table
2-Aminophenylarsonic acid isn’t just another footnote in chemical catalogs. It belongs to the group of organoarsenic acids, with a molecular structure containing an aromatic ring bonded to both an amino group and an arsonic acid group. At room temperature, it typically forms light-colored, odorless crystals that dissolve moderately in water. Chemists note a melting point close to 223°C, both a measure of its stability and its resistance to common breakdown during standard storage conditions. This unique arrangement of functional groups gives the compound interesting reactivity—researchers can attach or modify specific sites to spin off related chemicals, altering behavior or tweaking toxicity.
On Labels and List of Names
Names matter when tracking chemicals. 2-Aminophenylarsonic acid goes by several. Scientists often call it “roxarsone,” but other labels appear: 4-Hydroxy-3-nitrobenzenearsonic acid, NSC 34502, and more niche identifiers in some regulatory texts. The technical specifications—purity, maximum levels of trace arsenic, and elemental composition—reflect both its medical legacy and changing regulatory landscapes. Older specifications focused on ensuring consistent dosing for animal feed, while modern approaches look for residues in soils and food products, owing to growing awareness about bioaccumulation and chronic exposure risks.
Making the Compound: Preparation and Chemistry
Production of 2-Aminophenylarsonic acid usually starts with nitroaniline or direct nitration of aniline derivatives, followed by processes that substitute the nitro group with an arsonic acid group. The sequence demands careful temperature control and strict isolation of intermediates. What matters to the chemist isn’t just completion of synthesis but also removing unreacted arsenic species and controlling by-products. Later purification ensures a usable product with reliable properties for animal health or research. Researchers often tweak synthetic pathways in search of safer steps or higher yields, reflecting advances in green chemistry.
Chemical Reactions and Tweaks
The chemical reactivity of 2-Aminophenylarsonic acid lets it serve as a foundation for further modification. Scientists have explored methods to link the arsonic group to polymers for water treatment, or combine it with other biologically active molecules. The aromatic ring and amino group open doors to reactions with other organic compounds, giving rise to an array of analogues. Once these variants enter animal systems or soil, questions soon follow about rates of breakdown, persistence in meat or eggs, and possible conversion to inorganic arsenic—a known toxin. Environmental chemists test samples after exposure to sunlight, heat, and microbial action, tracking both obvious and subtle changes.
Standards, Safety, and the Human Angle
Handling anything with arsenic in its name demands respect. Standards now shape every aspect of 2-Aminophenylarsonic acid’s lifecycle, from production to use, disposal, and even transport. Regulatory bodies in North America, Europe, and Asia keep tight controls on concentrations allowed in feeds, limits for residues in edible tissues, and procedures for environmental monitoring. Accidents or lack of oversight once paved the way for exposures with uncertain consequences; now manufacturers must provide detailed labeling, user training, and disposal guidance. Workplace safety, ventilation, and protective equipment can’t fall by the wayside. Despite stricter standards, stories linger about improper disposal or illegal imports in regions where enforcement falls short, a reminder that rules matter most when applied everywhere.
Applications—From Barnyard to Bench
Most people who recognize 2-Aminophenylarsonic acid associate it with livestock feed, where it helped reduce infections and accelerate growth. Nations once applauded its use when food shortages loomed large. Regulations and shifting consumer views prompted many producers to withdraw it or seek alternatives, pushing researchers to uncover feed additives without long-term health risks. The compound still pops up in specialized applications. Lab scientists use it as a marker in trace element analysis, a test substance in arsenic metabolism studies, and a benchmark in toxicological research. Educational settings sometimes rely on it to demonstrate organometallic synthesis.
Research, Toxicity, and the Road Ahead
Science continues to probe the health effects of exposure. While routine use in feed has dropped in many regions, 2-Aminophenylarsonic acid’s environmental persistence remains a focus of major studies. Research shows that both the compound and the arsenic it releases can persist in soils, with by-products often more toxic than the original molecule. Chronic low-level exposure links to bladder, lung, and skin cancers in some human populations, raising alarm over cumulative effects in food chains. Animal model studies reveal differing breakdown rates in chickens, pigs, and soils—further complicating broad regulatory action. Amid these debates, new analytical techniques continue to deliver better measurements of arsenic species in meat, eggs, groundwater, and dust. Calls grow louder for transparent data on accumulation and for sharing findings with affected farming communities.
