Pentachlorophenol: A Deep Dive into Its Past, Present, and Unfolding Future
Historical Development
Pentachlorophenol’s story stretches back to the late 1930s, right into a world hungry for industrial innovation and robust wood preservatives. Folks once treated everything from railroad ties to utility poles with this chemical, seeing its ability to fight rot and fungal decay as groundbreaking. After World War II, demand skyrocketed, blending into the backbone of American infrastructure and expanding globally. Over decades, scientists started to observe problems sprouting up—contamination of soils, bodies of water, and the emergence of health issues among workers and surrounding communities. These warning lights led many countries to clamp down on its use, shifting the conversation from progress to protection.
Product Overview
Wandering through any industrial supply warehouse thirty years ago, you’d spot barrels of pentachlorophenol stacked beside other potent chemicals. This compound stood out for its efficiency in driving off pests and stopping mold in its tracks. It was widely available as an oily solution or dissolvable powder, often colored greenish or brown, reeking of phenol and chlorine. Despite its former ubiquity, tightening regulations have slowly pushed the product out of mainstream circulation, except in limited, approved cases managed under strict control. Its steady fade from the shelves reminds us that utility sometimes walks hand-in-hand with unintended consequences.
Physical & Chemical Properties
Pentachlorophenol comes with a heavy molecular weight, a crystalline form, and a melting point hovering near 190°C, giving it a sturdy presence under harsh conditions. As a chlorinated aromatic compound, it shrugs off most water, showing higher solubility in organic solvents. Its volatility runs low, so it tends to linger for years wherever spilled. Its reactivity with bases drives notable changes, like salt formation, while strong oxidizers break it down into less persistent molecules. Chemical stability in the environment has become both a blessing and a curse, slowing the processes needed to clean it from contaminated sites.
Technical Specifications & Labeling
Regulation forced the industry to mark every drum, bottle, or sack of pentachlorophenol with clear hazard designations. Labels display pictograms warning against inhalation, skin contact, or improper storage. Typical formulations aimed for purity above 85%, though trace contaminants like dioxins and furans often haunted quality standards and posed real health concerns. Safety data sheets outline the risks of acute poisoning and long-term exposure, sometimes painting a troubling picture of chronic health disruption and environmental persistence. Such documents draw a line between ignorance and accountability, making transparency a core requirement.
Preparation Method
Chemical manufacturers rely on direct chlorination of phenol under heat and pressure, pushing the reaction through multiple steps to reach the all-chlorinated product. Early production used open systems, releasing waste and byproducts into air and waterways, but modern plants shifted to closed reactors and tight emission controls. Chloroform serves as a solvent in some methods, boosting yield but also raising disposal challenges. Each ton of product carries an environmental footprint shaped by both raw material sourcing and waste management, testing the ingenuity of engineers and the vigilance of environmental regulators.
Chemical Reactions & Modifications
Chemists working with pentachlorophenol have found it stubbornly resistant to many benign degradation pathways. Strong bases strip away chlorine atoms under harsh conditions, while ultraviolet light or ozone treatments start to break it into smaller fragments. Notoriously, these breakdown products—such as polychlorinated dibenzo-p-dioxins—raise a fresh crop of problems, with toxicity sometimes eclipsing that of the parent compound. Researchers tinker with catalytic reduction and advanced oxidation, hunting for ways to neutralize its threat or convert it into less harmful substances before it escapes into soil or air.
Synonyms & Product Names
On the international market, pentachlorophenol has traveled under a patchwork of trade names and synonyms. Some labels call it PCP or “Penta,” making it a staple term in logging and railroad maintenance. Trade names like Permacide or Santobrite flashed through catalogs, especially in the heyday of routine wood treatment. Regulatory lists and chemical inventories may tag it under other identifiers, complicating both sales oversight and accident response for workers and inspectors. Connecting these dots helps prevent confusion when cross-referencing safety information and global trade documents.
