P-Nitrophenyl Isocyanate: Deep Dive into Science, Safety, and Application
Historical Development
Chemistry builds on curiosity, and P-Nitrophenyl isocyanate came to life out of pure experimental drive. It first started popping up in labs in the early 20th century. Chemists grabbed hold of isocyanates as soon as they realized their reactivity with amines, alcohols, and other nucleophiles could unlock whole new classes of materials. Specifically, para-nitrophenyl isocyanate stood out because of the nitro group on its aromatic ring, giving scientists new control over reactivity. Over time, the focus shifted toward clean, controlled preparation and detailed study of its properties, but the root motivation never changed—seeking new tools for building molecules that fuel pharmaceuticals, polymers, and specialty chemicals.
Product Overview
One glance at p-nitrophenyl isocyanate and you see a pale yellow, crystalline compound with a sharp odor. Its main selling point in the lab comes from that highly reactive isocyanate group perched on an aromatic ring decorated with a strong electron-withdrawing nitro group. This structure primes it for reactions, especially in making ureas, carbamates, and other critical intermediates. People in research and industrial settings rely on it to introduce specific functionalities into target molecules in a simple, robust fashion.
Physical & Chemical Properties
P-nitrophenyl isocyanate has a melting point between 56 and 58 °C and boils at roughly 170 °C under reduced pressure. It dissolves fairly well in common organic solvents like acetone, ether, and dichloromethane, but keeps itself mostly out of the water—something typical for aromatic isocyanates. The nitro group on the aromatic ring makes it more reactive than plain phenyl isocyanate. This helps in everything from peptide synthesis to the preparation of diagnostic reagents because the chemical will react quickly and cleanly in the hands of a careful chemist.
Technical Specifications & Labeling
Laboratory bottles should clearly state the compound’s name: p-nitrophenyl isocyanate. Other important items on the label include the chemical formula, C7H4N2O3, and the CAS number, 104-16-5. Labels show hazard warnings required by regulations—usually the red diamond pictogram indicating acute toxicity, along with statements reminding users to wear gloves, avoid inhaling vapors, and keep it in tightly closed containers in well-ventilated, dry spaces. Specifications list purity, which often climbs above 98% for research purposes, with makers providing information on melting range, color, and solvent compatibility to assure users of batch quality.
Preparation Method
Synthesizing p-nitrophenyl isocyanate starts by converting p-nitroaniline into the desired product. In practice, many chemists use phosgene or triphosgene as a reagent, which reacts with p-nitroaniline to form the isocyanate group. After mixing the precursors in an appropriate solvent, the operator slowly introduces phosgene under cool conditions to control the exothermic reaction. Careful temperature monitoring, good ventilation, and proper containment form the backbone of a safe prep, especially considering the risk posed by phosgene gas. Isolation involves removing solvents, washing, and recrystallizing the solid to yield product ready for further use or market.
Chemical Reactions & Modifications
Living in a world where tailored molecules solve many problems—diagnostics, medicine, coatings—p-nitrophenyl isocyanate earns its place by reacting with amines, alcohols, and thiols to build ureas, carbamates, and thiocarbamates. These linkages play starring roles in peptide synthesis and labeling of biomolecules, letting researchers attach tags or control activity with surgical precision. The electron-withdrawing nitro group not only boosts reactivity but offers anchor points for additional modifications, supporting new discoveries in enzyme kinetics or drug design.
Synonyms & Product Names
Around the world, chemists and industry refer to this compound by several names: 4-nitrophenyl isocyanate, para-nitrophenyl isocyanate, and simply PNPI among them. In catalogs, you’ll see “p-nitroisocyanatobenzene” and “1-isocyanato-4-nitrobenzene.” No matter the label, people seek it out because experience and reference papers point to this class of isocyanates as highly reliable reactive partners in synthesis.
Safety & Operational Standards
Working with any isocyanate takes serious commitment to safety. P-nitrophenyl isocyanate doesn’t let you cut corners. Short exposure may irritate eyes, respiratory tract, and skin, so fume hoods and gloves are non-negotiable. Safety data sheets recommend storing the powder away from acids, bases, moisture, and ignition sources. Accidental spillage demands immediate clean-up with absorbent materials while wearing full protective gear. Waste disposal follows strict local chemical guidelines—in my old lab, no one skipped the paperwork because one careless moment sends reactive fumes throughout a workspace. Reading up on both manufacturer instructions and broader chemical handling protocols ensures the safety net never slips.
