Perchloromethyl Mercaptan: A Closer Look at a Chemical Powerhouse
From Early Days to Modern Labs: A Story of Discovery
Few chemical compounds have sparked as much intrigue as perchloromethyl mercaptan. Chemists first isolated this sharp, pungent substance in the late nineteenth century, back when the appetite for innovation often outpaced the understanding of risks. Its significance grew over the past century, mainly thanks to its versatility in complex organic synthesis. In academic circles, folks who studied organosulfur chemistry found themselves drawn to this molecule’s unique mix of reactivity and selectivity. Its historical path runs in step with society’s push toward more effective crop protection and advanced manufacturing processes. Each era found fresh uses, and worldwide demand reflected the ebb and flow of shifting industrial priorities.
Perchloromethyl Mercaptan at a Glance
There’s nothing subtly anonymous about perchloromethyl mercaptan. This compound, known also as trichloromethanesulfenyl chloride, carries the formula CCl3SCl. Its distinctive smell and corrosive properties keep folks on their toes. Workers in chemical plants, my former colleagues among them, recall the unmistakable sting in the air during handling. Rather than fading into the background, it commands attention because it both opens doors for manufacturing possibilities and throws up glaring red flags for safety. Despite its hazards, it remains a workhorse in select fields where no clear substitute quite matches its chemical power.
Physical Grit and Chemical Punch
Perchloromethyl mercaptan usually shows itself as a yellowish or clear liquid. Its density sits a bit heavier than water, which lets it settle at the bottom when poured into a jar with water on top. It doesn’t bother trying to mingle gracefully—it hydrolyzes, and the resulting mix releases hydrochloric acid vapors and sulfurous compounds. The smell is both offensive and telling, recalling classic sulfur compounds that assault the senses before anyone even glances at the label. Its boiling point hovers around 147°C, which means it stands up to moderate heat, but its volatility still brings real fire and inhalation risks—a fact that more than one veteran of the chemical industry remembers all too well.
Technical Specs and Labeling: More Than a Checklist
Companies that deal in perchloromethyl mercaptan slap their technical specs straight onto packaging, warning anyone nearby that they’re not just carting around water and vinegar. The labeling not only highlights concentration and purity but often goes further, detailing decomposition products and emergency handling guidelines. Regular users know the label is their first line of defense. In my days in chemical safety training, I saw how even a small misreading of a label or a missing technical detail could send an ordinary workday spiraling into chaos.
How Chemists Make It: A Relentless Approach
Production methods didn’t just land on perfection by chance. The most common preparation involves reacting carbon tetrachloride with sulfur monochloride, sometimes employing an iron catalyst to speed up the process. This recipe sounds simple but demands careful temperature control: a runaway reaction risks violent pressure surges and dangerous emissions. The preparation’s success hinges as much on experienced hands and rigorous monitoring as on raw ingredients. I recall learning about the challenge firsthand during a university research internship, watching the blend of chemistry and discipline turn dangerous precursors into a valuable product.
Chemical Reactions and the Art of Modification
In the wild west of synthetic chemistry, perchloromethyl mercaptan carves out a distinct place. It’s reactive, yes, but in the hands of a skilled scientist, it becomes a reliable starting point for the construction of sulfenyl chlorides, dithiocarbamates, and certain pesticides. The molecule responds briskly with nucleophiles, offering up a range of derivatives depending on who’s doing the asking and how persistently. Its usefulness in making crop protection agents comes from this reactivity, giving manufacturers the tools they need to customize molecules to target particular insects or weeds.
By Any Other Name: Synonyms and Common Labels
The jargon around perchloromethyl mercaptan includes names like trichloromethanesulfenyl chloride and trichloromethylsulfenyl chloride. Experienced plant operators and researchers—especially those who’ve switched employers or research topics—recognize that a chemical’s name may change from one technical paper to the next. Regulatory documents and transport records sometimes list it under old or translated names, so the onus falls on chemists to keep a sharp eye for synonyms. This web of terminology means anyone serious about safety or research can’t afford to make assumptions.
Safety, Standards, and the Human Factor
Anyone who has worked with perchloromethyl mercaptan develops a deep respect for both the substance and the regulations built up around it. Its toxic fumes and potential for burns mean that personal protective equipment isn’t optional—even a passing whiff can lead to coughing or much worse. Industrial standards, written as they are by people who have lived through real accidents, extend beyond simple recommendations to include mandated ventilation, spill containment plans, and redundant safety checks. It’s not paranoia; it’s a response born from harsh experience. The stories that circulate among plant workers stay with you—the rushed cleanup gone wrong, or the near-miss that only a well-practiced evacuation drill turned into a learning moment instead of a headline.
