Octafluoropropane: A Clear-Eyed Look at a Key Modern Compound
Tracing the Path: A Brief Walk Through Its History
Octafluoropropane didn't enter the chemical landscape overnight. Developed in the backdrop of rising industrialization, this compound rose to prominence once scientists dug deeper into fluorinated materials in the twentieth century. Early uses grew from the need for inert, stable gases in electronics, an industry that found traditional alternatives lacking. Over the past few decades, engineers and chemists have experimented and refined both synthesizing methods and applications, spurred along by both technological opportunity and regulatory pushes. Even in my own work, I’ve watched this gas evolve from something whispered about in academic research to a standard item on the shelf of every microelectronics lab. Those who’ve tracked its story know that the motivations behind its emergence reflect just how a single chemical can shape and define an entire field.
Getting to Know the Compound
Octafluoropropane sets itself apart through its combination of stability and reactivity. This is a colorless, odorless gas at room temperature, which means it doesn’t alert you to its presence or give much trouble in terms of handling compared to nastier volatiles. Its chemical formula—C3F8—packs eight fluorine atoms, and that heavy fluorination is where its claim to fame starts. The resulting molecule resists breakdown and keeps heat at bay, so it works as a dielectric and plasma etching agent. Its ability to hang together through heat, electricity, and light means you can push it in situations where other gases bow out. That’s part of what made me trust it for critical work, especially knowing that stray reactivity won’t undo hours of careful prep or damage sensitive equipment.
Physical & Chemical Features That Matter
When handling octafluoropropane, anyone who’s been elbow-deep in a lab remembers how its low boiling point and high density compared to air turn otherwise routine plumbing into a puzzle. There’s no mistaking its inertness for weakness, though. This is a gas that rarely plays ball with water, acids, or bases, and that lack of participation gives systems builders real predictability. We count on its non-flammability, especially when safety or equipment warranties hang in the balance. Its presence in environmental debates isn't hard to understand, either, since it lingers in the air for centuries. Precision thinking is a must; cutting corners means ignoring solid evidence about how this chemical behaves once it’s released.
Behind the Label: Technical Specifications That Count
Every cylinder of octafluoropropane comes with a long list of ratings, from 99.99% purity grades for the chip factories to stricter specs crafted for the medical sector. Regulatory oversight sets firm rules: storage labels handle leak risk; transport containers demand heavy-duty alloys, and pressure thresholds guard life and limb. As someone who has read enough datasheets to make your eyes glaze over, I've found the real variable lives in how each sector tailors the gas to its end-use quirks. Some applications call for precise trace impurity control; others lean on the supply chain to keep costs down. There’s an art to matching the right gas lot to the right need, and the best techs will always keep one eye on both purity data and operational habits.
How Chemists Prepare the Stuff
Making octafluoropropane never struck me as glamorous, but it involves real discipline. Most commercial batches start from either hexafluoropropylene or trichlorotrifluoropropane, with specialized reactors using either direct fluorination or halogen exchange methods. The process steps require meticulous handling, not just for product yield but also in scrubbing out unwanted leftovers. Byproducts with fewer fluorines—that’s where headaches start—so plant operators continuously tune their process parameters to ensure what leaves the plant meets spec. It’s a game of finesse, with safety and waste handling taking center stage to avoid expensive setbacks or community headaches.
Chemical Shifts: Reactions and Modifications
For all its famed stability, octafluoropropane plays a role as both a shield and scaffold in chemical engineering. Plasma processes can break the molecule apart in semiconductor fabrication, creating fluorine radicals that clean or etch in microelectronics manufacture. Other fluorinated products borrow elements of its structure to create even more exotic compounds. I’ve seen research groups leverage carefully controlled decomposition to produce materials for medical contrast works and imaging enhancement, pushing the boundaries of what was once just an inert filler gas.
The Nomenclature Maze: Synonyms and Alias Names
Octafluoropropane sometimes hides behind other labels—perfluoropropane, C3F8, or simply R-218 in the compressed gases universe. Each term signals a different audience: engineers, chemists, regulators, and market managers. Confusion can trip up even seasoned professionals, especially as technical literature and procurement paperwork sometimes dance between names without warning. In my experience, breaking through the naming tangle clears paths both at the drawing board and in the field.
