Oxalyl Chloride: A Deep Dive
Historical Development
Oxalyl chloride rolled onto the scene just as the chemical industry began to shift into high gear in the 19th century. German chemists saw its potential and mapped out its structure with curious eyes. By the mid-1800s, in dusty labs filled with the tang of acid and hope, researchers first isolated oxalyl chloride through reactions with phosphorus pentachloride and oxalic acid. As science marched forward, demand for new chemical building blocks grew. By the 20th century, oxalyl chloride had stepped up, proving essential for pharmaceuticals, agrochemicals, dyestuffs, and advanced materials—a true workhorse for modern synthesis. The way companies scaled up production—managing those sharp fumes and corrosion—opened doors for new handling equipment, boosting safety standards that shaped the industry.
Product Overview
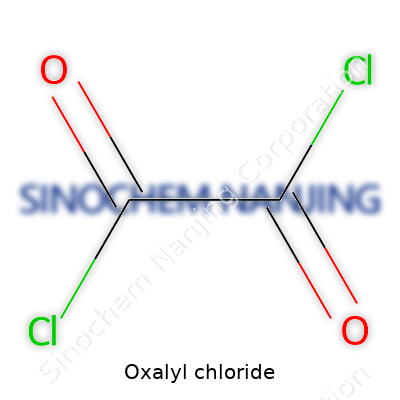
C2O2Cl2—those six atoms pack a serious punch. Oxalyl chloride isn’t just a colorless liquid with a sharp, choking smell; it’s a workhorse for chlorination, dehydration, and as a coupling agent. Those who spend time in research labs or production plants learn quickly that it's one of the go-to reagents whenever there's a need for acylation, especially with amines and alcohols. The demand for it keeps climbing, fueled by pharmaceutical pipelines and the creation of specialty plastics. Handling it isn’t for the faint of heart; reliable personal protective equipment stands between you and a noseful of acid vapors, and facilities need specialized ventilation to manage its aggressive reactivity.
Physical & Chemical Properties
What makes oxalyl chloride tricky boils down to the properties at room temperature. With a boiling point just under 64°C, it evaporates faster than you expect, yet solidifies below –10°C for anyone working in colder spaces. Its density hovers around 1.48 g/cm3. Solubility lies low—water sends it fizzing into a cloud of hydrochloric acid and carbon monoxide. Put it near alcohol or amine, and it fires off an exothermic reaction. Because it's such a strong electrophile, the odor sticks around long after you’ve capped the flask. Safety data sheets are filled for a reason—respirators, gloves, and goggles aren’t just a suggestion in any serious syntheses. It corrodes most metals quickly, so containers of glass or special high-grade plastics are the only thing standing between you and a nasty spill.
Technical Specifications & Labeling
Every drum or ampule carries its markings: C2O2Cl2 on one side, packed with handling warnings, UN numbers, batch receipts, and hazard pictograms. Makers keep to purity standards—99% and above for fine chemical work. Residual acidity, iron content, and water content sit within tight limits, so each lot passes a battery of tests—titration, Karl Fischer, colorimetric methods. Flammable liquid, toxic by inhalation, corrosive—these aren’t just on paper; they drive the way warehouses store and labs transport it. Manufacturers support end-users with detailed certificates of analysis, and global shipments have to clear regulatory hurdles in every country, so supply chains keep careful logs and documentation.
Preparation Method
Every chemist learns the old school way: take anhydrous oxalic acid and hit it with thionyl chloride or phosphorus pentachloride under dry, controlled conditions. The fumes chase you out if ventilation slips. Alternate routes use phosgene, though folks shy away from that for obvious safety reasons. Modern plants run closed systems with temperature control, scrubbing exhaust with alkaline traps, and recycle spent acid for environmental compliance. Scale-up means more automation—metering, mixing, and distilling, all with backup plans for leaks or overpressure. Careful monitoring at each stage ensures clean conversion without unwanted side products, which pays off in high yields and cleaner product for downstream chemists.
Chemical Reactions & Modifications
Anyone tasked with turning out pharmaceuticals or custom intermediates reaches for oxalyl chloride to transform carboxylic acids into acyl chlorides. The reaction water jets forth as gas, making for some lively flasks and plenty of ventilation. Drop oxalyl chloride into an alcohol solution, and esters spring up—a win for polymer precursors. In peptide chemistry, it links amino acids with surgical precision. Friedel–Crafts acylation, dehydration of amides, even the Swern oxidation for turning alcohols into aldehydes—all depend on this liquid’s punch. Even outside the lab, you can see the impact of its reactivity in specialty coatings and high-performance plastics, where durable bonds matter more than ever.
