Pentachloronitrobenzene: A Real-World Look at Its Journey, Use, and Impact
Historical Development
Pentachloronitrobenzene, or PCNB for short, started appearing in old chemistry journals just as scientists began tinkering with halogenated aromatics in earnest. The tale traces back to the early 20th century, as researchers tried to create stronger fungicides for agriculture. In those days, crops regularly suffered yield losses from tough fungi like Sclerotinia and Rhizoctonia. Chemistry labs got to work, and somewhere between the World Wars, several companies refined chlorination and nitration methods for benzene derivatives. The commercial value became impossible to ignore, especially as crop protection depended more and more on synthetic chemistry rather than sheer luck and labor. My own grandfather recalled how PCNB formulations lined shelves in local farm supply stores during planting season, a fixture of the pursuit to squeeze more from every acre. The history of PCNB shows a steady push for performance and a hope that new molecules could keep fields green and forests of food on the table.
Product Overview
Looking at a bag of commercial PCNB, you see the end product of lots of industrial chemistry, technical refinement, and—frankly—pragmatic compromise. Sold mostly as granules or wettable powders, PCNB remains geared for agricultural use, especially as a soil-treatment fungicide. It often shows up with trade names like Quintozene, Terraclor, and Brassicol. Each brand focuses on distinct crops, soil conditions, or application styles. The end user, usually a farmer or turf manager, gets a product promising robust control for certain fungal pathogens. For years, the agriculture industry leaned on PCNB because it did the job under a wide range of field conditions, where humidity, irrigation, and temperature can mess with lesser products.
Physical & Chemical Properties
PCNB comes off as a pale yellow solid, sometimes a powder, with a faint musty odor. This isn't some volatile chemical—melting point hovers around 140°C, and it barely dissolves in water. Solvents like acetone and ethyl ether work better. Thanks to the five chlorine atoms and one nitro group decorating the benzene ring, PCNB holds up in harsh settings. Its molecular weight crops up at 295.3 g/mol—not exactly light. The logP value tips it toward fat solubility, which can mean it stays in soils or plant tissues longer than some alternatives. In the dusty corners of the storage shed, bags of PCNB don’t degrade quickly, and this persistence helps explain its controversial reputation in environmental circles.
Technical Specifications & Labeling
Every commercial drum or package carries detailed labeling, not just to follow the rules but to steer clear of misuse. The label spells out purity—often above 99% for the active ingredient in most products—plus any inert fillers, dispersants, or sticking agents. Statement of active ingredient, batch code, and expiration date keep things transparent. Storage advice isn’t just formality; PCNB stays stable under cool, dry, and well-ventilated conditions, but humidity and sunlight speed up breakdown or caking. Most markets require warning symbols and proper hazard communications, reflecting the growing push for safe handling from warehouse through field application.
Preparation Method
Most PCNB on the market comes from industrial synthesis that starts with chlorination of nitrobenzene. This isn’t home chemistry—the process uses pressurized reactors, chlorine gas feeds, and careful monitoring of temperature and time. Alternatives sometimes work backward, starting with pentachlorobenzene and using mixed acid to nitrate the product. Chemical engineers who’ve spent time in the plant remember the dense haze and strict controls on leaks and emissions. The reaction’s a balancing act: too much heat or not enough residence time, and the yield tanks. Proper separation and purification—usually by distillation and filtration—brings the final product close to spec, ready for packaging and downstream blending.
Chemical Reactions & Modifications
The structure of PCNB, with its tough benzene ring fortified by five chlorines and a nitro punch, makes it resistant to most ordinary organic chemistry tricks. Still, under certain conditions, the nitro group can swap out for amines or even transform under intense reducing environments. Soil bacteria and fungi do manage to degrade PCNB, breaking it down stepwise to metabolites like pentachloroaniline and pentachlorophenol. In research labs, chemists sometimes graft PCNB rings onto polymers for material science or mess with the halosubstitution pattern searching for new bioactivity. Modifications rarely end up at scale, mostly because PCNB’s initial structure fits its target use so well that tweaks—so far—don’t bring big benefits for field application.