Working Safe: Standards and Practical Know-How
Factories, hospitals, and labs all recognize that safe handling of octafluoropropane calls for more than just following the letter of the law. Respiratory protection matters; accidental releases or poor ventilation risk replacing oxygen, even though toxicity sits at low levels. Staff need solid training around pressure-rated regulators, proper leak detection, and emergency protocols—this isn’t theory. I’ve seen seasoned plant workers double-check every connection before turning on the supply, knowing well that this caution determines both uptime and safety. Investing in routine inspection and owner-level responsibility makes a clear difference in operational security.
Where the Rubber Meets the Road: Application Areas
Semiconductor manufacturers rely on octafluoropropane as a plasma etching medium. Engineers can achieve precise patterning down to the nanoscale, critical for chips driving everything from laptops to smart cars. Medical teams deploy the gas as a contrast agent for advanced ultrasound imaging, drawing on its physical properties to illuminate tiny vascular structures. High-voltage electricity infrastructure sometimes benefits from its insulating power, though environmental debates stir ongoing research into greener alternatives. Watching this gas pull double duty across markets served as a constant reminder: the same bottle can power electronics or support a life-saving diagnosis.
Pushing Forward: Research and Future Improvements
Academics, industry labs, and startups all seek ways to stretch the usefulness of octafluoropropane. I’ve seen work exploring supercritical extraction for niche pharmaceutical separations, low-temperature refrigeration research, and more environmentally responsible take-back or decomposition solutions. Pushes for recycling programs mirror larger chemical sector efforts to minimize emissions. As regulatory and climate concern grows, garnering support for lower-impact manufacturing routes will mean marrying process efficiency to stewardship. Funding patterns today indicate that those who solve the problem of environmental longevity without losing technical capability stand to reshape the landscape.
It Isn’t Harmless: Toxicity and Environmental Impact
Despite its reputation for low acute toxicity in humans, octafluoropropane poses real issues on an ecological scale. The gas’s atmospheric lifespan stretches beyond what most industrial emissions controllers can tolerate, so it bears a daunting global warming potential. I remember participating in workshops discussing ways to cap emissions at the fab floor, realizing how perfect inertness can carry long-term downside. While acute workplace exposures rarely cause symptoms, chronic release stirs regulatory scrutiny, making both source reduction and advanced venting controls the clear solution set. Learning from this, large users now include end-of-life controls and lifecycle management in their audits, treating environmental discipline as both scientific requirement and operational reality.
The Road Ahead
The future of octafluoropropane sits at a crossroads. Technological breakthroughs keep it relevant, but sustainability questions aren’t fading away. Greater transparency across industries in monitoring, recycling, and substituting will mark progress for both producers and consumers. Regulatory guidance continues tightening, driven by both data and citizen activism. I expect next-generation materials—whether bio-based or purely synthetic—will siphon off some demand, but entrenched high-tech uses won’t vanish in a hurry. The challenge is to keep innovating, marrying legacy strengths to greener solutions, and keeping the collective eye on what legacy this compound leaves behind.
What is Octafluoropropane used for?
Why This Gas Matters
Octafluoropropane sounds like something only scientists need to care about, but life often proves otherwise. In reality, this colorless, odorless gas flows through a surprising number of modern systems. Looking at the facts, it finds its way from medicine to electronics, popping up in places you don’t expect.
From Medical Imaging to Saving Eyesight
Doctors rely on octafluoropropane for a procedure called retinal detachment repair. My own family member went through this surgery a few years ago. The gas played a quiet role—filling the eye temporarily, pushing the retina back in place, and giving tissues valuable time to heal. Surgeons trust this compound for its stability and non-toxic nature. You don’t hear about it in waiting rooms, but that single step can mean the difference between blindness and clear sight for many people every year.
Ultrasound specialists use it in the form of microbubbles, injected to highlight blood flow or make hidden vessels stand out. The better the imaging, the sharper the diagnosis. More lives get a fighting chance when doctors can detect blocked arteries or damaged organs early.
High-Tech Manufacturing Needs
Walk through any tech lab, especially in places building smartphones or computer chips, and octafluoropropane often sits in a gas cylinder at the edge of the action. Manufacturers have leaned on it to etch circuits into silicone wafers. As these gadgets shrink and become more complex, companies want gases that can work without contaminating sensitive electronics.
