Octafluoro-2-Butene: The Journey, Impact, and Future of a Modern Chemical
Historical Development
Octafluoro-2-butene traces much of its story back to the surge in interest around advanced fluorinated molecules during the hydrofluorocarbon boom. Throughout the twentieth century, researchers kept probing new fluorinated chemicals, drawn by their unique stability, volatility, and performance in demanding environments. The landscape started shifting as traditional chlorofluorocarbons faced environmental bans due to ozone-depletion and global warming risks. In the search for replacements, octafluoro-2-butene caught the eye of both scientists and engineers for its balance of low toxicity, thermal stability, and potential uses in high-tech applications. I remember the era in the early 2010s when a flurry of patents and academic papers surfaced, showing a growing urge in both industry and academia to master new fluoroalkenes. The development of octafluoro-2-butene fits right into this context, emerging as researchers realized that certain perfluorinated olefins could outperform older compounds in specialized fields without leaving behind the environmental damage of their predecessors.
Product Overview
Octafluoro-2-butene serves as a versatile tool in the hands of chemists and engineers. As a clear, non-flammable gas or liquid under the right conditions, it holds promise where traditional hydrocarbons struggle. This molecule speaks to the kind of innovation sparked by modern chemistry — pushing for properties that tackle tough engineering challenges, like chemical inertia and resistance to breakdown at high temperatures. Applications spread across the board, from advanced electronics to niche refrigerants and specialty polymers. For those who spend their days translating lab-scale findings into real-world products, octafluoro-2-butene represents a chance to introduce more durable, robust components into systems where other chemicals simply can't last.
Physical & Chemical Properties
Looking at octafluoro-2-butene under the lens of practicality, its resistance to chemical attack stands out. High electronegativity from the fluorine atoms enhances its persistence against acids and bases, while the perfluoro-structure stymies degradation pathways that plague standard butenes. Its boiling point, volatility profile, and inertness set it apart — especially in cleanroom environments where every contaminant has consequences. In personal experience, these features cut down maintenance cycles for equipment and give operators more confidence running continuous processes. The dense electron cloud gives the molecule a noticeable heft compared to unfluorinated analogs, lending stability without going as heavy as perfluorocarbons once popular as solvents or heat-transfer agents.
Technical Specifications & Labeling
Working with octafluoro-2-butene comes with strict labeling rules due to its specialized nature. Proper identification, concentration, and handling directions arrive on each container. Regulatory standards drive companies to provide detailed information — not just chemical formulae, but also recommended storage temperatures, necessary personal protection, and environmental handling tips. The kind of labeling that sticks in memory always prioritizes transparency, especially when dealing with highly fluorinated organics. Labeling today relies less on tradition and more on lessons learned, often written to avoid the confusion that older industrial-grade chemicals once caused. Keeping specification clear helps avoid dangerous mishaps and gives traceability back through the supply chain.
Preparation Method
Synthesizing octafluoro-2-butene demands skill and careful process control, largely due to the reactivity of fluorine gas and the trickiness of selective fluorination. Over the years, I’ve seen production shift away from brute-force fluorination toward more controlled, stepwise approaches that minimize by-products. Many laboratories and pilot plants now employ catalytic pathways or unique electrochemical cells, tuning temperature and pressure to steer the reaction toward a high yield of octafluoro-2-butene with minimal side reactions. Making this material at scale means more than just technical know-how — it asks for robust infrastructure and real respect for the hazards of handling elemental fluorine or specialty fluorinating agents. Only facilities with real experience in volatile organofluorine chemistry attempt such processes, and even then, process optimization never really stops.
