Perfluorooctanesulfonyl Fluoride: A Deep Dive into Its Past, Properties, Uses, and Uncertain Future
Historical Development and Product Overview
Perfluorooctanesulfonyl fluoride, often called PFOSF, stands out within the family of per- and polyfluoroalkyl substances (PFAS). Tracing the roots of PFOSF brings us to the post-war boom in fluorochemical research. Companies sought new materials for non-stick cookware, stain-resistant fabrics, and fire-fighting foams—the so-called "miracles" of modern chemistry. PFOSF served as the key intermediate for manufacturing a variety of PFOS-based compounds, which began appearing in consumer and industrial products during the late 1940s and early 1950s. Industry insiders often mention the transformative effect these substances had on personal and public safety products but also the elusive environmental cost that only began to surface decades later. As regulations started to focus on environmental impact and public health around the turn of the millennium, a new conversation started about the long-term footprint these chemicals leave behind.
Physical and Chemical Properties
PFOSF carries a hefty molecule—it’s dense, highly stable, and almost slippery in a chemical sense. Its chemical resilience comes from the carbon-fluorine bonds, recognized as some of the strongest in organic chemistry. The compound resists heat, water, oil, and many acids, which explains many of its past and present uses. In the lab, it usually appears as a colorless, oily liquid, noticeable for a certain harsh odor and a high boiling point, traits that show both its utility and the precautions necessary during handling. Its extreme stability means that once it enters the environment, it tends to linger for years without significant breakdown, a quality once hailed for safety but now cited as a long-term risk.
Technical Specifications and Labeling
Industrial users and researchers both pay close attention to the purity and concentration of PFOSF, since even trace impurities can affect the performance of downstream products. Most handling and shipping labels warn about skin and respiratory hazards, echoing hard-earned lessons from chemists who have accidentally inhaled vapors or suffered chemical burns. Labeling usually reflects a growing emphasis on tracking, since environmental authorities now keep a closer eye on shipments and storage. Detailed specifications might include explicit temperature, storage, and ventilation recommendations, but they always point back to the overarching caution that goes into working with such persistent chemicals.
Preparation Methods and Chemical Reactions
Most people outside the chemistry world would not expect the elaborate steps, high pressures, and aggressive reagents needed to produce PFOSF. Typically, electrochemical fluorination of octanesulfonyl fluoride forms this molecule, a process requiring specialized equipment and handling skills. This remains a hazardous undertaking—hydrogen fluoride byproducts, for instance, pose significant risks to workers. PFOSF itself reacts with a variety of chemicals, forming sulfonamide derivatives and PFOS salts. These reactions once made it a darling of the specialty chemical industry, opening the door to an array of fire-resistant and stain-repellent materials. Now, any discussion about PFOSF chemistry turns sharply to safe alternatives, reduced emissions, and containing leaks at every stage.
Synonyms, Product Names, and Terminology
Even seasoned chemists get caught off guard by the number of names PFOSF carries. Names change depending on historical usage, regional practices, and regulatory tradeoffs. In older literature, references often crop up under synonyms such as perfluorooctanesulfonic fluoride or even “FC-98.” This naming maze often frustrates people trying to interpret reports or regulatory documents, leading to inevitable confusion or miscommunication. The tangle of aliases makes calls for standardized chemical identifiers more urgent—not just for paperwork, but for tracing the fate of these compounds in international markets and environmental monitoring.
Safety and Operational Standards
Handling PFOSF safely calls for more than just standard gloves and goggles. Veterans in chemical manufacturing tell stories of relentless training and the need for airtight facilities with high-efficiency scrubbers and emergency wash stations. Accidental spills, even small ones, require fast action and proper containment, as the material’s persistence means it can contaminate water and soil quickly and last for decades. Those who work around these chemicals often express frustration about shifting safety guidelines, given that new toxicology data emerges every few years. Standards have tightened, now mirroring a deeper appreciation for long latency risks, especially occupational illnesses. Compliance often ties closely to regulatory reporting mandates that seek a full accounting of exposure and emission sources.