Safety & Operational Standards
People handling pentachlorophenol learned to trust gloves, respiratory masks, and covered skin long before such habits became standard industry practice. Showering at the end of a shift, using sealed storage tanks, and running regular air monitoring tests turned from recommendations into hard rules. U.S. OSHA and European agencies set clear exposure limits—thresholds designed to minimize both acute poisonings and chronic illnesses, like organ damage and certain cancers. Waste streams falling outside disposal codes can trigger heavy fines and cleanup costs, so responsible companies installed wastewater treatments and advanced scrubbers, reinforcing the idea that risk management has no shortcuts.
Application Area
For decades, pentachlorophenol guided wood poles, fence posts, and cross-arms through tough winters and muggy summers, defeating rot and tunnels bored by insects. Industrial use also crept into leather tanning, though those processes gradually faded as environmental audits exposed the toxic runoff. As regulations restricted its reach, the chemical found fewer new applications and mostly stayed in legacy infrastructure, slowly transitioning to more benign alternatives. The memory of contaminated soil in old treatment plants reminds communities how long this legacy can last, fueling interest in innovative cleanup and restoration efforts.
Research & Development
Scientists in public health and engineering spent decades poring over the environmental fate and biological impact of pentachlorophenol, building a rich library of case studies, lab tests, and field data. Research teams tracked how wastewaters migrate into aquifers, how chronic low-level exposure can disrupt hormone systems, and how breakdown products harm aquatic life. Companies poured resources into finding replacements—products delivering wood protection without leaving “forever chemicals” in their wake. Novel approaches like bioremediation and advanced oxidation break through some technical barriers, yet the real prize lies in prevention—engineering new materials and processes that sidestep the need for hazard altogether.
Toxicity Research
Doctors and toxicologists began noticing troubling health patterns among workers dipping railroad ties or handling treated wood. High doses can bring on sweating, fever, and convulsions, while lingering, lower exposure links to liver issues, nerve disorders, and increased cancer risk. Lab animal studies pile up, confirming that dioxins created as side products spell danger for animal and human cells alike. Water regulators keep close tabs on pentachlorophenol levels in surface and groundwater, striving to keep contamination below public health advisory limits. These efforts pull together the voices of scientists, regulators, and affected communities, building a consensus that lines up with protective action rather than delay.
Future Prospects
Looking forward, pentachlorophenol’s time as a staple in preservation chemistry winds down, making space for greener, safer wood treatments. As regulatory guardrails get taller and cleanup demands grow louder, investment shifts toward biobased preservatives, improved composite materials, and on-site detoxification. The history of pentachlorophenol offers more than just caution—it underscores the need to watch for side effects from chemicals woven into our daily lives. Thorough risk assessment, honest public dialogue, and continuous research form the clearest path toward a future where innovation stands alongside human and ecosystem health, without repeating the mistakes of the past.
What is Pentachlorophenol used for?
The Story of an Old Wood Preservative
Some names pop up, then vanish from the public eye. Pentachlorophenol falls in that camp, though anyone who’s driven by miles of utility poles has probably seen its work up close. Folks started using it back in the 1930s, not because it sounded catchy, but because, for a time, it protected wood from rot and bugs like nothing else on the shelf. Think about railroad ties or old barn timbers. Treating them with pentachlorophenol meant they stuck around—sturdy as ever—even in damp country stretches.
Where It Shows Up
I remember my uncle’s shed creaking under the weight of tools and the unmistakable smell that hit you when you opened the door. Wood treated with "penta" packs a punch. Utility companies, railways, and some builders liked it for that reason. Once applied, wood could weather storms, fungus, beetles, and more.
Records from the U.S. Environmental Protection Agency show nearly all pentachlorophenol used in recent decades went toward industrial wood preservation. Fence posts, power line poles, and waterway pilings gained the most. Folks working in agriculture might have seen it on cattle fences or barns, too. Paint strippers and herbicides once contained it, though newer laws have squeezed it mostly out of those products for home use.