Chemical Reactions & Modifications
Octafluoro-2-butene rarely enters chemical reactions as a simple reactant. The full shield of fluorine makes it sluggish, so modifications usually depend on strong conditions or extremely reactive partners. This stubbornness brings value in applications where stability matters more than reactivity. That said, niche transformations can happen. A handful of oxidative processes or strong metal-catalyzed conversions open doors to other fluorinated building blocks. In my own work exploring fluorinated monomers, direct functionalization often proved a dead end, but a combination of clever catalysis and controlled conditions could tease out just enough reactivity for polymer synthesis or specialty intermediates. Anyone hoping to modify this molecule needs to respect both its resilience and the risks of forcing reactions too hard.
Synonyms & Product Names
Across patents, journals, and chemical catalogs, octafluoro-2-butene sometimes hides under other names: perfluoro-2-butene, 2-butene, octafluoro-, or even shorthand labels reflecting its structure, like C4F8. These alternative names can trip up even seasoned researchers, so having a clear cross-reference list in the lab or the office avoids confusion and misplaced orders. Commercial distributors occasionally use trade names that play up performance or purity, but anyone working with specialty chemicals soon learns to hunt for the CAS number to ensure the right product shows up on the loading dock. Decoding these names forms part of the fluoro-organic chemist’s apprenticeship.
Safety & Operational Standards
Handling octafluoro-2-butene calls for vigilance. While less acutely toxic than some fluorinated gases, it requires proper ventilation, airtight fittings, and strict avoidance of open flames or sparks. Protective gear — gloves, goggles, and sometimes complete face shields — heads the safety checklist for every technician. Leaks or spills, though rare with careful work, could spell problems, particularly in closed spaces where vapors could displace breathable air. On the operational level, engineering controls such as gas cabinets and remote shutoff systems add another layer of defense. These protocols reflect both regulatory mandates and hard-earned lessons from years in the field, where minor lapses have taught sobering lessons. Workers in the industry value practical checklists, real drills, and a culture that prizes speaking up about hazards, no matter how specialized the job may seem.
Application Area
Octafluoro-2-butene draws interest in more fields than one might guess at first glance. In electronics, the molecule’s cleanliness and resilience against plasma make it a valued etchant and insulating layer precursor, carving ever-smaller features on semiconductor wafers. In refrigeration, its stability and negligible flammability put it on the radar as a next-generation working fluid, especially where traditional refrigerants fail tougher performance or environmental standards. The world of specialty polymers looks to octafluoro-2-butene as both monomer and modifier, creating surfaces that shrug off contamination and handle temperature swings with ease. I’ve seen design teams in aerospace consider fluorinated olefins for everything from lightweight insulation to seals that stay intact under hostile conditions. These uses all rely on the molecule’s blend of chemical stubbornness, volatility, and low toxicity — a combination that older chemicals rarely got right.
Research & Development
Academic and industrial labs keep pushing the boundaries with octafluoro-2-butene. New catalysts accelerate synthesis while reducing unwanted by-products. Analytical chemists refine detection methods, aiming to catch fugitive emissions that regulators now keep a close watch on. Cross-disciplinary teams explore new ways to harness the molecule’s stability in battery electrolytes, membrane films, and optical coatings. I remember the enthusiasm at fluorine-centered conferences as researchers compared notes on post-functionalization or microchip process integration. Pilot lines now churn out not just the base molecule but a range of derivatives, each crafted for an emerging market. This molecules’ history as a specialty ingredient keeps it at the edge of discovery, with commercial and academic players sharing the stage.
Toxicity Research
Toxicologists approach octafluoro-2-butene cautiously, designing studies that track both immediate and chronic effects. Compared to older halocarbons, the molecule shows lower toxicity, likely because its strong C–F bonds limit metabolic breakdown inside living systems. Animal studies and in vitro experiments remain ongoing, searching for any long-term risks, especially as volumes increase in industrial use. Chemists and safety officers rely on published thresholds, adjusting ventilation and workplace exposure limits based on evolving evidence. The data so far suggest risks can be managed with current industrial hygiene practices, but decades of experience have taught us not to downplay the need for vigilance — especially since widespread adoption often uncovers new hazards. Full transparency and peer-reviewed research build the knowledge base required to foster both innovation and safety.