The Reach of Perchloromethyl Mercaptan: Beyond the Factory Floor
The most well-known application of perchloromethyl mercaptan lands in agrochemicals, where it paves the way for products that protect fields from ravaging pests. Its derivatives power the effectiveness of established pesticides and fungicides. Out of the spotlight, it also plays supporting roles in dye manufacturing and specialty organic synthesis. No one really hunts for it on supermarket shelves, but its fingerprints turn up on finished products in ways the average person might never suspect. From textile colors to rubber processing aids, it serves quietly, often well behind the scenes.
Experiences in Research and Promoting Safer Practices
Research into perchloromethyl mercaptan doesn’t slow down. Teams keep probing its chemistry, seeking routes that trim risk or reduce the number of steps needed to reach a desired molecule. In academic circles, students learn from the old stories and the latest journal articles how to handle it responsibly. Every experiment that trims waste or turns up a less hazardous byproduct gets attention, because no one wants to see their name linked to an accident. Laboratories run drills, supervisors put pressure on newcomers to memorize emergency protocols, and research continues to test greener synthesis pathways.
The Steep Price: Toxicity and Its Implications
Some compounds promise utility but demand a significant cost in safety. Toxicity research into perchloromethyl mercaptan draws a grim picture—exposure means trouble, and high doses can lead to organ damage or death. Chronic exposure isn’t something most folks sign up for, so the medical literature fills with case reports and animal model studies. These cautionary tales shape the rules, reminding everyone who steps into the lab that safety infrastructure forms just as crucial a part of innovation as any new reaction. Public health authorities watch closely, updating permissible exposure limits and making sure employers keep workers informed and protected.
Eyeing the Future: Where Could It Go From Here?
With advances in green chemistry and growing societal pressure for environmental stewardship, the story of perchloromethyl mercaptan continues to change. Scientists who once focused just on output now collaborate with teams designing safer alternatives or improved containment technologies. There’s a push to develop replacement compounds for its hazardous roles, but that often comes with trade-offs between performance and environmental impact. Industry veterans admit that while the chemical remains valuable for specialty synthesis, newer approaches like high-precision catalysis and enzyme-based routes begin to shoulder some of the load. If the past decade has taught us anything, it’s that even old stalwarts face retirement or reinvention, especially in a world that prizes both progress and precaution. Investments in education, safer plant designs, and stricter regulation signal the way forward, because the safest chemical is always the one that never has to leave the bench without a backup plan.
What is Perchloromethyl Mercaptan used for?
From Lab Benches to Industry Giants
Perchloromethyl mercaptan, known to chemists as CCl3SCl, pops up in conversations more often than most folks expect. I first learned about it during an industrial safety training at a chemical plant. Anyone who’s ever popped the cap off a bottle of this stuff never forgets the pungent smell. To a handful of manufacturers, though, that strong odor signals potential. Perchloromethyl mercaptan draws huge demand from folks working with pesticides—especially insecticides. The world leans heavily on global food production, so shocking as it sounds, this stuff keeps crops going in regions where insects devastate yields. It serves as a building block for chemicals like ethyl parathion and fenthion, tools farmers have long relied on.
Here’s some perspective. In the seventies and eighties, pesticides made with intermediates like perchloromethyl mercaptan saved countless harvests. Today’s food supply chains evolved thanks, in part, to chemical solutions dreamed up by agricultural scientists. The “better living through chemistry” motto defined that era. The tide has shifted lately, with folks calling for more organic methods, but the world still hasn’t fully weaned itself off the tough stuff. Until new technologies step up, intermediates like perchloromethyl mercaptan still play a vital role on farms across continents.
Risks Few Can Ignore
Perchloromethyl mercaptan, for all its industrial uses, brings real risks. It’s not just the overpowering odor. I remember reading safety data sheets after a near-miss during a shipment inspection: exposure can attack the eyes, lungs, and skin. Repeated exposure for workers even in small amounts means bigger worries—headaches, shortness of breath, burning sensations. That’s why industrial hygiene teams I’ve worked with always treat this chemical as a top hazard with dozens of layers of safeguards. Spills turn into emergencies fast, and every moment counts.