Why It Matters
There’s no escaping the reason no one slaps pentachlorophenol on a shelf at the local hardware store anymore: health and environmental worries. Cancer risks, liver and kidney harm, and trouble for the immune system raised more than a few red flags. Soil and water stay tainted for years wherever it leaks. Think of the neighbors living near a treated lumber storage yard—fish die off, the garden turns risky, and kids get exposed just walking by.
Studies by the World Health Organization linked pentachlorophenol with cancer. Soil samples keep showing residues, sometimes decades after the first treatment. In my own county, an old sawmill site needed years of cleanup before anyone could safely open a park there again. This is more than a technical issue; it's people's health, farmland, and clean water at stake.
Looking for Answers
Tough questions come up as more folks speak out. Some say companies dragged their feet, knowing the risks for years. The EPA clamped down tighter over time, banning most residential uses in 1984 and phasing out the chemical by 2022 for almost everything else in the U.S. Even now, some countries let it ride, based on old safety claims that don’t hold up.
Being practical, I believe it takes more than government rules to handle chemicals like this. Utility firms can find safer treatments, even if costs climb at first. Creosote and copper-based products don’t come without problems, so rooting for new, less toxic methods makes sense. Local communities—especially those near old treatment plants—need solid testing, cleanup, and honest information.
Folks who worked with pentachlorophenol, like railroad and utility crews, deserve health monitoring, clear records, and support where there’s trouble. Residents near past spill sites need straightforward answers, not a wave of paperwork and broken promises. It’s just common sense to put health before old habits or profits.
Is Pentachlorophenol hazardous to health?
What Is Pentachlorophenol?
Pentachlorophenol, often called PCP, showed up for decades as a wood preservative. Utility poles, railroad ties, fence posts—many of them got treated with it to fight decay and insects. The chemical’s ability to keep wood from rotting seemed like a modern miracle. Yet, problems began surfacing soon after its widespread use began in the 1940s. By the time I first learned about PCP, it was already under heavy regulation, but its legacy lingered around the materials and sites where it had been used for so long.
How PCP Gets Into Our Bodies
Most people don’t handle pentachlorophenol directly. Still, it doesn’t stay put. PCP can seep into the soil, contaminating water or crops nearby. People living near wood treatment plants or using treated wood for home projects could breathe it in or get it on their skin. It also moves through food, especially in places where it contaminates water or soil.
Health Hazards Linked to Pentachlorophenol
The dangers tied to PCP aren’t subtle. Direct contact causes irritation—red, itchy skin, sore eyes, breathing trouble. Long-term exposure goes further. PCP is toxic to the liver, kidneys, blood, lungs, nervous system, and immune system. People working around this stuff used to complain of headaches, sweating, feeling weak, and even nerve issues. The U.S. Environmental Protection Agency (EPA) labels it a probable human carcinogen. The International Agency for Research on Cancer puts it in Group 2B, which means there’s reason to worry that pentachlorophenol raises cancer risks, especially for those exposed over a long time.
Back in my grad school days, we did a site study near a former wood treatment plant. One neighbor showed us his vegetable garden. He’d set up right next to the old fence. Soil testing showed PCP levels far above what you’d find in cleaner earth. He had no idea that the chemical moved from old wood into soil and into crops. This kind of accidental exposure adds up quietly, year after year.
Why Watchdogs Regulate It
Regulators began cracking down after studies kept piling up. Restrictions on pentachlorophenol in residential settings went into effect through the 1980s and 1990s. In the U.S., only trained professionals can use it, and only for wood that never touches food, homes, or playgrounds. Other countries banned it outright.
Disposal creates headaches, too. Burning or dumping old treated wood can throw PCP into the air or wash it into waterways. That’s why EPA guidelines tell people not to burn PCP-treated wood or compost it. Community collections for hazardous waste sometimes turn up big piles of the stuff, which needs special care for safe disposal.
Looking for Safer Solutions
Alternatives have gained traction over the years. Copper-based preservatives, heat treatments, and naturally rot-resistant woods have filled in gaps left by a PCP phase-out. But old structures still hang around. Dealing with these leftovers means education matters. Homeowners ought to ask before cutting, burning, or recycling pressure-treated wood.