Synonyms & Product Names
PCNB comes with a laundry list of names thanks to decades of international trade and branding. Some of the main synonyms include Quintozene, Pentachloronitrobenzol, 2,3,4,5,6-Pentachloronitrobenzene, and various trade names like Terraclor and Brassicol. This matters for supply chains and regulatory oversight, as some countries restrict one name but permit another. Back when I helped research global pesticide policies, this stew of synonyms made comparisons a headache, as each regulatory agency seemed to favor its preferred terminology.
Safety & Operational Standards
Handling PCNB calls for respect. Direct skin contact, inhalation of dust, or accidental ingestion can cause a range of health effects—from mild irritation to more severe systemic problems if exposure’s prolonged. Workers wear gloves, goggles, long sleeves, and, in confined spaces, dust masks. Storage must keep the chemical away from food, feed, and groundwater sources, not least because runoff after heavy rain presents an ongoing concern. Regulations around the world, including those set by agencies like OSHA, EPA, and their counterparts abroad, lay out strict standards for exposure, labeling, spill containment, and disposal. Regular equipment checks and worker safety trainings have become standard across responsible operations, acknowledging both the benefits and risks that come with using a persistent synthetic chemical.
Application Area
Farmers and turf managers consider PCNB a specialist’s tool for soilborne fungi like clubroot in brassicas or dollar spot in turfgrass. It enters the soil as granules or diluted sprays, usually before planting or early in the growing season. Countries with heavy vegetable, cereal, or cotton production lean on PCNB more than others. In golf course and sports turf circles, it’s an old standby for patch diseases that laugh at conventional treatments. The product doesn’t belong in every rotation, though—regulations and resistance concerns call for rotation among modes of action. In my years covering ag innovation, I’ve seen PCNB’s role shift from default to more strategic use, pushed by stricter residue rules and a focus on environmental stewardship.
Research & Development
Universities, public research institutes, and private labs keep looking for both better ways to use existing PCNB and greener alternatives. Studies dig into optimal timing, application rates, and combinations with cultural or biological controls. Some experiments dive into nanotech delivery for targeted release or reduce dust with novel encapsulation techniques. And every so often, a new formulation promises longer effectiveness or lower drift. Despite the long shadow of environmental concerns, investment still flows into PCNB research, either to make it safer, to degrade it faster in soil, or to replace it with next-generation chemistry. These efforts reflect both the dependence on reliable crop protection and the pressure to keep moving toward sustainability.
Toxicity Research
Research into PCNB’s toxicity covers multiple fronts: lab animals, farm workers, downstream consumers, and the broader ecosystem. Studies in rats and mice point to liver and kidney impacts at doses much higher than routine occupational exposure. Human health assessments focus on chronic exposure, with some worry about groundwater contamination and food residues. Aquatic life gets a lot of attention, as PCNB doesn’t break down rapidly in sediment and can affect fish and crustaceans. Regulators in the US, EU, and elsewhere keep updating risk assessments, balancing practical application with emerging data. Laboratories now run high-resolution analyses of metabolites, tracking PCNB persistence and movement from field to food to water table. Growing up near a river-fed valley, local anglers always asked whether the water was “clean,” and the questions around persistent chemicals like PCNB remind us that responsible use and honest science go hand in hand.
Future Prospects
The future for PCNB won’t rest on past performance. Demand exists where control of resilient soil fungi makes or breaks a season. Yet, the growing emphasis on environmental health, food safety, and sustainable agriculture narrows the window for chemicals with persistence and hazard profiles like PCNB's. Researchers test biopesticides, crop rotation schemes, and integrated pest management systems to reduce reliance. Some companies explore biodegradable analogs that marry effective disease control with a softer footprint. The regulatory climate grows stricter year by year, with many countries phasing out or tightly restricting PCNB. For those who grew up seeing Quintozene drums as a field fixture, this transition feels both necessary and bittersweet. The challenge now becomes how to maintain food security and crop quality while caring for the soil, the water, and the wider world we all share.
What is Pentachloronitrobenzene used for?
Digging Into Pentachloronitrobenzene
Pentachloronitrobenzene, or PCNB, doesn’t often pop up in everyday talk, but its footprint can be found across farm fields and sometimes even on dinner tables. This chemical comes from a batch of processes that bond chlorine and a nitro group to a benzene ring, creating a tough molecule that doesn't break down easily. Its role as a fungicide made it a staple for farmers dealing with fungus problems. For years, folks who grew rice, peanuts, and vegetables counted on it to keep crops free of blight and rot.
