P-Menthane Hydroperoxide: An Editorial Look at a Forgotten Powerhouse
Historical Development
Around the middle of the twentieth century, the chemical industry saw a considerable push for new organic peroxides. P-Menthane hydroperoxide rose out of that energy, having roots in both academic curiosity and industrial necessity. Researchers at the time were driven not just by the pursuit of profit, but also by the postwar era’s optimism and a desire to harness chemicals that could push forward polymerization, cure resins faster, and open up new product lines. It’s easy to forget how much patience and grit chemists needed when synthesizing and isolating unstable materials. Each attempt with this compound built up not only a more efficient molecule, but a better collective sense of risk assessment and equipment design. This growth didn’t happen overnight, either—the chemical journey for p-menthane hydroperoxide mirrored the expansion of plastics and advanced manufacturing, with lab-based breakthroughs often racing to keep up with demand.
Product Overview
Many think of p-menthane hydroperoxide simply as another industrial oxidizer or initiator, but that barely scratches the surface. Its value lies in a delicate balance between reactivity and practicality. In everyday practice, it's added as a key ingredient in the polymerization of unsaturated polyester resins and silicone rubber. The material bridges a gap between older, less stable peroxides, and the safer, better-behaved modern versions. What personally catches my attention is its relative stability compared to similar organic peroxides. With careful handling, this compound has served industries for decades, underpinning products as varied as boat hulls and wind turbine blades—each application a reminder of how small tech advances ripple out for years.
Physical & Chemical Properties
To those who have spent time with chemicals in real-world settings, physical cues matter. P-Menthane hydroperoxide usually shows up as a colorless to pale yellow liquid, with a noticeably sharp, potent smell. Anyone working around peroxides knows you respect that odor—it's the chemical's way of warning you not to take liberties with basic safety. In terms of chemical structure, it features a hydroperoxide group attached to a p-menthane skeleton, which gives it both its oxidizing nature and its moderate volatility. The compound can decompose in contact with impurities, heat, or even light, underscoring why storage and transport regulations are strict. In my experience, keeping such substances temperature-controlled and away from contamination is not just a box to check; it’s the kind of discipline that keeps disasters off the record books.
Technical Specifications & Labeling
On the practical side, technical specifications for p-menthane hydroperoxide often focus on concentration, purity, and stabilizer content. What seems like paperwork ends up being a lifeline when things go wrong. Labels don’t just mark the bottle—they communicate shelf life, storage conditions, and emergency procedures. The safety data, the red diamonds, the hazard codes—people in the field treat these not as legal hurdles, but as living documents. My own career taught me that accidents usually trace back to skipped or misunderstood labels rather than exotic chemistry. Nothing instills good habits like seeing the consequences of sloppiness up close.
Preparation Method
Making p-menthane hydroperoxide typically begins with the oxidation of p-menthane, which itself comes from abundant natural sources like turpentine. Industrial settings use controlled reaction conditions with hydrogen peroxide or air and a suitable catalyst. What sounds cut-and-dried on paper often gets much trickier in the plant: Raw material quality, impurities, and stir times all shape the final output. I’ve seen how careful monitoring—flow rates, temperature, pH—can mean the difference between a consistent batch and a costly waste stream. The process also yields side products, making separation and purification a daily challenge and fertile ground for process innovation.
Chemical Reactions & Modifications
Chemically, this peroxide likes to give up its oxygen atom, which is why it's found a niche as an initiator and oxidizing agent. Processes that call for strong, yet not wild, oxidation—such as in controlled polymerizations or epoxidations—showcase its value. Modified versions of p-menthane hydroperoxide allow chemists to tweak performance or safety based on the application's demand. Better stabilizers, optimized purity, and blend formulations all stem from deeper research into its reaction profile. The best minds in the field keep tinkering at the edges, making small tweaks that, over time, add up to safer workplaces and better output.
Synonyms & Product Names
Over the years, this hydroperoxide has collected a laundry list of aliases. It’s not uncommon for the same material to pop up in literature under different names, especially across different regulatory regions. Chemistry textbooks might call it peroxy-p-menthane, while industry catalogs often slice it by its functional group or blend, such as PMHP. This can create confusion for procurement teams and researchers sifting through decades of papers and product bulletins. My tip: Double-check the CAS number and structural formula before making any buy or use decision.
