Orthophosphoric Acid: Past, Present, and Future
Historical Development
Back in the 18th century, chemists first managed to isolate orthophosphoric acid, a discovery that completely changed the way industries managed food preservation, fertilizers, and cleaning agents. Johann Wolfgang Döbereiner, a German chemist, heated phosphorus with nitric acid and brought phosphorus chemistry into a new light. Over time, people started to understand more about phosphoric acids, and industries started using orthophosphoric acid on a commercial scale around the late 19th and early 20th century. As industries boomed, especially agriculture and soft drink manufacturing, demand for this acid shot up, fueling global trade and innovation.
Product Overview
This acid shows up everywhere, from cola drinks to rust removers. Chemically known as H3PO4, it’s a triprotic acid, so it can donate three protons. Companies supply it mostly as a clear, syrupy liquid, with concentrations going up to 85%. Some folks might call it phosphoric acid too, but that name also covers its various molecular cousins, so using “orthophosphoric acid” helps avoid confusion. In hardware stores, its practicality draws in everyone from homebrewers to metalworkers, making it an item you would struggle to avoid at some point in your life.
Physical & Chemical Properties
At room temperature, orthophosphoric acid stays as a thick, colorless liquid. It’s odorless and blends easily with water, making it useful in food processing. When cooled below 21°C, it solidifies into a crystalline mass. The pH plummets when added to water, creating a highly acidic environment, and the acid acts as a moderate oxidizing agent. It’s non-volatile and lacks that chest-clenching fume you get from hydrochloric or nitric acid. Its melting point sits at about 42°C, and the boiling point creeps up to over 200°C as water evaporates out. This stability helps companies safely transport it in bulk tankers or drums.
Technical Specifications & Labeling
Commercial orthophosphoric acid often comes at 75% or 85% by weight. Production for food and pharmaceutical use sticks with stricter impurity limits — heavy metals and arsenic can’t sneak over set thresholds. Labeling includes the standard UN number (1805), hazard pictograms for “Corrosive,” and warnings about possible burns or eye damage. Take the SDS (Safety Data Sheet) seriously. It’s common sense, but easy to ignore when dealing with something that looks like a thick syrup.
Preparation Method
Industrially, most companies have settled on two processes. The wet process dominates, using sulfuric acid to react with phosphate rock. This method spits out a product suited to making fertilizers, not food – too many impurities. The thermal process uses furnace-grade phosphorus burned with air, producing a vapor that mixes with water to create acid that meets food and technical standards. The thermal route remains cleaner and costlier, reserved for cases needing higher purity. Years ago, labs made small batches by burning phosphorus and catching vapors, but now the scale grew so big, only industrial plants bother.
Chemical Reactions & Modifications
Orthophosphoric acid displays versatility, handing off hydrogen ions to react with metals, alkalis, and organic compounds. Mixing with sodium hydroxide creates sodium phosphates for detergents and water treatment. Mix with calcium and you land with the bone-building calcium phosphate used in fortified foods. Reacting it with basic oxides leads to fertilizer ingredients, which have fed the world for generations. Dehydration of the acid gives metaphosphoric or pyrophosphoric acids, both useful for specialty applications. Under heat, phosphoric acid becomes a key phosphorus donor for organic synthesis, fueling R&D in drug design and food science.
Synonyms & Product Names
You might spot this acid under several names: orthophosphoric acid, phosphoric acid, E338 (food additive code), and even just “fosforzuur” in Dutch-speaking countries. These labels appear across drums, totes, and ingredient lists, so knowing them reduces mistakes when ordering in bulk or researching MSDS sheets.
Safety & Operational Standards
Handling this acid means gloves, goggles, and sometimes a face shield. It burns skin and eyes, and if splashed, can cause lasting damage. Storage means keeping it away from strong bases, combustible materials, and moisture-sensitive chemicals. Emergency showers and eyewash stations should never be far away from storage areas. Many local safety codes sync with OSHA’s requirements, focusing on reducing accidental releases and making sure every employee reads the labels and safety data sheets. Plenty of folks overlook long-term risks, so health and safety training must become routine and not just a “check the box” exercise.