Application Area
P-nitrophenyl isocyanate runs the gamut of applications in the lab, from tagging amino acids for HPLC analysis to building custom polymers. In protein chemistry, attaching the reagent to terminal amino groups allows detection at low concentrations—essential for mapping the fine details of structure and sequence. Scientists in pharma use the reactivity of the isocyanate to produce potential drug candidates by building urea linkages linked to biological activity. Polymers benefit from tailored monomer additions, giving coatings and adhesives properties not possible with off-the-shelf chemicals. This compound’s reach keeps expanding with the question, “Can we tag, connect, or functionalize that with PNPI?” often getting a confident yes.
Research & Development
From my own stints in synthetic chemistry and bioconjugation, research with p-nitrophenyl isocyanate always circles back to reactivity and selectivity. Scientists push for milder, greener synthesis routes to cut down hazardous byproduct waste, since traditional routes rely heavily on phosgene. On the analytics side, new labeling methods using isocyanates let researchers spot subtle differences in biomolecule structure that guide everything from understanding diseases to designing next-gen therapeutics. The industrial sector explores broader utility in specialty polymers, improving durability or adhesiveness without sacrificing cost or processability. These research streams keep bringing up new uses as more teams share their tricks and successes.
Toxicity Research
Most people don’t spend time pondering chemical toxicity, but researchers certainly do. Toxicity studies with p-nitrophenyl isocyanate show respiratory and dermal hazards, much like other aromatic isocyanates. Animal exposure studies point out risks such as bronchial irritation and potential sensitization reactions. Chronic exposure links to occupational asthma, drawing clear lines for threshold limits in the workplace. On the molecular level, the nitrophenyl group raises flags regarding environmental persistence and bioaccumulation. That means regulators keep a close eye and labs take no chances—every incident of exposure gets documented, and personal stories of chemical burns remind scientists to respect both the power and risk of their tools.
Future Prospects
A lot rides on how safely and sustainably we use and manufacture p-nitrophenyl isocyanate. The chemical’s value spreads across fields like diagnostics, pharmaceuticals, and materials. Continued demand for precision labeling in proteomics and drug design fuels improvements in production. Calls for “greener” synthesis—using enzymes, milder reagents, or recyclable catalysts—inspire a new generation of chemists to rethink long-standing recipes. At the same time, advances in automation and in-line detection help reduce worker exposure and streamline large-scale production. Balancing function with safety, chemists collaborate across disciplines, seeking methods that cut risks while delivering on performance. Stories from the lab floor, like mine, fit into a long arc of research, where persistence and innovation gradually turn challenging chemicals into safer, more effective tools.
What is the chemical structure of P-Nitrophenyl Isocyanate?
Getting to Know the Molecule
P-Nitrophenyl isocyanate isn’t something you come across at your local hardware store, but it plays a quiet role in labs and industries that shape materials, drugs, and coatings. Its structure blends two attention-grabbing groups: a nitro group and an isocyanate group, each attached to a benzene ring. Picture a six-sided carbon ring, the benzene base. At one spot, you find a nitro group (NO2) sticking out. On the exact opposite side, you find an isocyanate group (NCO). Working in a lab, I struggled to remember the orientation early on—turns out, the position of those groups defines how the molecule acts and who it reacts with.
The Layout in Plain Terms
You might hear chemists toss around the phrase “para-substituted” for this setup. That just means the two groups sit across from each other, not bunched up on the same side. Connecting those dots helped me wrap my head around P-nitrophenyl isocyanate’s layout. Other molecules, like explosives or pharmaceutical reagents, often turn wild depending on where those nitro or isocyanate groups land. Get the placement wrong, and you don’t just get a different molecule—you sometimes get totally opposite chemical behavior.
The Nitty-Gritty: Why Structure Matters
The isocyanate group (NCO) lets P-nitrophenyl isocyanate hook up with amines or alcohols to make ureas or carbamates. If a chemist wants to connect two completely different molecules, this isocyanate can work like a sharp tool in a toolbox. The nitro group does more than just sit pretty—it pushes electrons through the ring. That tiny change shifts reactivity, changing what sorts of bonds the molecule forms and how quickly the reactions run.
