Octabromobiphenyl: Deep Dive into an Old Flame Retardant
Historical Development
Looking back, the story of octabromobiphenyl jumps straight out of the mid-20th century push for fire safety in consumer products. Concerns over house fires and accidents led to a huge demand for flame retardants that could slow down or stop ignition in everyday items, from electronics to plastics. Manufacturers chased after solutions that could easily blend into polymer matrices without wrecking their performance. Octabromobiphenyl, a polybrominated biphenyl, emerged from this race, gaining popularity through the 1970s, especially in electrical components. Its structure—a biphenyl backbone fully loaded with bromine—made it stand out, not just for effectiveness but also for persistence in the environment. The rush for safer homes and workplaces gave it a spotlight, but that attention brought scrutiny, and so the octabromobiphenyl journey took some unexpected twists as science caught up with safety and toxicity concerns.
Product Overview
Octabromobiphenyl carries a heavy bromine load, which explains why it works so well in applications demanding fire resistance. The substance features two benzene rings joined together with eight attached bromine atoms. People in the plastics and electronics industries once leaned into its capabilities to enhance fire safety rules, seeing it as a straightforward additive for thermoplastics. Over time, though, attention shifted from its technical convenience to larger environmental consequences as research exposed the downside to wide usage. This chemical rarely appears in products today, mostly due to regulatory restrictions and its naming on several watch lists, but learning its story sheds light on how industry, safety, and environmental health all collide.
Physical & Chemical Properties
Octabromobiphenyl usually comes as a white or off-white powder, sometimes with a pale yellow tinge. Its most striking feature is its low solubility in water and most organic solvents, which means it hangs around in the environment and in products long after its job is done. Melting point statistics have consistently shown high thermal stability—key for flame retardants—because once you hit the high hundreds in temperature, the compound starts breaking down. That thermal durability makes it tough on fires but also tough to break down naturally, which creates headaches for safe disposal.
Technical Specifications & Labeling
Technical specifics for octabromobiphenyl always point to its molecular formula—C12H2Br8—and a molar mass over 943 g/mol. As manufacturing ramped up in past decades, labeling requirements typically included its hazardous classification and personal protective equipment recommendations. Labels flagged the substance as a potential persistent organic pollutant and included pictograms signaling toxic hazards. Since regulatory measures have tightened, you’re not likely to run into this compound outside of reference libraries or legacy equipment, but the emphasis on clear, hazard-aware labeling remains important for anyone handling archived material.
Preparation Method
Octabromobiphenyl comes from direct bromination of biphenyl. Industrial synthesis involved treating biphenyl with elemental bromine in the presence of a catalyst like iron, under controlled conditions to avoid overbromination or undesirable side products. The bromination reaction is exothermic. Factories took great care to optimize temperature and pressure, chasing after a high yield of the desired octa-substituted product. Purification usually required a solvent extraction or crystallization step, which took time and careful adjustment of reaction parameters. Years of chemical manufacturing lessons came together in perfecting these reaction conditions, yet the resulting environmental and human health risks changed how people weighed the value of this chemistry.
Chemical Reactions & Modifications
For the most part, octabromobiphenyl doesn’t engage in many everyday reactions because of all those bromine atoms—those bulky halogens protect the molecule and slow down further chemical attacks. Burning or high-temperature treatment cause it to degrade, but that process releases a range of hazardous byproducts, including brominated dioxins and furans. Some academic groups have tried breaking it down with advanced oxidation or dehalogenation techniques, but these approaches come with logistical hurdles and haven’t seen broad adoption outside of specialized remediation projects.
Synonyms & Product Names
Octabromobiphenyl has picked up a handful of names over its commercial life, including “BB-209” and “OBBP.” Chemists sometimes reach for the more systematic “1,1′:4,4′:2,2′:3,3′:5,5′:6,6′-octabromobiphenyl.” Old technical sheets and patents may refer to it simply as octaBB or via trade names owned by long-gone manufacturers. These different aliases reflect how products migrate between research labs, factories, and regulatory watchlists.