Environmental risk runs deep, too. With pesticides, traces of perchloromethyl mercaptan and the compounds made from it can leach into groundwater sources or stick to food crops. Some of these residues can carry long past the fields where they started. Overuse in poorly regulated regions sometimes means soil damage, biodiversity loss, and serious harm to local communities. Folks who grew up near heavy chemical zones know what happens when corners get cut.
Pushing Toward Safer Solutions
The future of perchloromethyl mercaptan ties directly to industry innovation and accountability. Companies, large and small, face tough scrutiny from environmental groups, local governments, and neighbors. As someone who’s worked at companies balancing efficiency and safety, I’ve watched approaches evolve over the years. Engineering controls, such as local exhaust systems and chemical vapor detectors, became non-negotiable. Regular worker health screenings save lives. Training goes beyond checklists—everyone from operators to transport drivers needs to spot leaks and symptoms early.
Research points to food scientists and chemists alike searching for “greener” alternatives—biological controls, integrated pest management, and better crop genetics. Still, not every region can afford these advances at scale yet. Global efforts must support knowledge transfer, worker protections, and investment in better technologies so every community can move past reliance on potentially hazardous chemicals.
Perchloromethyl mercaptan remains a staple in some sectors of crop protection chemistry. Its significance to agricultural output can’t be overstated. Responsibility sits squarely with manufacturers, governments, and every worker who handles these compounds. With better safeguards and a push toward cleaner ways of farming, the hope is future generations won’t face the same choices between productivity and safety.
What are the safety precautions when handling Perchloromethyl Mercaptan?
The Reality of Handling a Potent Chemical
Few chemicals make professionals stop and double-check their safety habits like Perchloromethyl mercaptan. This stuff isn’t famous, but anybody who has spent time in an industrial lab or in chemical manufacturing knows its reputation. Even before the scientific explanations start rolling in, most folks notice its intense, garlic-like odor—the warning sign that things are about to get serious. My first real job involved helping with inventory where this compound was present, and the atmosphere tightened immediately once anyone mentioned it.
Not Your Average Safety Protocol
Breathing in the fumes from Perchloromethyl mercaptan can leave you with burning lungs and watering eyes. Too much exposure can knock you out cold, trigger asthma-like symptoms, and tear up your respiratory system. Even simple contact with your skin brings out a nasty reaction— redness, irritation, sometimes chemical burns. And let’s not forget that just a little goes a long way. The effects don’t wait for someone to get careless two or three times, so being careful on "most days" isn’t enough.
One key lesson: never trust a half-mask respirator with this compound unless it’s fitted right and you keep your filters fresh. Go for full-face protection if there’s even a chance of a spill or splash. In labs where I’ve worked, we had strict written rules about changing out gloves every hour—nitrile or neoprene only—since breakthroughs aren’t theoretical stories, they actually happen. The minute there’s a drip on your glove, you toss them and wash your hands. It sounds strict, but the risk isn’t worth saving a few bucks in disposable PPE.
Proper Storage: No Room for Shortcuts
It’s tempting to treat smaller chemical bottles with less caution than giant drums, but Perchloromethyl mercaptan brings out the “treat it like acid” instincts in people who know the risks. These bottles stay in tightly-sealed, clearly marked containers, far from heat or ignition sources. Putting them in a locked chemical cabinet with solid ventilation isn’t just about following rules. The vapor can sneak out, so relying on ventilation and restricted access saves both property and lives if there’s a leak.
What Happens on Cleanup Day
Mistakes still happen. One coworker once knocked over a sealed bottle and a small leak started. Alarms went off, everyone followed the drill, and the spill kit for toxic organosulfur compounds came out right away. Nobody went back into that area without a proper respirator, chemical splash goggles, and fresh gloves. We isolated the ventilation, neutralized the spill with sodium bicarbonate, and double-bagged every contaminated item as toxic waste. Even the floor got scrubbed, and no one complained about the time spent. Watching firsthand how people reacted to the incident showed me that a tight routine isn’t just a paperwork thing.
Cultivating Respect Through Training
People handling dangerous chemicals count on good training and real stories, not only handouts and safety posters. One good supervisor talked through close calls and walked us through emergency showers and eyewash stations before we even saw the chemical. New staff shadowed experienced handlers every day for a week. These steps build confidence and healthy fear—exactly what complex and risky chemicals demand.
