P-Bromophenyl Isocyanate: Perspectives from a Practical Eye
Historical Roots and Shifting Approaches
Traces of p-Bromophenyl Isocyanate begin to show up in the scientific records during an era marked by the rapid growth of organic chemistry. Back in those decades, chemists were hungry for new building blocks, and aromatic isocyanates like this one stood out for their versatility. By the 1960s, the distinct role of brominated aromatic compounds in synthesis and industry had started to gain traction. Over time, as focus shifted toward more advanced pharmaceuticals and specialty coatings, so did the interest in compounds that could unlock new reaction possibilities. Researchers drew inspiration from early discoveries by German chemists who explored isocyanates for medicinal and polymer applications. Today, industrial labs continue to revisit these roots, applying decades of accumulated knowledge about reactivity and hazards.
Product Fundamentals: What Sets p-Bromophenyl Isocyanate Apart
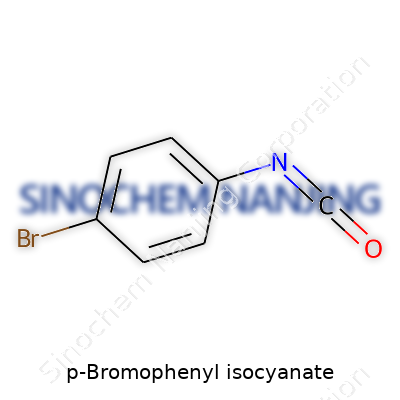
A glance at this compound reveals a white to pale beige crystalline solid. Its molecular formula, C7H4BrNO, tells the story: a phenyl ring topped with bromine at the para position and an isocyanate group tagging along. The Para orientation of bromine doesn’t just affect reactivity—its electronic effects play a big role in reactions and performance in end products. Chemists looking for reliability in chemical modifications have often favored it because of its straightforward substitution pattern. In the practical sense, I’ve noticed suppliers pride themselves on pinpointing low impurity levels, which speaks to the compound’s importance in sensitive syntheses where trace contamination can send a whole batch off course.
Physical and Chemical Properties at the Workbench
This compound doesn’t mess around when it hits the bench. Melting occurs at roughly 62 to 65°C, and volatilization at higher temperatures releases sharp odors typical of isocyanates. Its solubility offers straightforward handling in solvents like dichloromethane, chloroform, and ethyl acetate, but don’t expect miracles in water. This hydrophobicity means you need to keep things dry—moisture easily triggers unwanted reactions, leading to sticky messes and hazardous byproducts. The para-bromine atom not only aids detection via spectroscopic methods but also subtly tunes the reactivity, which helps in fine-tuning polymer and drug precursor synthesis. I’ve seen many R&D chemists in the lab rely on its predictable FTIR and NMR signals for clear batch verification.
Technical Standards and Labeling Practice
In the chemical world, clear labels save lives. Every commercial pack signals danger: corrosive, harmful by inhalation, and sensitizer. The regulatory language from GHS and REACH demands bold pictograms and signal words, a legacy of the product’s reactivity and toxicity. Each certificate of analysis documents moisture, purity (98%+ typical for research grade), residual solvents, plus identification hurdles met by NMR and GC-MS. Labs demand exact CAS number (13903-89-6) documentation for compliance checks. Data sheets outline emergency procedures, yet too often, I see under-trained staff overlook these crucial details in daily operations. Proper labeling and tracking cut risks, especially where sensitive analytical results drive the process.
The Chemistry Behind the Preparation
Synthesis in the lab typically uses p-bromoaniline as a starting point. Reaction with phosgene or its safer substitutes under anhydrous conditions delivers the isocyanate. Tinkering with bases and solvents lets chemists steer yields and minimize the stubborn byproducts that can trip up scaleups. Even though modern labs respect the hazards of phosgene, many still use the classic approach for its efficiency. Green chemistry efforts have begun to probe safer carbonylating agents and continuous-flow reactors to slash waste and improve worker safety. None of these tweaks replace the steady hand and attention needed to control exotherms and handle overpressures. Each batch tells its own story, shaped by operator vigilance.
Chemical Modifications and Reactivity in the Real World
The isocyanate group jumps at nucleophiles. This opens doors for creating ureas, carbamates, and other building blocks, essential from the lab bench to industrial lines. Polyurethanes spring to life when reacting with diols. Pharmaceuticals bank on transformations that introduce tailored side chains or bridge units through the isocyanate group. That para-bromine grabs the spotlight in cross-coupling chemistry: Suzuki, Heck, and Buchwald-Hartwig reactions ride on its presence, expanding access to complex biaryls and heterocycles. I’ve seen process chemists use this reactivity not just for making intermediates, but for innovating in diagnostics—attaching marker groups or designing new probes for biological assays.