Why PCNB Took Root in Modern Farming
Crop loss from soil-borne diseases pushes growers to look for reliable shields against fungi. PCNB answered that call. On golf courses and large lawns, grounds crews applied it to prevent turf diseases. Seeds dusted with this compound had better odds of sprouting healthy and strong, as it helped fend off threatening fungi early on. Its persistence in soil and water created a dependable barrier that lasted through growing seasons.
Catching the Side Effects
My time helping at a community garden taught me how easily chemicals leach into the soil and, in turn, reach the food we eat. Frequent use of PCNB left soils with lingering residues. Some break down products, including pentachloroaniline (PCA), flag concerns for both people and the critters that live underground. Drinking water monitoring in farming regions sometimes showed traces, signalling that the molecule’s tough-to-break nature brought lasting worries.
University research in the 2000s started tying PCNB exposure to possible health problems. Studies found certain breakdown products could trigger cancer in lab animals. The United States Environmental Protection Agency started putting limits on its use, reflecting mounting doubts about safety. Still, some countries continued using it, facing the tough balance of fighting crop loss versus managing long-term risks.
Searching for Safer Methods
One lesson stands clear from years around the garden: heavy chemical reliance rarely stays harmless for long. As folks want cleaner and safer food, growers have tried switching to crop rotation and disease-resistant seeds. These options don’t give instant results, but through trial and error, many small farms now manage soil health without needing strong chemicals.
Groups like the World Health Organization and Food and Agriculture Organization stress better training for handling farm chemicals. They push for regular water and soil testing, making sure PCNB levels don’t build up unchecked. That demands public investment, so families near farms don’t pay the price down the line.
Piecing Together Solutions
Organic practices show promise, cutting chemicals out of the equation by using compost, natural predators, and smart planting choices. Not every farmer can swap out PCNB overnight; new farming methods bring learning curves and costs. People who steer research—scientists, universities, and local governments—must listen to what farmers see and experience out in the field. Solutions stick best when they blend knowledge from soil scientists, experienced growers, and input from local communities.
PCNB’s story speaks to more than just one chemical. Its long reach in soil and water reminds us to keep close watch on what we use to grow food and how those choices ripple out. Shifting away from chemicals like PCNB won’t happen fast, yet small changes in practice and policy can start new traditions, making food production both safer and more sustainable for future generations.
Is Pentachloronitrobenzene hazardous to health or the environment?
Understanding the Chemical
Pentachloronitrobenzene, often called PCNB, shows up in places most folks overlook. It’s not just a line of text on a shipping label; this chemical goes into some fungicides and is common in fields where food grows. Farmers have used it to tackle mold, especially for soil treatment around vegetables such as cabbage and beets. At first glance, this approach looks efficient — keep out fungus, and crops make it to market in better shape. Scratch the surface, though, and bigger questions step into the light.
Health Hazards in Plain Sight
During my years covering public health and agriculture, I’ve spoken with people who rarely think of pesticide residue. That changes fast when a neighbor’s pet falls ill after digging in treated gardens. Lab research illustrates that PCNB doesn’t just rinse away from root vegetables. It stays put — sometimes it even seeps down to groundwater. Scientists have flagged it as causing liver damage to rodents, with suspicions it brings cancer risk over long exposure. Human cases are harder to document, but that’s routine for chemicals first developed decades back.
If you’ve ever held a package of store-bought potatoes and spotted a warning about washing, there’s a reason. Chronic exposure to chemicals like PCNB links to organ dysfunction and immune suppression. Children usually face higher risks than adults since their bodies process toxins differently. The chemical’s ability to linger in both food and dust means it can sneak into the body under everyone’s radar — not just through carrots, but through the very air near spraying zones.
Environmental Impact Grows Over Time
Walk through an old field treated with PCNB and look for wildflowers or earthworms. Their numbers tend to dip, based on studies from environmental agencies in the US and Europe. Once PCNB soaks into the soil, it breaks down slowly and releases other chemicals, several of which hit even higher marks for toxicity. That’s a risk for anyone who eats from community gardens downstream or who draws water from local wells.