Safety & Operational Standards
Handling octabromobiphenyl calls for a heavy dose of caution. The compound’s persistence and potential health risks make it a hazard in occupational settings. Anyone working with it, even in tracing legacy contamination, needs gloves, eye protection, and fume hoods to control dust and accidental inhalation. Waste protocols demand secure, high-temperature incineration, and many regional regulations now treat octabromobiphenyl as hazardous waste. Past industrial accidents offer a reminder: slow responses and loose protocols have led to environmental cleanups that spanned decades, underlining the need for airtight operational standards.
Application Area
Earlier decades saw octabromobiphenyl loaded into thermoplastics, particularly in housings for electronics—think televisions, insulation panels, or wiring sheaths. Its fire retardance made it popular among manufacturers trying to meet stricter insurance and safety codes after high-profile fires. Automobile interiors and certain building materials also benefited from its properties before substitutes with lower toxicity and higher environmental compatibility took over. Now, its legacy is mostly found through contamination in old products or soils near legacy manufacturing sites, with clean-up efforts driven by public pressure and regulatory science.
Research & Development
Much of the research activity around octabromobiphenyl now focuses on environmental tracing, toxicity mechanisms, and cleanup techniques. Scientists track its breakdown products, movement in soil and water, and bioaccumulation in the food chain. Early R&D prioritized better flame retardance, but later work turned toward structure-toxicity relationships and safer alternatives, driving innovation in greener chemicals. In labs, the molecule serves as a test case for understanding persistent organics and building tools for risk assessment. It’s often cited as a warning for what happens when toxicology lags behind scaling up commercial usage, pushing today’s researchers to develop flame retardants that solve one problem without creating two more.
Toxicity Research
Health studies highlight a stark reality. Octabromobiphenyl can build up in tissues and stick around for years in both humans and animals. Workers exposed during production have shown increased risks for liver and thyroid issues, reproductive harm, and even certain cancers. Animal studies back up those findings—higher brominated biphenyls disrupt hormone systems and neurological development. Environmental researchers keep digging into how these compounds move through sediment, water, and wildlife, linking contamination hot spots to historic dumping or outdated industrial practices. The compound’s sheer persistence means even a ban can’t erase its impact overnight, showing the need for vigilant long-term monitoring and remediation.
Future Prospects
Octabromobiphenyl’s story offers a hard-won lesson. Industry once hailed it as a fire safety fix, but the ensuing environmental and public health hangover shifted the calculus. Its future lies less in new production and more in legacy management: tracking residues, restoring contaminated sites, and supporting the transition to safer alternatives. Researchers focus on developing and deploying remediation technologies that break down persistent organic pollutants, whether through advanced thermal techniques or new chemical processes. Policy work continues as environmental watchdogs keep pressing for global phaseouts and tougher controls on similar substances. In the bigger picture, the octabromobiphenyl saga pushes chemists and manufacturers to look beyond short-term technical gains, centering safety, sustainability, and transparency at every step.
What is Octabromobiphenyl used for?
The Story Behind Octabromobiphenyl
Octabromobiphenyl belongs to a group known as polybrominated biphenyls (PBBs), chemicals that took center stage in the drive to make our homes and workplaces safer from fire. In the ‘70s and ‘80s, the world looked for new ways to reduce the risk of deadly blazes in electronics and household goods. PBBs like octabromobiphenyl held the promise of safety, adding a powerful barrier against flames to everything from plastics in TVs to circuit boards in office machinery.
My father worked maintenance at an old electronics plant upstate, and I remember the harsh smell wafting from the molding area. He explained the chemicals helped electronics not catch fire so easily, but even back then, the guys on the floor wore masks and gloves as much as possible. It wasn’t only about safety from fire—it was about staying healthy, too.
What Makes Octabromobiphenyl a Problem?
Manufacturers leaned on octabromobiphenyl for its flame-fighting reputation, blending it into plastics, appliances, and sometimes building materials. Unfortunately, these chemicals don’t just stay where they’re put. Over time, they leach out of products and drift into dust, soil, and even food sources. Because they linger for decades, they settle into ecosystems and, more troubling, into our bodies.