Application Area
Few chemicals feature as widely in daily life. Farmers depend on phosphate fertilizers to boost yields, especially in soils starved of phosphorus. Orthophosphoric acid adjusts the pH in food processing, adds tartness to soft drinks, and prevents calcium build-up in beverage lines. Metal finishers rely on it for rust removal and phosphating, laying down a corrosion-resistant layer. Water treatment engineers use phosphate salts derived from this acid to prevent lead leaching. Even dental hygienists benefit: dentists etch tooth enamel using it before applying fillings. Every one of these applications depends on the ability of the acid to react, chelate, or adjust acidity.
Research & Development
Industry and academia focus attention on greener processes for manufacturing, seeking ways to extract phosphorus from waste and recover acid from used streams. Work in nanotechnology explores phosphoric acid-based surface coatings. Pharmaceutical labs build on it as a reagent for synthesizing key drug intermediates. Researchers dive into finding substitutes for food and beverage acidulants, yet keep circling back due to its safety track record at low doses and lack of off-flavors. Projects in battery technology examine phosphate derivatives for stable, non-flammable electrolytes.
Toxicity Research
At the concentrations found in sodas or food, this acid is considered safe, but misuse and overexposure spark concerns. Animal studies link excessive phosphate intake with kidney damage and metal leaching, raising questions about heavy drinkers of soft drinks. Inhaling mists or ingesting large amounts corrodes tissues, so industrial exposure remains tightly regulated. NIOSH details workplace exposure limits, tracking worker health to catch any chronic problems. Toxicology keeps shifting as new research clarifies roles of dietary phosphate in chronic disease; policymakers adjust allowable levels to keep up, balancing industry needs with public health.
Future Prospects
Moving ahead, the industry faces mounting pressure to recycle phosphorus and reduce mining. Efforts ramp up to recover phosphates from wastewater and urban refuse, aiming for a more sustainable supply chain. As food safety regulations grow tighter, demand for higher-purity acid rises, presenting a challenge to manufacturers targeting both fertilizer and food-grade markets. The search for alternative acidulants in beverages presses on, but orthophosphoric acid's reliability and familiarity push it to the front of the line. Look to see more investment in closed-cycle production, green chemistry, and novel applications in high-tech electronics and environmental clean-up. Experience with handling chemicals and watching regulatory trends suggests companies that lead in safety, environmental recovery, and adaptability are set to define the role of orthophosphoric acid in tomorrow’s world.
What is orthophosphoric acid used for?
Unpacking Orthophosphoric Acid’s Place in Daily Life
Orthophosphoric acid doesn’t grab headlines, yet just about everyone crosses paths with its work, whether through a sip of cola or the food on their plate. In the food business, this clear, sour-tasting liquid acts as an acidifier—not only does it bring a tangy edge to colas, it also slows spoilage and helps preserve color and freshness in processed foods. The soft drink industry pulls vast quantities each year, and it’s one reason that fizzy drinks manage to keep such a satisfying punch. Unfortunately, guzzling too much comes with its downsides—a diet heavy in phosphoric acid-laced sodas can lower calcium in bones, a fact that troubles public health researchers.
Fertilizers: Fueling Global Harvests
In farming, orthophosphoric acid makes the leap from lab beaker to vast fields. Food production in every corner of the globe depends on phosphate fertilizers, like monoammonium and diammonium phosphate. Both come from reacting orthophosphoric acid with ammonia. Plants growing in well-fertilized soils turn out stronger, with fatter grains and brighter leaves, marking the acid’s influence on the dinner table. Yet heavy fertilizer use threatens rivers and lakes, feeding toxic algae blooms from runoff. Farmers and scientists continue searching for smarter ways to feed crops without poisoning waterways—soil testing, slow-release fertilizers, and better application timing all offer promise.
