Pentachlorobenzene: A Closer Look at a Controversial Chemical
Historical Development
Pentachlorobenzene traces its roots back to the expanding chemical industry of the early 1900s. At the time, researchers sought chlorinated aromatic compounds for their potential in pest control and manufacturing. Demand for new industrial chemicals soared during the Second World War, and pentachlorobenzene emerged as both a precursor and a byproduct in the production of more complex chlorinated compounds. Large-scale production flourished throughout the 1950s and 1960s, a time when regulations were looser and public concern about persistent organic pollutants had yet to take hold. Researchers studying persistent organic pollutants flagged pentachlorobenzene for its ability to stick around in the environment and its tendency to accumulate in living tissues, setting the stage for today’s strict scrutiny and ongoing research.
Product Overview
As a crystalline solid, pentachlorobenzene earned a niche spot in chemical manufacturing as well as in the formulation of dielectric fluids and pesticides. Chemists valued it for its stability and effectiveness as a chemical intermediate. Though few intentional uses remain, small amounts still exit laboratories and legacy supply chains, mostly as waste or side products in chlorinated solvent production.
Physical & Chemical Properties
Holding a distinct yellow-white appearance, pentachlorobenzene resists water yet dissolves in common organic solvents such as benzene and ether. At room temperature, it sits solidly in the non-volatile category, thanks to five chlorine atoms locked onto a single benzene ring. The chemical formula, C6HCl5, shows just how far chlorination runs: hydrogen all but disappeared, making the molecule tough to break down. With a melting point hovering near 85°C, flame isn’t the main risk. Instead, its environmental persistence and potential for bioaccumulation stand out.
Technical Specifications & Labeling
Bottles and containers of pentachlorobenzene usually wear international hazard labels. Every shipment displays the UN 3077 code (environmentally hazardous solid, n.o.s.), a skull-and-crossbones warning, and the typical indication of toxic substance. Specifications must meet purity thresholds—industrial grades rarely exceed 99% purity and analytical grades reach only slightly higher. Each datasheet ticks off boiling and melting points, solubility, pH range in solution, and storage conditions (cool, dry, tightly sealed). These aren’t just formalities. Chemical handlers rely on precise documentation to manage risks, meet regulatory requirements, and clarify disposal protocols for a hard-to-destroy compound.
Preparation Method
Industrial chemists typically synthesize pentachlorobenzene through direct chlorination of benzene in the presence of a Lewis acid like ferric chloride. The process demands careful control to avoid over-chlorination, which can tip the reaction toward hexachlorobenzene—another highly persistent chemical. By fine-tuning temperature, chlorine flow rates, and catalyst loading, operators get pentachlorobenzene in decent yield, though byproducts shadow every batch. This production method, used for most of the last century, leaves an environmental footprint that’s become a focus of both remediation and policy reform.
Chemical Reactions & Modifications
Once formed, pentachlorobenzene resists most attempts at chemical transformation. Its chlorine-heavy benzene ring fends off many common oxidizers and reducers, though it does open up under extremes—ultraviolet light can prompt some breakdown, and strong nucleophiles pry off chlorine atoms, sometimes generating less persistent but still toxic byproducts. These limited reactions nudge researchers toward bioremediation or high-energy treatments for detoxification. For chemists, handling pentachlorobenzene mostly means managing its transformation into hexachlorobenzene or using it as a step in the synthesis of specialty chemicals and dyestuffs.
Synonyms & Product Names
Known by a list of monikers that speak to its ubiquity and chemical structure, pentachlorobenzene also appears as PCBz, Benzene pentachloride, and 1,2,3,4,5-pentachlorobenzene. Common database entries also list “QCB.” In trade documents and supply lists from past decades, it’s referenced with varying hyphenation or abbreviations that can sabotage digital searches for safety info; this patchwork of synonyms complicates inventory audits and risk assessments, especially for companies trying to root out legacy stocks.
Safety & Operational Standards
Pentachlorobenzene handling requires full chemical protective suits, nitrile gloves, and splash-proof goggles. Ventilated enclosures cut down on inhalation risk, though the low volatility of the solid helps a little. Companies must install airtight storage systems and maintain rigorous logs for inventory tracking, minimizing worker exposure and loss through leaks. Disposal guidelines mandate incineration at high temperatures well above typical hazardous waste standards—anything less risks sending residues into landfills where leaching can contaminate groundwater for decades. The strictness of these guidelines springs from the compound’s proven ability to persist both in the human body and in the environment long after its original use has faded into memory.