Future Prospects
Looking ahead, octafluoro-2-butene stands to become a staple in advanced engineering and environmental stewardship. Stricter environmental rules keep accelerating the push for alternatives to legacy refrigerants and insulators, and policy shifts worldwide reflect growing pressure to curb greenhouse gas emissions. Chemists and material scientists keep finding new roles for robust, non-flammable, low-reactivity molecules in renewable energy storage, composite manufacturing, and next-generation electronics. From energy to transportation, every gain in efficiency or lifespan translates to lower costs and less waste. Researchers remain alert to any signs of unanticipated risks, keen to build on a foundation of solid science, ethical choices, and respect for both human and environmental health. Adopting octafluoro-2-butene places real demands on companies: invest in worker training, stay mindful of emissions, and commit to ongoing research, not just for compliance but for the sake of long-term progress. This story remains unfinished, and its next chapters will depend on collaboration and a willingness to learn not only from triumphs, but from setbacks as well.
What is the chemical formula of Octafluoro-2-Butene?
Diving Into Chemical Names
A name like "Octafluoro-2-Butene" sounds complex, but breaking it down reveals a simple structure. The root "butene" points to a four-carbon chain with a double bond somewhere in the middle. Throw "octafluoro" on top, and every hydrogen on that backbone gets replaced by fluorine atoms, which is a classic trick chemists use to boost stability or change how a compound reacts. This swap ramps up resistance to chemical wear and delivers strong environmental effects, good and bad.
The Formula Itself
Octafluoro-2-butene carries the formula C4F8. No hidden hydrogens. No oxygen, nitrogen, or other stowaways. Just carbon and fluorine atoms, rigged for a specific physical profile. If you've done any organic chemistry labwork, you know C4 means four carbons long. F8 matches up with the octafluoro label; eight fluorines anchor themselves where hydrogens would normally sit.
Why Fluorinated Compounds Matter
Fluorine brings more than just high scores on the periodic table. In the lab and out in the field, molecules rich in fluorine have shown toughness against fire and chemicals alike. That’s part of why these compounds often show up in refrigerants and certain industrial processes. Carbon-fluorine bonds offer world-class resilience, sometimes to our benefit, but sometimes to the detriment of ecosystems—think about PFAS and all the trouble caused by their persistence.
Environmental Impact and Caution
I’ve seen the persistence of perfluorinated compounds firsthand. The tough carbon-fluorine bonds don’t break down easily, leading to groundwater issues near manufacturing plants. This concern spans from obscure lab chemicals right to well-known household products. Anyone working with octafluoro-2-butene must approach it with respect: The resilience that makes these compounds attractive calls for responsible use and proper disposal.
Applications and Industry Use
Octafluoro-2-butene doesn’t turn up in kitchen cabinets or car interiors, but specialty applications value its unique mix of stability and reactivity. The semiconductor industry seeks such molecules for etching processes. Cooling systems sometimes need a compound that won’t break down under pressure or high temperatures, and highly fluorinated chemicals fit that bill. At the factory or in research, those strengths line up with performance standards only a handful of chemicals can meet.
Challenges and the Road Forward
Regulators have started looking harder at whole classes of persistent fluorinated compounds. Society faces a choice: keep riding the wave of technological benefits, or turn toward alternatives that offer lower environmental risk. Some engineers experiment with fewer fluorine atoms per molecule, hoping to find a sweet spot that protects both industrial goals and water basins. Anyone using or developing octafluoro-2-butene joins this conversation, contributing to research, safe practices, and responsible innovation.
Finding Solutions
Change starts with transparency about waste and emissions. Scientists and industry leaders share the job of chasing down biodegradation pathways and investing in safer molecule design. If a substitute falls short on performance, clear data can drive new rounds of innovation. Working in labs and collaborating with environmental chemists, I’ve seen progress emerge from small test tubes and real pilot runs, not government mandates alone. As a community, we keep raising the bar for what responsible chemical stewardship means.