Application Area
For years, industries prized PFOSF derivatives for their ability to impart resistance to oil, water, staining, and heat. These features shaped countless consumer products: textiles that shrugged off red wine, carpets that tolerated years of kids and pets, firefighting foams that saved airfields and fuel depots from catastrophe. The same properties that protected people and assets later appeared in electronics manufacturing, pesticides, and even in photographic film production. As awareness of contamination risks grew, many applications phased out or leaned on alternative chemistries, but some niche uses, including certain types of industrial surface treatments, still hold out as regulations and supply chains recalibrate.
Research and Development
Ongoing research into PFOSF mirrors a broader search for alternatives that match its performance but without long-term toxicity and environmental impact. Scientists face a tough challenge here. Even minor changes to the molecule often reduce chemical stability or increase production costs, creating tough choices for companies depending on these materials to meet strict durability standards. Some research pivots toward bio-based or degradable fluorinated molecules, but the record shows no easy chemical shortcut. Research teams also focus on better methods for detection and remediation of PFOSF in the environment, realizing that historic and legacy contamination requires as much innovation as developing new molecules.
Toxicity Research
Debate about PFOSF toxicity sharpened as studies produced troubling findings in wildlife and human populations. PFOSF and its derivatives resist breakdown, accumulating in blood, tissue, and the broader ecosystem. Researchers found links between exposure and problems such as thyroid disruption, immune changes, developmental delays, and some cancers. The path from laboratory rodents to clear public health guidelines doesn't come easy, as effects depend on both dose and duration. Yet mounting evidence prompted regulatory bodies to restrict production and push for stricter cleanup standards. Researchers believe there’s more to discover, especially about low-dose, long-term exposure. What sets this apart from other chemical debates is how slowly some effects might surface, demanding vigilance even after use tapers off.
Future Prospects
PFOSF stands at an uneasy crossroads. Even as countries tighten regulations, stockpiles linger, and older sites continue to release trace amounts into rivers, groundwater, and the air. Switching to safer alternatives brings its own technical, regulatory, and cost challenges, giving the chemical industry little margin for error. Stronger investment in green chemistry and improved safety protocols offers the best shot at phasing out legacy compounds while also uncovering more about degradation processes and possible methods for cleaning up what’s already out there. People and policymakers need to keep their eyes on both the science and the changing realities of global regulation, especially given the slow, difficult task of environmental restoration. The PFOSF story demonstrates the risks of relying too heavily on one class of molecules simply because they seem impervious—nature often reveals the hidden liabilities only after decades of widespread use.
What is Perfluorooctanesulfonyl Fluoride used for?
A Closer Look at PFOSF’s Reach
Perfluorooctanesulfonyl fluoride—let’s call it PFOSF—plays a bigger role in industry than most people imagine. Long before anyone started worrying about “forever chemicals,” companies relied on this chemical to solve tough problems. I spent time working in a lab environment and later reporting on industrial chemistry, so I’ve seen firsthand how certain compounds gain almost a legendary status among manufacturers. PFOSF belongs to that league, serving as a building block for products that needed to deal with oil, water, stains, or major temperature swings.
PFOSF and Waterproofing
My earliest encounter with PFOSF came from the textile world. Rain jackets that held up year after year owed some of their resilience to treatments based on PFOSF derivatives. Once, while researching outdoor gear supply chains for a story, I learned that many mills used these treatments because consumers expected clothing that shrugged off downpours and mud. Fabric finishers preferred PFOSF chemistry over old-fashioned waxes that broke down fast. Carpets did, too—companies provided stain-proof promises across entire product lines, banking on the power of this fluoro-based barrier. The trouble is, what makes PFOSF good for resisting spills also keeps it from breaking down in the environment.
Industrial Uses and Everyday Perks
Fluorosurfactants based on PFOSF bring value to firefighting foams. These foams helped put out fuel fires at airports and plants, where nothing else worked as reliably. A lot of firefighters I met during my reporting swore by older foams, even though they knew the environmental concerns. In plating shops, PFOSF derivatives made chrome look smooth, keeping mists of toxic stuff out of the air. Electronics factories relied on them for etching delicate circuits—splashing acids don’t mix well with contamination, and PFOSF held everything in check.