Labeling, Synonyms, and Market Names
Ask for “4-Bromophenyl Isocyanate” or "para-Bromophenyl Isocyanate" and you’ll get the same bottle. Trade catalogs might throw in abbreviations like 4-BPI or 4-Br–C6H4–NCO for clarity. Names such as (p-Bromophenyl) isocyanate are common in analytical specs. This specialization reflects the precise needs of researchers trained to differentiate among positional isomers. Synonyms aren’t just for tradition—they save confusion and help in regulations, ensuring imports and handling don’t grind to a halt from a misnamed invoice.
Putting Safety and Operation Front and Center
Handling this compound tests a chemist’s discipline. The isocyanate’s fumes irritate eyes, mucous membranes, and can trigger severe respiratory reactions. Skin exposure brings rashes and, with time, potential sensitization that forces some workers out of the lab for good. Operational best practices mean sealed vessels, good gloves, tight masks, and trained personnel. I’ve watched teams improve outcomes just by revising air handling in small-scale production, moving from open beakers to closed-loop reactors. Spill management has gained more attention: neutralizing agents like ammonia solutions stand at the ready, and waste needs careful segregation to avoid unplanned releases. Real safety comes not from reading the manual, but from keeping routines tight—constant improvements, regular drills, and learning from near-misses.
Where It Counts: Application Areas
Demand isn’t limited to one sphere. Ingredient blending for agricultural chemicals uses p-Bromophenyl Isocyanate to create potent ureas. Polymer industries call for it in designing hard segment chains for specialty polyurethanes. Drug discovery keeps it handy for its double-act as an aromatic and isocyanate handle, making scaffold modifications much smoother. Diagnostics developers have built labeling agents from it, exploiting the bromine’s mass for detection in complex mixtures. Even in materials science, it finds service in functionalizing organic semiconductors or as cross-linkers in coatings engineered for extreme conditions. My experience follows the trail of literature—custom applications spring up wherever chemists need a rugged, reactive aromatic intermediate.
Progress in Research and Development
Interest in derivatives often points to the broader discovery pipeline. Green synthesis projects aim to drop hazardous reagents and find recyclable catalysts. High-throughput platforms screen possible combinations for better performance in catalysis and stability. A lot of teams have tried swapping out phosgene for less toxic carbonyl donors, chasing reduced emissions and tamer reaction profiles. Biologists dig into the potential for modified isocyanates as protease inhibitors or “clickable” probes, transforming the compound from an industrial cog to a biological tool. I’ve noticed that success in these spaces usually comes to groups who understand both the theoretical chemistry and the practical day-to-day juggling of process constraints.
Taking Toxicity Seriously
Toxicologists have chronicled the hazards. This isn’t just another strong-smelling lab reagent—it can push immune systems over the edge, especially with repeated handling. Isocyanate-induced asthma doesn’t announce itself right away; it sneaks up on workers lacking proper PPE or fume controls. Chronic animal studies have underlined risks of organ damage at higher doses, but even short-term exposures command respect. Environmental toxicologists file reports on aquatic effects. Legal limits on air concentrations reflect risks to both direct handlers and bystanders. Younger researchers often underestimate the compound’s hazards—mentorship and real-life incident case studies keep the awareness from dropping.
What’s Next: Future Prospects
Innovation circles back to sustainability and smarter process design. Developers actively search for ways to build in benign-by-design principles—the sort that cut hazard without cutting utility. Novel uses in electronics, such as designer cross-linkers for next-generation displays and wear-resistant materials, look promising. Pharmaceutical groups continue to probe new derivatives for activity, banking on both the bromine and isocyanate groups for molecular recognition. Regulatory agencies clamp down harder on processes with toxic byproducts, spurring development of closed systems and safer alternatives. Investing in AI-driven reaction optimization may soon lighten the load of trial-and-error, letting skilled teams focus on what matters most: delivering results with fewer risks.
What is P-Bromophenyl Isocyanate used for?