A troubling twist — PCNB builds up in aquatic ecosystems. Fish exposed to runoff can show stress or mutations, and that only gets worse up the food chain. Birds, frogs, pets: all stand in line for a dose they never chose. The story often comes out after the fact, pieced together from sick animals and changed insect patterns.
What Can Be Done?
Real progress depends on two things: transparency and safer alternatives. In my experience reporting rural health problems, trust climbs when people know exactly what’s been applied nearby. Labels and records shared at the community level start important conversations. For farmers, the best options often blend old tricks with new science: rotating crops, boosting soil health, seeking out less harsh fungicides backed by field data.
Regulators need to keep pushing for strict limits and updated safety testing. Most importantly, folks living near treated areas deserve resources on safe gardening and prompt pollution cleanup. Information can save lives in ways that chemicals alone never will. PCNB’s story isn’t just about compounds in the dirt — it’s a lesson that choices in food production ripple out to every corner of the community.
What are the safe handling and storage guidelines for Pentachloronitrobenzene?
Understanding the Substance
Pentachloronitrobenzene doesn’t show up in regular household conversations, but step into the world of agriculture or chemicals, and its name carries weight. Known by some as quintozene, this compound sees use as a fungicide in crop production. Information from the Environmental Protection Agency identifies hazards like skin, eye, and respiratory irritation. I’ve learned through onsite training that even brief encounters with dust or fumes left colleagues coughing or seeking fresh air.
Handling Precautions
Direct contact turns risky fast. Workplaces use gloves made from nitrile or rubber instead of standard latex, which doesn't hold up against such chemicals. Eye protection goes beyond simple safety glasses — chemical splash goggles add a better barrier. Think about long-sleeved clothing, too. One spill on a forearm reveals why. Dust from opening containers drifts, so face masks or respirators rated for organic vapors serve a practical purpose. Ventilation matters. Cracking windows or relying on small fans falls short; professionally engineered local exhaust keeps contaminated air away from workers and prevents accidental buildup.
Safe Storage Principles
I’ve seen too many storage mishaps in my time. Keeping pentachloronitrobenzene anywhere near ignition sources creates unnecessary danger. Static electricity jumps or a stray cigarette can light up dust. The right place looks like a cool, dry, well-ventilated storeroom, set apart from direct sunlight or radiators. Heavy-duty, corrosion-resistant containers seal the deal, locking out moisture and preventing leaks. Accidentally mixing with acids or strong bases creates hazardous byproducts, so segregation matters.
Shelving height counts in real situations. Chemicals up high tumble with the slightest shake, which can mean a cracked package and an emergency clean-up. Keeping the containers at waist level or lower means fewer slips and spills. Clear labeling also helps — workers know what they’re grabbing, and first responders waste less time in emergencies.
Workplace Habits and Emergency Measures
Cleanliness. Leaving residue on workbenches or floors opens the door to repeat exposure. Designated areas for mixing and transferring make clean-up quicker. Sudden spills still happen. Having an emergency kit stocked with absorbent materials, neutralizers, and protective gear by the worksite takes the pressure off during a rush. Employees benefit from accessible eyewash stations and showers — it only takes seconds to flush away harmful dust after an accidental splash.
Regulatory Requirements
Employers bear the duty to train staff on the risks and the safety data sheets—no one wins by skimping on orientation or recurring drills. Keeping pentachloronitrobenzene storage areas under lock and key keeps unauthorized hands out, helps with inventory control, and tracks quantities before they expire. Documentation supports traceability in case something ever goes wrong. State and federal rules often restrict quantities and set strict reporting if thresholds get crossed. The EPA and OSHA both monitor compliance, and fines add up quickly after a violation.
Facing the Future
Talking with those in the trenches—whether on farms, in packaging plants, or in labs—leaves no confusion. Respect for chemical safety helps avoid mishaps that can hurt people and the environment. Investing in robust safety practices today keeps the workplace running and the community safe tomorrow.
What are the physical and chemical properties of Pentachloronitrobenzene?
A Closer Look at the Substance
Pentachloronitrobenzene, often called PCNB in labs and agriculture circles, stands out due to its heavy load of chlorine atoms and a single nitro group attached to a benzene ring. People might recognize it more as a fungicide than as a chemical curiosity, but its properties deserve some unpacking.
