O-Nitrobenzenesulfonic Acid: An Experienced Look at a Specialty Chemical
Historical Development
O-Nitrobenzenesulfonic acid has a story going back over a century. Chemists in academic labs worked with sulfonic acids and nitroaromatic compounds before many major industries even existed. By the early twentieth century, practical routes for adding a nitro group to a benzene ring were already standard fare. Introducing a sulfonic group in concert transformed the baseline chemistry, pushing lab and pilot-plant efforts beyond simple batch reactions. Over time, production shifted from classic batch methods to continuous innovation—engineering controls got better, reaction conditions more tightly managed, and awareness of byproduct management started shaping how these chemicals found their way onto workbenches and into industry catalogues. In graduate school, friends worked on related derivatives, often talking about how this family of molecules opened doors to other transformations. O-Nitrobenzenesulfonic acid became a recognizable name for those training in organic synthesis, not just for its own features, but also for the history built alongside its chemistry.
Product Overview
The acid sits as a strong, niche reagent with roles that show up in dye manufacturing, pharmaceuticals, and research-scale organic synthesis. Not every scientist keeps it stocked, but when a synthesis calls for a sturdy, electron-withdrawing group paired with a sulfonic acid, this compound lands on the shortlist. Its performance as a coupling agent or intermediate helps break new ground in both academic articles and commercial projects. As markets for advanced materials and specialty dyes grew, demand for unique aryl sulfonates increased too. The consistent behavior in classic tests—good stability under normal working temperatures, distinct color for easy monitoring—convinces many chemists to use it where specificity counts. Communities working in aromatic substitution or charged dye chemistry have come to rely on its predictable profile to move projects forward.
Physical and Chemical Properties
O-Nitrobenzenesulfonic acid stands out as a pale yellow to yellow-brown crystalline solid. Its structure brings together two powerful groups—one nitro, one sulfonic—attached to a benzene core. It dissolves well in water, which isn’t always the case with nitroaromatics. Measuring out the powder never gets old: the faint, distinctive aroma and quick clumping around moisture in the air remind you of its sulfonic roots. The molecule doesn’t catch fire easily, but strong acids and oxidizers must be kept out of the way—side reactions can get messy. Its melting point and solubility make purifications a little easier, at least compared to trickier analogues. I remember during undergraduate research, we checked absorbance at precise wavelengths: its pale color doesn’t shout, but enough to mark its presence in solution. Its reactivity profile means it holds its own against base but submits quickly to strong reducing agents.
Technical Specifications & Labeling
Labs handle O-nitrobenzenesulfonic acid with clear, consistent standards. Chemical suppliers print hazard codes and regulatory information up front because nobody wants mistakes in inventory or benchwork. The compound comes labeled with warnings about skin and eye contact, instructions for safe disposal, and clear molecular data—no guesswork for researchers swapping bottles mid-experiment. Technical data sheets typically state purity, batch number, and spectral verification. Over time, as regulatory compliance deepened, I noticed an uptick in detail: accurate labeling and traceable batch history reduce headaches for both quality assurance teams and the grad students doing the work.
Preparation Method
Real-world production of O-nitrobenzenesulfonic acid usually involves direct sulfonation or nitration of benzene derivatives. In practice, companies and research labs choose a path based on what’s easier to scale or what suits the downstream chemistry. Sulfonation before nitration can manage regioselectivity; other times, chemists find benefit in timed addition and temperature control to nudge the yield higher for the ortho isomer. Bringing a nitro group onto already sulfonated benzene means strict temperature regulation and close management of acid concentrations—cutting corners leads to tars, byproducts, and wasted time. My own experience running similar reactions confirmed that watching every parameter pays off. Work-ups often include careful neutralization, followed by crystallization to true purity. Where scale allows, continuous flow chemistry is starting to find a home, especially for controlling exotherms and product isolation.
Chemical Reactions & Modifications
This acid acts as a lynchpin in many transformations. Its nitro group primes the ring for further reductions, leading to amino-sulfonate compounds—an important class for dyes and pharmaceuticals. The sulfonic acid itself provides a handle for ion exchange, salt formation and, in the right conditions, displacement reactions. I’ve run reductions with iron and acid during internships to yield amino derivatives ready for further chemistry, sometimes pushing into azo dye routes. It rarely acts alone: teams develop modifications to attach side chains, convert functional groups, or insert radioactive isotopes for tracing in medical research. Wherever precision in regioselective transformation matters, O-nitrobenzenesulfonic acid provides an entry point.
Synonyms & Product Names
Ask for O-nitrobenzenesulfonic acid in different countries or labs, and names shift a bit. Many call it ortho-nitrobenzenesulfonic acid, others abbreviate it as O-NBSA. The synonyms track its IUPAC roots and industrial traditions—terms include 2-nitrobenzenesulfonic acid, or for the chemically inclined, 2-nitrobenzene-1-sulfonic acid. Some suppliers build brand nicknames around their own catalog, but the chemical backbone stays unchanged no matter what’s printed on the bottle.