If you’re mixing reagents in a factory or setting up a safety protocol, that nitro group on the para-position demands attention. It makes the molecule tougher to ignite and raises questions about health and environment. Splashes or vapors from isocyanates can make you cough, irritate your eyes, and, if things get out of hand, lead to breathing trouble. One day in the lab, a cracked stopper released just a noseful of fumes—my own lungs would not let me forget the consequences of a simple mistake.
Building on Trust and Safety
Trust comes into play because the smallest shift—like moving that nitro group around the ring—can mean the difference between a useful reagent and a mischievous hazard. Every trusted chemical supplier posts detailed safety data sheets that name all the reactive sites and outline how to handle each molecule in real-world language. That isn’t paperwork for its own sake; it means fewer accidents and more reliable batch-to-batch results. The health sector, in particular, leans on this kind of precision: immunoassays rely on reaction specificity, and the right isomer is everything.
Practical Steps Forward
Factories could design scrubbers and better ventilation to control fumes, reducing chemical accidents and environmental leaks. Training helps too: the first time I learned to spot the para- versus ortho- isomer using simple color reactions in a hood, I understood the difference between a safe workspace and a risky shortcut. Labs should use in-line monitoring, allowing real-time checks on reaction progress. Digital modeling adds another layer, letting chemists predict how a tweak to the structure will ripple through a supply chain or change how a medicine behaves in the body.
Everyone along the chain, from the person drawing the first structural diagram to the one tightening that last valve, shapes how P-nitrophenyl isocyanate performs in practice. In chemistry, details matter twice as much as they seem.
What are the common applications of P-Nitrophenyl Isocyanate?
Turning Chemicals into Tools in Modern Research
P-Nitrophenyl Isocyanate doesn’t pop up in everyday conversations, but in the world of labs and product development, it plays a powerful role. It’s not just a complicated name—behind those twelve syllables lies a backbone ingredient for chemistry that links molecules together in precise ways. Sometimes, the smallest changes in a molecule can spark big breakthroughs, and this compound helps bring those changes to life.
Paving the Way in Pharmaceutical Synthesis
Drug discovery often depends on coupling one molecule to another, and this is where P-Nitrophenyl Isocyanate steps in as a key partner. Medicinal chemists harness its tendency to react quickly with amines and alcohols, leading to ureas and carbamates—structures you’ll find in a host of drug candidates. Over the past decade, more advanced antibiotics and anti-cancer agents started with reactions that used this compound as a linker. Joining these building blocks with such efficiency saves both time and resources, letting researchers focus on real-world testing and delivery.
Reliable in Quality Control and Protein Chemistry
In the world of biotech, consistent and accurate testing stands as the foundation for progress. This is especially clear in protein sequencing, where P-Nitrophenyl Isocyanate supports amino acid detection. During Edman degradation—a method that sequences proteins step by step—this reagent tags amino termini so the individual building blocks can be analyzed. That’s not just science jargon; it’s how we unlock everything from genetic disorders to the next protein-based therapies. Having reliable detection tools keeps lab teams one step ahead of problems down the line.
Catalyst for Fine Chemicals and Dyes
Beyond healthcare, paints and plastics also benefit from this chemical’s unique properties. Synthetic dyes require stable bonds, and P-Nitrophenyl Isocyanate creates these by linking aromatic amines. Today’s vibrant pigments and color-fast textiles can trace some of their durability back to stable connections forged with this compound. In specialty plastics and resins, it steps in to cross-link molecules, improving toughness and heat resistance in finished products.
Sustainability and Safety Issues in Chemical Handling
With great synthetic tools come real challenges. P-Nitrophenyl Isocyanate is reactive, and health agencies like OSHA and EU Reach have flagged it for safe handling practices. Acute toxicity, primarily by inhalation, grabs the attention of specialists working with it. It’s crucial to use fume hoods, checked ventilation, and keep personal protective gear close at hand. I’ve watched teams struggle with old or poorly maintained equipment—little lapses can turn into major hazards, emphasizing the need for up-to-date safety checks.