What are the main uses or applications of Octafluoro-2-Butene?
Understanding Octafluoro-2-Butene
Octafluoro-2-butene sounds like something cooked up only for chemists, but it touches many corners of modern technology and industry. Speaking plainly, this is one of those chemicals that has escaped the narrow corridors of academia and found work where real-world problems need tackling. Its chemical formula, C4F8, gives it some special properties, making it more than just another industrial chemical.
High-Performance Dielectric Gas
One of the standout uses for octafluoro-2-butene comes up in the electric power world. Power companies look for gases that can break down less, spark less, and keep energy moving where it’s supposed to. Electrical transformers and circuit breakers tend to work with a lot of force and nobody wants unwanted sparks flying. Engineers have relied on sulfur hexafluoride (SF6) for years, but SF6 lingers in the atmosphere far too long, wreaking havoc on the climate. Octafluoro-2-butene turns up as a newer and less climate-damaging way to keep those high-voltage systems safe and running. Thanks to a much lower global warming potential, the energy sector can keep the lights on without locking in more future climate troubles.
Refrigeration and Cooling Solutions
Cooling offices, supermarkets, data centers, and cars draws on chemistry that most folks never see. Though the best-known refrigerants keep changing due to environmental rules, octafluoro-2-butene has seen research as a next-generation candidate. Old formulations, like hydrofluorocarbons (HFCs), drive up global warming and fall under tough new bans. Octafluoro-2-butene answers the call for safer, effective cooling. Chemists look at its stability and climate impact, giving hope for chillers and refrigerators that keep things cold without trading that comfort for a hotter planet. There’s still debate and ongoing testing, but the interest signals confidence in this molecule’s cooling power.
Specialty Chemicals and Industrial Manufacturing
Some people might be surprised to learn that chemical manufacturing can get even more complex than pharmaceuticals or plastics. Octafluoro-2-butene gives other industries new tools. Its strong fluorine bonds make it helpful as a specialty reactant in chemical synthesis, especially in the world of advanced materials and electronics. Think microchips or coatings that have to last through extreme conditions. The fluorinated backbone sticks around when lesser molecules break down, serving as a backbone in industry recipes where failure is not an option. I worked alongside a team exploring alternatives for specialty etchants, and octafluoro-2-butene’s clean reactions and tight tolerances gave them freer rein to adjust formulas without nasty byproducts.
Environmental and Safety Considerations
The push for lower-emission alternatives has thrown new light on chemicals like octafluoro-2-butene. Unlike some stubborn gases, this one breaks down faster in the air, slicing down its climate punch. Still, every fluorinated compound raises questions about health, safety, and the best ways to handle leaks or spills. Safe storage, proper ventilation, and careful handling remain key talking points. Chemical safety teams dig deep into published toxicity and exposure data before signing off, but so far, no widespread red flags have popped up in regulated uses.
The Path Forward
Whether you work in electric utilities, refrigeration, or advanced materials, octafluoro-2-butene chemistries promise a step forward. The world keeps looking for alternatives that don’t just work but work better—cleaner, safer, and with less long-range baggage. Octafluoro-2-butene shows that good science can keep pace with the pressure to do better by the planet and by ourselves, all while keeping systems running smoothly.
Is Octafluoro-2-Butene hazardous or toxic?
Looking Past the Chemical Name
Octafluoro-2-butene isn’t a name you’ll stumble across in day-to-day conversations. Still, folks working in refrigeration, specialty chemistry, or materials manufacturing might cross paths with it. You see, the industrial world is hungry for chemicals that can help cool, clean, or propel without tearing up the ozone layer or causing runaway greenhouse effects. Octafluoro-2-butene looks like one of those hopeful contenders, especially when compared to older, notorious fluorocarbons. Yet, some questions about its safety keep popping up.