My time as an intern in a semiconductor plant showed how tight those margins can be. The entire process revolves around precision. If the wrong gas gets involved, millions of dollars evaporate overnight. Octafluoropropane helps keep the etching sharp and the components pure, while producing fewer dangerous byproducts than some old-school chemicals.
Environmental Concerns and the Search for Balance
While octafluoropropane gets the job done in medical and manufacturing spaces, there’s always a tradeoff. Its chemical stability—what makes it so reliable—means it lingers in the atmosphere. The gas acts as a potent greenhouse contributor. Data from the Environmental Protection Agency puts it among gases with high global warming potential.
Scientists struggle with this exact problem: efficient, safety-minded processes on one side of the scale, long-term planetary health on the other. A few companies have started investing in better abatement equipment to trap and break down emissions before they escape into the wild. Governments across Europe and North America ask industry leaders to reduce leakage. In my own life, I think about how every improved filter or stronger regulation can help us use smart chemistry without ignoring the bill nature hands us later.
A Call for Smarter Use
No one can wave a wand and drop octafluoropropane overnight, since millions rely on the results it delivers. Engineers I’ve spoken to describe real progress: finding alternative gases for manufacturing, updating aging hospital stockrooms, and making sure emissions actually get captured.
It’s easy to overlook what keeps hospitals running or silicon chips working, but a look under the hood always reveals questions worth asking. As technology and medicine keep evolving, everyone involved—scientists, lawmakers, and each of us as citizens—benefits from keeping the conversation grounded in facts and a willingness to adapt.
Is Octafluoropropane safe to handle?
What Is Octafluoropropane?
Octafluoropropane, which you’ll often hear called C3F8, tends to pop up in electronics manufacturing, plasma etching, medical imaging, and as a refrigerant. The clear, odorless gas looks like it could be harmless, but many chemicals hide their risks behind a neutral appearance. In labs, when we had to work with various industrial chemicals, complacency always led to someone dashing for the eyewash or, worse, the emergency room. So I tend to approach unknowns with a healthy dose of skepticism.
Gas Under Pressure: Risk on Multiple Fronts
Octafluoropropane isn’t flammable. That lowers the heat in my worry circuit. It doesn’t react violently with water or oxygen. Still, it sits under pressure when stored in cylinders. Gas leaks can displace air in closed areas. Breathing gets tough, unconsciousness can hit, and all those warnings about asphyxiation on the safety sheet come to life if you relax your safety guard. The U.S. National Institutes of Health singles out how exposure in poorly ventilated spots can crowd out oxygen—with fatal consequences. OSHA points to simple fixes, like keeping tanks secure and checking for leaks before each use, but it's easy to overlook these steps once you get used to the routine.
Health Hazards Go Beyond Breathing
C3F8 doesn’t burn skin or eyes. But it won’t do you any favors if you inhale a heavy dose. As someone who has worked in a facility where compressed gases could escape, I learned that not every dangerous exposure gives you dramatic symptoms. Breathing mostly C3F8 means breathing less oxygen. Since this chemical is colorless and odorless, nobody notices until the headache and dizziness kick in. If you or your crew don’t pay close attention, the risk sneaks up. NIOSH recommends handling it in well-ventilated spaces, so old labs with poor airflow should make upgrades. Keeping oxygen monitors handy helps warn of any leaks.
Engineering Controls and PPE
Most folks in the semiconductor world meet C3F8 behind fume hoods, in exhausted enclosures, or paired with snaking vent lines. I remember seeing an engineer skip the vented bench "just for a minute." That shortcut led to alarms blaring and everyone evacuating a room the size of a classroom. Handling this gas safely means leaning on good engineering controls and not trusting luck. Personal protective equipment—eye protection, gloves, maybe a face shield—offers another layer, even if it sometimes feels unnecessary.
Safe Storage and Transport: Lessons Learned
Storing octafluoropropane means keeping cylinders upright, chained, and away from heat or direct sunlight. Those requirements always sounded tedious, but the aftermath of one toppled tank in our old lab taught me the hard way. Welding gas lines tightly, watching regulators, and never leaving containers where anyone can bump them changes accidents from likely to rare. Precision handling isn’t about paranoia but habit.