The Trouble with Longevity
No conversation about PFOSF can skip its dark side. Studies show that variants like PFOS and related substances persist in soil, water, and bodies for years or decades. The EPA and independent researchers have linked exposure to issues like hormone disruption and some cancers. Few folks grew up thinking about “bioaccumulation,” but scientific evidence doesn’t care if a word feels remote or technical—what matters is that these molecules get everywhere. I remember talking with a fishery manager who had to inform people about contaminated catches; local families just wanted clean food, not a chemistry lecture or a legal fight.
Solutions Moving Forward
Some chemical giants are trying to turn the page by producing substitutes with shorter environmental tails. Regulation is picking up, with governments limiting old-school PFOSF use. I’ve seen companies invest in research to clean up sites, swapping out gear for alternatives. It’s not as easy as banning a bad ingredient—engineers and business owners need tough, affordable replacements. Fire chiefs want assurance that new foams won’t put their teams at risk. The push for transparency is real; consumers and advocacy groups press brands to share what goes into their products. Everyone along the chain—chemists, regulators, shoppers—has a role in turning momentum into safer practices. Innovation follows when lives and water sources end up on the line.
What are the safety precautions when handling Perfluorooctanesulfonyl Fluoride?
The Real Risks Behind the Science
Perfluorooctanesulfonyl fluoride, often called POSF, isn’t something most folks run across at home. Laboratories and factories feature this chemical, and hazards follow close behind. In my years chatting with chemists and touring facilities, stories about strict protocols become almost routine. The reason stands out: POSF can hurt people and the environment if ignored. Data from the Occupational Safety and Health Administration points out that accidental inhalation or skin contact can spark health problems, from burns to long-term systemic damage. Stories of folks bypassing rules only to end up at the clinic stick with me. The chemistry looks impressive, but real people count on safety every shift.
Personal Protection: More Than Just A Reminder
High-quality gloves matter. Nitrile gloves stand up to chemicals better than the flimsy latex ones in my desk. Goggles shouldn’t slip around or fog up. Lab coats need to reach wrists and fall below the waist. Chemical splash suits, boot covers, and full-face respiratory masks round out the gear. Workers I’ve spent time with never settle for shortcuts. They know if POSF makes skin contact, it can bring burns and severe irritation. Breathing in vapor? Permanent lung injury. These dangers cannot be shrugged off. Using proper personal protective equipment gives science a real-world safety net—and that net saves lives.
Ventilation: Crucial for Air Quality
Hood fans in research labs aren’t just there for show. Engineers install powerful filtration systems to keep the air clean and free from dangerous fumes. Inhalation of POSF isn’t just uncomfortable—it can scar lungs or trigger chemical pneumonia. I remember lab techs recounting drills where air flow got monitored before the day's experiments even started. Knowledge and vigilance form the backbone of safe working spaces. Sensors in the ceiling and at benches catch trouble before symptoms hit.
Storage: Locking Down the Risks
POSF stays in tightly closed containers marked with clear, indelible labels. Steel cabinets bolted to the floor or wall raise the bar. These containers keep humidity, light, and heat away. Heat transforms even small spills into a room-wide hazard. Acids and oxidizers never share a shelf with POSF—mixing those together amps up the risk. After a local company faced an explosion from poor storage, word traveled fast: safety rules come before productivity.
Spill Response: Action Over Panic
No one wants to scramble after chemicals hit the floor, but spills do happen. I’ve watched teams trained to handle emergencies lock down an incident in minutes. Absorbent material, sealable drums for cleanup, and well-marked wash stations stand nearby. Once, someone told me about a minor POSF spill where response and good training made all the difference—nobody got exposed, and the facility faced no downtime. The Environmental Protection Agency requires reporting even small spills in some regions, so every move counts. These policies stem from real cases of lingering contamination that sickened workers years after carelessness.