Not Just Another Inert Gas
It’s true that fluorinated gases can behave in very stable ways, which suggests lower flammability and less explosive risk. Manufacturers often highlight such properties, leaning into claims that suggest safety. My years in the lab taught me that the shelf-life or non-reactivity of a gas doesn't guarantee safety for people or ecosystems. Toxicity isn’t always about fire or explosions.
Let’s get clear: specific toxicology research on octafluoro-2-butene remains thin. Data from closely related fluorocarbons suggests that many of these compounds can produce nasty byproducts when heated—hydrogen fluoride among them. That’s an acid with a reputation: eats through tissue, causes severe burns, and lingers long after the initial exposure.
Trouble Along the Supply Chain
In industrial handling, a chemical may arrive at a plant in perfectly sealed drums, handled by folks wearing gloves, goggles, and respirators. These protocols reduce risk for direct exposure—at least on paper. Cut a few corners, slack on the training, and accidents find their way in. Once a leak happens, inhalation becomes the biggest worry for anyone nearby. Even if the gas feels odorless and non-irritating, breathing in concentrated levels of these substances can damage the lungs and nervous system.
Beyond the factory, disposal and degradation form a real concern. Perfluorinated compounds hang around in the environment. They drift into water, settle in soil, and work their way up food chains. Studies reveal links to health issues that include hormone disruption, immune system impacts, and some cancers. European regulators, for example, continue to tighten reviews of these “forever chemicals.”
Weighing the Evidence and Moving Forward
Nobody enjoys moving backward to old, more damaging chemicals, but playing fast and loose with replacements barely counts as progress. Most researchers want comprehensive testing—both acute (short-term) and chronic (long-term)—on potential new refrigerants, solvents, or propellants. Octafluoro-2-butene deserves a seat in that process. Government agencies like the EPA or ECHA should be pushing for more animal studies, environmental fate trials, and better transparency from manufacturers.
Companies sitting on safety data shouldn’t hide it behind trademarks or proprietary claims. In my experience, open sharing leads to better public health outcomes. Workers deserve thorough training and protective gear as standard, not just as a recommendation. Engineers can design better leak controls and invest in monitoring and alarms for storage rooms.
If there’s one lesson from the last fifty years of chemical innovation, it’s that early caution pays off. Folks using or living near industrial sites deserve honesty about risks—both known and possible. No chemical, no matter how “advanced,” replaces the need for solid, open science and practical protections.
How should Octafluoro-2-Butene be stored and handled?
Why Proper Storage Matters
Octafluoro-2-butene isn't something most folks bump into outside specialty industries. Folks working with it, though, know this stuff requires some real care. It’s a colorless gas that’s heavier than air and, if you’re not paying attention, can leak or collect in low spots. Safety data from chemical manufacturers underline that inhalation brings fast-onset symptoms like dizziness or headaches. Eyes and skin don’t take well to direct contact. There’s a genuine risk sitting in plain sight: not handling or storing it right can lead to leaks or unwanted chemical reactions. Those risks don’t just stay in the lab – they follow folks home, too.
Getting Storage Right
Storing chemicals rarely means stacking them out of traffic and locking the door. I learned early on in technician training that temperature swings push pressure levels up and down, which can stress tanks past safe limits. Octafluoro-2-butene works best in cylinders or pressure-rated containers rated for low temperatures. It isn’t flammable, but the gas can displace oxygen. Store it in a cool, well-ventilated place—no crowded closets or warm machinery rooms. Regular checks on valves, seals, and gauges prevent surprises. If you see any corrosion or stains, swap the part and log it, rather than hoping for the best.
Handling With Care
A mistake with toxic gases sticks with you, long after the cleanup crew packs up. Protective gloves and goggles protect exposed skin and eyes, and folks should suit up with a lab coat or coveralls. Work should happen in fume hoods or spaces with spot ventilation so the air always flows out and away—not swirling around your head. I’ve seen people rush to refill or adjust containers without checking their equipment. Regulator fittings and hoses deserve checks every single time. Gasket leaks or loose couplings won’t give you a second chance.