Public agencies and health experts continue to track environmental and health effects, push for clear labels, and expand safe disposal options. Staying alert and leaning on science shields families and communities from the worst health impacts tied to pentachlorophenol.
How should Pentachlorophenol be stored and handled?
Real-World Concerns About Pentachlorophenol
I first learned about pentachlorophenol—most folks call it PCP—while helping on a farm years ago, scraping old wood for repainting. The label on a faded can warned of skin burns and an overwhelming chemical odor. These memories come back any time someone asks about storing or working with this stuff. PCP isn’t paint thinner and shouldn't be treated like it. The risks hang in the air—quite literally, with fumes that can wreck the lungs and skin contact that burns deep.
Why Storage Matters More Than People Think
Pentachlorophenol breaks apart quickly in sunlight and heat. If it leaks, groundwater and soil won’t recover for years. There’s no shortcut; chemical-proof drums or tightly sealed containers do the job right, and folks should put them inside cool, locked buildings. Ventilation shouldn’t just be a buzzword—it’s life or death for workers and neighbors. I’ve seen barns where everything, including the family dog, picked up the stench after a leaky can sat forgotten. Anyone using PCP owes it to the land and local water to keep it dry and sealed. Storing it away from food, animal feed, and anything edible might sound like common sense, but cutting corners happens fast when no one checks up.
The Health Dangers Hit Home
Factory workers and fence builders used PCP for decades without respirators, scrubbing their bare hands with kerosene. These stories don’t always make headlines, but the health outcomes stick. Besides the immediate threat to skin and eyes, regular breathing of PCP fumes links straight to liver and kidney damage. Even tiny amounts over time matter. Storing gear in labeled, chemical-resistant bins reduces mix-ups and accidental contact.
Everyday Handling: What Really Helps
Folks picking up a brush or operating a sprayer full of PCP need gloves made for chemicals—not just gardening gloves—and full face shields. Tossing on a cotton mask won’t block vapors. Real-world experience says workers change gloves after every break, and wash up outside the storage area. Long sleeves, tough boots, and aprons help, but only if kept clean, far from the canisters. Dirty clothing left in the family laundry spreads the hazard.
Spills and Waste: No Shortcuts Here
Cleaning up after a drip isn’t about grabbing paper towels. Clay absorbents, tarps, and dedicated bags keep the mess contained. Local governments often make strict rules about dumping waste, and for good reason. I’ve seen people bury old containers or burn them, sometimes out of ignorance, often to save a buck. Both spread toxins. Bringing waste and tools to certified facilities protects neighborhoods and wildlife downstream. Storing spill kits alongside the chemical is plain good sense—it cuts down response time when every second matters.
Setting Up a Better System
Cities and rural counties need regular oversight. Health and fire inspectors should walk through sites every season, not just after a complaint. Training sessions could make a world of difference—explaining the risk, not just ticking boxes. Farmers, construction crews, and warehouse staff all benefit from real talk about symptoms and emergency contacts posted on-site. Industry leaders have a responsibility, not just a legal one but an ethical one, to cover safety gear and proper facilities in the budget, treating it as essential as tractor fuel or payroll.
Well-handled chemicals protect more than profits—they keep families, water, and land healthy for years to come.
What are the environmental impacts of Pentachlorophenol?
A Closer Look at Contamination and Consequences
Pentachlorophenol has stood out for decades as a wood preservative, keeping railroad ties, utility poles, and lumber from rotting away. Still, beneath all that utility, the chemical leaves a lingering stain on land and water, one often overlooked in discussions around toxic substances. As someone who grew up fishing along streams near a lumber mill, I saw signs warning us to stay away, but nobody explained why. Later, I learned runoff and dust could mean real trouble for animals and the people who live nearby.
Polluting More Than Just the Water
Once released, pentachlorophenol doesn’t just vanish into thin air. It sticks around — infiltrating soil and water, getting caught in sediment at the bottom of rivers and lakes. Over time, it finds its way into the food chain. Fish pull in toxins from murky water; birds and mammals eating those fish inherit something they never bargained for. According to the EPA, pentachlorophenol can persist in soil for months to years, depending on sunlight and microbes in the ground. So, once it enters an area, it waits out any quick fix.