Years of research connected PBBs, including octabromobiphenyl, with a long list of health worries. Michigan, in the 1970s, saw a massive agricultural contamination. Entire herds had to be destroyed. People living near those farms reported rashes, hormonal issues, and worse. Studies today link PBBs to liver damage, issues with immune systems, thyroid disruption, and even certain cancers.
Why This Still Matters
Brominated flame retardants, like this one, stick around in older computers, TVs, even insulation inside our walls. Governments in North America and Europe have phased out their use, but traces remain, especially where old waste gets recycled or dumped carelessly. Growing up, my family always threw out old electronics with the garbage, never thinking of toxic leftovers lingering in landfills or drifting down wind.
Communities pay the price later. Waste sites often stand near towns or rivers. Chemicals used in products decades ago seep into soil or water, showing up in fish, wildlife, and—eventually—our dinner plates. Once in the body, they’re tough to clear out. Childrens’ developing bodies, in particular, face greater risk. Studies show kids’ blood can still contain measurable levels, especially in communities with long industrial legacies.
What’s Next?
Switching away from toxic halogenated flame retardants remains a priority. Newer fire-resistant materials, like certain minerals and safer polymers, have dropped into the market. Companies need strong oversight to prevent a new generation of “safer” chemicals causing the same old problems. Recycling old electronics responsibly, instead of letting them pile up in landfills or shipped to developing countries, cuts risk for everyone.
People have a role here, too. Learning about what’s inside the items we use, pushing for transparency, and backing recycling programs keeps the pressure on makers and lawmakers alike. Octabromobiphenyl shows what happens when safety takes a shortcut. Protecting our families starts with not forgetting these hard lessons.
Is Octabromobiphenyl hazardous to health or the environment?
Where It Shows Up
Octabromobiphenyl, often just called OBB, landed in factories and electronics as a flame retardant. Plenty of older office equipment, plastics, and foam-filled chairs contain it. The message from its early years: make products safer, stop fires, and protect homes or workplaces. Over time, though, more people started looking at what this chemical does outside the lab.
Health Isn't Just a List of Symptoms
I remember stories from workers at electronics plants in the '80s. Hands chapped by new compounds, headaches that wouldn’t go away, and odd stomach aches. Back then, science wasn’t catching every culprit, but OBB sat on the list of suspects in studies linking certain flame retardants to nerve or liver issues.
Research into similar chemicals points to some big problems. The body doesn't flush these compounds out quickly. They build up in fat tissue, in mother’s milk, and even in the blood of infants. With OBB, that bioaccumulation brings real worries. Populations near dump sites or manufacturing plants report reproductive problems and possible immune issues. It’s not just about the person in the factory; these compounds slip into food chains and water supplies.
The Environment Doesn't Hit 'Reset'
Walking along the edge of a landfill near my old apartment, I once found coils of old wiring and scorched plastic housing. No obvious fire, just the leftovers of things with nowhere to go. Studies back up what was obvious that day: OBB sticks around in water, sediment, dust, and soil for decades. Fish take it up in rivers near industrial zones. Birds eat fish, then predators eat birds. That entire chain absorbs a chemical that only gets more concentrated up each link.
North America's Great Lakes show what happens when persistent substances like OBB get loose—large fish with so many toxins in their flesh that some health agencies warn against eating them often. Scientists have even detected these retardants in polar bears, hundreds of miles from any likely source, carried by winds and water currents.
Regulation and The Search for Alternatives
Europe banned OBB in the mid-2000s after multiple studies. The United States and other countries followed with restrictions or outright bans. Even so, the stuff that made it into sofas, TVs, or office carpets didn’t just disappear. Many homes and junkyards still hold these chemicals. New flame retardants have taken center stage, but some of those run the same risks, creating a cycle that drags on.
Fixing this means we can't just swap one mystery chemical for another. Manufacturers need to show their replacements really pose less risk before shipping them out en masse. Workers deserve stronger protections from dust and fumes. Household electronics need clearer labeling about old chemicals used inside.