Disposal: Respect for the Environment
Disposing of POSF means calling qualified chemical waste handlers. You won’t find it down any regular drain. Specialized facilities can safely incinerate or neutralize this compound. Recent EPA findings show that water tables near improper disposal sites carry traces of POSF years later. Once this stuff gets out into the world, it refuses to leave. The danger isn’t just to people inside the lab—it’s to their cities, water supplies, and wildlife. Strict adherence to legal disposal standards protects everyone in the community, not just the professionals within facility walls.
What is the chemical structure of Perfluorooctanesulfonyl Fluoride?
A Closer Look at the Molecular Blueprint
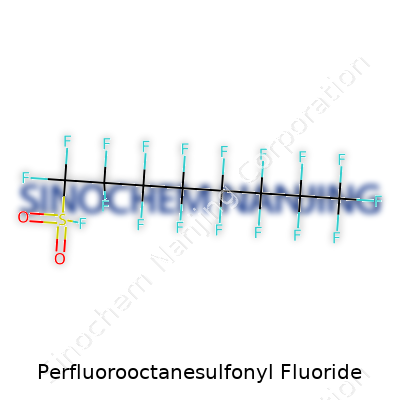
Perfluorooctanesulfonyl fluoride, also known as PFOSF, carries weight in both chemistry labs and environmental circles. Its structure looks simple on the surface: an eight-carbon chain, each carbon fully loaded with fluorine atoms, and a sulfonyl fluoride group tagged onto one end. In chemistry jargon, you see it written as C8F17SO2F. This string of carbons and fluorines isn’t an example of nature's handiwork. Human scientists designed it for jobs where most other molecules fail.
Those fluorine atoms, tightly clinging to the carbon backbone, matter because they make the entire molecule tough and slippery. PFOSF doesn’t break down in water, sunlight, heat, or most common chemicals. The sulfonyl fluoride group attached—SO2F—becomes reactive during manufacturing, opening doors to turn this basic skeleton into all sorts of useful products. The reaction chemistry here sets PFOSF apart, making it a building block for a whole family of chemicals.
What Sets PFOSF Apart
In job sites like electronics, firefighting foams, and the textile industry, folks often reached for PFOSF-based ingredients. The reason ties back to its structure. Those fluorinated chains love to repel pretty much everything—water, oil, even dust. Regular detergents can’t wash these bonds away, and the environment has a hard time breaking them down too. From water-repellent jackets to stubborn stains on carpets, PFOSF often left its mark in ways both useful and problematic.
Living in a community downstream of a chemical plant, I once saw firsthand how local waterways continued to test positive for PFOS derivatives long after manufacturing stopped. The structure that makes PFOSF so useful is exactly what lets it stick around for decades. You never see it degrade in a backyard compost pile or break down after a rainstorm. Real problems show up when these molecules accumulate in fish, wildlife, and even human blood—raising big questions about our long-term health.
Paths Toward Safer Chemistry
PFOSF’s story isn’t just about clever design. It highlights how chemists and policymakers both need to consider the whole life cycle of a molecule. Long carbon-fluorine chains don’t just wash away. Industry spent years searching for safer molecules that offer the performance of PFOSF without the legacy of environmental damage. Some replacements swap out parts of the chain, making the molecule shorter and a bit easier to degrade. The move toward so-called “short-chain” alternatives isn’t perfect; it’s a work in progress with researchers and watchdog groups still pushing for better answers.
Lighting the path forward, scientists use high-resolution mass spectrometry and other tools to track how new molecules move through water, soil, and food webs. This data gives us the proof needed to shape laws and guide companies toward smarter formulas. We don't have to accept that high performance always goes hand-in-hand with long-term pollution. By digging deeper into the chemical structure of molecules like PFOSF, future generations have a shot at building chemicals that do the job and leave no trace behind.
How should Perfluorooctanesulfonyl Fluoride be stored?