Safety & Operational Standards
Anyone planning to use p-menthane hydroperoxide needs a dogged commitment to safety. Like other organic peroxides, it can cause fire and explosive hazards if mishandled. Experience has taught the industry hard lessons about respect for basic housekeeping and training. Closed containers, inert atmospheres, refrigerant storage, and non-reactive containers are non-negotiable habits. PPE isn’t just a poster on the wall; it’s a shield against burns and toxic fumes. Health agencies and environmental watchdogs continue to tighten standards, often after preventable accidents highlight gaps in protocol. These heightened regulations reflect a shift in culture—one that values human life and environmental safety over cutting corners for speed or cost.
Application Area
P-Menthane hydroperoxide steps up as a workhorse in polymer and resin industries. Its mainstay involves catalysis of polyester resin curing—a process that underpins products from kitchen counters to car parts. Boat fabricators, construction suppliers, and electronics encapsulation outfits all rely on its predictability and performance. The chemical’s ability to drive free-radical polymerization without excessive heat makes it a key player in settings where other initiators present bigger risks. Its role continues to grow as companies look for safer, more sustainable alternatives to older peroxides, often under pressure from toughened worker-safety rules.
Research & Development
Researchers examining p-menthane hydroperoxide often search for ways to boost both productivity and safety. Polymer scientists tinker with formulations to lower risk and improve storage stability. Others hunt for greener synthesis paths with less waste and energy demand, reflecting the ongoing pivot toward sustainability. Toxicologists regularly return to its breakdown products, mapping out potential hazards for workers and the environment. There’s still a long runway for research—especially in understanding subtle reaction pathways, blending techniques, and byproduct management. Each new study, even the dead-ends, matter. That kind of patient, iterative discovery has always driven chemical innovation.
Toxicity Research
Handling p-menthane hydroperoxide requires respect for its toxicological effects. Skin contact leads to irritations or burns, and inhaling the vapor brings respiratory risks. Over years, toxicology studies have mapped dose-response curves and highlighted chronic exposure concerns. The seriousness echoes in the way plants monitor air quality and invest in better ventilation. Environmental impact also raises alarms: improperly disposed or spilled hydroperoxide can harm aquatic life and stress wastewater systems. These realities shift the conversation from quick fixes toward long-term solutions—smarter containment, more robust worker training, and faster leak detection.
Future Prospects
Looking ahead, p-menthane hydroperoxide appears poised to evolve alongside regulatory and environmental trends. There’s growing pressure to phase out peroxides with higher toxicity or instability, pushing innovation across the supply chain. Researchers are already working on safer blends, process intensification, and greener feedstocks. Some envision improved recycling and recovery systems to minimize waste. On a personal level, I see an opportunity for industry and academia to collaborate more closely—not just to meet compliance, but to genuinely minimize risk from the ground up. If the lessons of recent decades stick, the next chapter for this chemical could balance both industrial progress and public well-being.
What is P-Menthane Hydroperoxide used for?
P-Menthane Hydroperoxide in Manufacturing
P-Menthane hydroperoxide shows up often in the industrial world. Factories that make plastic rely on this chemical to start chain reactions that help build polymer structures. In the process of creating plastics like polypropylene and polyethylene, initiators are essential. My visits to production plants revealed how a slight adjustment in initiator concentration shifts the texture and durability of plastic goods we use daily—think bottles, containers, even the lining in our cars.
This chemical has made its way into several leading methods for making foamed plastics and coatings. A production manager once described how it helps meet the deadlines for sports shoe soles and automotive panels. Precision and speed matter on modern assembly lines, especially with rising demand. Supervisors monitor every step, understanding that improper handling or dosing can halt entire lines. Knowing the difference between a properly cured polymer and one that fails under strain keeps everyone on their toes.
Role in Cross-Linking and Vulcanization
In the world of rubber, p-menthane hydroperoxide takes on another role. Rubber isn’t much use straight from the plant since it needs to become stronger and more elastic. This happens by linking the rubber molecules together, a process called vulcanization. Many rubber goods, from gaskets to medical tubing, depend on precise chemical reactions for safe performance in critical environments.