Industry at Work: Metal Cleaning and Beyond
Pick up almost any household cleaning product for rust and you’ll probably spot phosphoric acid on the label. Metal fabrication shops count on it too. The acid cuts through rust fast, turning flaky orange into a harmless gray surface that paint grips tightly. In my own experience, working on an old bicycle frame, nothing stripped the rust like a solution mixed at the local hardware store. Car shops often dip automotive parts in tanks filled with the solution for spotless results before finishing. Still, the acid’s corrosive touch doesn’t know friend from foe—a splash on bare skin can sting, so workers and do-it-yourselfers gear up with gloves and goggles.
Medicine and Water Treatment
Hospitals and dental clinics reach for orthophosphoric acid every day. Dentists use it to etch teeth before fillings, helping materials hold tight for years. Blood testing labs use it in sample preparation. City water departments turn to orthophosphoric acid to stop lead leaching from pipes. By coating pipes with a protective layer, the acid locks out the metal, keeping families’ water safer. Communities that have switched to treated water cut lead exposure in the following months without expensive pipe replacements—though ongoing monitoring remains crucial.
Striking a Balance With Safety
The benefits stack up, from higher crop yields to safer drinking water. Yet, nobody should shrug off the risks—unprotected handling causes burns, and spills in factories need swift clean-up and careful disposal to avoid hurting people or wildlife. Regulators demand safety labeling, training, and regular inspections. As green chemistry advances, new ways may ease reliance on harsh acids, but for now, orthophosphoric acid keeps earning its place in manufacturing, medicine, and food.
What is the chemical formula of orthophosphoric acid?
Getting to Know H3PO4
Every now and then, a name pops up that sounds more complicated than it actually is. Orthophosphoric acid fits that bill, but its formula, H3PO4, couldn’t be more straightforward. It’s almost odd to realize how often this compound shows up in daily routines. As someone who’s taught high school chemistry, I’ve reached for the bottle of “phosphoric acid” plenty of times for experiments that help students see theory turn practical.
Why H3PO4 Matters Beyond the Lab
It’s easy to dismiss certain chemicals as “just” school material, but H3PO4 impacts lives outside textbooks. The food industry leans on this acid to add tartness in sodas. Open the ingredient list on a can of cola and there it is—orthophosphoric acid adding that distinct tang. In agriculture, fertilizers depend on the phosphorus that comes from this source. Crops soak up nutrients, and ultimately, people eat better food because of the role this acid plays.
Clear Facts: Chemistry Driving Real-World Benefits
Phosphorus from orthophosphoric acid isn’t just good for plants. It’s a backbone for healthy bones and teeth. Medical uses tap into its ability to keep calcium in check, where it’s needed. Teeth, for instance, get cleaned with preparations containing a bit of this acid to prep them for fillings. There’s nothing abstract about its impact—healthier crops, tastier drinks, well-kept teeth all owe something to H3PO4.
Looking at the Environmental Picture
With so much benefit comes a responsibility. Factories making or using orthophosphoric acid cannot just disregard the environment. Too much phosphate drifting into lakes or rivers triggers huge algae blooms. Those blooms pull oxygen out of the water, fish suffocate, and the whole ecosystem can wobble out of balance. The modern fertilizer industry faces real pressure to control how much phosphorus reaches fields, then rivers.
Striking a Balance: Safer Use and Smart Innovation
Learning from mistakes, the scientific community pushes for better stewardship. Technologies now help monitor runoff in real time. Farmers adopt precision application techniques guided by soil testing—applying just enough fertilizer, in the correct places. In food and beverage manufacturing, strict quality controls keep consumer safety front and center. Regulatory bodies like the Food and Drug Administration check and re-check that no dangerous levels make it into groceries.
Connecting the Dots: Science Meets Daily Life
It turns out, the formula H3PO4 carries its story far and wide. Each time someone enjoys a fizzy drink, tends their garden, or gets a dental checkup, that chemical makes a subtle but important difference. Taking stock of these connections, it’s hard not to respect the ripple effect a simple acid can have.
Is orthophosphoric acid safe to handle?