Application Area
At its height, pentachlorobenzene filled a surprising range of roles—in pesticides, as an intermediate for the manufacture of dyes, and even as a heat transfer medium in some commercial processes. Dielectric fluids took advantage of its high stability, though concerns about toxicity have erased those applications from new markets. Today, most intentional uses have vanished, replaced by less persistent, less toxic alternatives. Environmental monitoring teams still encounter the compound, though—remediation engineers, landfill auditors, and hazardous waste handlers know its profile well, both in soil and industrial runoff.
Research & Development
Current research on pentachlorobenzene pivots on two poles: understanding long-term ecological impacts and finding effective remediation strategies. Trace residues show up in fish, soil, and even remote Arctic ice, fueling studies on transport, accumulation, and ultimate human exposure. Teams use gas chromatography and mass spectrometry to track even minuscule residues in complex matrices. Bioremediation holds promise—engineered consortia of bacteria have started breaking down pentachlorobenzene in lab-scale studies, but upscaling to real-world conditions often falters. Funding agencies increasingly push chemical and process engineers to design greener substitutes for legacy chlorinated aromatics, but replacing pentachlorobenzene’s unique properties isn’t a simple swap.
Toxicity Research
Long-term studies show that pentachlorobenzene accumulates in fatty tissues of both animals and humans. Ingestion and inhalation studies on rodents yield results ranging from liver hypertrophy to hormonal disruption. The compound’s persistence means even jurisdictions with outright bans continue to wrestle with legacy contamination. Human health agencies point to evidence of immunotoxicity and possible carcinogenic effects. The World Health Organization groups pentachlorobenzene among other persistent organic pollutants that demand global action, citing measurable traces in breast milk and remote wildlife. The slow, chronic exposure pathway underscores just how insidious these compounds can be—even at concentrations that appear harmless in short-term lab tests, cumulative effects mount up. Public health researchers point to pentachlorobenzene as a poster child for why strict oversight remains essential long after industrial enthusiasm fades.
Future Prospects
Interest in cleaning up pentachlorobenzene isn’t going anywhere. Governments press for better mapping of contaminated sites and safer, cheaper remediation options—in situ thermal destruction, advanced oxidation, and innovative bioaugmentation draw real investment. Regulators prioritize removal from wastewater and landfill leachate, weighing tighter controls for legacy installations in developing economies. International treaties already limit production and trade, but technical loopholes and illegal dumping keep the threat alive. Cyclists in major river deltas still catch whiffs as sediment stirs, and groundwater surveys turn up traces even far from the original manufacturing footprints. The movement for a chemical circular economy, where hazardous compounds no longer become permanent fixtures in soil and water, relies on new technology and persistent political will. Scientists and advocates push for more research into the long-term impacts on both people and ecosystems, aiming to learn—and teach others—how to avoid repeating past mistakes.
What is Pentachlorobenzene used for?
From Manufacturing to Environmental Headlines
Pentachlorobenzene often shows up in conversations about pollution or legacy chemicals, but this compound didn’t just appear out of thin air. Industries have put it to work for decades. Its main action happens behind the scenes, usually in the production of other chemicals. Factories once used pentachlorobenzene to create substances like polychlorinated biphenyls (PCBs) and pentachloronitrobenzene (PCNB), which then found their way into electrical equipment and fungicides.
With its five chlorine atoms hooked onto a benzene ring, this substance holds together well, resisting breakdown from both water and sunlight. At one time, this made it prized as an intermediate step in chemical manufacturing. I remember reading about its role helping to keep some crop soils free from fungus, especially in the 1960s and 1970s, when the agricultural industry leaned heavily on chemicals to boost harvests.
Risks Outweigh Benefits
Talk with almost any researcher focused on environmental health, and pentachlorobenzene quickly raises red flags. One challenge: this compound hangs around in the environment, long after companies stop making or using it. Its persistence threatens water, soil, and air quality. Over time, it builds up in living tissue—a process known as bioaccumulation. Fish caught in polluted lakes can show levels dozens of times higher than the water they swim in.