Factories that make technical rubber parts rely on p-menthane hydroperoxide because it enables fast, energy-efficient cross-linking. Some chemists explain that the chemical triggers free radicals, those short-lived, reactive particles that rearrange molecular bonds. The difference shows up in final products that resist heat, stretching, and breakdown. I remember handling rubber O-rings on an assembly line; a poor batch cracks or falls apart after barely any use, causing downtime no car mechanic wants to see.
Risks and Handling Considerations
P-Menthane hydroperoxide isn’t something you leave unguarded on a shelf. Chemical safety officers stress its potential to cause burns or fires if not managed properly. Safe storage practices aren’t just talk; a fire in a storage drum can take down an entire plant. Proper labeling, temperature control, and trained staff matter. Safety data sheets provide the facts, but practical experience counts for more. Stories circulate among plant workers—about near misses, quick reactions, and hard lessons learned from overlooked details.
Despite the risks, the benefits drive adoption. Proper training changes behavior. Regular drills, up-to-date gear, and clear communication between shifts stop mistakes before they start. These lessons stick, shaping a workplace culture where everyone feels responsible.
Environmental Responsibility and Moving Forward
Regulation plays a big part in how manufacturers use chemicals like p-menthane hydroperoxide. Environmental agencies monitor spills and emissions. Workers watch for leaks because small mistakes often have big consequences. Proactive companies invest in containment and process automation, lowering chances for exposure and accidents.
The push for greener chemistry pushes industry leaders to find new ways to work with or replace hazardous components. Advancements in process control and engineering support safer, cleaner manufacturing. Sharing best practices across companies leads to better products and a safer workplace.
Final Thoughts
Understanding p-menthane hydroperoxide isn’t just for chemists. From factory floors to the goods on store shelves, its uses shape more of daily life than most people realize. By following safety standards and embracing innovation, the benefits can reach society without sacrificing health or the environment.
What are the safety precautions when handling P-Menthane Hydroperoxide?
Why Extra Attention Matters
I’ve seen a lot of chemicals pass through warehouse doors. Most don’t command much worry, but P-Menthane Hydroperoxide gets a different set of rules. It acts as a strong oxidizer and packs a real punch in the wrong conditions. Because it likes to decompose fast and can react without much provocation, a careless move can lead to fire, nasty fumes, or even an explosion. In 2021, a laboratory fire in Asia traced its root back to a mislabeled container of this compound—nobody got hurt, but the smoke damage shut work down for months. Stories like this underline why few safety steps get skipped when dealing with this stuff.
Personal Protective Equipment Is Non-Negotiable
You don’t want P-Menthane Hydroperoxide on your skin. Gloves—nitrile or heavy-duty chemical-resistant kinds—go on before the container comes out. Even cotton gloves can sometimes fuel a reaction. Lab coats or aprons keep street clothes safe, and eye protection stays strapped on, always. Splash goggles do the trick better than simple safety glasses. Respirators fit the bill for spot cleanup jobs or work in small rooms. For people with asthma like me, going in without a respirator would be asking for trouble, since fumes from this compound bite right into your lungs.
Storage—A Few Feet Make All the Difference
A bad storage spot can turn a bottle of P-Menthane Hydroperoxide into a disaster waiting to happen. This compound breaks down faster in the heat—my old workplace set chemical refrigerators just for it, never letting it climb above 20°C. Never stack it next to acids, bases, metal powders, or paper. I’ve watched careless stacking send a lower shelf crashing, setting off alarm bells in the whole plant. Only keep what you need in the open—store the rest under lock and key, where curious hands can’t accidentally knock things over or open a cap for a sniff.
Handling Spills and Accidents Face-On
No one plans a spill, but preparing for one saves lives. In my experience, spill kits must include absorbents that won’t start a fire—vermiculite works better than sawdust or cloth. Flood the spill area with water if any splashes threaten clothing or shoes and keep an emergency shower unhindered. Forget mops with wooden handles—the last thing you want is a reaction from an absorbent tool. Never ignore the “buddy system” rule, either. Working alone with reactive compounds just adds risk. In bigger operations, designating a single exit route before opening a bottle of P-Menthane Hydroperoxide never feels excessive.