Practical Safety Questions About Orthophosphoric Acid
Handling chemicals in daily work calls for straight talk. Orthophosphoric acid sits on plenty of inventory shelves, whether in labs, water treatment, or food manufacturing. It shows up as a clear, syrupy liquid. Folks reach for it to adjust pH, clean metals, and blend fertilizers. Yet, with all this use, its safety often draws questions—some based on scary stories, others on real burns and accidents.
Why It Matters To Take Care
I have learned through hands-on experience that cutting corners grows risky fast. Spilling this acid on your skin leads to burns—redness, pain, sometimes blisters. Breathing its fumes will irritate your lungs and throat. If it gets in your eyes, the result often means a trip to the doctor, not a quick rinse at the sink. Even small amounts left on a workbench corrode tools and chew up clothing.
People sometimes shrug off these possibilities, thinking the acid isn’t as nasty as sulfuric or hydrochloric. Still, the numbers back up caution. The Globally Harmonized System rates orthophosphoric acid as corrosive. The Occupational Safety and Health Administration (OSHA) requires splash protection in workplaces that use the acid, setting exposure limits to keep air clean. The European Chemicals Agency recognizes it as causing severe skin burns and eye damage.
People at Home Aren’t Immune
Orthophosphoric acid shows up in household rust removers and concrete cleaners. A parent or a cleaning crew may not always check the label, but that doesn’t make spills any kinder. Children have landed in emergency rooms after accidental contact. It doesn’t take much—a knocked-over jug, a curious hand, and seconds later, real harm.
Safe Handling Steps That Make A Difference
Lab training drills in basic steps. Gloves built to resist acid should go on before opening the bottle. Goggles keep splash out of your eyes. Clothing covering skin cuts down exposure. Working under a fume hood clears the air so you’re not breathing in sharp vapors. Having a proper spill kit nearby gives quick response if anything tips over.
At home, the same habits matter. Never pour in a small sink. Open windows if you need to use a cleaner with orthophosphoric acid. I still remember seeing a neighbor’s paint come off their basement shelves after they cleaned with the wrong brush—damage like that adds up fast if left unchecked. Triple-check childproof locks and keep chemicals far from food prep areas.
Why Talk About Substitutes
Safer alternatives exist for many jobs—citrus-based cleaners, milder acids, abrasive pads. If less hazardous options perform as well, I reach for those first. In industry, switching might involve extra training, or suppliers digging up new product sheets. This upfront effort beats long-term health claims or paying for injury treatment later.
If the work truly calls for orthophosphoric acid, sticking with regulations and common sense pushes accidents way down. Emergency eyewash stations, clear labels, and consistent training all help. Rushed jobs and missing gloves? That’s usually how people get hurt.
Key Takeaways From Real-World Use
Having seen both careful and careless handling, I side with the experts who recommend treating orthophosphoric acid with the same respect you’d show any corrosive chemical. Even if the liquid looks harmless, your skin, lungs, and eyes know otherwise. Safety steps aren’t extra—they form the backbone of long, healthy careers and families free from chemical burns.
How should orthophosphoric acid be stored?
Pursuing Safety Beyond the Lab Manual
A lot of folks hear the word “acid” and picture bubbling vats and lab coats. In the real world, orthophosphoric acid works its way into far less dramatic places—fertilizer plants, food industries, car workshops. It’s the same chemical no matter who handles it, but storage mistakes can quickly snowball, turning a routine task into a hazard.
Experience has shown that storage never gets the same attention as handling or transport. It should—since most incidents spark from simple oversights like damaged containers or misunderstanding what not to store together. I’ve watched a seasoned warehouse crew rush a quick transfer, dribbling acid down a dusty drum that later corroded through. Cleaning it up after hours in safety gear hammered home the risks that come from cutting corners.
What Proper Storage Really Looks Like
Safe storage of orthophosphoric acid means more than tossing it on a sturdy shelf and locking the door. Acid containers work best in cool, dry spots—somewhere far from direct sunlight and any chance of extra heat. Heat speeds up corrosion and draws in moisture, especially if you crack the seal too often, and that changes the acid’s strength over time.