Communities living near waste sites call for real clean-ups, not just paperwork or press conferences. Air and water testing could alert families to danger before someone gets sick. That early warning could make all the difference, since prevention always beats cleanup.
Everyday Choices Matter
Sometimes, the best step comes down to people getting informed. I always check old labels on electronics at thrift shops and avoid suspicious plastic foam in old furniture. Local recycling programs—if they exist—make a dent in bringing these relics out of circulation. Conversations with neighbors about household waste add up. Each little effort helps tame a problem that once felt like someone else’s responsibility.
What are the safety precautions for handling Octabromobiphenyl?
Recognizing the Risks
Octabromobiphenyl has a track record as a flame retardant, but its history doesn’t shine. It sticks around a long time in the environment, and it moves up the food chain, which means people and wildlife can get exposed. I'm not a stranger to stories of chemicals that caused harm in factories and in homes, so this is something to take seriously. Exposure carries real health concerns—skin irritation, impacts on the nervous system, possibly even cancer over years. Keeping folks safe calls for practical steps, not just checklists on a wall.
Choosing the Right Protection
Working with powders and dust means you can’t trust your eyes and nose to warn you. I always reach for gloves made from nitrile or neoprene. They give your hands a barrier. Long sleeves and full pants become the uniform, and tossing on a disposable lab coat helps stop anything from hitching a ride home. I keep goggles handy—for me, a face shield works best if things get splashy. If you’re in an area with shaky ventilation, I go for a P100-rated respirator. It grabs fine dust particles before they reach my lungs.
Ventilation and Air Quality
Fresh air counts more than most folks realize. Setting up a fume hood or local exhaust system keeps dust out of breathing spaces. Lab tests I’ve seen show that a closed-off room without airflow lets these tiny particles settle everywhere. Open windows don't fix the problem on their own. Equipment that captures air near where you work, then pulls it through a HEPA filter or straight to the outside, makes a big difference. I check that fans run before opening a container. It seems basic, but it cuts risks sharply.
Safe Storage and Spill Cleanup
I always choose a sturdy, labeled container for anything hazardous. Keep Octabromobiphenyl in a cool, dry storeroom with clear signs. It doesn’t belong on open shelving or next to anything combustible. Spills get handled with care—never sweep or use a dry broom. That turns dust into airborne threats. Wet methods work: I use damp cloths or a vacuum fitted with filters built for hazardous dust. Afterward, I bag everything in double-sealed trash and flag it for proper disposal. The rules aren’t just paperwork; they keep everyone from seeing a small hiccup turn into a big mess.
Reducing Exposure Outside Work Areas
Most accidents I hear about happen after work ends—on doorknobs, in cars, or at home. I set aside time to wash hands and arms up to the elbows, even if gloves stay on the whole time. I toss work clothes in a separate bag for laundry; they don’t mix with regular clothes. These steps keep the stuff from sneaking into kitchens or bedrooms.
Training and Communication
Having the best safety gear in the world won’t help if folks don’t know how to use it. Every time I started a job with a tricky chemical, the best introduction came from stories of what can go wrong and what to do right. I check in with coworkers before starting any tricky task. Pointing out hazards as they come up, plus regular refreshers, build habits stronger than any printed manual. Real safety comes from everyone pitching in and paying attention, not just one person in charge.
What are the physical and chemical properties of Octabromobiphenyl?
The Look and Feel of Octabromobiphenyl
Octabromobiphenyl walks into any lab as a pale, almost ghostly powder. The dust lingers long enough on gloves to make you reach for a new pair. It holds together until a gust or a fumble sends a light haze rising — never pleasant, but easy to spot. I once saw a chemist mention the solid is heavier than winter road salt. He was right. Its density packs over 2 grams per cubic centimeter, which gives it a weighty presence for its size.
You won’t catch a whiff of any scent from it. No pungent tang or offputting odor signals danger. That changes once heat comes in, though. At a little under 380°C, the substance starts to give off fumes and breaks down well before anything like melting happens. It can’t handle a direct flame and definitely should stay away from unventilated workspaces.