Emergency Planning Isn’t Optional
No one expects an accident, yet stories ripple across industries about small leaks escalating quickly. I once watched a demo where a minor hiss at a valve triggered a panic—luckily, protocols worked just like the safety officer promised. Facilities should install gas detectors for this reason. Training folk on how to turn off the main lines, use emergency showers, and alert responders all feed into good practice. Spill kits and fresh air masks need to be right at hand, never tucked away or locked up for “just in case.”
Reducing Risks and Building Culture
Some companies put safety talks front and center, reviewing labels, storage rules, and proper disposal steps before anyone cracks open a cylinder. Experience shows that routine, not reaction, makes the difference. OSHA and EPA guidelines both stress a cradle-to-grave approach to handling chemicals. That means tracking deliveries, monitoring usage, and logging every disposal step, not glossing over paperwork because a clock runs late.
People will always look for shortcuts under pressure, yet the real fix relies on slow, steady effort. Safe storage and handling of octafluoro-2-butene aren’t just regulatory hoops—they cut down on injury, environmental harm, and hefty bills that follow after a chemical mishap. Every person in the chain, from delivery to disposal, holds a piece of the puzzle. Experience, teamwork, and a willingness to speak up make the job safer for everyone.
What are the physical properties of Octafluoro-2-Butene?
What Sets Octafluoro-2-Butene Apart
Working with chemicals like Octafluoro-2-butene, I learned how essential it is to know what you’re handling before you uncap a single cylinder. This compound stands out because it packs eight fluorine atoms onto a simple butene backbone. The presence of all that fluorine changes everything – not only for reactivity but also for basic physical traits you notice in the lab.
Appearance, State, and Odor
Most folks don’t expect a gas from a compound that bears such a hefty name, but at room temperature, Octafluoro-2-butene comes as a colorless gas. No dramatic odors, just a faintly sweet scent that might only get noticed in a very concentrated spill. The lack of color or warning smell has caught people off-guard, especially if procedures for leaks become too casual.
Boiling and Melting Point: How It Handles Temperature
In practice, you can watch Octafluoro-2-butene change state with a little pressure or a drop in temperature. Its boiling point hangs around -4 to -5°C, so you won’t see it as a liquid unless you chill things close to the freezing point of water or raise the pressure. In a busy plant, that makes storage and transfer more challenging, since you’re working with pressurized cylinders much like a refrigerant. The melting point lands below -80°C, far below most lab freezers. This property means you never wind up with unwanted clumps or crystals during handling, which helps keep things running smoothly.
Density: Heavier Than Air
In the warehouse, density sets the tone for safety planning. Octafluoro-2-butene weighs in at roughly 1.47 grams per cubic centimeter in its liquid state and carries a vapor density significantly heavier than air. If a leak occurs, this gas collects in low spaces. I’ve seen teams overlook this and later need to rethink ventilation and detector placement. This is not a gas to take lightly around confined spaces; it hugs the ground and displaces oxygen before you even notice.
Solubility and Stability: Interacting With the Environment
This compound resists dissolving in water, which matches expectations given the non-polar nature of fluorinated hydrocarbons. Instead, it mixes better with organic solvents. That low solubility in water limits how much accidental spills spread into groundwater, but any release into the environment remains tough to remediate due to chemical persistence. With standard handling, Octafluoro-2-butene stays stable, resisting breakdown, and does not spontaneously ignite or corrode metal tanks.
Uses and Handling: Safety At the Forefront
Octafluoro-2-butene shows up where stability under severe conditions is a must — for example, as a specialty refrigerant, reaction intermediate, or in niche electronics processes. Its chemical inertia makes it valuable, but also hard to clean up. Engineering controls, proper ventilation, and clear labeling in all storage and transport areas must remain a top priority. Personal experience reminds me that regular detector checks, floor-level gas monitors, and working in pairs help prevent small leaks from becoming big incidents. Small changes in storage protocol make a noticeable difference in both efficiency and safety.