Green chemistry is pushing for alternatives that achieve the same coupling strength but break down harmlessly. Academic labs and industry teams are testing reagents with fewer risks, though replacing tried-and-true reactions involves trial, error, and a willingness to question standard protocols. Incentives for sustainable chemical research can drive innovation, making our labs safer while still delivering the products people rely on daily.
Looking Forward: Smarter Use, Better Outcomes
P-Nitrophenyl Isocyanate has held its place because it does a complicated job with high reliability, whether in medicine or materials. By staying informed on both its applications and safety issues, chemists get the best of both worlds—innovation and responsibility. Shifting to greener alternatives and strict adherence to handling protocols invites a better, safer future, not just for professionals but for everyone that science touches.
What are the storage and handling recommendations for P-Nitrophenyl Isocyanate?
P-Nitrophenyl Isocyanate Isn’t Just Another Chemical
Few things draw attention in the lab like the presence of compounds with strong reactive groups, and p-nitrophenyl isocyanate is no exception. Over the years, I’ve seen a fair share of people assume it can be treated like benign white powder sitting on the shelves. From its powerful isocyanate group to the lively nitro substituent, this material asks for respect and care.
The Daily Realities of Storing P-Nitrophenyl Isocyanate
Labs are often bustling with activity, but storage practices for chemicals like p-nitrophenyl isocyanate make all the difference between safety and disaster. This compound reacts with water, so direct contact with moisture creates a risk not worth taking—ranging from formation of hazardous byproducts to ruined experiments. Storing it in a tightly sealed container, kept in a dry, cool place such as a designated chemical storage cabinet, has become second nature for those who’ve learned from past mistakes.
A lot of isocyanates require protection from light, but p-nitrophenyl isocyanate especially suffers from prolonged exposure. Sunlight or even overhead lab lighting can degrade it. Storing it in amber or opaque containers adds that layer of protection against light-triggered breakdowns. Simple steps like labeling with hazard information and storing away from incompatible materials speak louder than any written protocol in a folder.
The Risks Aren’t Just Theoretical
Anyone who has handled reactive isocyanates knows the smell and sting that hits you if vapors escape. I still remember the nervous energy the first time a bottle wasn’t closed fully—there’s nothing quite like that sharp chemical bite in the air calling you back to proper procedure. That’s why local exhaust, gloves, goggles, and lab coats aren’t just for show. Routine exposure to isocyanates has been linked to respiratory issues, skin sensitization, and irritation of the eyes.
Spills, even small ones, can turn ugly fast. Rapid action with absorbent material, followed by careful containment and disposal, limits spread. Neglecting this lands people and experiments on the losing side: imagine one unplanned reaction wiping out weeks of work or sickening colleagues. It’s a mess best avoided by double-checking every handling step.
Fact-Based Solutions Rooted in Experience
Training matters. A lab that drills its team on spill management, personal protective equipment, and emergency procedures faces fewer incidents. Chemical suppliers provide safety data sheets for a reason—they hold practical advice vetted by experience and regulatory science. Reading them isn’t a chore; it’s insurance.
Putting the right absorption materials and neutralizing agents in reach is invaluable. I once observed a clean-up handled smoothly only because the sorbents weren’t locked away, and that memory sticks. Regular checks on inventory, containers, and labels help fend off surprises. If containers or labels degrade, replacing them immediately protects everyone down the line.
Waste management, too, takes center stage. It pays off to develop arrangements with hazardous waste handlers so residues and expired stocks exit the lab safely rather than accumulating out of sight. Even something as minor as encouraging team communication about low stocks or spent containers contributes to a culture of safety.
The Stakes Remain High
Every lab professional carries stories of near-misses and lessons paid for in full. Storing and handling p-nitrophenyl isocyanate safely isn’t just a box to tick. It means investing effort, sharing experience, and building routines that keep everyone moving forward. The minor inconvenience of vigilance holds off the far bigger inconvenience of injury or lost research. In chemistry, the right habits stick, because the alternative never pays.
What are the safety precautions and hazards associated with P-Nitrophenyl Isocyanate?
Understanding the Hazards
P-nitrophenyl isocyanate may sound like another long chemical name, but it brings real dangers for people handling it. My first time stepping into a lab that used this stuff, caution signs covered every cabinet. P-nitrophenyl isocyanate damages the lungs when inhaled, and even a small spill—if someone wipes their eyes—can burn and seriously injure. Inhaling the dust or vapor releases sharp, pungent fumes, and that’s where the trouble really starts: coughing, tightness in the chest, and sometimes worse. Chemical burns and skin irritation knock people out of work for weeks.