Why Smart Storage Makes a Difference
Perfluorooctanesulfonyl fluoride (POSF) carries a reputation for being both useful and risky. Plenty of chemical plants use this compound to make fluorinated surfactants, fire-fighting foams, and specialty materials. The upside looks attractive until you see how easily a slip-up can send people or the environment down a tough road.
My work in chemical safety has shown me that shortcuts with storage never stay hidden. One day an old drum’s seal shows stress; next thing, you’re dealing with fumes nobody in the building wants to breathe. Stories like these explain why storage isn’t just a technicality—it’s central to the health of workers and the neighborhoods nearby.
What Real Storage Looks Like
Some sections of the industry keep POSF in carbon steel drums lined with chemical-resistant coatings. That’s basic, but alone, it doesn’t cut it. At temperatures above 40°C, this compound gets eager to decompose and can eat up sealing materials. At the same time, moisture invites slow hydrolysis, leading to the production of perfluorooctane sulfonic acid, a compound linked to serious health concerns and environmental persistence.
A climate-controlled warehouse cuts this risk sharply. Think cool, dry, and shaded, with strict access control. It’s important to install ventilation, not just circulate air but capture stray fumes. In my experience onsite, basic fans spread fumes faster than they clear them unless filters built for acid gases back up the system.
Corrosion-proof bunds, spill kits built for fluorinated materials, and ready-to-grab personal protective equipment show respect for the risks. Labels that fade or peel wipe away hard-earned trust with regulators. I watched one audit spiral fast when an inspector couldn’t read a hazard symbol. Nobody needs that kind of stress—or the hazard that goes with it.
Routine Monitoring Beats Regret
Chemicals change over time, especially when exposed to warmth or wetness. Monitoring temperature and moisture content around storage drums saves time, money, and possibly lives. Leaks rarely announce themselves loudly. Drip trays, regular drum inspections, and logbooks written by hand—not just entered by a bot—keep track of things that automated systems miss.
Plenty of places skimp on training. I’ve watched teams coast for years without a safety drill. When a real emergency hit, only muscle memory saved the day—trained responses, neighbors helping each other before first responders even pulled up. That’s something you only pick up if everyone in the facility gets honest about what’s actually stored there.
Solutions That Keep Risk Small
Shifting toward smaller batch storage, using double-walled containers, and keeping raw stock and waste streams clearly separated helps reduce mistake odds. Transparency counts. Sharing incident reports across facilities fast-tracks learning. Real-time data loggers on drums let facility managers check readings on a smartphone, alerting them to danger points before a problem grows.
So much of what keeps POSF safe in storage comes down to attention and care. Facilities that stay open with workers about hazards, update procedures when regulations change, and keep communities nearby in the conversation set a standard everyone should copy. Risk scales fast in the dark, but it shrinks where people shine a light and take responsibility together.
Is Perfluorooctanesulfonyl Fluoride hazardous to the environment or human health?
What Poses a Threat?
Perfluorooctanesulfonyl fluoride, usually shortened to POSF, might sound technical on paper but causes real and lasting problems in the world around us. POSF is a key chemical in making substances called PFOS and other per- and polyfluoroalkyl substances (PFAS). These compounds became famous for their ability to resist heat, water, and oil, fueling the success of products like nonstick pans, water-repellent clothing, and firefighting foams. But the story goes way beyond shiny marketing promises.
The Staying Power of POSF
In my experience working with environmental surveys, very few substances show the stubborn staying power of PFAS chemicals. POSF does not break down easily, not even over decades. Scientists often call PFAS “forever chemicals.” They collect in soil, groundwater, and rivers—wherever they end up, they stay. Once POSF enters an ecosystem, fish and wildlife show traces in their bodies, and water supplies sit tainted for generations. People often find out about contamination too late, after poisons make their way into drinking water or fish from a local creek.
Impact on Human Health
Everyday routines start to seem less safe when invisible substances float in the things people use or consume without a second thought. Research from the US Environmental Protection Agency links PFAS, including POSF-derived compounds, to an array of health problems—some subtle, others potentially life-altering. Exposure can lead to higher cholesterol, problems with the immune system, thyroid disruption, and more. Some studies link extended exposure to a higher risk for certain cancers and reproductive harm. Often, it is the people living near manufacturing facilities or firefighters handling foam who face the highest risks, but virtually anyone can be exposed through contaminated water or food.