Scientists have connected pentachlorobenzene to a range of health concerns. Studies link exposure to problems in liver function, thyroid balance, and even the nervous system. Evidence in both lab animals and human case studies led governments to regulate or ban its use for many applications. As more stories emerged about families living near old dump sites with higher rates of disease, the message grew clearer: what happens upstream lands downstream in our food chain.
Searching for Solutions
Limiting new releases helps, but the real test lies in cleaning up what's already out there. Cleanup teams face some tough decisions. Mechanical removal works in hotspots, but spreading the cost across vast regions tainted by chemical runoff often needs a different approach. Some new methods look at using bacteria or plants to digest pentachlorobenzene found in soil or water, a process called bioremediation. Results show promise, though these fixes often take years, especially in cold climates or remote areas.
Regulatory agencies play a vital role. In my own city, tighter rules on industrial waste shifted focus toward better monitoring and transparency. Tracking hazardous waste more carefully, holding polluters responsible, and supporting research into safer alternatives has already driven down levels of pentachlorobenzene and related chemicals worldwide. Education helps too. As more people understand how chemical choices ripple through ecosystems, calls for smart, sustainable alternatives grow louder.
Learning From the Past for a Healthier Future
History offers countless examples of chemicals that seemed useful at first, only to create more problems down the road. Whether pentachlorobenzene shows up in old equipment, pesticides, or in contaminated rivers, keeping a close watch matters. It takes ongoing commitment from scientists, regulators, and the communities affected by pollution. Many experts now focus on green chemistry, designing new materials that don’t linger or harm wildlife. One thing’s certain: learning from legacy chemicals like pentachlorobenzene shapes smarter choices for tomorrow.
Is Pentachlorobenzene hazardous to human health?
What Is Pentachlorobenzene?
Pentachlorobenzene shows up in the world as a man-made chemical. Factories once used it to make other chemicals, serve as a fungicide, and sometimes as a byproduct and impurity in the production of certain pesticides. No one would expect it on a grocery shelf, but traces have made their way into the environment, clinging to soil, sliding into waterways, or lingering in air—often far from where it started out.
How People Can Encounter It
Once in a while, chemical names sound so distant they barely connect to daily life. Pentachlorobenzene isn’t like that. Small amounts can leave factories in wastewater, drift from hazardous waste sites, or leach from discarded products. People might breathe it in air, drink water carrying it, or eat food, especially fatty fish, where these chemicals accumulate. Some workers in chemical plants risk more intense exposures, a reality that’s hard to ignore if you’ve known anyone in industrial jobs.
What Science Says About Its Hazards
No one in their right mind wants chemicals that build up in tissue. Pentachlorobenzene stacks up in fat, and sticks around longer than you’d like. Environmental researchers have studied its effects over decades. Animal studies found it harms the liver and thyroid, two organs that run the show for energy, growth, and waste removal. Exposure in large doses caused weight loss, and it made a mess of normal organ structure. The World Health Organization has recognized the risks, and many countries have worked to limit its production for good reason.
People can’t shrug off these animal studies. Chemicals that damage the liver and thyroid in rats and mice often spell trouble for humans too. The U.S. Environmental Protection Agency classifies pentachlorobenzene as a possible human carcinogen. That sounds technical but carries weight. Cancer, after all, doesn’t look at backgrounds or habits before striking. Even though direct, one-to-one human data stays limited, strong evidence points to a need for caution.
Regulation and the Role of Clean-Up
No one wants chemicals silently sticking around in backyards, drinking water, or playground dirt. Many countries, including the United States, list pentachlorobenzene as a persistent organic pollutant. The Stockholm Convention targeted it for elimination because these pollutants can travel across borders, settle in Arctic ice, or build up in food webs. Once something makes it into a global treaty for phase-out, you know there’s real cause for concern.
Communities living near old industrial sites have pushed for stronger clean-up and tighter laws. Seeing firsthand how lack of environmental controls leaves families at risk creates urgency. Safer disposal practices, better emissions tracking, and honest information about soil and water contamination help keep people aware and protected. Public health agencies publish exposure guidelines to keep drinking water clean and provide advice on testing wells and soil in suspected areas.