Synonyms & Product Names
Industry insiders know oxalyl chloride by many faces: ethanedioyl dichloride, oxalic acid dichloride, and ethane-1,2-diyl dichloride top the list. Old catalogs and modern SDS sheets might call it Perkloryl or Perchloroxalic anhydride. International shipments move under UN number 3290. Specialty suppliers list in English, German, Chinese, and Japanese, each with its preferred spelling but always the same two-carbon backbone with four reactive ends. This makes ordering consistent, despite language barriers, so no cross-ups land the wrong bottle in a research program.
Safety & Operational Standards
Anyone who works with oxalyl chloride learns respect fast. Splash suits, full-face respirators, and gloves—no exceptions. Air monitoring for hydrochloric acid and carbon monoxide levels backs up exhaust fans, with emergency showers on standby. Start-up and shutdown procedures are drilled until routine, and transfer lines get checked for every batch. Storage tanks sit tucked away, vented to scrubbers, remote-operated to keep people safe. Even the waste drums need special labels, since leftover oxalyl chloride reacts with stray water, raising the risk of pressurization and release of toxic vapors. Regular safety drills and clear signage protect not just the chemists but anyone in the building. Vendors supply printed manuals for a reason—more than one accident has been traced to a missing page or ignored checklist.
Application Area
Day-to-day, oxalyl chloride reshapes industries ranging from medicine to polymers. Drug makers depend on it for forming active ingredients in antibiotics, antivirals, and cancer therapies. In the agrochemical sector, it drives the synthesis of herbicide and pesticide backbones—critical for feeding the world. High-performance plastics—like polycarbonates and polyimides—rely on acyl chlorides for chain extension and crosslinking, credits oxalyl chloride with making those materials tough and thermally stable. Electronics manufacturers dip into the same chemical for etching and specialty resins, while dyes and pigments get brighter and longer-lasting thanks to clean, efficient acylation chemistry.
Research & Development
In research, the hunger to push oxalyl chloride’s limits never stops. Medicinal chemists chase new drugs with purer, more targeted structures, relying on acyl chlorides for building blocks. Polymer scientists play with chain length, branching, and crosslinking, using every tool available to tune materials for aerospace, automotive, or wearables. Environmental chemists seek better ways to contain, neutralize, or recycle the byproducts, hoping to make synthesis both efficient and gentle on the planet. Startups explore microreactor systems, reducing scale to grams and taking pressure off big plants, while developing sensors and controls to keep processes lean and safe.
Toxicity Research
Anyone unlucky enough to get a lungful of oxalyl chloride remembers it—coughing, difficulty breathing, burning that lingers. Acute inhalation leads to pulmonary edema, so oxygen monitoring and onsite medical response are musts. Chronic exposure raises questions about long-term lung, eye, and skin effects. Animal studies show dose-dependent damage to airway tissues and suggest that low-level exposure over time can impact organ function, possibly linking to increased cancer risk. Toxicologists track exposure times, dose limits, and breakdown pathways in the body, hoping to protect both plant workers and local residents from unexpected releases. Regulatory agencies update permissible exposure limits as new data rolls in.
Future Prospects
Innovation doesn’t let up. Greener chemistry directions keep researchers busy, looking for replacement reagents and closed-loop processes to reduce emissions and waste from oxalyl chloride production. Advances in personal protection and automation improve workplace safety, while new synthetic protocols cut down side-products and energy use. The push for sustainable sourcing means feedstocks and manufacturing pathways need tightening, with digital twins of plants predicting leaks, inefficiencies, and exposure hotspots. Pharmaceutical and materials science doesn’t show any signs of dropping oxalyl chloride for key routes, though and there’s fresh demand for even more precise chemistry, using machine learning to map out optimal reaction pathways. Health and safety regulations evolve, raising both the bar and the cost, yet every breakthrough in process intensification or substitution brings benefits measured in cleaner air, lower risk, and stronger products.
What is oxalyl chloride used for?