The Real Story Behind a Complex Chemical
P-Bromophenyl isocyanate barely registers in everyday talk, but anyone who’s worked in a chemistry lab recognizes the scent—sharp, almost electrifying, carrying memories of late hours mixing solutions and measuring out powders with stubborn static clinging to gloves. This compound, simple-looking as it is, has a way of showing up on important to-do lists for researchers who need to make molecules that won’t come together any other way.
Sparking Connections in Organic Synthesis
In my years at the bench, I’ve seen how isocyanates solve stubborn synthetic puzzles. P-Bromophenyl isocyanate brings together two reactive worlds—an aryl group and the notoriously lively isocyanate functional group. This lets it act as a bridge in building complicated drug molecules or specialty chemicals that don’t just fall off the shelf. Medicinal chemists rely on these bridges. If you want to make ureas, carbamates, or custom-made pesticides, you need a starter like this—not necessarily mass-produced, but reliable for specific reactions.
The bromo part of the structure doesn’t just look good on paper. It lets chemists tweak the reactivity—one small group changes how fast the isocyanate grabs onto amines or alcohols. That fine control helps with selectivity. In real language: you get fewer unwanted byproducts, which saves time and cuts down cleanup.
Research That Needs More Than the Basics
Drug discovery teams sometimes complain about how slow it feels to optimize a “hit”—the molecule that shows some early promise in fighting a disease. Getting the side chains just right can mean the difference between a breakthrough and a dead end. P-Bromophenyl isocyanate gives scientists a tool to tack on chemical groups in ways that influence the way a new drug candidate folds, binds, or holds together in water. Papers published in journals like Journal of Medicinal Chemistry show work where this compound acts as a stepping stone in anti-cancer and anti-inflammatory projects. Patents document it in the recipes for new agrochemicals where designers want targeted weed control with less effect on other plants.
Safety Isn’t Just Another Warning Label
Many years ago, I watched a coworker drop a flask and freeze as a vapor cloud drifted up. Isocyanates, including this one, aren’t “mild”—they sting the nose, can burn the throat, and sometimes trigger nasty allergies. There’s a reason most labs keep the isocyanate stash locked in dry cabinets and restrict access. Before handling it, you want fume hoods on, gloves checked for holes, and no excuses about protective goggles. Chronic exposure can sensitize the lungs—symptoms don’t always show up for months. The stories I hear about missteps have made me cautious and turned me into that person who double-checks the vent is running before opening the vial.
What Could Make the Work Safer and Cleaner?
The biggest headaches come from handling and disposal. Many industrial teams have started looking at safer substitutes or processes that need less of the reactive stuff. Automation helps: remote-controlled samplers and built-in sensors track vapors before they reach the nose. Some companies have poured research money into alternatives, but so far, nothing does quite what p-bromophenyl isocyanate does in the same set of reactions. That probably won’t change overnight, but calling it out is better than pretending substitutes exist for every job.
No lab runs just on textbook formulas. Each new target molecule brings its own curveballs, and a building block like p-bromophenyl isocyanate sticks around because it does things few other chemicals can match. The trick is using it smart, with respect for both its utility and its risks.
What are the safety precautions for handling P-Bromophenyl Isocyanate?
Why P-Bromophenyl Isocyanate Matters in the Lab
P-Bromophenyl isocyanate shows up in many research and manufacturing labs because it helps scientists build creative new molecules and materials. But this is the kind of compound that demands respect. We’re not talking about sugar or salt here. Handling this chemical asks for careful attention to safety because things can get serious if it touches your skin, eyes, or lungs.
Personal Protective Measures Make the Difference
No one wants to deal with chemical burns or breathing trouble from a splash or a whiff. In my years working with reactive chemicals, I’ve learned not to take shortcuts with gloves, goggles, or protective clothing. For this specific chemical, nitrile gloves and a lab coat protect the skin, while splash-proof goggles shield the eyes. For work that might raise dust or vapor, a chemical fume hood goes from optional to mandatory. Never trust regular room ventilation: fumes or dust from p-bromophenyl isocyanate irritate eyes and airways fast, so open benches don’t offer enough protection.
Respiratory protection steps up when large amounts or heated samples enter the picture. Fit-tested respirators with organic vapor cartridges—these protect when a hood isn’t available or if there’s a spill.
Practical Handling at Every Step
The number one rule around p-bromophenyl isocyanate is to keep it contained. Pouring slowly and never working near food or drinks helps avoid accidents. Even a tiny spill can cause a problem, especially if it lands on a surface where someone else might lean their arm or rest a tool. In case of contact, immediate washing with lots of water makes a huge difference. The difference between a light rash and a hospital trip often comes down to seconds, not minutes.