Human Health Risks Are Part of the Package
The evidence from studies on workers exposed to pentachlorophenol paints a troubling picture. People handling treated lumber, or living close to storage yards, see a higher risk of cancer and serious damage to their liver and immune system. I remember a neighbor who fell sick after working in a wood treatment plant, and that sickness brought questions nobody managed to answer until years later. The CDC lists pentachlorophenol as a possible human carcinogen, which should make us all pause and think about what goes unmeasured in the soil in children's parks or under a power pole.
Solutions Grow from Monitoring and Safer Choices
Turning the tide on persistent contamination calls for more than cut-and-dried regulations. Cities can map places where pentachlorophenol treated wood gets used and then check those soils often, using modern sensors and clear documentation. I’ve seen community scientists near my town run their own water tests, pushing the local authorities to act when toxins show up.
Industries have alternatives, though some cost more up front. Borate-based treatments, or modified wood products using heat or pressure, show promise without leaving behind toxic residues. Construction companies also can clean up their disposal methods, making sure old utility poles aren’t just tossed or left to weather near rivers. Pushing for stronger recycling and safe removal rules can break the cycle of exposure for people and animals. Pressure from buyers helps too — if utility districts demand cleaner choices, suppliers look for lower-risk preservatives.
No One-Size-Fits-All Fix, Just Commitment
Too many communities wind up dealing with leftover chemicals in forgotten storage sites or shallow burial pits. Clean-up groups, regulators, and regular people bring energy and ideas to the table. Pentachlorophenol's presence in the environment delivers a message about the consequences of short-term thinking. Real progress comes from facing the risks honestly, putting health first, and backing up science with straightforward, public solutions.
Is Pentachlorophenol banned or regulated in my country?
Looking at Pentachlorophenol’s Story in Our Communities
Pentachlorophenol, often called "penta," doesn’t come up in daily conversations, but its fingerprint lingers in fence posts, utility poles, and old barns across many countries. For decades, penta helped preserve wood against insects, fungi, and weather. Its effectiveness earned it a spot on farms, railways, and neighborhoods. The legacy, however, carries more than just weathered pine and cedar. Growing up near stacks of treated wood, I saw how the chemical smell settled into the air, especially after rain. My parents never thought twice—like everyone else, they trusted the system said it was safe enough.
Over time, research began to link pentachlorophenol to serious health concerns. The U.S. Environmental Protection Agency classified penta as a probable human carcinogen after studies showed possible risks of cancer, liver damage, and endocrine disruption. Exposure doesn’t just happen on-site—runoff and leaching find their way into water and soil, changing local ecosystems. Aquatic life, already struggling, picks up the load. Communities living near treatment plants face higher risks. Childhood memories with the sweet tang of soaked telephone poles turn bitter with each new piece of evidence.
How Countries Tackle the Problem
Rules around pentachlorophenol look different depending on where you stand. Countries in the European Union stopped penta’s use back in the 1990s. Canada phased it out for most purposes, keeping strict restrictions only in very specific cases. In the United States, penta's production took a final bow in 2021 after pressure from both scientists and advocates. Still, old stock and previously treated wood stay in place, stretching the impact over generations.
For several countries across Asia, Africa, and Latin America, the story isn’t as clear-cut. Regulatory oversight often lacks teeth, and enforcement gets tricky. Trade in used utility poles and recycled lumber means the chemical can resurface far away from its starting point. Demand for affordable, durable materials sometimes outweighs concern for hidden risks. My own experience with local hardware shops showed shelves stocked with treated planks, warnings buried in dense labels.