Chemical Power at Work
Oxalyl chloride may not be in every household, but its fingerprints are on quite a few things that matter, especially for people who have ever had a medicine prescribed or have marveled at modern plastics. As a chemical reagent, oxalyl chloride owns a seat in the front row of organic chemistry labs and factories. People who make pharmaceuticals value it for how easily it helps build the building blocks of many drugs. In fact, it often serves as a core ingredient to activate carboxylic acids, allowing chemists to “stitch” together molecules in ways water just can’t compete with.
From Lab Bench to Pharmacy Shelf
Ask any synthetic chemist about painkillers, antibiotics, or even cancer drugs, and many will mention oxalyl chloride without skipping a beat. Its knack for producing acid chlorides fuels the creation of not just simple medicines, but complex compounds that actually fight tough diseases. For example, manufacturing pharmaceuticals often involves protecting and modifying delicate groups within big molecules. Oxalyl chloride handles these tasks with precision. Its reaction releases gases like carbon monoxide and carbon dioxide, but doesn’t dump water into the mix, which makes a big difference for reactions that would otherwise grind to a halt in a wet environment.
Polymers, Dyes, and More
I have met folks working in plastics and dye plants who have stories that start with oxalyl chloride. Its value spikes big time when producing high-performance polymers such as polycarbonates and polyether ketones, prized for strength and clarity in everything from optical lenses to aerospace parts. In industrial dye manufacture, it acts as a reliable partner for making colorants more vibrant and stable. Without it, industries would need to lean on slower, messier chemicals that waste more energy and time.
Risks Behind the Curtain
Of course, its story is not all bright lights and smooth solutions. Even the folks I know who swear by it keep their masks close and their ventilation fans on. Oxalyl chloride releases fumes—think choking, pungent clouds of hydrogen chloride gas—so safety culture is essential. In the hands of pros, it transforms lives by powering new discoveries. In careless or inexperienced hands, it turns hazardous fast. Factories worldwide invest heavily in sealed equipment, air scrubbers, and staff training not only to protect workers but also to keep the chemical out of waterways and air.
Some Roads Forward
I have seen researchers push for alternatives that keep efficiency alive while reducing health risks. Some labs now use flow reactors that tame these harsh reagents, cutting down exposure. Regulatory agencies encourage real-time monitoring and better containment. At the same time, teaching new chemists about respect and caution maintains a steady supply of skilled hands. Sustainable chemistry trends also push for greener substitutes, even though matching oxalyl chloride’s performance proves tough. Science leans on innovation, and trained people—working with facts, deep understanding, and practical safety measures—keep progress on track.
What is the chemical formula of oxalyl chloride?
The Formula and Its Meaning
Anyone with a background in chemistry usually learns chemical formulas by rote, but the story behind them opens up plenty more. Oxalyl chloride carries the formula C2O2Cl2. Just by looking at it, you can spot two carbon atoms, two oxygens, and a pair of chlorines attached. Sure, those letters and numbers paint a basic structure, but the real impact of this compound steps in during actual chemical work.
Why Oxalyl Chloride Stands Out in Labs
In the world of organic synthesis, oxalyl chloride gets as close to “essential” as a reagent can be. I’ve seen it used countless times to convert carboxylic acids into acyl chlorides—an indispensable step. This step opens doors for making pharmaceuticals, dyes, and even specialty polymers. Speed and reliability of this transformation help keep projects on track.
Its significance grows once you realize what goes wrong without it. Making acid chlorides without oxalyl chloride often means switching to less efficient, messier alternatives. Oxalyl chloride keeps the lab work flowing, so chemists don’t spend hours dealing with stubborn byproducts.
Why Safety Matters So Much
Working with oxalyl chloride comes with a sharp learning curve. It reacts viciously with water, tossing out dangerous fumes of hydrogen chloride and carbon monoxide. In my own experience, I’ve had to double-check the airflow and protective gear every single time before opening a bottle. There isn’t room for shortcuts—one careless whiff and your throat seizes up. That memory sticks.
Good training in handling these reagents lays the foundation for lasting safety habits. A strong exhaust system, a tidy workspace, and fast access to safety showers or eyewash stations matter far more than any expensive glassware. I recall the peace of mind in my grad school lab only after we replaced a dodgy old fume hood with a new high-powered one. Accidents dropped sharply. Sharing tips about these practical changes often made bigger differences than lectures.