Disposal—Shortcuts Create Headaches
A lot of folks want to dump leftover hydroperoxides down the drain or toss them with regular lab trash. That’s a recipe for a blocked pipe, corrosion, or an unwelcome call from emergency services. Follow the waste protocol set by local hazardous waste programs. Sometimes, only small amounts get neutralized on-site under supervision. Anything significant waits in a vented steel can, sealed up and labeled, for pickup by certified disposal crews. An ex-coworker tried shortcutting the system once; the corrosion in the pipes shut our facilities down for days.
Training and Mindfulness Keep Troubles Away
No amount of safety gear replaces clear procedures and solid training. Every staff member, from entry-level techs to seasoned supervisors, gets a walkthrough at my facility before they go near hydroperoxides. Labels get checked. Exposure plans get drilled. The key always comes down to attitude—a healthy respect for what the stuff can do, and no urge to cut corners. This compound acts unforgivingly with mistakes. Take the right steps every time, and the risk drops sharply.
How should P-Menthane Hydroperoxide be stored?
Understanding the Risks Involved
P-Menthane hydroperoxide is no harmless chemical. It brings some serious risks to any facility handling it. This isn’t because of some obscure regulatory point or academic concern—it boils down to what this compound can do when left unchecked. Left exposed to heat, friction, or contamination, it cranks up the chances of fire, explosion, and personal injury. History reminds us: similar organic peroxides have caused incidents from minor fires to catastrophic plant losses. Ignoring safe storage can push people and businesses into real danger.
Why Temperature and Ventilation Aren’t Optional
I’ve seen chemical storerooms treated like afterthoughts in older facilities: no working thermometers, ventilation systems on the fritz, barrels stacked near windows. That’s just asking for trouble. P-Menthane hydroperoxide breaks down fast above room temperature, and the products of that breakdown fuel combustion. Smart labs and warehouses keep this one under 30°C and use dedicated chillers if the local climate creeps higher. In an ideal setup, constant airflow yanks away any vapors before they reach a dangerous level. It’s a basic rule: a little investment in decent ventilation and temperature control saves lives and product.
Keep It Separated—No Shortcuts
Stacking different chemicals on the same shelf may save space, but with peroxides like this, that space-saving rarely ends well. P-Menthane hydroperoxide really hates being close to metal ions, acid, catalysts, or organic materials like cardboard and wood dust. Even a forgotten iron nail in the wrong drum can spark decomposition. Segregating storage using separate cabinets, secondary containment, and real inventory tracking lets everyone know what’s stored where. The costs mean little compared to the price of an industrial accident.
Packaging and Spill Preparedness
Poor packaging opens the door for leaks. The right drums—HDPE, lined steel, or specialized containers—do more than tick a regulatory box. They cut risks of accidental mixing and slow down decomposition if temperatures rise. Good facilities keep containers closed tight except during use, label them clearly, and never transfer contents to unlabeled vessels. A leak still happens sometimes, so proper spill kits, neutralizers, and personal protective equipment should always stay close by. I once watched a team stop a one-liter spill from causing a disaster simply because the right absorbents and gear were within arm’s reach.
Regular Checks Are Key
Documentation isn’t just bureaucratic busywork—it’s the habit that keeps track of every container’s age and origin. Workers scan the dates, check for swelling, discoloration, or crusting on the lids, and remove any suspect drums before problems grow bigger. Training new staff to spot trouble is as crucial as installing fire sprinklers. Companies that skip these checks pay later, often in insurance claims or worse.
Getting Safer—A Work in Progress
The science around organic peroxides like P-Menthane hydroperoxide keeps moving forward. Safer stabilizers, better packaging materials, and tougher testing put pressure on companies to ditch outdated habits. Sharing lessons from accidents, updating procedures, and listening when a worker points out a storage problem help prevent tomorrow’s headlines. It isn’t just about keeping regulators off our backs; it’s about preventing harm before it happens.
What are the main hazards of P-Menthane Hydroperoxide?
The Chemical’s Bite
P-Menthane hydroperoxide tends to pop up in a number of industrial labs where it does the job as an initiator or catalyst. But make one mistake with storage, handling, or disposal, and you’re rolling the dice with more than spilled liquid. The big trouble sits in its stubborn reactivity. This chemical has a reputation for catching fire or bursting apart on its own, especially when exposed to heat or shock. For anyone working near the stuff, that’s always in the back of their mind.