Prospects—Change Hinges on Awareness and Innovation
Any journey toward less hazardous agriculture benefits from examining chemicals like 2-Aminophenylarsonic acid. A broad shift away from arsenic-containing compounds already pushes innovation in animal husbandry and food production. Biologists pursue probiotic solutions, botanists breed more resistant animal strains, and some companies develop entirely synthetic antimicrobial peptides. Environmental engineers join the fray, designing filters or treatments to break down arsenic residues before water reaches rural communities. Lasting solutions call for blending chemical knowledge with revamping regulatory frameworks and prioritizing open, accessible science. Without collective scientific memory and vigilant standards, lessons of the past can fade, risking cycles of rediscovery and loss. Ethical research, proactive regulation, and engaged communities offer hope for managing not just one challenging substance but the larger catalogue of chemical legacies that shape daily life.
What is 2-Aminophenylarsonic Acid used for?
Uncovering the History
Many are surprised to hear the name 2-Aminophenylarsonic acid come up, especially outside of a chemistry lab. It doesn't make headlines often, but those who grew up around agriculture in the mid-1900s might remember the conversations. This compound found its way into farming as a feed additive called arsanilic acid. The goal was to help livestock, mostly poultry and swine, grow faster and fight off certain infections. The positive effects on animal weight were backed by studies, which showed animals grew about 5% faster on average. Families who depended on livestock for their living watched production go up, so there was a sense of progress and hope.
Concerns with Use
After years of relying on 2-Aminophenylarsonic acid, questions started to pile up. People began to ask what happens to all that arsenic once the animal eats it. Arsenic, after all, is not friendly stuff. Scientists started finding residues in meat, leading to heated debates and community meetings packed with concerned parents. The U.S. Food and Drug Administration studied it further and eventually decided the risks to human health outweighed the growth benefits. In 2013, the FDA pulled approval for its use in feed. Modern day, you won't find it in any legal livestock operation in the U.S.
Legacy in Agriculture and the Environment
Even after a ban, traces linger for a long time. Manure from animals once fed arsanilic acid ended up on fields and in water supplies. I remember growing up in a rural Midwestern town where kids swam in ponds fed by runoff from nearby farms. In those days, no one thought twice about possible chemicals in the water. It turns out, some forms of arsenic can stay in the ground for decades. This brings up problems for anyone growing vegetables, whose crops soak up what’s left behind. In several studies, arsenic levels in soil stuck around above recommended safe levels for years after use ended.
Science and Safer Options
Animal agriculture has always been an industry looking for ways to improve. Farmers work hard, and any tool that promised easier gains would get a chance. Today, focus shifts towards feed supplements made with safe, natural ingredients. Probiotics and carefully balanced vitamins help support animal health without dangerous side effects. If science turns up new information, researchers need a path to publish and share openly without political pressure. Honest, peer-reviewed research helped bring an end to 2-Aminophenylarsonic acid in feed in the first place.
Solutions for Impacted Communities
For rural communities who lived through its use, fixing soil and water problems means money and patience. Phytoremediation—planting certain species that naturally “pull” arsenic from the soil—offers some hope, though progress moves slowly. Better testing of water in vulnerable spots should come before any new farming project. State and local governments need to be ready to help clean up the legacy of chemicals that fall out of favor over time. If more people share stories and pay attention to the science, the next generation will carry a little less of this chemical past.
What are the safety precautions when handling 2-Aminophenylarsonic Acid?
Real Risks Behind the Long Name
2-Aminophenylarsonic acid might sound like something found only in dusty lab textbooks, but it pops up in research, pharmaceuticals, and sometimes even agriculture. This chemical, though, packs a real punch when handled loosely. In my years around labs, I've seen smart people caught off guard because they forgot what even common chemicals could do when not respected.
Personal Protective Equipment: An Everyday Shield
Anyone working with 2-Aminophenylarsonic acid starts by gearing up. Nitrile gloves serve as a solid barrier. They keep your skin clear of contact, and since many compounds can seep right through thin latex, nitrile often wins out. Good goggles shield your eyes from splashes. If dust or fumes might get kicked up, a well-fitted respirator with an appropriated-rated cartridge changes the game. Lab coats—even the old, patched-up ones—do more than show you’re on the job. They prevent accidental contact with your arms or torso, especially after a quick spill or splatter.
Training Isn’t a Box-Checking Exercise
Every serious lab invests time teaching staff and students to respect chemicals like 2-Aminophenylarsonic acid. I used to think safety briefings dragged on, but skipping details can mean missed steps. Understanding the symptoms of arsenic exposure, for instance—things like headache, confusion, or stomach trouble—means you notice problems before they grow.