Oxalyl Chloride and Green Chemistry
Concerns about health and the environment have pushed researchers to hunt for safer substitutes. Newer protocols focus on minimizing waste and cutting toxic byproducts, but so far, oxalyl chloride’s efficiency remains hard to match in many reactions. The trick lies in smart lab management—recycling solvents, keeping reaction scales minimal, and constantly looking for redesigns that shave down usage. Initiatives encouraging greener practices aren’t about blame but collaboration, where sharing practical ideas makes the science better for everyone.
The Bigger Picture: Responsibility in Chemistry
For researchers and workers in chemical industries, knowing that C2O2Cl2 isn’t just a string of atoms, but a potent tool with risks, makes all the difference. Every decision, from measuring out a fresh batch to safe disposal, becomes a reflection of care for both health and progress. Taking oxalyl chloride seriously means working smarter, staying alert, and searching for methods that respect both the molecule and the people handling it.
How should oxalyl chloride be stored safely?
Understanding What Makes Oxalyl Chloride Tricky
Anyone who’s worked in a synthetic chemistry lab recognizes oxalyl chloride right away—pungent smell, steely warnings on every bottle. A potent chlorinating agent, it reacts violently if you splash a drop of water into it. Vapor exposure tears up your eyes and burns your lungs. The danger isn’t just theoretical, either. One spill near a faucet or a cracked cap in the wrong drawer can start a real emergency.
Dry, Cool, Secure: The Essentials
Old habits in storage—sliding hazardous liquids into any open cabinet or choosing the nearest shelf—don’t cut it with oxalyl chloride. Dry environments make a noticeable difference, since moisture triggers a sharp reaction. The right approach calls for a desiccated chemical storage cabinet or a fume hood with limited airflow, away from sinks or active plumbing. This isn’t just about following a rulebook; it’s about stopping a potential release of choking fumes before it starts.
Chilled storage slows down deterioration. Labs that can afford specialty refrigerators set aside a section, away from anything aqueous or flammable. Not every setup matches this ideal. If cooled cabinets aren’t available, room temperature suffices as long as there's no risk of temperature spikes or direct sunlight. In southern climates, rooms lacking proper ventilation compound the hazard.
The Importance of Right Containers
Every experienced lab tech checks packaging before putting oxalyl chloride away. Factory-sealed amber glass does a good job. Never transfer into unlabelled containers or use plastic, which can corrode or degrade. Tight-sealing caps—free of cracks or residue—keep vapors locked inside and water out. Once, during a marathon week of syntheses, I saw a forgotten bottle with residue around the neck cause headaches for everyone in a shared lab. After that, I started dating every bottle and double-checking seals personally.
Separation Prevents Catastrophe
Oxalyl chloride turns disaster-prone if stored with common chemicals like alcohols, amines, or even simple cleaning agents. Mixing with bases or oxidizers isn’t just risky—it produces toxic fumes in seconds. Separate storage keeps these dangers at bay. A locking storage cabinet limits unwelcome access. Posting clear hazard labels and laying out index cards with emergency contacts cuts confusion during a spill.
Training Matters More Than Checklists
Safety rules only work if people on the ground know the risks and the reasons behind every step. Training everyone from the graduate students to seasoned researchers builds real muscle memory: not just where to store oxalyl chloride, but how fast to act if something spills. Nobody forgets the sting of phosgene odor, which can form if the bottle isn’t sealed right. Regular drills, walkthroughs, and spot checks make it a team habit.
Solutions for Resource-Limited Labs
Not every university or startup lab has the luxury of high-end safety cabinets, but practical steps tighten up safety. Use silica gel packs to pull moisture from storage spaces. Place secondary containment trays beneath bottles to catch leaks. Rotate stock, use oldest bottles first, and get rid of degraded material using a certified hazardous waste contractor. Encourage an open-door policy for safety concerns—if someone spots condensation on a bottle, treating it as an immediate priority averts disaster.
Expert Recommendations, Real-World Protection
Every recent accident report points to lapses in discipline or short-cutting safety routines. Laboratories that make a habit of inspecting containers, separating incompatible chemicals, and prioritizing training show far fewer incidents. Consulting materials safety data sheets (MSDS) with every new order and adopting procedures recommended by organizations like OSHA makes the work not only compliant but a safer environment for everyone in the lab.