Ventilation matters more than most realize. Acid fumes creep out, sometimes almost invisible. Without airflow, those vapors build up and corrode metals, irritate skin and lungs, or trip an alarm on a sensitive detector. Industrial setups often use acid cabinets made of polyethylene or fiberglass, not metal. Plastic doesn’t rust or react, and it shrugs off spills—unlike steel racks or basic concrete floors.
I’ve seen how someone’s carelessness with storage can damage more than just the packaging. Strong acids latch onto any water source in the room, even the humidity in the air. Over time, this pulls water into the container, changes the mixture, and sometimes balloons the container until it splits open. Setting up a simple humidity monitor near storage marked a turning point at my workplace. The readings brought home how much things can shift over the course of a year, especially with summer storms or a busted HVAC system.
Pay Attention to Compatibility
Orthophosphoric acid never plays well with every chemical neighbor. Storing it next to ammonia, bleach, or strong alkalis sets up a recipe for hazardous reactions. Fire codes require a separation zone—usually a clearly-marked section or specific shelf far apart from bases and oxidizers.
Labels shouldn’t fade or peel. A fresh hazard label helps keep staff aware, especially rotators who might not have handled acid in months. Containers with damaged seals, odd bulges, or mysterious stains often hide bigger risks inside. Regular checks catch these issues before there’s a spill on the floor.
The Human Factor: Training and Oversight
It takes more than following rules written on a checklist. At most small facilities I’ve visited, formal training happens once a year at best—but nobody really absorbs safety from slides in a fluorescent-lit room. Ongoing reminders, quick drills, open conversations about close calls, those make a bigger difference. Some of the safest plants keep a whiteboard in the acid room—staff jot down stories or tips when they spot trouble, and everyone learns from real mishaps instead of textbook warnings.
Protective gear such as gloves and face shields should sit right by the entrance. Convenience means they actually get used. A spill kit and an eyewash station nearby reduce the stakes if mistakes happen. My own hands have felt how quickly orthophosphoric acid burns unprotected skin, and I learned the hard way that delaying a rinse, even for a few seconds, can lead to nasty scars.
Small Fixes Make a Big Difference
Storing orthophosphoric acid safely doesn’t take expensive gear or specialist know-how—just a commitment to routine checks, clear labeling, and basic chemistry smarts. It starts with respecting the risk, looking out for the next person who might grab that drum or bottle, and owning the little habits that steer everyone clear of accidents.
What are the health hazards associated with orthophosphoric acid?
What Orthophosphoric Acid Does to the Body
Orthophosphoric acid pops up in a lot of places you might not expect. It's in fertilizers, detergents, and even soft drinks. Its powerful acidic nature makes it useful but also risky when safety slips. I’ve seen cases at work where a forgotten pair of safety goggles or careless handling meant a trip to the nurse, and sometimes, follow-ups with the doctor.
This acid burns skin and eyes on contact. I’ve witnessed a lab tech freeze for a second before rinsing off a splash, a delay that ended up costing her weeks of discomfort and scarring. It can chew through skin with just a quick spill, especially in higher concentrations. Eyes need even less exposure; even a droplet can cloud vision or burn deep, meaning a permanent change in eyesight. Inhaling its fumes can set off coughing fits and can irritate the throat and airways. At higher exposures, it can make it hard to breathe thanks to swelling or even chemical burns inside the lungs.
Long-Term Exposure Worries
Here’s where things get personal. Decades in industrial environments taught me that folks exposed daily, especially without real attention to personal protective equipment, wind up with more than just the occasional red patch. Chronic exposure, often through low-level leaks or frequent cleaning with phosphoric-acid-based products, has triggered asthma-like symptoms for some workers. Others report persistent irritation of the eyes and sinuses, which doesn't fade over weekends.
Drinking or ingesting it—rare, but possible if children get their hands on cleaning products—can result in stomach pain, vomiting, and burns in the mouth and throat. This isn’t just a hypothetical; poison control centers have stories about parents who left bottles uncapped.