Direct contact with this compound isn’t just annoying; it can leave lasting scars. With repeated exposure, skin becomes more sensitive. Allergies build up over time, and next thing you know, someone can’t go near the chemical at all. Studies confirm links to respiratory problems and dermatitis in settings where strict controls get ignored. In older plants, stories circulate about workers suddenly developing asthma-like symptoms—turns out, not everyone took ventilation seriously.
Real-World Safety Measures
In smaller labs, just one lapse—like not sealing a container properly—quickly fills a room with choking fumes. New safety rules mean everyone wears fitted respirators and nitrile gloves. The thin latex gloves I used to rely on? Useless against this kind of chemical. Swapping them out made a difference right away, especially as gloves showing even a pinhole break down fast. Well-designed ventilation matters—a working fume hood isn’t optional. Room air changes every few minutes cut down on harmful build-up before it reaches anyone’s lungs.
Eye protection gets overlooked far too often. A friend once got a tiny splash in his eye—he flushed it immediately, but the pain lasted days. No one makes that mistake twice. Full goggles and face shields stay non-negotiable in our lab now, even for routine transfers. Storage means more than just a locked cupboard: everything must stay dry, away from acids, water, and heat. Isocyanates break down and can explode if mixed with the wrong stuff or left in sunlight.
Practical Steps for Risk Reduction
Good training makes the biggest difference. At my current workplace, we added weekly safety talks specifically about dangerous chemicals like this one. People learn from hearing the gritty details of what went wrong elsewhere. Training must cover emergency washes, spill cleanup (never with water, always with dry absorbents), and clear step-by-step guides for what happens if someone feels sick.
Labeling every container stops mistakes before they start. We color-code everything and double-check labels at the end of the day. Disposal takes center stage, too. Waste goes in sealed, dedicated bins, and regular audits mean fewer slip-ups. Nobody trust their memory—everyone uses written checklists.
Fostering Responsibility
Working with hazardous chemicals always comes back to a shared sense of responsibility. I’ve seen how one person’s shortcut risks everyone else’s safety. To me, real safety culture means walking out at the end of a shift knowing the right gear, habits, and vigilance kept everyone healthy. P-nitrophenyl isocyanate doesn’t take a day off, so neither do our precautions.
What is the purity and available packaging size of P-Nitrophenyl Isocyanate?
High Standards of Purity
P-Nitrophenyl isocyanate finds a spot on the benches of chemistry, pharmaceutical, and materials science labs. Every bottle of this stuff spells out the same priority: purity. For most research and synthesis needs, suppliers tend to offer it at a minimum purity of 97%. Some reputable names push that up to 98% or even 99%. This detail isn’t just for show. If you’ve worked through a day’s worth of organic syntheses or spectroscopic analyses, impurities translate to headaches—yield drops, byproducts creep in, reactions stall. Anything less than 97% tends to mess with results and can cost both time and money in lost experiments.
I’ve seen even small impurities throw off a full week’s worth of chromatograms and force a painful round of troubleshooting. Contaminants aren’t just inconvenient—they sometimes change how the isocyanate reacts, especially with amines or alcohols, or else they show up during crucial steps of scale-up work. So, those small numbers next to the label, those 98s and 99s, matter more than they seem from the outside. Companies tend to include a certificate of analysis to back up their claims since nobody wants mystery peaks showing up on an NMR right before a deadline.
Packaging Size: Practical Options
Sourcing chemicals for research often feels like a balance game—big enough for the job, not so big it wastes precious shelf space or budget. P-Nitrophenyl isocyanate usually comes in glass ampoules or bottles, sometimes in small HDPE containers if it’s meant for industry rather than academia. Common sizes include 1 gram, 5 grams, 10 grams, and sometimes larger increments like 25 or 50 grams for bigger labs or industrial needs. For small academic projects, a 1-gram bottle often suffices, especially since this compound reacts well in relatively low concentrations.