What safety precautions should be taken when handling oxalyl chloride?
A Glimpse into the Risks
Oxalyl chloride isn’t the sort of chemical to treat lightly. Years in research settings introduced me to its sharp, stinging scent—the sign to step back and double-check every step. It reacts fast and violently with water, sending out hazardous fumes. Think about hydrochloric acid gas and corrosive smoke filling the air. Those risks aren’t theoretical; one slip can mean a real emergency.
The Reality of Personal Protection
Lab coats and safety goggles set the baseline. They’re not just for show. Oxalyl chloride can burn skin and throw off fumes that hurt eyes and lungs. Nitrile gloves form a steady shield, as this chemical passes straight through latex. Splash-proof goggles and a full-face shield are worth it. On more than one occasion, goggles have saved me from a faceful of trouble during simple transfers or extractions.
Workplace Ventilation: No Shortcuts
Any use of oxalyl chloride must happen in a working, certified fume hood. Open lab benches invite disaster. A good fume hood protects everyone in the room, not just the person running the reaction. You’ll see signs of poor practice right away—persistent odors, irritated throats, and unusual corrosion on metals. Those red flags never end well, so hoods and exhaust fans demand regular inspection and maintenance. Ignoring them makes the entire workplace unsafe.
Storage and Handling—Never an Afterthought
Oxalyl chloride likes dry, cool spots away from direct sunlight. Moist air breaks it down, forming corrosive byproducts and pressure inside bottles. In labs, brown glass containers seal out light and keep the compound stable longer. Forgetting to tighten the cap has ruined work and equipment overnight. Always keep it away from water sources; just a single drop can trigger fumes.
Accident Prevention Begins with Training
People unfamiliar with oxalyl chloride often underestimate the risk. Proper training covers much more than reading a safety data sheet. Regular drills, up-to-date spill kits, and seasoned mentors change how newcomers approach dangerous chemicals. Only by witnessing a real-life spill do most folks realize how important preparedness feels—scrambling for a respirator while the air grows sharp and thick isn’t a lesson you want.
First Aid Response Saves the Day
Spills on skin demand running water, not just a quick rinse. After flushing for at least fifteen minutes, medical attention comes next, regardless of visible injury. For inhalation, getting to fresh air remains the top priority. Rescue attempts can quickly expose others, so communication and clear emergency plans help keep everyone safe. Workers know which doors lead quickest outside; practicing escape routes isn’t just regulation—it shapes muscle memory in the panic of the moment.
Better Solutions in Design and Practice
Engineering controls keep danger in check. Closed-system transfers, sealed fittings, and splash guards all cut risk. Automation now lets labs handle transfers without ever opening a container to air. For smaller operations, step-by-step protocols help stop shortcuts—clear instructions taped beside the fume hood have stopped more than one mistake in its tracks. Investing in backup supplies of gloves, masks, and eyewash pays for itself ten times over the first time they’re needed.
Building a Culture of Respect
Safety relies on people, not just gear. Facing up to the hazards of oxalyl chloride means open conversations about near-misses, constant checks on safety gear, and leadership that never tolerates “just this once.” Only through experience do these practices settle in for good. Sharing stories, reviewing incidents, and checking each other’s protocol help everyone walk out of the lab healthy at the end of the day.
What are the hazards associated with oxalyl chloride exposure?
Why Oxalyl Chloride Matters in Real Life
Oxalyl chloride often turns up in chemical labs, especially when people produce medicines, plant protection products, and various polymers. It looks like a watery liquid, but don’t let that fool you. My first time handling a bottle of this chemical stayed in my memory for one reason: the unmistakable, irritating vapor that hit my face even before I uncapped it. It becomes very clear how serious things could get with poor precautions.
Health Risks You Can’t Ignore
Breathing in the fumes is where the biggest risk starts. The vapor is sharp and strong—almost like standing near hydrochloric acid, except the stinging goes deeper. Within moments, anyone in the room feels it in their nose, throat, and lungs. I read a study where even low doses caused coughing and wheezing almost immediately. In some cases, lab workers needed hospital care for breathing trouble. Over time, people exposed without tight controls risk asthma-like symptoms and more chronic breathing issues.
Splashes on skin or into eyes lead to instant pain. One colleague got a tiny droplet on his glove; the exothermic reaction burned right through the material, raising a painful blister. Contact with eyes can threaten vision. The chemical’s reactivity doesn’t only destroy organic samples—it eats into living tissue just as fast.