I believe the difference between a minor event and a tragedy often rests on solid routines, honest experience sharing, and not letting comfort sneak into the workplace. We can’t take shortcuts with Perchloromethyl mercaptan, and the lessons learned carry over to safer handling across the board.
What is the chemical formula of Perchloromethyl Mercaptan?
Understanding the Chemical Formula
Perchloromethyl mercaptan, known by chemists for its pungent odor and notable toxicity, is a compound carrying the chemical formula CCl3SCH3. There’s no mystery behind the structure; each molecule consists of a trichloromethyl group (CCl3-) joined to a methylthio group (-S-CH3). Chemists often abbreviate it as PCM for convenience, but there’s nothing convenient about handling or working with it.
The Real Impact of Perchloromethyl Mercaptan
In the chemical world, this substance stirs up strong feelings, largely because of the safety risks tied to its use. Its applications stretch mainly into the production of pesticides and herbicides—industries that lean hard on efficient and reactive compounds to do tough jobs. The compound’s unique structure makes it valuable for creating chemicals that protect crops, but that same structure also introduces real hazards. Exposure to its vapor can lead to headaches, nausea, and, at higher levels, respiratory problems or even more serious health issues. In my own chemistry work, respect for this molecule starts the moment it’s on the reagent list; its reputation for being both reactive and noxious is no exaggeration.
Why Formula Knowledge Matters
Recognizing the structure CCl3SCH3 does more than just satisfy scientific curiosity. It means technicians, researchers, and workers can anticipate how it’ll react with other substances. The compound’s carbon, chlorine, and sulfur atoms interact in ways that can release harmful gases or cause unexpected reactions if mishandled. I remember during my training seeing a fume cabinet set up specifically to contain even the smell of PCM—ventilation, gloves, and closed bottles were mandatory, without exception. Those protocols exist for good reason. Anyone coming into contact with PCM, whether in a factory, lab, or at a cleanup site, can’t afford to overlook the details of its chemical makeup.
Risk Management and Solutions
In places where PCM gets manufactured or used, health and safety training takes center stage. The Material Safety Data Sheet (MSDS) for PCM reads like a checklist of everything that could go wrong—spill kits, respirators, and full-body protective gear are part of standard procedure. Companies have started to review their use of such chemicals on a regular basis, balancing the requirements of crop protection with the need to keep workers safe. Substitutes with less severe health impacts can reduce risk, but switching isn’t always simple. The chemistry is precise; not every replacement does the job as well. Still, investment in better engineering controls, improved training, and research into safer alternatives points the way forward.
Supporting Safer Science
Transparency around chemicals like PCM feeds public trust. Regulations—the kind set by OSHA, the EPA, and international bodies—haven’t just popped up out of nowhere. They were shaped through real cases, studied responses, and, sometimes, tragic mistakes. Sharing up-to-date facts about these chemicals supports better habits and smarter choices, not just for those in the labs, but also for anyone affected by what happens as a result of using them. Understanding what CCl3SCH3 is, what it can do, and how it can be managed responsibly isn’t just technical know-how; it feels like a basic obligation to anyone working with complex chemistry in the modern world.
How should Perchloromethyl Mercaptan be stored?
Handling the Risks Up Close
Perchloromethyl mercaptan packs a dangerous punch—sharp, irritating odor, toxic fumes, burns that linger. This isn’t just another industrial chemical; anyone who’s ever worked around it knows a casual approach isn’t just careless, it’s risky. One mishap with this stuff and you never forget the lesson.
Space and Containment
A solid plan starts with where you keep it. I’ve seen both small labs and large plants underestimate ventilation and spacing for chemicals like this. An isolated, locked room with forced ventilation always gets top marks. The less foot traffic, the better. Metal cabinets lined with corrosion-resistant coating beat flimsy storage every time. Any sign of leakage means immediate cleanup; these odors aren’t just unpleasant—they signal a real threat.
Temperature Matters
Heat always ramps up volatility. I always recommend a cool, stable climate. I’ve watched folks cut corners by shelving chemicals near heat sources—machinery, sunlight creeping through windows, even tea kettles on the next bench. It’s a gamble that doesn’t pay. Thermometers and routine checks cut down on surprise reactions. Experts point to storage temperatures below 30°C as the line between safe and “call emergency services.”