Physical Details Worth Noting
PCNB appears as a solid, usually yellowish crystals or powder. At room temperature, it stays stable, not melting until hitting roughly 190°C. Not many substances give off such a sharp, earthy odor either. Trying to dissolve it in water leads to disappointment—only a tiny bit dissolves. If you turn to solvents like chloroform, acetone, or benzene itself, it gives in more easily.
People in labs handle it carefully, partly since it's denser than water (almost 1.7 times heavier) and doesn't float around easily. Its stubbornness toward dissolving in water creates big challenges for cleanup or accidental spills, especially outside the lab where it sometimes ends up as runoff from treated soils.
Chemical Personality on Display
With five chlorine atoms locking onto the benzene ring, plus one nitro group, PCNB shows serious stability. Want to break it down? That takes strong acids or bases, high heat, or long exposure to sunlight. Under ordinary conditions, this stuff barely budges. Its stability explains why it lingers in soil, doing its work against fungi but also sticking around much longer than many modern alternatives.
From personal experience working on research projects, this environmental persistence worries me. Even small amounts can collect over years, and there's not much a regular rainstorm can do to wash it out of farm fields. PCNB doesn’t play nice with microbes either—those tiny soil critters don't chew through it quickly.
Health and Environmental Impact
There’s a flip side to effectiveness. PCNB has been restricted or banned in some regions. The main reason: its breakdown products, like pentachloroaniline, have raised long-term health questions in humans and local wildlife. Some research points to potential carcinogenic effects and nervous system problems with long-term exposure.
Every time farmers or groundskeepers choose to use PCNB, they face a tradeoff—better crop yields or greener golf courses come with the risk of long-lasting residues. Spotting it in soil or water samples signals a need for closer scrutiny about what’s drifting beyond field edges.
Seeking Safer Solutions
Faced with these risks, scientists and regulators keep searching for safer options. Rotating crops, planting disease-resistant breeds, and working microbes into the soil have started picking up the slack where PCNB once dominated. Where PCNB use continues, people need better handling protocols. Storing it securely, labeling clearly, and training people thoroughly lowers chances of accidental contamination in storage rooms or sheds.
Newer chemistries are chipping away at PCNB’s old dominance. Many of these break down quicker and target pests more precisely, avoiding the “one-size-fits-all” chemical solutions of decades past. Researchers are also testing simple methods—composting crop debris, using less chemical-heavy treatments, and keeping a closer watch on soil and water quality around treated areas.
How should Pentachloronitrobenzene spills or exposure be managed?
Facing the Hazards Head-On
Dealing with a nastier chemical than pentachloronitrobenzene (PCNB) proves tough. PCNB lingers in soil and water, it accumulates, and it brings risks to both people and wildlife. Nobody wants to spend their day worrying about toxic spills, but ignoring a leak doesn’t make things safer. One bad day with PCNB can create long-term damage, so public health and environmental stewardship demand real action.
Personal Stakes and Lessons Learned
My family raised vegetables. Once, a nearby farm runoff carried a batch of PCNB into the stream behind our property. The water stopped supporting frog eggs, and a summer later the crayfish vanished. You learn quickly: even small spills bring outsized effects, especially for rural people with wells and local gardens.
Science backs up what farmers figured out decades ago. The EPA considers PCNB a probable carcinogen, and studies link it to liver and thyroid problems in animals. Workers using pesticides with PCNB face headaches, skin irritation, and a list of long-term health problems. That isn’t theoretical: in the cotton fields, my neighbor spent weeks with unexplained rashes before noticing a broken sprayer line.
Taking Quick Action
Nobody should treat spills like minor headaches. Calling local hazmat responders tops any list—waiting for a “better time” risks everyone’s health. Soil needs to come out, sealed up, and hauled to a permitted hazardous waste landfill. Watching contractors wear suits and breathing gear drives the point: this chemical doesn’t belong in backyards or waterways.
Shoveling up dirt by hand just spreads the mess. Using heavy machinery, especially when rain threatens, reduces the risk of runoff and deeper groundwater contamination. On a practical note, landowners must press agencies for groundwater and well testing. Letting soil sit untouched invites slow, hidden poisoning—a preventable tragedy.