Keep hands away from your face and clean up every surface after working. Double-bagging waste in chemical waste containers, labeling everything, and tracking inventory keep surprises to a minimum. Once, in a shared lab, I saw a bottle completely unlabeled except for a sticky note—bad move. Proper labeling saves you and the people around you from a world of hurt.
Good Storage Habits Matter
Store this chemical in a cool, dry area, away from acids, bases, and moisture. Anyone who has ever seen a reactive chemical react with a leaky pipe or an old shelf knows the mess and risk that can follow. Sealed containers—preferably glass or heavy-duty plastic—stay upright and clearly marked, so coworkers know what’s inside. Being able to spot a hazard at a glance helps prevent accidents.
Spill and Emergency Preparedness
No lab can guarantee zero accidents, so knowing the spill kit’s location and how to use it beats hunting for instructions in a crisis. Absorbent pads, proper neutralizing agents, and lots of water for decontamination support a safer cleanup. Reporting the event right away keeps the group alert, and ensures everyone checks their own health or exposure.
Having an eyewash station and safety shower ready can take a scary moment and make it manageable. A co-worker once tripped while moving a bottle, and the lab’s response—immediate rinse and calling for medical support—kept things from getting much worse.
Training and Teamwork Back Up Lab Safety Culture
Good habits don’t come from reading rules—they stick from doing things right day after day, mentoring each other, and talking openly about close calls. Regular training, making sure new team members buddy up with someone experienced, and real-time reminders go further than just hanging signs on the wall. Respect for p-bromophenyl isocyanate, and for everyone sharing the workspace, builds a culture that looks out for everyone at the bench.
What is the molecular formula of P-Bromophenyl Isocyanate?
Understanding Chemical Identity
Most chemists and students working with organic compounds know that the language of molecules always starts with the formula. For P-Bromophenyl Isocyanate, the molecular formula tells a lot about what you’re dealing with. It reads as C7H4BrNO. Each symbol stands for a specific element: C for carbon, H for hydrogen, Br for bromine, N for nitrogen, and O for oxygen. Reading that, I remember the rush the first time I learned how these building blocks create something much more than a sum of their parts.
Molecular Structure Tells a Story
The formula on its own gives a clear code. P-Bromophenyl Isocyanate has a benzene ring at its backbone. That ring holds a bromine atom in the para (opposite) position against the reactive isocyanate group. The “para” placement influences how the molecule behaves in reactions. For somebody mixing chemicals or planning syntheses, knowing this structure matters as much as the formula itself. Substituting at the para-position can shift a reaction from sluggish to sharp—or make it safer or riskier to handle. Researchers count on consistency, and knowing the position of each atom delivers that predictability in the lab.
Every Atom Has a Job
The isocyanate group (–N=C=O) on this molecule grabs attention. Isocyanates pop up across research papers and patents because they’ve got a knack for reacting with amines, alcohols, and water. That versatility leads to a mix of benefits as well as risks. Anyone who’s ever opened a bottle of isocyanate knows the importance of gloves and a fume hood—they can snap into reactions with just a trace of moisture in the air. It isn’t just about making plastics and drugs. It’s about handling something that wants to change and connect, sometimes suddenly.
Why This Matters Beyond the Lab
Every detail—like that bromine attached to the benzene ring—shapes the properties that matter in manufacturing and drug development. With bromine, the compound often takes on extra heft and reactivity. Companies use derivatives like this for making specialty chemicals, certain pharmaceuticals, or advanced materials. If the formula changed by just one atom, it could set off a ripple across safety protocols, environmental impacts, and even pricing for customers downstream.
Fact-Based Awareness Makes a Difference
Accurate chemical knowledge keeps labs and workers safer. Just knowing C7H4BrNO tells a chemist they’re up against a molecule weighing about 198 grams per mole. That number makes a difference in calculating reaction yields or estimating how to store or ship the compound. The bromine atom, with its unique mass and reactivity, guides environmental planning too. Regulators in the EU or US look for formulas just like this to flag chemicals for monitoring or restriction. Ignoring a formula can mean missing a hazard or missing out on a chance to innovate responsibly.
Solutions Built on Trust and Precision
Chemical transparency begins with a correct formula. Courses and safety trainings should show students how to interpret and respect these codes. For businesses, open supply chains benefit from clear labeling to reduce mix-ups and legal risks. In practice, scanning a package for “C7H4BrNO” speeds up communication between labs and helps track what flows through a facility. That sort of clarity saves lives and keeps the science honest.