The Building Blocks
The real intrigue comes from the eight carefully added bromine atoms. Their presence fully transforms the biphenyl backbone. I remember my first research paper covering organobromines; bromine-stuffed molecules always showed greater unwillingness to dissolve in water. True here, too. Octabromobiphenyl resists every attempt by water to thin it out. Drop it in and you’ll get a film that sits on top instead of swirling away. Only powerful organic solvents — especially those with a taste for halogenated compounds — can handle the job. Toluene or dichloromethane usually do the trick.
Those bromines aren’t just for show. Their bulk and electronegativity help prevent the molecule from burning easily. That’s why industries once leaned so heavily on these chemicals for fire suppression in plastics and wiring casings. Take the flame away, and you kneecap the risk of electrical fires.
Tough By Design
Octabromobiphenyl laughs at microbes and does not budge under sunlight. Incineration or strong acids are about the only forces that break it down with any speed. This persistence earned flame retardants like this a wide berth in plastics and textiles made in the last century. But the features that make it survive everything also create real headaches for soil and water if it leaks out. It clings to dust, settles in sediment, and breaks down too slowly for comfort.
Health Hazards Behind the Science
Every solid with eight bromines comes attached with ethical baggage. Its resistance to fire also means resistance to metabolic breakdown in people and animals. I learned early in my chemical safety training: treat halogenated biphenyls with real caution. Studies have linked exposures in factories — sometimes through handling or inhaled dust — to developmental delays, hormone disruption, and even cancer. The world’s health researchers spent years following up on the problems, and the evidence got strong enough to push for sharp restrictions in places like the EU and US.
Better Ways Forward
It’s impossible to ignore octabromobiphenyl’s legacy. Many products already out there still shelter these molecules deep inside. The solution comes in finding alternatives that scrub out fire risks but skip the lifetime persistence and health fallout. Some new flame retardants turn out less toxic and more willing to break down, at least under industrial composting or with sunlight and air. Switching manufacturing over remains expensive and complex, not every company makes the leap fast enough. Still, the message from toxicology is clear: take care with what’s added to plastics, push for safer substitutes, and invest in cleanup for the spots left behind.
Is Octabromobiphenyl regulated or banned in any countries?
Looking Closely at Octabromobiphenyl
Octabromobiphenyl, a mouthful to say, once played a pretty big role as a flame retardant in everyday products. Think plastics for electronics, cables, and other things you might find in an older office building or inside aging gadgets. Run through lab tests and scissors of regulators, scientists kept noticing its persistence in the environment, and that spilled over into worries about health effects.
Regulation and Bans in Different Countries
Europe doesn’t waste time with chemicals that raise red flags. The European Union entirely banned octabromobiphenyl under the POPs (Persistent Organic Pollutants) Regulation and included it on the REACH list—one of the world’s strictest chemical control regimes. This isn’t just paperwork: under these rules, companies can’t make, sell, or even use products containing the substance unless they’ve got an exception, which almost nobody does for this compound.
The United States stopped new production or import of octabromobiphenyl decades ago. The EPA took a clear stance as early as the late 1970s, after animal studies showed links to liver tumors and changes in the immune system.
Japan, Canada, and Australia also treat it as too risky for any new use. In those places, strict bans or restrictions mean you won’t find new electronics or furniture featuring it. Countries that signed on to the Stockholm Convention—a global treaty dealing with persistent pollutants—made an agreement to phase it out because it crosses borders in the environment and keeps turning up in wildlife and people.
Why Does All of This Matter?
I remember seeing the ways chemicals like octabromobiphenyl, used for just a bit of added fire safety, echo far beyond the factories that first mixed them. The issue just keeps getting bigger: These substances don’t break down quickly. Birds pick them up in far-off marshes, and they can sneak into breast milk or turn up in fish thousands of miles from wherever somebody melted down some plastic. It isn’t only a problem for the heavily industrialized countries.
Health impacts never stand still. Studies over the years found that it messes with hormones, may cause cancer, and lingers in our fatty tissues. Europe’s willingness to outlaw it, and America’s early retreat, didn’t wait for every bit of possible proof. Governments saw enough to switch off the tap.