What Gets Stored Nearby
Mixing this chemical with others amplifies the danger. Once, someone left a reactive acid on the same cart—luck worked in our favor that day, but luck isn’t a plan. Strong bases, reducing agents, and oxidizers belong nowhere near perchloromethyl mercaptan. Even small spills can set off chain reactions that bring operations to a halt and endanger lives. Frequent audits of storage practices help prevent costly mistakes. A simple printed chart of incompatibles taped to the storage door works better than any digital inventory.
Container Choices
Leaks don’t start big—they start with small oversights. Glass and sealed HDPE containers keep this chemical stable and contained. I remember a technician once choosing a standard plastic jar; corrosion ate through in days. Proper labeling, too, plays a bigger role than people think. Day-to-day, it’s easy to forget which bottle holds the nastiest stuff. Worn or unclear labels invite disaster. Every shipment gets a dedicated check for intact seals and clean, bold warnings.
Containing Fumes
Any container, even a sealed one, can let out trace vapors over time. Vapor-proof secondary containment—think a tub or tray made for spill capture—catches slow leaks before they spread. After one incident where fumes sent two workers to the onsite medic, management never skipped this safeguard again. Carbon filtration in exhaust systems takes cleanup a step further, lessening the risk for anyone passing through.
Training and Emergency Planning
People, not just protocols, hold the line on safety. Everyone who touches or moves dangerous chemicals needs direct, hands-on training. Watching a video once a year won’t cut it. Buddy systems for handling, clear escape routes, and regular drills make safety second nature. Keeping up-to-date Material Safety Data Sheets within arm’s reach serves as a quick guide when nerves hit and decisions matter most.
Pushing for Continuous Improvement
Every close call, every spill, brings a lesson. Reviewing incident reports, swapping stories with industry peers, and staying plugged into new research can sharpen storage routines. The folks who work on the ground offer insights textbooks miss—they remember which labels peel, which rooms get hottest in July, and which gloves actually stand up to spills. Their experience keeps workplaces accountable, safe, and always one step ahead of disaster.
What are the health hazards associated with Perchloromethyl Mercaptan exposure?
Understanding Exposure and Immediate Risks
Perchloromethyl mercaptan isn’t a chemical that gets tossed around in everyday conversations, though you’ll find it across different manufacturing fields, especially in making pesticides and pharmaceuticals. The stuff gives off a nasty smell and looks like a pale yellow liquid. Just a little bit in the air, and your nose and throat start to burn. Last summer, a friend working at a chemical warehouse called after an accidental spill. The irritation, headaches, and coughing kept him off his feet for days. Even if you’re not standing right over an open drum, vapor exposure can happen fast and cause real breathing trouble.
Beyond Short-Term Effects
Most folks worry about the immediate sting or burning in the eyes, nose, and throat. Perchloromethyl mercaptan can inflame airways and even cause chest tightness and wheezing. In places with poor ventilation, small leaks turn into big health threats. The real danger creeps in with longer exposures or higher concentrations. Over time, repeated contact can damage the lungs and lead to asthma-like symptoms, making day-to-day breathing harder.
Dermal contact brings on burns and rashes. Anecdotally, several workers in the chemical industry told me they’ve needed steroid creams after just a splash. There’s a temptation to brush off minor skin irritation. But with chemicals this toxic, the body doesn’t always bounce back. One safety officer at a pesticide plant mentioned cases where chronic exposure prompted allergic reactions, making it impossible for workers to return to the same job.
Toxic Load on Organs
Most authorities, including the U.S. EPA, flag perchloromethyl mercaptan as “dangerous to life and health,” especially at high concentrations. Liver and kidney damage count among the more severe problems. These organs filter toxins from the blood, and overloading them with harsh chemicals leaves lasting scars. Inhalation isn’t the only concern; accidental ingestion or significant skin contact ramps up the risk for long-term harm.
Animal studies serve as a wake-up call: repeated exposure produced clear evidence of organ toxicity, raising tough questions about what even low-level, “safe” exposure means for workers who clock in day after day.
Community and Environmental Impact
While most stories focus on workplace safety, leaks can slip past facility walls. Folks living near production or storage sites deal with contaminated air and the constant worry about a bigger release. Prolonged low-level exposure in a neighborhood can add up in ways that are harder to track, especially among kids or people dealing with asthma. And while direct data about perchloromethyl mercaptan in groundwater or soil remains limited, the record with related chemicals suggests vigilance isn’t optional.