Waste Management: The Final Step
Disposing of unused or spent octafluoropropane should fall to licensed hazardous waste handlers. Just venting to the open air adds greenhouse gases, and some studies highlight its role in climate change. At our workplace, any gases leaving spent cartridges headed straight to collection points. This not only protects people but also the local environment.
Building a Safer Workplace
Nobody wants a trip to the ER or unexplained fatigue slowly creeping into their crew. If handling octafluoropropane, proactive steps—upgrading ventilation, monitoring oxygen, running regular safety drills, and reinforcing protocols—all prove their worth quickly. Mistakes with compressed, invisible gases only have to happen once before safety becomes everyone’s top priority for good reason.
What are the storage requirements for Octafluoropropane?
Why the Right Storage Changes Everything
Octafluoropropane, sometimes called C3F8, shows up in all sorts of places—from electronics manufacturing to medical procedures. This gas won’t just drift away into the background unnoticed; it comes with some heavy risks if folks skip precautions. I’ve spent years seeing what happens when chemical storage is handled with care, and also when companies cut corners. Anyone working with octafluoropropane should see it as a priority and not a checklist item.
The Real Dangers of Leaks and Mishandling
A high-pressure gas like this doesn’t forgive mistakes. Pressurized cylinders can turn into missiles under the wrong conditions—think of a dropped tank shearing its valve. I’ve seen a cylinder launch clear into the ceiling after a valve sheared off. This wasn’t just bad luck; it happened because the storage space doubled as a storeroom for all kinds of stuff, and proper restraints weren’t used.
Octafluoropropane boasts excellent chemical stability, but that doesn't mean it’s safe to relax around it. The gas displaces oxygen in the air, raising the risk of suffocation in smaller, sealed spaces. Even those who handle hazardous gases regularly sometimes underestimate just how stealthily asphyxiation can hit—by the time you feel something’s wrong, it’s too late.
Handling Temperature and Pressure Right
Storage in a cool, dry, well-ventilated space can prevent most accidents. Temperatures above 50°C can ramp up pressure inside cylinders until they fail—so outdoor sheds under the summer sun are a bad call. In my time working with compressed gases, I’ve stepped into supply rooms that felt like saunas; it scared me every time. If you see sweating pipes and feel stifling air, it’s time to move those cylinders fast.
Building in Layers of Safety
Some of the best-run labs I’ve seen use sturdy racks or cylinder cages firmly anchored to the ground. Keeping tanks upright with chains or straps stops them from tipping. Every tank gets a clear label with hazards spelled out. Separating octafluoropropane tanks from flammable materials keeps small mistakes from becoming full-blown disasters.
It bears pointing out: installing leak detection monitors is not overkill. In one outfit I consulted for, sensors flagged a slow leak that nobody caught by smell alone. If they’d checked only after workers felt dizzy, someone might have ended up in a hospital.
Regulations and Training
Every worker—even the guy sweeping floors—benefits from a walkthrough of the risks and responses. I’ve watched people fumble at safety valves simply because no one showed them the basics. The Occupational Safety and Health Administration (OSHA) in the US spells out rules for storing compressed gases like C3F8. Regular inspections and following regional chemical storage standards don’t just cover you legally—they save lives.
The Value of Investing in Upgrades
Putting off replacing aging valves or shaky racks to save cash only works until something goes wrong. One shop upgraded to modern pressure relief devices and double-checked their fittings every six months. The peace of mind paid for itself in fewer near-misses. Anyone dealing with octafluoropropane owes it to their crew—and the neighborhood downwind—to treat storage as an investment, not overhead.
What is the chemical formula of Octafluoropropane?
Octafluoropropane and Its Formula: Not Just Numbers and Letters
Octafluoropropane carries the chemical formula C3F8. That set of characters means more than chemistry trivia. It tells a bigger story about how small molecules shape big industries and daily routines. I’ve spent years combing through chemical data and noticed that people outside science circles often miss how those formulas connect to real-world impact.