Good Habits Make the Biggest Difference
Fume hoods aren’t just furniture. When weighing or mixing 2-Aminophenylarsonic acid, I always work inside one, trading convenience for lungs that work tomorrow just as well as today. Any spills go straight onto absorbent pads, instead of desk surfaces. Everything stays labeled: old jars with hand-written labels sometimes lead to big mistakes, so updating them keeps everyone safe.
Washing hands well after work—soap, water, no exceptions—cuts down risks. I once saw a colleague recall too late that he’d touched his sandwich after cleaning beakers. The result was a mild case of exposure that sent him home dizzy. It never happened to him again.
Storage and Waste: Keeping Problems Small
Lock away 2-Aminophenylarsonic acid in sealed, clearly labeled containers. Cool, dry conditions slow down unwanted reactions. Never set such chemicals near heat sources or strong acids. If powders escape into the workspace, they tend to linger and spread, so seal bags and jars tight every time.
Disposing of the waste means following rules strictly. Dumping anything with arsenic into regular trash isn’t just bad practice; it draws hefty fines and real harm to the environment. Most places set up hazardous waste protocols, and all of us handling chemicals should use them every time.
Small Actions Lower the Odds
At the end of the day, handling 2-Aminophenylarsonic acid safely depends on vigilance and respect for how harmful it can get. Nobody enjoys more paperwork or slower workdays, but trading caution for convenience proves expensive in the long run. It always helps to slow down, check labels twice, and keep protection gear in reach.
What is the chemical formula and structure of 2-Aminophenylarsonic Acid?
Unlocking the Details
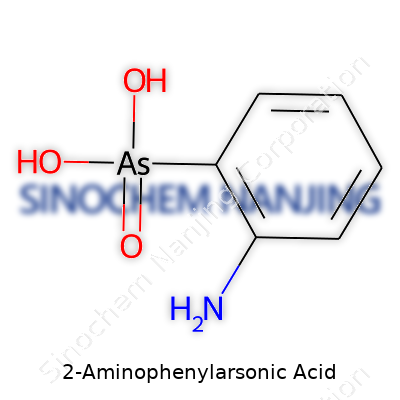
Chemistry has a way of drawing invisible lines between fundamental science and how the world operates. 2-Aminophenylarsonic acid delivers a clear example of that connection. At first glance, the name sounds intimidating, but it breaks down more simply than most would expect. The chemical formula for this compound is C6H8AsNO3. It brings together a benzene ring, an amino group, and an arsonic acid group in a single structure.
Understanding the Structure
Imagine a benzene ring – that familiar six-carbon ring every organic chemist gets introduced to early on. Now, attach an amino group (-NH2) at the second position and an arsonic acid group (-AsO3H2) at the first position. The amino and arsonic acid groups sit on neighboring spots, earning this molecule its “ortho” or “2-” label. Its structure isn’t just convenient shorthand for chemists; it makes a visible impact on how the molecule behaves in practice.
Looking at molecular drawings, there’s a central hexagonal ring. One carbon hugs an NH2 group, and right next door, another carbon latches onto the arsonic acid group. Moving atoms on paper changes how compounds act in the real world, from solubility to acidity. That brings up a key point: small differences in structure drive big changes in practical use, and ignoring the blueprint leads to real-world headaches.
Importance and Where Chemistry Meets Life
2-Aminophenylarsonic acid lives at the intersection of organic and inorganic chemistry. It caught the attention of researchers and veterinarians because it landed roles in animal feed additives for decades. Its arsonic acid group contains arsenic, which carried concerns as evidence mounted about the lingering effects of arsenic-based compounds in the food chain and environment. Having worked with regulatory reviews and farm operations in the past, I saw debates heat up over feed additive safety. The structure of this acid makes it more water-soluble than some other arsenic compounds, leading to concerns about leaching into groundwater.
The chemical isn’t just a historical footnote. Tracking its use raised important questions about managing residues in soil and meat, and what alternatives can take its place. Public health research showed how even low-level arsenic exposure raises risks for both workers and consumers. That put pressure on regulators, producers, and researchers to team up and address real gaps.
Solutions, Research, and Future Choices
Safer feed systems stand on science-backed choices. Understanding the exact formula and structure of each additive, including 2-Aminophenylarsonic acid, gave scientists the clues needed for smarter regulations. I’ve found that taking the time to map out these molecules aids early risk detection. Replacing old additives with safer options, including non-arsenic-based compounds, means better outcomes for everyone.