Chemical Behavior in Real Settings
Most chemical professionals know that oxalyl chloride reacts fast with water or even just moist air. That reaction produces clouds of hydrogen chloride and carbon monoxide. These fumes don’t just make breathing tough; they carry real emergency room stories with them. In poorly ventilated labs, a small spill or accidental opening of a container can force an evacuation. I’ve talked with engineers who recalled sensors tripping alarms and everyone scrambling to fresh air.
This chemical leaves its mark on metal, plastics, and everyday work clothes. Over time, even trace spills corrode metal workbenches, making equipment replacement a cost nobody wants to explain to their boss. Chemical-resistant gloves, face shields, and proper fume hoods aren’t just box-ticking; they save skin, lungs, and paychecks.
The Route Toward Safety
Some folks argue that good lab techniques alone keep risks down, but my experience proves layered safety works best. Strong ventilation, emergency showers, and smart storage mean fewer scary moments. Always keep the original bottle tightly closed with secondary containment, away from sources of moisture. I’ve seen many labs switch from older glass stoppers to PTFE-lined caps after too many accidents caused by stuck or leaking closures.
Training makes the biggest difference. Every new lab worker should watch videos—real accident stories, not just procedures. Regular drills for chemical spills or gas leaks mean people jump in, not freeze with panic. Before reordering this chemical, safety teams should ask if a less aggressive reagent will do the job. Many modern syntheses rely on alternative acids or conversion agents, reducing the need for such a risky compound in the first place.
Strong oversight and a culture of safety help. I always encourage open reporting—nobody gets blamed for a close call if it teaches the team how to work smarter next time. That kind of support turns a hazardous environment into a learning one, where mistakes push everyone toward better habits.
Looking Ahead
Oxalyl chloride will stay part of advanced manufacturing and research, but careful handling separates ordinary workdays from disasters. Respect for the risks, backed up by solid preparation and the right attitude, keeps people safe wherever these chemicals turn up.
| Names | |
| Preferred IUPAC name | ethanedioyl dichloride |
| Other names |
Ethanedioyl chloride Oxalic acid chloride Oxaloyl chloride Oxalic chloride |
| Pronunciation | /ɒkˈseɪlɪl ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 79-37-8 |
| Beilstein Reference | 1209220 |
| ChEBI | CHEBI:64276 |
| ChEMBL | CHEMBL1367 |
| ChemSpider | 54634 |
| DrugBank | DB13917 |
| ECHA InfoCard | 100.011.822 |
| EC Number | 201-197-0 |
| Gmelin Reference | 821 |
| KEGG | C18627 |
| MeSH | D005078 |
| PubChem CID | 8220 |
| RTECS number | KI1100000 |
| UNII | 1R8V47101D |
| UN number | UN3261 |
| Properties | |
| Chemical formula | (COCl)2 |
| Molar mass | 126.93 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent penetrating odor |
| Density | 1.48 g/mL at 25 °C |
| Solubility in water | Reacts violently |
| log P | -0.07 |
| Vapor pressure | 19.4 kPa (20 °C) |
| Acidity (pKa) | -0.7 |
| Basicity (pKb) | -5.0 |
| Magnetic susceptibility (χ) | -47.5 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.445 |
| Viscosity | 0.54 cP (20 °C) |
| Dipole moment | 1.41 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -507.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -483.9 kJ/mol |
| Pharmacology | |
| ATC code | Oxalyl Chloride does not have an ATC code. |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; causes severe burns and eye damage; reacts violently with water, releasing toxic gases (hydrogen chloride and phosgene). |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331, H314, H335 |
| Precautionary statements | P261, P271, P280, P305+P351+P338, P303+P361+P353, P304+P340, P310, P312, P330, P391, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Autoignition temperature | 275 °C |
| Lethal dose or concentration | LD50 oral rat 826 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 146 mg/kg |
| NIOSH | QK9625000 |
| PEL (Permissible) | PEL: 1 mg/m3 (ceiling) |
| REL (Recommended) | 1 ppm |
| IDLH (Immediate danger) | 2 ppm |
| Related compounds | |
| Related compounds |
Oxalyl bromide Oxalyl fluoride Oxalic acid Thionyl chloride Phosgene |