How a Simple Formula Shapes Everyday Reality
At first glance, C3F8 sounds like it belongs in a lab or a textbook. The reality looks a bit different. In my time working with refrigeration experts, I’ve seen Octafluoropropane turn up in unexpected ways—sometimes inside cooling systems, other times in medical imaging. This molecule shrugs off chemical reactivity, thanks to its eight fluorine atoms. That makes it tough, stable, and useful in places where breakdown could spell disaster.
Getting the formula right can mean the difference between safety and risk. One of my mentors in the chemical industry drove home the lesson that a wrong formula could translate to dangerous product substitutions or failed experiments. C3F8 doesn’t just offer stability; it also helps build confidence among engineers who count on predictable outcomes. This is vitally important in semiconductor manufacture, where one wrong move turns expensive materials into waste.
Octafluoropropane in Action: From Factories to Hospitals
Seeing this molecule in the wild, I’ve talked to technicians who use it for plasma etching—carving circuits onto silicon chips that power smartphones and computers. There’s precision in every step. Any impurity, any misstep with chemical identity, and a whole batch ends up ruined. The same precision comes into play in hospitals, where Octafluoropropane serves as a contrast agent for ultrasound imaging. Doctors use it to look at blood flow in the heart—a task where accuracy can be a matter of life and death.
Environmental Questions: Addressing Responsibility and Solutions
Experience in industrial settings has taught me not to ignore the environmental side. Like many fluorocarbons, Octafluoropropane hangs around in the atmosphere, resisting breakdown. That’s a good thing for technical processes but a major downside for the climate. Its global warming potential is far higher than carbon dioxide. In conversations with sustainability officers, concern over emissions comes up again and again. The industry has a responsibility to control leaks, apply recovery processes, and push for viable alternatives with a lighter environmental footprint.
R&D teams work on recycling techniques that capture Octafluoropropane after industrial use, keeping it out of the air. This isn’t a magic solution, but it’s a step forward. Making sure teams stay trained and equipment stays tight helps, too. Companies that put these safeguards front and center often see fewer accidental releases plus a boost in consumer trust—a win for business and planet alike.
Why It Matters
Understanding the formula C3F8 goes beyond chemistry class. It means knowing the responsibilities that come with handling powerful molecules and making choices that shape technology, healthcare, and climate. Getting those basics straight benefits everyone from engineers to patients to anyone who shares the air.
How should Octafluoropropane be transported?
Octafluoropropane doesn’t show up in most people’s daily conversations, but this colorless, odorless gas plays a critical role in several industries. Found in medical imaging, semiconductor manufacturing, and even fire suppression, folks who handle it understand its benefits and risks. Transporting it isn’t just about following the rules—it’s about protecting people, property, and the environment. My time working in a facility that borrowed plenty of specialty gases taught me that treating rare chemicals like household items is a recipe for disaster.
Safety Isn’t Just for the Rulebook
Some treat chemical safety as a box to check. That mindset falls apart the moment something leaks or someone gets sick. Octafluoropropane falls under the category of liquefied compressed gases. The tanks are usually shipped pressurized, which means a knock or a drop can turn a simple mistake into an accident. Inhaling high concentrations pushes out oxygen, causing dizziness or worse. I’ve watched old colleagues shrug off cylinder handling guidelines, only to panic when a valve stuck open or a regulator failed.
What does responsible transport look like? Securing each cylinder upright, valve covers tightly in place, labels clean and legible. Drivers who haul this material should learn how to use protective equipment, understand emergency procedures, and keep a spill kit within reach. Once, our team spent three hours tracking down a misplaced cylinder. Knowing where hazardous materials are at all times isn’t just a paperwork task—it gives everyone peace of mind, especially if evacuation ever comes up.
Law Isn’t Just Red Tape
The Department of Transportation lays out pretty direct rules for materials like Octafluoropropane. The gas rides in certified pressure cylinders, each with paperwork stating what’s inside and who owns it. Even the vehicle needs the right placards. Making assumptions leads to fines, and those fines sometimes pale next to what happens if a cylinder bursts. Not every town wants trucks full of specialty gases rolling through residential areas. Proper routing helps, along with alerting local authorities about what’s coming their way. Following legal requirements prevents confusion and delays, especially if a truck winds up stopped during transit.