Access to clear, accurate chemical data also matters for emergency response. Knowing the specific arrangement of atoms in 2-Aminophenylarsonic acid shapes proper handling, packaging, and disposal. Industry, farmers, and consumers benefit when detailed knowledge leads to practical changes: stricter residue limits, cleaner farming practices, and investments in research for replacements. The story of this compound proves that a simple formula written on a chalkboard leaves a long shadow across labs, farms, and our daily meals.
Is 2-Aminophenylarsonic Acid hazardous or toxic?
Understanding the Risks in Simple Terms
2-Aminophenylarsonic acid — sometimes called arsanilic acid — often raises eyebrows because of one word: arsenic. Most folks hear arsenic and picture dangerous poisons, and with good reason. Many compounds that use arsenic can harm health if not handled with real care. This particular chemical gets used in some industrial settings, research, and even animal production.
Why Toxicity Isn’t Just a Buzzword
Personal experience has taught me that factory chemicals with ‘arsenic’ in the name need distance and respect. At one farm where I worked in college, I watched how some animal feeds relied on old-school growth promoters. Arsanilic acid used to pop up in these lists to help poultry and pigs grow faster or deal with infections. New studies and regulations led most producers to drop it, but for years, workers and animals alike got exposed. Symptoms didn’t always show up right away. Over time, it’s the slow build-up that does damage. Chronic exposure can cause skin problems, nerve issues, and at higher amounts, cancer. Even after stopping use, arsenic-based chemicals can linger in the soil, sometimes turning up in crops or water wells later.
The Science Behind the Danger
University research points out that 2-aminophenylarsonic acid doesn’t act all at once. Instead, it breaks down over time, especially in soil or animal waste. The by-products don’t just evaporate. They stick around, and some can move into groundwater, getting into places far from where they started. The World Health Organization lists arsenic as a human carcinogen. Breathing in dust or splashes from this acid opens the door to risk — for everyone from science lab workers to people who live near run-off zones. Some cases line up with respiratory problems or headaches linked to airborne particles. The biggest problem is that you can’t see or smell the risk. It’s a hidden hazard, quietly building up unless folks stay on top of prevention.
Old Habits Die Hard
Regulations have made a dent in use across many countries, but not everywhere. Some regions lag behind with enforcement, especially in lower-income areas, where growth promoters still get sold. Farmers rarely get training on chemical safety, and masks sit unused on dusty shelves. If you’ve handled poultry or worked near feed mills in such places, you’ll know how easy it is to get careless. Sometimes, it’s only much later, after illness, that the connection with arsanilic acid shows up. Full hazard awareness doesn’t trickle down as fast as it should.
Looking for Safer Ways Forward
Saying goodbye to 2-aminophenylarsonic acid calls for more than a rule change or a lecture. Farmers need tools and support for safe disposal. Soil and water must be tested long after the switch, because old contamination could linger. Safer alternatives exist. Producers using organic farming practices skip these chemicals entirely, but transitioning costs money and training. Government agencies can step up with funding and education, especially in places where the chemical still shows up. Investing in protective gear, teaching workers how to spot risks, and sharing simple disposal tips can keep families and communities safer. Small, steady steps beat big promises every time in keeping toxins out of our food and water.
How should 2-Aminophenylarsonic Acid be stored and disposed of?
Why Storage and Disposal Matter
After years spent working in labs and around chemicals, I learned that the dangers from some compounds don’t disappear just because the bottle is back on a shelf or tossed in the trash. 2-Aminophenylarsonic acid falls in that group. It contains arsenic, which can harm people and wildlife if handled carelessly. The headaches from an accident or slip-up last much longer than the few extra minutes it takes to play things smart.
Smart Storage Practices
Every reliable lab I’ve known keeps their hazardous reagents in dedicated areas, and 2-Aminophenylarsonic acid belongs right there. Store this chemical in a tightly sealed glass or high-density polyethylene bottle. The spot should be cool, dry, and shielded from sunlight, since heat and light can speed up decomposition or cause pressure build-up. Don’t place the container near acids or bases; the fumes can interact and spell trouble.
A real risk shows up from leaky containers. I once found out the hard way: a few drops seeped out overnight and ate through the shelf paint. A simple tray under each bottle, something made to catch drips, keeps a minor issue from growing. Labels matter, too. Don’t trust fading sharpie—use a chemical-resistant label with the full name and date it entered storage. If someone unfamiliar comes across it years later, they’ll know exactly what they’ve got.