Risk and Responsibility: What Matters Most
Octafluoro-2-butene isn’t a household name, yet its physical properties demand respect from anyone in the chemical field. All the high-end lab analytics or glossy MSDS sheets won’t make up for slack habits or fuzzily defined safety routines. Solid knowledge, routine verification, and a healthy suspicion that gases can settle where you least expect them go further than any manual.
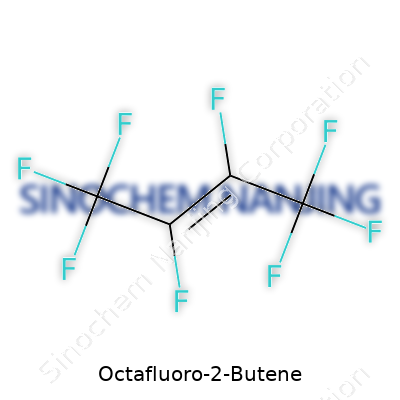
| Names | |
| Preferred IUPAC name | 2,3,3,4,4,4-Hexafluorobut-2-ene |
| Other names |
2,3,3,3-Tetrafluoro-2-(trifluoromethyl)prop-1-ene Octafluoro-2-butene 1,1,1,4,4,4-Hexafluoro-2-butene C4F8 |
| Pronunciation | /ˌɒk.təˌfluː.rəʊ.tuːˈbjuːtiːn/ |
| Identifiers | |
| CAS Number | 307-36-0 |
| Beilstein Reference | 1854382 |
| ChEBI | CHEBI:139858 |
| ChEMBL | CHEMBL1631462 |
| ChemSpider | 123153 |
| DrugBank | DB16672 |
| ECHA InfoCard | 03c1d423-b35a-4f05-86bb-9024655cb682 |
| EC Number | 601-798-6 |
| Gmelin Reference | 61336 |
| KEGG | C19392 |
| MeSH | D000077581 |
| PubChem CID | 9910429 |
| RTECS number | RZ2060000 |
| UNII | K8B5A2Q0WP |
| UN number | UN3163 |
| CompTox Dashboard (EPA) | DTXSID10917782 |
| Properties | |
| Chemical formula | C4F8 |
| Molar mass | 180.04 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 1.51 g/cm3 |
| Solubility in water | Insoluble |
| log P | 1.99 |
| Vapor pressure | 99.11 kPa (at 20 °C) |
| Acidity (pKa) | pKa ≈ -8.2 |
| Basicity (pKb) | pKb > 10 |
| Magnetic susceptibility (χ) | -53.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.252 |
| Viscosity | 1.06 cP (25°C) |
| Dipole moment | 2.39 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 354.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –1340.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2307 kJ/mol |
| Pharmacology | |
| ATC code | |
| Hazards | |
| GHS labelling | GHS02, GHS04 |
| Pictograms | GHS04 |
| Signal word | Warning |
| Hazard statements | H280: Contains gas under pressure; may explode if heated. H351: Suspected of causing cancer. |
| Precautionary statements | P210, P261, P271, P304+P340, P403 |
| NFPA 704 (fire diamond) | Health: 1, Flammability: 4, Instability: 2, Special: |
| Flash point | -37 °C |
| Autoignition temperature | 285°C |
| Lethal dose or concentration | Lethal dose or concentration (LD50/LC50): Inhalation LC50 rat 2000 ppm/4H |
| LD50 (median dose) | LD50 (median dose): >5000 mg/kg (rat, oral) |
| NIOSH | KZC042500 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Octafluoro-2-Butene: Not established |
| REL (Recommended) | Not established |
| Related compounds | |
| Related compounds |
cis-1,2,3,3,4,4,4-Heptafluoro-1-butene 1,1,1,4,4,4-Hexafluoro-2-butyne Perfluoro-2-butene Octafluorocyclobutane 1,1,2,2,3,3,4,4-Octafluorobutane |