Why Pentachlorophenol Stays Relevant
The real trouble with pentachlorophenol lies in its persistence. It breaks down slowly, so contamination lingers in soil and groundwater long after the last drop leaves the treatment tank. Research out of the CDC and WHO points at risks that don’t shrink just because rules changed. Kids climbing old jungle gyms or gardeners working in treated soil keep this chemical close, whether they know it or not. Stories from friends who work along railroad tracks or in construction say the same: gear protects skin, but stubborn dust and vapor get everywhere.
Getting a handle on pentachlorophenol starts with transparency about where it turns up. Governments can tighten inspections and push utilities to replace aged infrastructure safely. Community-led reporting helps document where exposure risks pop up, especially in rural areas. As a consumer, I buy plants from nurseries that don't use treated wood and press for clear policies from local government. Industry can turn towards alternatives like copper-based or borate formulas, since modern options offer decent performance with less harm. In every case, stopping future contamination means rewriting the rulebook around hazardous leftovers, not just banning the next new thing.
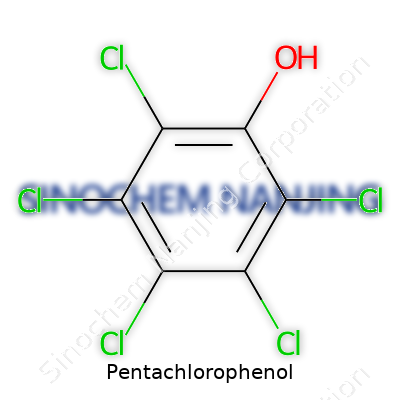
| Names | |
| Preferred IUPAC name | 2,3,4,5,6-pentachlorophenol |
| Other names |
PCP Penchlorol Penta-Kil Permaclor Cereclor Santofen Dowicide EC-7 Lumber Pro NCI-C01863 |
| Pronunciation | /ˌpɛn.tə.klaɪˈrɒ.fə.nɒl/ |
| Identifiers | |
| CAS Number | 87-86-5 |
| Beilstein Reference | '1711394' |
| ChEBI | CHEBI:39382 |
| ChEMBL | CHEMBL1436 |
| ChemSpider | 986 |
| DrugBank | DB03775 |
| ECHA InfoCard | 03e4b8b8-21de-4d80-8380-3900c2ef8c14 |
| EC Number | 201-778-6 |
| Gmelin Reference | 727 |
| KEGG | C01323 |
| MeSH | D010406 |
| PubChem CID | 992 |
| RTECS number | SN3500000 |
| UNII | 'LXMCU1N771' |
| UN number | UN3155 |
| Properties | |
| Chemical formula | C6Cl5OH |
| Molar mass | 266.34 g/mol |
| Appearance | White to tan crystals or needles |
| Odor | Medicinal; Phenolic |
| Density | 1.981 g/cm³ |
| Solubility in water | 0.014 g/100 mL (20 °C) |
| log P | 5.01 |
| Vapor pressure | 0.0001 mmHg (25°C) |
| Acidity (pKa) | 4.7 |
| Basicity (pKb) | 6.9 |
| Magnetic susceptibility (χ) | \-49.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.640 |
| Viscosity | 1.75 cP (20°C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 174.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -146.4 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2412 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D08AE01 |
| Hazards | |
| Main hazards | Toxic by inhalation, ingestion, and skin absorption; causes liver, kidney, and nervous system damage; suspected carcinogen; environmental hazard. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301, H311, H331, H372, H400 |
| Precautionary statements | P260, P261, P262, P264, P273, P280, P284, P301+P310, P302+P352, P304+P340, P305+P351+P338, P310, P330, P361+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-0-☠ |
| Flash point | 69°C |
| Autoignition temperature | 132°C |
| Lethal dose or concentration | LD50 (oral, rat): 50 mg/kg |
| LD50 (median dose) | LD50 (median dose): 230 mg/kg (oral, rat) |
| NIOSH | PCP |
| PEL (Permissible) | PEL = 0.5 mg/m3 |
| REL (Recommended) | 0.5 mg/m³ |
| IDLH (Immediate danger) | 3 ppm |
| Related compounds | |
| Related compounds |
Pentachloroanisole Pentachloronitrobenzene Chlorophenol |