What Can Make a Difference
Responsible disposal of hazardous wastes, especially by industries, makes a world of difference. Supporting local and national rules that keep chemicals like pentachlorobenzene out of the environment pays off in health and safety. Testing old industrial sites, cleaning up contaminated soil, and discouraging the reuse of abandoned sites for homes or schools without proper remediation reduces the risks directly. Learning about what lingers in the dirt, water, and air of a community brings a sense of control. Knowledge and action, grounded in solid science, make the biggest difference for real people, not just policy makers or researchers.
How should Pentachlorobenzene be stored and handled?
Recognizing the Risks
Pentachlorobenzene, a chlorinated aromatic compound, exudes a sense of seriousness in any workplace that stocks hazardous chemicals. Decades of industrial and scientific work have made one thing stand out: it pays to treat this chemical with more care than some of its cousins. Anyone who’s spent time around chemical labs or warehouses learns the hard way that casual storage of substances like pentachlorobenzene is a fast-track to trouble.
Acute and chronic health risks from exposure remain well documented. The EPA considers pentachlorobenzene a probable human carcinogen, and animal research points to nervous system effects, liver and kidney trouble, and interference with thyroid function. People handling it need to know its hazards before they walk anywhere near a barrel or flask. Not everyone heeds those warnings, but ignoring regulations and best practices invites real harm.
Separation and Site Selection
Using knowledge from years navigating chemical storerooms, it becomes clear that location matters more than label-checking. Pentachlorobenzene can volatilize over time. Keep it apart from heat sources and from chemicals that could start a reaction—acids, alkalis, and oxidizers belong as far away as possible. Storage areas demand locked entry, secure shelving, and clearly marked hazard signage, leaving no room for confusion. Storing it outside, exposed to weather, sets the stage for leaks and wider environmental impact. Tucked safely indoors, with routine climate control, pentachlorobenzene stays contained.
The Right Containment: Practical Decisions
Glass containers with tight, chemical-resistant lids work best for laboratory quantities. For industrial storage, choose steel drums lined with a compatible coating. Rely on personal experience here—plastic vessels sometimes react or degrade, releasing both the chemical and microplastics into the air or soil. No one wants an accidental spill, so solid containment makes life easier. Line floors with sealed, non-porous materials, because cracked concrete and old wood open the door to seepage and contamination. Keep spill kits and fire extinguishers nearby, stocked and in reach. Over the years, I’ve learned that repeated checks (not just annual audits) catch problems that paperwork misses.
Personal Protection and Training on the Ground
For all the talk about containers, it’s people who shoulder the risk. That means wearing nitrile gloves, chemical splash goggles, and properly rated respirators—even for brief tasks. In smaller companies, PPE sometimes gets skipped under time pressure, but the fallout from a single splash or inhalation can haunt a worker for years. Training beats lucky guesses every time. Employees who understand exposure pathways—skin absorption, inhalation, accidental ingestion—recognize why every precaution matters. Posting visible, straightforward handling instructions near storage spots keeps routines sharp even during hectic workdays.
Disposal and Emergency Response
Pentachlorobenzene ranks as a persistent organic pollutant. Dumps, drains, or incinerators without specific hazardous waste permits are out of the question. Build a working relationship with licensed waste handlers who follow the law and document disposal thoroughly. Spills call for immediate cordoning and cleanup by trained personnel, never by untrained hands or with improvised materials. A good storeroom always features eyewash stations and emergency showers within a few steps, not tucked away on another floor.
Commitment Beyond Compliance
Safe storage and handling of pentachlorobenzene shows respect for colleagues, communities, and the environment. For chemical workers, these routines bring peace of mind and keep doors open for ethical business. In my years around chemicals, cutting corners always led to close calls—so I stand firm: thorough planning and consistent vigilance form the backbone of real safety with pentachlorobenzene.
What are the environmental impacts of Pentachlorobenzene?
Hidden Problems Lurking in the Ground and Water
Pentachlorobenzene, usually called PeCB, has a nasty tendency to stick around. It shows up in soil and sediment for years because it breaks down very slowly. Decades-old chemical dumps still leak PeCB into the earth, which means even “retired” contamination zones keep spreading concern. This stuff drifts into rivers and groundwater, and there’s a real risk of it ending up in crops or drinking water supplies.