How should P-Bromophenyl Isocyanate be stored?
Understanding P-Bromophenyl Isocyanate
Chemicals like P-Bromophenyl Isocyanate usually come with long safety sheets, but the real concern comes down to everyday storage. This isn’t just about legal requirements — it’s about keeping people safe and property intact. In laboratories and chemical plants, ignoring basic practices turns small mistakes into serious incidents. As someone who’s spent time around chemicals, I’ve seen how the little details matter the most.
Why Proper Storage Really Matters
Exposure to isocyanates carries health risks. Workers can develop skin irritation, respiratory issues, or worse. I’ve watched new hires breeze past PPE out of habit — until a close call forces them to rethink. Storing hazardous chemicals properly doesn’t just protect individuals, though; it keeps business operations running smoothly and reputations intact.
Many isocyanate accidents come from improper storage conditions. So, what’s the root cause? It usually starts with poor labeling or containers placed somewhere they shouldn’t be. In labs I’ve worked, a simple oversight once left a highly reactive compound too close to an incompatible material. Quick intervention averted disaster, but not everyone gets that lucky.
Key Storage Practices for P-Bromophenyl Isocyanate
Sturdy, chemical-resistant containers with tight seals form the backbone of safe storage. These limit moisture entry and lower the risk of vapors escaping. P-Bromophenyl Isocyanate reacts with water or alcohols, producing carbon dioxide and heat. That’s not something you want happening on a shelf or workspace. I always reinforce the importance of double-checking lids right after use, never leaving things to chance.
Temperature makes a difference. Cool, dry rooms away from direct sunlight help keep the compound stable. I’ve seen storage rooms overheat in summer, making accidents much more likely. Ventilation cuts down on fume buildup, giving an added layer of safety. Facilities with modern fume hoods keep the air clear, but even basic exhaust fans cut down on low-level exposures.
Segregation and Documentation
People new to the field sometimes treat chemical storage like kitchen cupboards, tossing bottles wherever space exists. This mindset risks dangerous reactions. Isocyanates should always stay away from acids, bases, and oxidizing agents — separate shelves, separate spill containment. Clear labeling prevents confusion. Hard-to-read handwriting or missing labels almost always spark emergencies. I stress using bold, water-resistant markers so there’s no room for doubt.
Keeping up-to-date records of purchases, usage, and disposal makes tracking easier. Digital logs help, especially in busy labs where dozens of samples pass hands daily. Reviewing stock and expiry dates during routine checks reduces surprises. One place I worked rotated stock every quarter. We caught old containers before they became liabilities.
Emergency Readiness and Team Training
Spill kits with absorbent material, gloves, and goggles should sit close. Success in crisis situations depends on preparation before anything spills. Training drills, even short ones, help teams move quickly and correctly. Watching a team respond confidently during live drills gives reassurance. Quick, calm action means small leaks stay small — this mindset can’t come just from safety posters.
Good storage practices for chemicals like P-Bromophenyl Isocyanate start with respect for the risk. Small habits, from checking lids to keeping logs, protect health and keep doors open for business as usual. These details transform a hazardous material into something that can be managed safely, day in and day out.
What is the purity level of the P-Bromophenyl Isocyanate product?
Why Purity Isn’t Just a Technical Detail
In the chemical world, purity isn’t just some fancy number on a certificate. Take p-bromophenyl isocyanate as an example. A lab technician can spot a difference in yields, a safety officer catches the fumes from an unexpected by-product, and a chemist feels the sting when the data sheet doesn’t match reality. That purity percentage tells you about risk, consistency, and trust—all rolled into a clean line on a report.
What Good Purity Looks Like
With p-bromophenyl isocyanate, manufacturers often claim a product reaches a purity of 98% or more. Reputable supplies list this purity, confirmed by chromatographic analysis, and make sure impurities such as residual solvents or by-products are down at fractions of a percent. A reputable chemical company usually supports these figures with a certificate of analysis, using techniques like HPLC or NMR. End users trust these reports because any slip can show up as a toxic gas, weird solidification, yellow-brown tinge, or lower yields.
Risks of Cutting Corners
Low-grade materials invite trouble. Imagine handling a batch with only 93% purity. You breathe a bit easier knowing how nasty aromatic isocyanates get with the wrong contaminants. I’ve listened to chemists grit their teeth after reactions fail for reasons that only make sense after a full impurity profile. Solvents left behind, byproducts clinging to the main compound, or a touch of moisture—they all can pull a reaction off track or introduce hazards you didn’t sign up for. Even slight differences may create volatile situations. No one likes tracking down a mysterious fume hood leak.