Preventing Future Exposures
So many of these accidents trace back to old barrels, rusty sprayer tanks, and hurried handling. Proper training and equipment cost money, but one spill can run a family dry. Talking to farm supply dealers, attending workshops, and taking pesticide safety courses beats reading fine print on a warning label after something goes wrong.
Regulation helps too. Some countries put strict controls or outright bans on PCNB use, for good reason. Farmers have options—crop rotation, resistant seed varieties, and fungal control strategies that don’t rely on chlorinated chemicals. Pushing hard for non-toxic alternatives means fewer emergencies for the next generation.
Community and Accountability
Trust grows fastest between neighbors who look out for each other. Local authorities need to let people know where to call, who pays for clean-up, and where replacement water can be found. States and counties can set up public databases so communities track past spills and know what chemicals lurk underground. Real transparency makes it easier to spot problems before they escalate.
Staying safe with chemicals like PCNB boils down to common sense, vigilance, and a willingness to ask tough questions. No field, stream, or family benefits from secrecy and shortcuts. Owning up to spills and fighting for safer alternatives show respect for both the land and the people living on it.
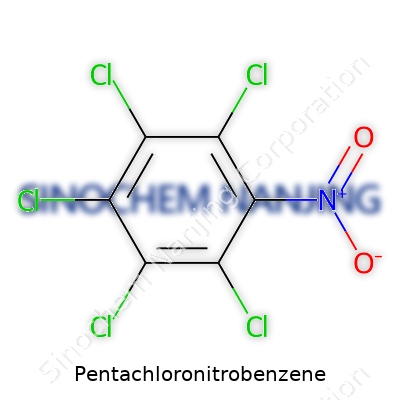
| Names | |
| Preferred IUPAC name | 1,2,3,4,5-Pentachloro-6-nitrobenzene |
| Other names |
PCNB Quintozene Nipacide Fonem Terracur Terrachlor Pentachloronitrobenzol |
| Pronunciation | /pɛnˌtæk.lɔːr.oʊˌnaɪ.trəʊˈbɛn.ziːn/ |
| Identifiers | |
| CAS Number | 82-68-8 |
| Beilstein Reference | 1367546 |
| ChEBI | CHEBI:38621 |
| ChEMBL | CHEMBL21614 |
| ChemSpider | 15702 |
| DrugBank | DB11206 |
| ECHA InfoCard | 03c4fca4-6e04-47dd-a1f2-9f5d318a47bd |
| EC Number | 208-060-1 |
| Gmelin Reference | 52968 |
| KEGG | C13395 |
| MeSH | D010478 |
| PubChem CID | 992 |
| RTECS number | GL7750000 |
| UNII | NN8I7SZO0J |
| UN number | UN1663 |
| CompTox Dashboard (EPA) | DTXSID8023467 |
| Properties | |
| Chemical formula | C6Cl5NO2 |
| Molar mass | 288.34 g/mol |
| Appearance | White to yellow crystalline powder |
| Odor | Odorless |
| Density | 1.692 g/cm³ |
| Solubility in water | Insoluble |
| log P | 3.94 |
| Vapor pressure | 0.00011 mmHg (@ 25 °C) |
| Acidity (pKa) | 12.9 |
| Basicity (pKb) | 15.33 |
| Magnetic susceptibility (χ) | −66.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.656 |
| Viscosity | 1.93 mPa·s (at 20 °C) |
| Dipole moment | 2.95 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 367.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -44.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -627.6 kJ/mol |
| Pharmacology | |
| ATC code | D08AE21 |
| Hazards | |
| Main hazards | May cause cancer; harmful if swallowed, inhaled, or absorbed through skin; causes skin and eye irritation; may cause damage to liver and kidneys. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H332, H351, H410 |
| Precautionary statements | P261, P272, P273, P280, P302+P352, P333+P313, P362+P364, P391, P501 |
| NFPA 704 (fire diamond) | 2-2-0-☣ |
| Flash point | Flash point: 181°C |
| Autoignition temperature | 405°C |
| Lethal dose or concentration | LD50 oral rat 7500 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 7500 mg/kg |
| NIOSH | PCNB |
| PEL (Permissible) | PEL 0.1 mg/m³ |
| REL (Recommended) | 0.5 mg/m3 |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
Hexachlorobenzene Nitrochlorobenzenes Chloronitrobenzenes Pentachlorobenzene Trichloronitrobenzenes |