What Works to Limit Harm?
Personal experience lines up with official advice: strict safety training, reliable personal protective gear, and real-time air monitoring in plants make a difference. One shift supervisor described how new alarm systems for vapor detection cut down on emergency medical calls. Rotating staff to shorter shifts in higher-risk zones also helps reduce individual exposure. Companies willing to report incidents quickly and transparently tend to keep workers healthier and community relations steadier.
On the legislative side, tighter rules about manufacturing, transporting, and storing this chemical can’t come soon enough. Funding better research means the people tasked with handling the material aren’t left with guesswork about what they’re breathing or touching.
Bottom line—perchloromethyl mercaptan isn’t something to take lightly or handle with guesswork. Focusing on worker voices and hard data will do more to protect health than any after-the-fact fixes.
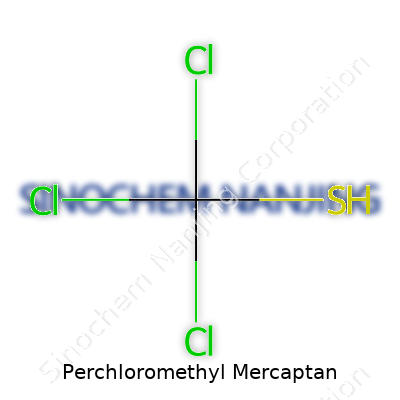
| Names | |
| Preferred IUPAC name | Trichloromethanesulfenyl chloride |
| Other names |
Trichloromethylthiol Trichloromethyl mercaptan Perchloromethylthiol Trichloromethanethiol TCMT Trichloromethylsulfide |
| Pronunciation | /pərˌklɔːroʊˈmɛθɪl mərˈkæptæn/ |
| Identifiers | |
| CAS Number | No. 594-42-3 |
| 3D model (JSmol) | `load =C(Cl)(Cl)(Cl)SC(Cl)(Cl)Cl` |
| Beilstein Reference | 82423 |
| ChEBI | CHEBI:34910 |
| ChEMBL | CHEMBL185635 |
| ChemSpider | 6130 |
| DrugBank | DB11240 |
| ECHA InfoCard | 03f88a3a-1d33-43c6-8ea7-fe6e8bfab2db |
| EC Number | 200-932-3 |
| Gmelin Reference | Gmelin 2095 |
| KEGG | C19316 |
| MeSH | D010482 |
| PubChem CID | 6618 |
| RTECS number | TJ4900000 |
| UNII | 07V15YZ3T4 |
| UN number | UN1670 |
| Properties | |
| Chemical formula | CCl3SCl |
| Molar mass | 185.384 g/mol |
| Appearance | Colorless or light yellow liquid with a pungent odor |
| Odor | Unpleasant, penetrating odor |
| Density | 1.857 g/cm³ |
| Solubility in water | insoluble |
| log P | 2.6 |
| Vapor pressure | 0.079 mmHg at 25°C |
| Acidity (pKa) | -4.2 |
| Basicity (pKb) | 7.3 |
| Magnetic susceptibility (χ) | -34.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.596 |
| Viscosity | 4.62 mPa·s (25 °C) |
| Dipole moment | 2.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.6 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -44.7 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -655.7 kJ/mol |
| Pharmacology | |
| ATC code | '' |
| Hazards | |
| Main hazards | Toxic by inhalation, ingestion, and skin absorption; causes severe irritation to eyes, skin, and respiratory tract; may cause delayed pulmonary edema; emits toxic fumes when heated. |
| GHS labelling | GHS02, GHS05, GHS06, GHS09 |
| Pictograms | GHS06,GHS05,GHS09 |
| Signal word | Danger |
| Hazard statements | H331, H311, H301, H314, H400 |
| Precautionary statements | P210, P261, P264, P271, P273, P280, P301+P310, P304+P340, P305+P351+P338, P320, P330, P361+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-4-2-W |
| Autoignition temperature | 550°C |
| Lethal dose or concentration | LD50 oral rat 47 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3.42 mg/kg (oral, rat) |
| NIOSH | TT4300000 |
| PEL (Permissible) | PEL: 0.1 ppm (0.7 mg/m³) |
| REL (Recommended) | 0.01 ppm |
| IDLH (Immediate danger) | 2 ppm |
| Related compounds | |
| Related compounds |
Carbon tetrachloride Chloropicrin Thiophosgene |