Fire and Explosion: Real-World Risks
Physical hazards edge out other risks. This organic peroxide has a short temper. Even minor bumps can trigger a chain reaction you won’t walk away from. People have seen drums rupture after someone left them near a steam vent, and there’s nothing theoretical about a fireball in a closed space. The decomposition releases oxygen, feeding any blaze. Fire departments and EHS professionals all keep their protocols ready for a reason—these accidents tend to get messy.
On the Human Side: Health Hazards
Get this chemical on skin or in your eyes, and you’ll remember its corrosive nature in a hurry. Skin burns aren’t rare, and inhaling vapors can hit your lungs hard. In the worst cases, folks wind up at the hospital with severe irritation or even permanent damage. P-Menthane hydroperoxide acts fast and doesn’t take much to cause a problem. Workers report breathing problems after clean-ups that skipped the right masks. Ignoring the personal protective equipment almost always ends badly.
Storage and Handling: Where Most Go Wrong
Plain, cold words from a manual mean little without backing them up in the real world. I’ve seen untrained staff leave containers too close to sources of heat, or stack them past their safe limit. Leaks often come from old gaskets or rusty caps—stuff that sits low on a checklist until something drips. Storage in well-ventilated spaces, away from direct light, cuts down risk. Separate P-Menthane hydroperoxide from combustibles and acids, and you cut your worry in half.
Waste: The Hidden Trap
Cleanup isn’t just about tossing gloves and wiping down the bench. If you treat this chemical like a typical solvent, you’ll make things worse. I once watched a co-worker pour out a small amount with other organic waste, and we caught the whiff of heat rising right away. Specialized disposal partners know how to neutralize or safely incinerate waste—trying home-brew methods at work has helped nobody over the years.
Tackling Prevention and Solutions
Putting in hazard training pays off more than any printed warning. New staff should run drills with real scenarios using less risky stand-ins before ever handling the real stuff. Smart companies update their safety data sheets and run inspections with fresh eyes every month. Choosing vendors who provide clear batch histories and proper shelf-life info takes out some nasty surprises too.
Investing in the right PPE, customized ventilation, and alarms that alert to heat or pressure changes can save both lives and dollars. Sharing stories and outcomes across the industry helps everyone work smarter. P-Menthane hydroperoxide will keep showing up in production, but the edge can be taken off its hazards with care and hard-earned experience.
What is the shelf life of P-Menthane Hydroperoxide?
Understanding Real-World Shelf Life
P-Menthane hydroperoxide, a versatile organic peroxide, plays a big role in polymer production and other synthetic processes. Anyone who’s worked in an industrial or laboratory setting knows how the vitality of a chemical can shift long before the printed expiry date runs out. The so-called shelf life depends on how the material gets handled, how it’s stored, and even the weather conditions in the warehouse. In the case of p-menthane hydroperoxide, things like storage temperature, container compatibility, and exposure to air really matter. Left at room temperature in a steel container, it can last 6 to 12 months with minimal degradation. Kept cold, away from sunlight and sealed tight, it may last closer to two years.
Why Shelf Life Really Matters
Decomposition can creep up without warning. Anyone who’s pulled a vial from the back of a cabinet recognizes the sour feeling of uncertainty. Peroxides don’t just weaken—they can turn hazardous fast. Over time, hydroperoxides can self-decompose, releasing flammable gases or raising pressure inside containers. I’ve seen what happens after a container has been left too long—the cap may swell, or solid residue builds up around the rim. Those are signs you’re tempting fate.
Chemically, p-menthane hydroperoxide molecules break apart under heat or direct sunlight, speeding up the breakdown. According to highlights from chemical safety boards, improper storage accounts for a vast number of dangerous lab incidents. The Centers for Disease Control offers data backing up this claim: a sizeable percentage of peroxide accidents come from stock kept past its prime or left without periodic testing.
The Importance of Safe Handling and Storage
Safety data sheets from major suppliers stress three clear things: store it cool, keep air out, block the light. Minimize opening the bottle and always seal the container afterward. In shared labs and on industrial sites, label the container, date it, and build rotation into your stock system. Set a review every 6 or 12 months, so people actually check those labels and use up older supply first.