Living near an old industrial site, I still hear neighbors wonder about what might lurk beneath the topsoil. And for good reason. PeCB doesn’t stay put. Heavy rain moves particles through surface water and drainage ditches, and fish in polluted creeks pick it up. Scientists have measured PeCB in fish tissue around the world—evidence that food chains are picking up the slack when basic site cleanup gets ignored.
Air Quality Concerns Nobody Wants
Some people imagine that banned chemicals fade from our lives, out of sight and out of mind. But PeCB makes its way into the air, especially near waste incinerators and old manufacturing sites. Once it’s airborne, winds carry it far from where it started. Tiny particles reach places that seemed untouched: Arctic ice fields, high-mountain lakes, even forests on different continents.
I remember a science news story about researchers finding decades-old industrial compounds in remote snow samples. PeCB particles, though invisible, travel long distances, changing the makeup of places that never produced any themselves. Families out camping or hiking have no way of spotting these threats, and local wildlife gets exposed year-round.
Risks to Wildlife and Local Health
Wildlife takes a bigger hit than we notice day to day. PeCB tends to build up in living things. Fish and birds living in tainted waters collect more of it over time, and the effects don’t stop with a single generation. Some studies draw links between PeCB exposure and changed behavior, trouble reproducing, and weaker immune systems in wild animals.
Communities relying on subsistence fishing shouldn’t have to worry about invisible toxins. I’ve seen the anxiety at community meetings, folks bringing in fish samples and asking if the catch is safe to eat. In regions where food sources already feel stretched thin, away from large supermarkets, toxic buildup just adds another layer of risk families can’t afford to ignore.
Looking at What Can Help
PeCB has shown up as a byproduct of making pesticides, solvents, and other chemicals. Even though regulations banned much production, some remains get released from industrial and municipal waste burning. Agencies like the EPA and international groups included PeCB on banned or restricted lists, but monitoring only works if backed by enough resources and action.
Local communities and environmental health workers have found success in tracking contaminated hotspots before the threats spread farther. Testing soils near old dump sites, restoring wetlands to slow down runoff, and improving controls at waste incinerators bring PeCB exposures lower. It isn't enough to just fence off an old site. Restoring contaminated land for safe community use usually comes only after long local effort and pressure.
Public awareness can make a difference. People in affected areas share information, demand stronger cleanup standards, and work with scientists to track where pollutants are showing up next. Talking with neighbors and staying informed through local news helps bring attention to issues regulators may miss. That push, more than anything, drives real cleanup and keeps new generations a bit safer.
How can Pentachlorobenzene exposure be detected or measured?
Real-World Stakes of Measuring Toxic Chemicals
I’ve spent years reviewing environmental health reports, and one thing that stands out is the sense of urgency that comes with tracking persistent organic pollutants like pentachlorobenzene. This chemical lingers in the environment and builds up in living things. Not many folks talk about it at the dinner table, but pentachlorobenzene moves quietly through soil, water, air, and right into people and animals. Once it’s in, it tends to stick around. That alone sets the stage for a bigger conversation about detection.
How Professionals Actually Test for Pentachlorobenzene
People usually think environmental testing only happens in the wake of industrial accidents or massive spills, but routine checks happen every day in labs across the world. Technicians collect everything from air samples near factories to human blood and urine samples. Lab pros look for pentachlorobenzene using instruments like gas chromatographs linked up with mass spectrometers. These machines act like bloodhounds, sniffing out the faint signature of the chemical in a complex mix. Scientists can measure levels in parts per trillion, which still matters for something this toxic.
Finding pentachlorobenzene in blood or fat tissue usually means a person has been around contaminated soil, food, or water. Most people never get these kinds of lab tests unless they live near hazardous waste sites or work in jobs with an elevated chemical risk. This leaves the broader population with less clarity on whether they’re being exposed. It’s more than a laboratory problem—it’s a public awareness one.
The Facts Around Exposure
Decades of research have shown pentachlorobenzene can move long distances in the air, settle in fatty tissues, and create a toxic domino effect up the food chain. Chronic exposure has been linked to issues like liver damage, neurological effects, and even cancer. The World Health Organization includes pentachlorobenzene among the top chemicals of concern worldwide. With that risk on the table, skipping detection only hands down the problem to the next generation.