Searching for Better Answers
Looking around at safer alternatives, manufacturers now have big incentives to pick different flame retardants, or even drop chemical treatment altogether in some cases. Sometimes the very design of the product lessens fire risk, dodging the trade-off between safety and toxicity. That’s the direction electronics and furniture makers go now, steering clear of legacy chemicals with shadows hanging over them.
Still, plenty of older items stay in use worldwide, especially where money is tight and regulations lack teeth. It takes concerted action to phase these things out—regular inspections, education, and affordable pathways to safe disposal. When recycling, electronics can leak old fire retardants unless the process stays tightly controlled.
Octabromobiphenyl’s story feels familiar: A technical fix that looked clever at first, only to stick around where it’s neither wanted nor useful. Bold bans and firm regs turned the page. The lesson that sticks with me is that taking early evidence seriously—acting before disaster piles up—ends up protecting everyone’s health and environment.
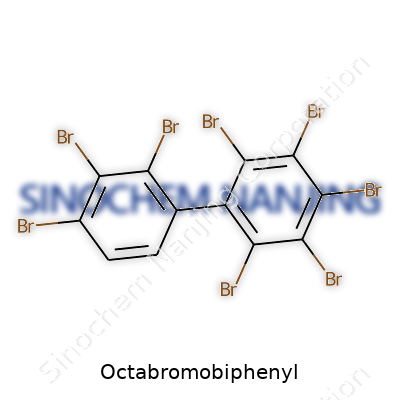
| Names | |
| Preferred IUPAC name | 3,3',4,4',5,5',6,6'-Octabromo-1,1'-biphenyl |
| Other names |
Biphenyl, octabromo- Octabiphenyl bromide Octabromodiphenyl |
| Pronunciation | /ˌɒk.təˌbroʊ.moʊ.baɪˈfiː.nɪl/ |
| Identifiers | |
| CAS Number | 27858-07-7 |
| Beilstein Reference | 12097894 |
| ChEBI | CHEBI:34733 |
| ChEMBL | CHEMBL430238 |
| ChemSpider | 17687 |
| DrugBank | DB13928 |
| ECHA InfoCard | 100.030.088 |
| EC Number | 251-087-9 |
| Gmelin Reference | 38808 |
| KEGG | C10968 |
| MeSH | D010025 |
| PubChem CID | 11907 |
| RTECS number | TD9275000 |
| UNII | 47C7M16X6F |
| UN number | UN3077 |
| CompTox Dashboard (EPA) | DTXSID4036792 |
| Properties | |
| Chemical formula | C12Br8 |
| Molar mass | 687.427 g/mol |
| Appearance | White to off-white powder |
| Odor | Odorless |
| Density | 3.36 g/cm³ |
| Solubility in water | insoluble |
| log P | 6.8 |
| Vapor pressure | 1.32E-8 mmHg at 25°C |
| Acidity (pKa) | 18.51 |
| Magnetic susceptibility (χ) | -1060·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.760 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 560 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -497.6 kJ/mol |
| Pharmacology | |
| ATC code | V09AM04 |
| Hazards | |
| Main hazards | May cause cancer. May damage fertility or the unborn child. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. Harmful to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS06, GHS08 |
| Signal word | Danger |
| Hazard statements | H351: Suspected of causing cancer |
| Precautionary statements | P280, P308+P313, P273, P305+P351+P338, P501 |
| NFPA 704 (fire diamond) | 1-2-0-NA |
| Autoignition temperature | 400 °C |
| Lethal dose or concentration | Lethal dose or concentration (LD50/LC50): LD50 (oral, rat): >10,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral >15 g/kg |
| NIOSH | SA9350000 |
| PEL (Permissible) | PEL: 5 mg/m3 |
| REL (Recommended) | 0.5 |
| IDLH (Immediate danger) | IDLH: 5 mg/m3 |
| Related compounds | |
| Related compounds |
Polybrominated biphenyl Hexabromobiphenyl |