If the container smells odd, looks pale or crusty, or seems pressurized, it’s not wise to open it. Dispose of it safely, following local hazardous waste rules. Many major chemical companies work with return programs. I’ve helped set up collection bins in one production facility so expired or questionable peroxides never get lost in the shuffle.
What Could Help Improve Shelf Life?
Refrigeration can double or triple usable lifespan. So can better container choice—HDPE or glass, not metal. Suppliers could include clear prep dates and explicit expiry guidelines, not just batch numbers. Automated tracking could flag expiring materials by email alert. Regular risk audits, frequent training, and clear policies keep accidents down and reduce waste.
Chemicals like p-menthane hydroperoxide are useful and safe only when they’re fresh and treated with respect. Ignoring shelf life creates unnecessary risks, wastes money, and invites disaster. Treating chemical storage as a living process—one that takes vigilance and organization—makes safer labs, cleaner work, and better end products for everyone.
| Names | |
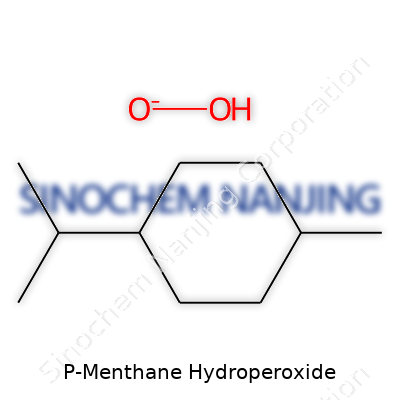
| Preferred IUPAC name | 2-Isopropyl-5-methylcyclohexyl hydroperoxide |
| Other names |
P-Menthane hydroperoxide Menthyl hydroperoxide 2-Isopropyl-5-methylcyclohexyl hydroperoxide p-Mentha-1,4-diene hydroperoxide |
| Pronunciation | /ˈpiː ˈmɛnθeɪn haɪˌdrɒpəˈrɒksaɪd/ |
| Identifiers | |
| CAS Number | T124-12-7 |
| Beilstein Reference | 1341047 |
| ChEBI | CHEBI:135717 |
| ChEMBL | CHEMBL1318092 |
| ChemSpider | 21414 |
| DrugBank | DB11240 |
| ECHA InfoCard | 100.017.836 |
| EC Number | EC 208-731-1 |
| Gmelin Reference | 9333 |
| KEGG | C14302 |
| MeSH | D008440 |
| PubChem CID | 67673 |
| RTECS number | SD8750000 |
| UNII | 16O8X7B71D |
| UN number | UN3115 |
| CompTox Dashboard (EPA) | DTXSID5022220 |
| Properties | |
| Chemical formula | C10H20O2 |
| Molar mass | 152.23 g/mol |
| Appearance | Colorless liquid |
| Odor | slight characteristic odor |
| Density | 1.06 g/cm³ |
| Solubility in water | Soluble in water |
| log P | 2.85 |
| Vapor pressure | <0.05 hPa (20°C) |
| Acidity (pKa) | 11.2 |
| Basicity (pKb) | 10.2 |
| Magnetic susceptibility (χ) | -74.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.453 (lit.) |
| Viscosity | 30 mPa·s |
| Dipole moment | 3.06 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 389.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -251 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6287.7 kJ/mol |
| Pharmacology | |
| ATC code | D01AE23 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS02,GHS05,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H242, H302, H314, H332, H335, H412 |
| Precautionary statements | H242-H302-H314-H332-H335 |
| NFPA 704 (fire diamond) | 3-4-1-OX |
| Flash point | 47 °C (117 °F) |
| Autoignition temperature | Autoignition temperature: 276°C (529°F) |
| Lethal dose or concentration | LD₅₀ (oral, rat): 1,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 495 mg/kg |
| NIOSH | SN3900000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for P-Menthane Hydroperoxide: "No specific OSHA PEL established |
| REL (Recommended) | 400 ppm |
| IDLH (Immediate danger) | 250 ppm |
| Related compounds | |
| Related compounds |
Menthyl hydroperoxide Diisopropylbenzene hydroperoxide Tert-butyl hydroperoxide Cumene hydroperoxide |