Limits of Current Detection
Surveillance systems often struggle to keep up. Most community-level monitoring happens around known industrial hotspots. In the rest of society, large blind spots exist. This leaves everyday folks who might grow their own food or collect rainwater with few resources to check for toxic exposure. Lab testing stays expensive, and most doctors do not order tests for pentachlorobenzene without a strong reason. If exposure does not show up in national surveys, communities fall off the radar.
Building a Safer System
People shouldn’t have to worry about invisible chemicals invading their homes. Making detection more accessible starts with better public information. Community health clinics, schools, and local governments can stock easy-to-understand resources that explain environmental test results. Affordable and reliable home testing kits are on the horizon, though most don’t cover pentachlorobenzene yet. Some countries, like Sweden and Canada, have created public monitoring programs that let citizens know what’s turning up in their own neighborhoods. Upping this level of transparency builds the groundwork for better personal and environmental choices.
Scientists keep improving detection. Cutting-edge laboratories now use portable detection tools at field sites, offering real-time updates for communities at risk. People working with soil, water, or food production benefit from this kind of development. These steps prove that measuring exposure is not reserved for specialists or big budgets—it’s a goal communities can tackle with the right willpower, expertise, and trustworthy information.
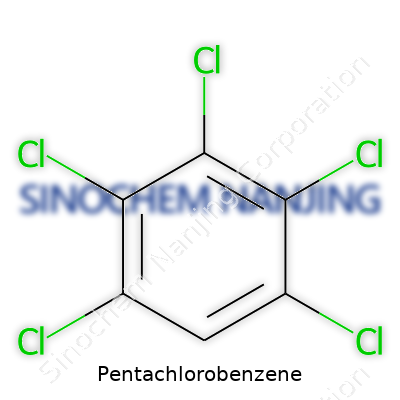
| Names | |
| Preferred IUPAC name | Pentachlorobenzene |
| Other names |
PCBz Pentachlorobenzol |
| Pronunciation | /ˌpɛntəˌklaɪrəˈbɛnzən/ |
| Identifiers | |
| CAS Number | 608-93-5 |
| Beilstein Reference | 1209241 |
| ChEBI | CHEBI:36474 |
| ChEMBL | CHEMBL232482 |
| ChemSpider | 5322 |
| DrugBank | DB13125 |
| ECHA InfoCard | 03f9eb4a-8ea7-4c73-a546-f6bdb4710c6c |
| EC Number | 601-055-00-7 |
| Gmelin Reference | 619 |
| KEGG | C14522 |
| MeSH | D010426 |
| PubChem CID | 10037 |
| RTECS number | GZ9625000 |
| UNII | A9D7FGN8G6 |
| UN number | UN3079 |
| Properties | |
| Chemical formula | C6HCl5 |
| Molar mass | 251.32 g/mol |
| Appearance | White crystalline solid |
| Odor | Aromatic. |
| Density | 1.675 g/cm³ |
| Solubility in water | Insoluble |
| log P | 5.18 |
| Vapor pressure | 0.13 mmHg (25°C) |
| Acidity (pKa) | >6.2 |
| Basicity (pKb) | 12.45 |
| Magnetic susceptibility (χ) | -0.00032 |
| Refractive index (nD) | 1.562 |
| Viscosity | 1.32 mPa·s (at 25 °C) |
| Dipole moment | 2.58 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 151.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -51.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3012.6 kJ/mol |
| Hazards | |
| Main hazards | Suspected of causing cancer; very toxic to aquatic life with long lasting effects |
| GHS labelling | GHS02, GHS07, GHS08, GHS09 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301+H331: Toxic if swallowed or if inhaled. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P260, P273, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P313, P501 |
| NFPA 704 (fire diamond) | 2-2-0-〡 |
| Autoignition temperature | 560°C |
| Explosive limits | Upper: 7.2% ; Lower: 1.2% |
| Lethal dose or concentration | LD50 orally in rat: 2500 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 750 mg/kg |
| NIOSH | GV9450000 |
| PEL (Permissible) | 1 mg/m³ |
| REL (Recommended) | 0.5 mg/m³ |
| Related compounds | |
| Related compounds |
Benzene Hexachlorobenzene Tetrachlorobenzene Chlorobenzene Polychlorinated biphenyls |