Purity’s Impact on Results
For pharmaceutical work, synthesis, or making specialty polymers, minor drops from 98% to 96% can force a process change or spark a chain of paperwork. Certain syntheses use every part of the starting material—any impurity isn’t just waste, it's a potential contaminant in the next product. For researchers, that means reviewing safety protocols, adding filtration steps, or just trashing impure batches. I’ve had whole afternoons spent justifying unexpected results, only to trace things back to a small shift in raw material quality.
How to Get Reliable Purity
Finding reliable p-bromophenyl isocyanate starts with sourcing. Pick suppliers who document every step and don’t dodge questions about analysis or stability. Good labs run spot checks and keep records up to date. Analytical methods like HPLC, FTIR, and mass spectrometry let you pinpoint what’s actually there—not just what a data sheet promises. In one project, after getting a supposedly “pure” shipment, our team checked in-house and found enough impurity to spark a round of refund requests. Ever since, double-checking everything before committing to a big run feels less like red tape and more like common sense.
Making Quality a Habit
In most research and industrial labs, purity never takes a back seat. If a supplier offers a higher margin but skirts analytical detail, that’s a warning sign. It’s always smarter to ask for profiles, challenge numbers, and even audit a batch now and then. That careful approach makes the difference between planned progress and surprise troubleshooting. In my experience, a few extra minutes of due diligence beat hours fixing mistakes from an unexpected impurity, especially with sensitive reagents like p-bromophenyl isocyanate.
Building on Experience
Each time a team checks and confirms high purity, confidence grows. Factoring in real-world hiccups—supply chain delays, unexpected process quirks, new regulations—it becomes clear that demanding quality from the start prevents headaches. Purity isn’t a luxury; it’s a foundation for safe, successful work in any lab using this kind of material.
| Names | |
| Preferred IUPAC name | 1-Bromo-4-isocyanatobenzene |
| Other names |
4-Bromophenyl isocyanate Isocyanic acid, 4-bromophenyl ester p-Bromophenylisocyanate |
| Pronunciation | /ˌpiːˌbrəʊməˈfiːnəl aɪˌsəʊˈsaɪəneɪt/ |
| Identifiers | |
| CAS Number | 586-97-8 |
| Beilstein Reference | 1369074 |
| ChEBI | CHEBI:34209 |
| ChEMBL | CHEMBL472895 |
| ChemSpider | 80824 |
| DrugBank | DB08634 |
| ECHA InfoCard | 100.018.620 |
| EC Number | 202-451-8 |
| Gmelin Reference | 86938 |
| KEGG | C19160 |
| MeSH | D016665 |
| PubChem CID | 69110 |
| RTECS number | GV8575000 |
| UNII | 7L4I7E7047 |
| UN number | UN2206 |
| Properties | |
| Chemical formula | C7H4BrNO |
| Molar mass | 256.04 g/mol |
| Appearance | White to pale yellow crystalline powder |
| Odor | Pungent |
| Density | 1.7 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.9 |
| Vapor pressure | 0.001 mmHg (25°C) |
| Acidity (pKa) | 6.51 |
| Basicity (pKb) | 11.01 |
| Magnetic susceptibility (χ) | -72.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.614 |
| Viscosity | 20 cP (20°C) |
| Dipole moment | 1.5681 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 333.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -54.0 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -380.4 kJ/mol |
| Pharmacology | |
| ATC code | Not assigned |
| Hazards | |
| Main hazards | Toxic if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02, GHS06, GHS05 |
| Signal word | Danger |
| Hazard statements | H302, H315, H317, H319, H332, H334, H335, H410 |
| Precautionary statements | H315, H317, H319, H334, H335 |
| NFPA 704 (fire diamond) | 2-2-1 |
| Flash point | 102°C |
| Autoignition temperature | 260 °C (500 °F) |
| Lethal dose or concentration | LD₅₀ Oral Rat 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): 6400 mg/kg (oral, rat) |
| NIOSH | NIOSH: ST1350000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.2 mg/m3 |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Phenyl isocyanate P-Chlorophenyl isocyanate P-Fluorophenyl isocyanate P-Methylphenyl isocyanate O-Bromophenyl isocyanate |