Environmental Stakes Run High
Octafluoropropane doesn’t break down easily in the atmosphere. It traps much more heat than everyday carbon dioxide. A single release sticks around for thousands of years, so a leak during transport writes a story that lasts generations. Shipping with reinforced containers, solid tracking systems, and trained handlers lowers that risk. My team once needed to ship leftover gas for destruction. The logistics company they picked specialized in hazardous materials, using insulated containers and strict drop-off protocols. That decision paid off—the transfer ran without a hiccup and everyone could breathe easy, literally.
Better Tools for Better Outcomes
Technology has changed how we move hazardous materials. GPS tracking, remote sensors, and automated notifications turn a risky job into a routine one. Some newer cylinders even feature pressure monitors that send alerts to drivers if something goes off spec. Investing in equipment upgrades saves money and headaches. Back in the day, managers cut costs by using old tanks or skipping regular inspections. Equipment failures cost more in the long run than proper planning ever does.
Training Makes All the Difference
Every worker along the route should know what they’re dealing with. Regular refreshers keep safety guidelines clear and everyone ready in case things don’t go as planned. Sharing near-misses or lessons learned creates a strong safety culture. Nobody ever expects a problem until it’s staring them in the face. Staying humble and vigilant keeps accidents rare.
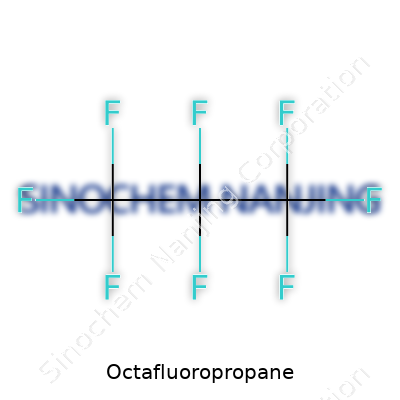
| Names | |
| Preferred IUPAC name | 1,1,1,2,2,3,3,3-Octafluoropropane |
| Other names |
Perfluoropropane C3F8 Freon-218 R-218 FC-218 |
| Pronunciation | /ˌɒk.təˌfluː.rəˈprəʊ.peɪn/ |
| Identifiers | |
| CAS Number | 76-19-7 |
| Beilstein Reference | 1361773 |
| ChEBI | CHEBI:38238 |
| ChEMBL | CHEMBL1209984 |
| ChemSpider | 7222 |
| DrugBank | DB09130 |
| ECHA InfoCard | 03d044d2-b7a0-4e54-8cd7-5e93c81941ad |
| EC Number | 204-075-2 |
| Gmelin Reference | 800 |
| KEGG | C06442 |
| MeSH | D017902 |
| PubChem CID | 9205 |
| RTECS number | TN8575000 |
| UNII | J9WNT6N25P |
| UN number | UN1976 |
| Properties | |
| Chemical formula | C3F8 |
| Molar mass | 188.02 g/mol |
| Appearance | Colorless gas |
| Odor | Odorless |
| Density | 6.62 kg/m³ |
| Solubility in water | Insoluble |
| log P | log P = 2.1 |
| Vapor pressure | 3340 mmHg (25°C) |
| Basicity (pKb) | > 24.08 |
| Magnetic susceptibility (χ) | -44.9e-6 cm³/mol |
| Refractive index (nD) | 1.222 |
| Viscosity | 0.0126 cP at 25 °C |
| Dipole moment | 0.00 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 300.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -2334 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2220 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V08DA06 |
| Hazards | |
| Main hazards | Extremely cold liquid and gas under pressure; may cause asphyxiation by displacement of air. |
| GHS labelling | GHS02, GHS04 |
| Pictograms | GHS04 |
| Signal word | Warning |
| Precautionary statements | Keep away from heat, hot surfaces, sparks, open flames and other ignition sources. No smoking. Do not breathe gas. Use only outdoors or in a well-ventilated area. Protect from sunlight. Store in a well-ventilated place. Store locked up. |
| Autoignition temperature | 460°C |
| Lethal dose or concentration | LCLo human inhalation 75000 ppm/15M |
| LD50 (median dose) | > 4,200 mg/m3 (rat, inhalation, 4 hours) |
| NIOSH | RA7690000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | 1000 ppm |
| Related compounds | |
| Related compounds |
Perfluorocyclopropane Hexafluoropropylene Perfluoropropane Trifluoropropane |