Storage comes up a lot with this chemical. The compound reacts easily with moisture, so suppliers choose air-tight and light-protective bottles. This isn’t paranoia but hard-won wisdom; anyone who’s opened a poorly-sealed isocyanate knows the sharp, eye-watering smell that tells you hydrolysis has already started. Good packaging also includes clear hazard labeling. P-Nitrophenyl isocyanate isn’t something you want out in the open, and it has a nasty tendency to irritate skin and lungs. Properly sealed bottles ensure minimal exposure and longer shelf life.
Access and Assurance
Certificates of analysis, real-time inventory checks, and rapid shipping now matter as much as the chemical itself. Labs can’t afford year-long stockouts in 2024, nor do students have the budget for wastage. The reputable suppliers, like Sigma-Aldrich or TCI, keep up standards by offering varying batch sizes and documentation. If a research group needs larger volumes, they arrange bulk discounts and safer packaging to minimize hazards during transport.
Transparency matters more than ever. It’s not just about what’s in the bottle but what backs it up. I remember a time when a delayed certificate forced manual purity validation, taking hours better spent elsewhere. Reliable data and clear communication from suppliers help research teams stay focused on results rather than logistics. That’s real value in a chemical order, whether the bottle contains 1 gram or 50.
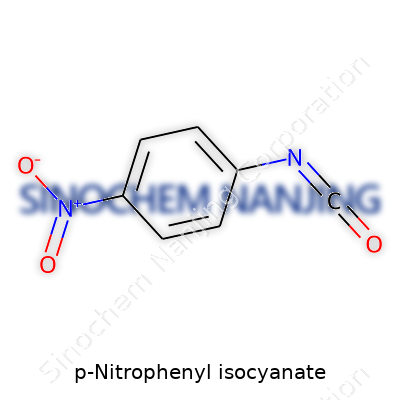
| Names | |
| Preferred IUPAC name | 4-nitrophenyl isocyanate |
| Other names |
PNPIC 4-Nitrophenyl isocyanate p-Nitrophenylisocyanate Isocyanic acid, 4-nitrophenyl ester |
| Pronunciation | /paɪˌnaɪtroʊˈfiːnɪl ˌaɪsoʊsaɪˈəneɪt/ |
| Identifiers | |
| CAS Number | 103-71-9 |
| Beilstein Reference | 1209226 |
| ChEBI | CHEBI:131769 |
| ChEMBL | CHEMBL153602 |
| ChemSpider | 141174 |
| DrugBank | DB08327 |
| ECHA InfoCard | 100.018.535 |
| EC Number | 221-838-2 |
| Gmelin Reference | 79008 |
| KEGG | C18634 |
| MeSH | D009600 |
| PubChem CID | 71153 |
| RTECS number | NL9300000 |
| UNII | 28C1YF460F |
| UN number | UN1851 |
| Properties | |
| Chemical formula | C7H4N2O3 |
| Molar mass | 181.13 g/mol |
| Appearance | Yellow crystals or powder |
| Odor | Aromatic |
| Density | 1.2 g/cm³ |
| Solubility in water | Insoluble |
| log P | 1.9 |
| Vapor pressure | 0.0015 mmHg (25°C) |
| Acidity (pKa) | 12.13 |
| Basicity (pKb) | 11.08 |
| Magnetic susceptibility (χ) | -56.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.554 |
| Viscosity | 1.504 cP (20°C) |
| Dipole moment | 3.67 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 325.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 261.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | Std enthalpy of combustion (ΔcH⦵298) of p-Nitrophenyl Isocyanate: -1841 kJ/mol |
| Pharmacology | |
| ATC code | V03AB37 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H317, H319, H332, H334, H335 |
| Precautionary statements | P261, P264, P271, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P332+P313, P337+P313, P362+P364 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | 95 °C |
| Autoignition temperature | 460°C |
| Lethal dose or concentration | LD50 oral rat 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 640 mg/kg |
| NIOSH | NIOSH: SN 1225000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for P-Nitrophenyl Isocyanate: "0.02 mg/m³ (as isocyanates, OSHA PEL) |
| REL (Recommended) | 0.05 mg/m3 |
| IDLH (Immediate danger) | IDLH: 25 mg/m3 |
| Related compounds | |
| Related compounds |
Phenyl isocyanate m-Nitrophenyl isocyanate o-Nitrophenyl isocyanate p-Nitroaniline p-Nitrophenol |