Making Work and Home Safer
Limiting problems starts with the basics: gloves that fit, face shields in splash zones, good ventilation. Factories use local exhaust systems to suck fumes away from workers—something I wish had been standard in earlier years. I’ve recommended and seen success with secondary containment bins and locked chemical storage for home settings, especially to keep kids and pets away.
Training matters more than people think. Fast, clear safety briefings with real-life mistake stories stick with folks longer than a printed pamphlet. I always urged coworkers to keep rinse stations free from clutter and close to work areas. It’s not enough to have rules; you need habits. Routine drills with eyewash stations save eyes and reduce panic when real accidents happen.
Thinking Beyond Industry
The acid turns up in food and soda, too, especially cola-flavored drinks. While the concentrations are lower, concerns have popped up linking regular high intake to lower bone density. Some researchers argue that, over years, high phosphate levels could edge out calcium and slowly thin bones. Medical communities suggest moderation, especially in kids and the elderly, to avoid risking bone strength.
Orthophosphoric acid helps drive much of modern manufacturing and food processing, but it's never harmless. The acid’s hazards never really fade; handling it with respect, building safety into daily routines, and keeping it out of reach at home lessen the chance of regretful accidents.
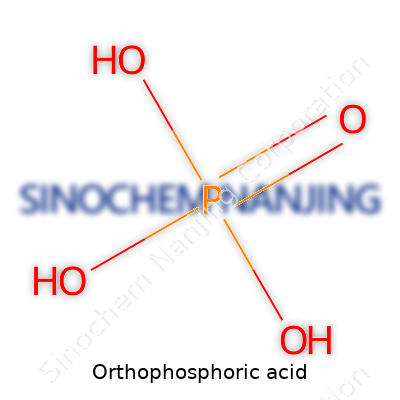
| Names | |
| Preferred IUPAC name | Phosphoric acid |
| Other names |
Phosphoric Acid E338 O-Phosphoric Acid Phosphoric(V) Acid Orthophosphoric(V) Acid Aqueous Phosphoric Acid |
| Pronunciation | /ˌɔːr.θə.fɒsˈfɔːr.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 7664-38-2 |
| Beilstein Reference | 1711077 |
| ChEBI | CHEBI:43474 |
| ChEMBL | CHEMBL1403 |
| ChemSpider | 534 |
| DrugBank | DB04310 |
| ECHA InfoCard | 03d6476d-4211-410a-a3e2-ce7e5f89183d |
| EC Number | 231-633-2 |
| Gmelin Reference | Gmelin Reference: 1662 |
| KEGG | C01081 |
| MeSH | D010818 |
| PubChem CID | 1004 |
| RTECS number | TB6300000 |
| UNII | VW10A02I9X |
| UN number | UN1805 |
| Properties | |
| Chemical formula | H₃PO₄ |
| Molar mass | 97.994 g/mol |
| Appearance | Colorless, odorless, syrupy liquid |
| Odor | Odorless |
| Density | 1.88 g/cm³ |
| Solubility in water | Miscible |
| log P | -1.05 |
| Vapor pressure | <0.01 mm Hg (20°C) |
| Acidity (pKa) | 2.14, 7.20, 12.37 |
| Basicity (pKb) | 1.6 |
| Magnetic susceptibility (χ) | −36.6×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.433 |
| Viscosity | Viscosity of Orthophosphoric Acid is 1.685 cP (at 25°C) |
| Dipole moment | 1.13 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 110.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1271.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1284 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | A09AB12 |
| Hazards | |
| Main hazards | Corrosive, causes severe skin burns and eye damage, harmful if swallowed, may cause respiratory irritation. |
| GHS labelling | Danger; H314, H290 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H290, H314 |
| Precautionary statements | P234, P260, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P363 |
| NFPA 704 (fire diamond) | 3-0-2-Acid |
| Autoignition temperature | 450°C |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat: 1,530 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1,250 mg/kg (oral, rat) |
| NIOSH | KW2090000 |
| PEL (Permissible) | 1 mg/m³ |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | 1000 mg/m3 |
| Related compounds | |
| Related compounds |
Polyphosphoric acids Pyrophosphoric acid Triphosphoric acid |