Handling and Personal Protection
Skin and lung exposure can happen in seconds, so no shortcuts here. Standard nitrile gloves, a splash-proof lab coat, and safety goggles go on every time. If pouring or mixing, a fume hood pulls away vapors and dust. Some labs post a “PPE checklist” on the cabinet door to remind anyone reaching in what they should be wearing.
Disposal Without Shortcutting Safety
Disposal gets tricky with anything involving arsenic. Pouring 2-Aminophenylarsonic acid down a drain or tossing it in the regular trash isn’t just irresponsible; in most places, it’s illegal. Many states list arsenic wastes as hazardous under EPA regulations, so expect inspectors to ask about your practices during audits. Fines pile up quickly if you get caught dumping it the wrong way.
Collection in a clearly marked, leak-resistant waste container—usually glass or special plastic—makes transport safe. Add a label: “Hazardous Waste: Contains Arsenic Compound.” Keep it separate from non-arsenic waste to prevent surprise reactions if contents mix. Most labs arrange for a certified hazardous waste disposal company to handle pickup. They track the waste with paperwork from the lab to the incinerator or treatment site. Your local environmental or university safety office can list licensed contractors for pickups. Don’t be tempted to shortcut: every step leaves a paper trail.
Community Responsibility
No one wants their water or soil tainted by carelessly handled chemicals. One time, a simple oversight meant a batch of runoff from an old bottle wound up sparking a lengthy remediation. Neighbors, students, and wildlife all pay the price when hazardous chemicals leave the control of the folks who use them. Storing and disposing of 2-Aminophenylarsonic acid with care shows respect for everyone’s health. It only takes one mistake for the headlines to turn ugly.
Learn from my experience—always ask for the official process if you’re unsure, keep records, and maintain a culture that values safety over speed. The extra effort today prevents bigger regrets tomorrow.
| Names | |
| Preferred IUPAC name | [4-(Aminophenyl)arsonic acid] |
| Other names |
Orsanilic acid o-Aminophenylarsonic acid o-Arsanilic acid 2-Aminobenzenearsonic acid |
| Pronunciation | /tuː əˌmiːnoʊˌfɛnɪlɑːrˈsɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 98-50-0 |
| 3D model (JSmol) | Here is the **JSmol 3D model string** for **2-Aminophenylarsonic Acid**: ``` C1=CC=C(C(=C1)N)[As](O)(=O)O ``` *This is the SMILES string compatible with JSmol for generating its 3D model.* |
| Beilstein Reference | 1369673 |
| ChEBI | CHEBI:28512 |
| ChEMBL | CHEMBL502158 |
| ChemSpider | 14191 |
| DrugBank | DB14024 |
| ECHA InfoCard | 03d44b52-9c15-4779-8331-099434d3eb77 |
| EC Number | 213-668-5 |
| Gmelin Reference | 85743 |
| KEGG | C01086 |
| MeSH | D000701 |
| PubChem CID | 71184 |
| RTECS number | AS8925000 |
| UNII | 3T8XG6G2GI |
| UN number | UN3439 |
| Properties | |
| Chemical formula | C6H8AsNO3 |
| Molar mass | 249.07 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 1.7 g/cm3 |
| Solubility in water | slightly soluble |
| log P | -2.0 |
| Acidity (pKa) | 4.18 |
| Basicity (pKb) | 7.38 |
| Magnetic susceptibility (χ) | -59.0e-6 cm³/mol |
| Dipole moment | 2.24 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 186 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -489.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4196 kJ/mol |
| Pharmacology | |
| ATC code | J01XE01 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. Suspected of causing cancer. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | [Ar](=O)(=O)c1ccccc1N |
| Signal word | Danger |
| Hazard statements | H302, H319, H332, H373 |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P305+P351+P338, P330, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | > 230 °C |
| Lethal dose or concentration | LD50 (Rat, oral): 4100 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1810 mg/kg |
| NIOSH | NA0450000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2-Aminophenylarsonic Acid: 0.5 mg/m³ |
| REL (Recommended) | 0.05 mg As/m³ |
| IDLH (Immediate danger) | IDLH: 25 mg/m³ |
| Related compounds | |
| Related compounds |
Arsanilic acid Carbasone Nitarsone Roxarsone Acetarsone |