Regulatory Push and Public Pressure
Communities have raised their voices, demanding safer products and cleaner water. Governments have started to pay closer attention, with agencies worldwide setting tighter rules. For example, the European Union keeps pushing for stricter controls on both the use and release of PFAS. In the United States, the government updates drinking water standards and funds cleanup projects in contaminated regions. Despite these steps, the market keeps churning out products made with chemicals that trace their origins to POSF. It takes time to phase out chemicals; my own conversations with industry experts suggest that finding safe, affordable alternatives isn't always simple—but it’s hardly impossible either.
Solutions and the Road Forward
Banning or sharply limiting POSF in manufacturing stands out as one way forward. Some companies are switching to safer alternatives, using green chemistry approaches to develop new water- or stain-resistant materials. Grassroots groups have forced greater transparency, so people know if their local water or environment faces a threat. On an individual level, people are taking more care, reading product labels and asking deeper questions about how goods get made. Scientists keep pushing for better detection methods that spot contamination faster and cheaper, which means less time before a problem gets fixed.
Living with the legacy of POSF showcases the challenge of balancing industrial progress with long-term health. The only sustainable choice requires action at every level—industry, government, and the public—so the next generation doesn’t face an even larger toxic burden.
| Names | |
| Preferred IUPAC name | Perfluorooctanesulfonyl fluoride |
| Other names |
Perfluorooctylsulfonyl fluoride PFOSF Heptadecafluorooctanesulfonyl fluoride 1-Perfluorooctanesulfonyl fluoride Perfluorooctane sulfonyl fluoride |
| Pronunciation | /pɜːrˌflʊəroʊˌɒk.teɪnˈsʌl.fə.nɪl ˈfluː.ə.raɪd/ |
| Identifiers | |
| CAS Number | 307-35-7 |
| Beilstein Reference | 1718733 |
| ChEBI | CHEBI:39040 |
| ChEMBL | CHEMBL236474 |
| ChemSpider | 78996 |
| DrugBank | DB11258 |
| ECHA InfoCard | 03f18053-8c5f-416d-a0d1-fc5caf40b6b8 |
| EC Number | 206-397-9 |
| Gmelin Reference | 82116 |
| KEGG | C18507 |
| MeSH | D010528 |
| PubChem CID | 87716 |
| RTECS number | YV9625000 |
| UNII | UC679VDG4S |
| UN number | UN2424 |
| Properties | |
| Chemical formula | C8F17SO2F |
| Molar mass | 503.13 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Odorless |
| Density | 1.88 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 2.8 |
| Vapor pressure | 67.5 mmHg (20 °C) |
| Acidity (pKa) | -2.8 |
| Basicity (pKb) | -5.1 |
| Magnetic susceptibility (χ) | -32.6e-6 cm³/mol |
| Refractive index (nD) | 1.299 |
| Viscosity | 2.2 cP (25°C) |
| Dipole moment | 4.63 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 489.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1671 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -9804 kJ/mol |
| Hazards | |
| Main hazards | Toxic if inhaled, swallowed, or in contact with skin; causes severe skin burns and eye damage; may cause respiratory irritation. |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P260, P262, P273, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P310, P363 |
| NFPA 704 (fire diamond) | 3-0-2-W |
| Lethal dose or concentration | LD₅₀ (oral, rat): >2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): "200 mg/kg (oral, rat) |
| NIOSH | WA8400000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Perfluorooctanesulfonyl Fluoride: Not established |
| REL (Recommended) | 0.005 mg/m3 |
| IDLH (Immediate danger) | IDLH: Not established |
| Related compounds | |
| Related compounds |
Perfluorooctanesulfonic acid Perfluorooctane sulfonamide Perfluorooctane sulfonic acid potassium salt Perfluorooctyl iodide Perfluorooctanoic acid |