Safety & Operational Standards
Chemists and plant operators take O-nitrobenzenesulfonic acid safety seriously. Any compound with a nitro group and sulfonic acid asks for careful PPE selection: gloves, goggles, good ventilation. Skin contact can bring irritation, and inhaling dust isn’t something anyone wants. Storage away from strong reducers and bases lands as a best practice. Disposal routes follow local hazardous waste rules—neutralization and controlled incineration, never down the drain. My own lab routines drilled in the connection between careful measurement and safe outcomes—one slip, and risk jumps. On the industry side, modern plants use contained mixing and transfer systems to keep workers safe and environmental releases minimal.
Application Area
Demand tracks with specialty manufacturing more than with bulk commodity chemistry. O-nitrobenzenesulfonic acid finds its major applications as a key intermediate in azo dye synthesis, as a precursor for substituted anilines, and in certain pharmaceutical syntheses. In my exposure to dye plant operations, the sulfonic acid groups anchor molecules onto fibers, improving wash resistance and vibrancy. Some medicinal chemistry groups explore these compounds for enzyme probing or as tagging agents. Analytical labs may use the acid as a standard or in derivatization protocols where a strong electron sink changes a molecule’s reactivity. Its sharp profile means it rarely ends up in consumer goods, but it influences the quality and reliability of ingredients in specialty formulations.
Research & Development
The research world keeps finding new wrinkles in an old compound. For a long time, the application for O-nitrobenzenesulfonic acid focused on dyes and colorants, but recent trends show rising interest in advanced material synthesis and pharmaceutical precursor work. Some university labs investigate catalytic reduction methods or green chemistry approaches to minimize secondary waste from sulfonation. Industry and academia often collaborate, particularly in developing more efficient routes for mass production or functional group switching. Experienced chemists look for more selective reactions—those that give the right ortho/para isomer ratio, or that handle increasing environmental regulations without driving up costs. Graduate students and postdocs compare notes across conference posters and Zoom seminars, sharing the tweaks that shave hours off syntheses or boost yields by a few percent. Open literature reflects this drive for improvement through hundreds of articles each decade.
Toxicity Research
Studying the toxicology of nitroaromatic sulfonic acids alerts operators and researchers to the need for vigilance. Acute toxicity studies and chronic exposure research inform the mandatory safety labels and wearing of protective equipment. Based on available case studies, ingestion or significant exposure to skin and eyes causes irritation or worse; inhalation adds another vector for harm. Several regulatory agencies monitor its environmental breakdown—slow for most nitroaromatics, and with potential byproducts that run afoul of discharge limits. My own readings in toxicology taught me to keep these risks front-of-mind: even low-volume specialties demand respect, not complacency. Environmental persistence and potential for accumulation shape operational rules in both research and manufacturing sectors.
Future Prospects
Looking ahead, the arc for O-nitrobenzenesulfonic acid aligns with trends in specialty chemicals. Green chemistry and environmental stewardship push for cleaner synthesis, higher atom economy, and safer byproducts. Real progress means moving beyond legacy approaches—better catalysts, solvent recovery, and real-time monitoring of emissions. Researchers developing new dyes and pharmaceuticals still find the acid essential for stepping into novel areas of aromatic substitution. Automated and flow chemistry platforms promise more consistent yields and fewer surprises, especially as batch-to-batch variability matters more for high-value products. Continued investment in research on toxicity and environmental fate will keep the compound viable for years to come, provided operators and scientists keep balancing innovation with responsibility.
What is O-Nitrobenzenesulfonic Acid used for?
O-Nitrobenzenesulfonic Acid: More Than Just a Chemical Name
O-Nitrobenzenesulfonic acid doesn’t show up in everyday conversation, but it holds real value in labs and factories. The chemical name sounds complicated, but the uses stand out for those working with advanced materials and sophisticated manufacturing. People who create dyes, pharmaceuticals, and specialty chemicals tend to know it well, as it plays a role at the root of many essential reactions.
The Role in Dye and Pigment Production
Anyone who works in dyes and pigments has seen O-Nitrobenzenesulfonic acid or its derivatives on ingredient lists. Chemists use it to introduce specific sulfonic acid groups to aromatic rings, making certain colors more stable or soluble. Companies that make dyes for textiles, inks, and plastics often turn to this compound for bringing out color consistency. Synthetic dye manufacturing sometimes feels mysterious to outsiders, but processes involving O-Nitrobenzenesulfonic acid have built their reputation on reliability and repeatable results.
Pharmaceutical Synthesis
It’s easy to overlook the amount of chemistry behind a tablet or a capsule. For some pharmaceutical makers, O-Nitrobenzenesulfonic acid appears early in production as a reagent that helps make other molecules more reactive. Taking the nitro group to sulfonate rings can set the stage for important chemical changes further down the manufacturing line. These transformations let researchers link complex molecular pieces together or fine-tune a drug's function. This has mattered to scientists chasing improved treatments, as the tiniest change in a chemical pathway sometimes leads to better medication for patients. Real-world results often come from these tweaks, even if the initial steps seem routine.
Replacement and Modification Chemistry
Chemical synthesis in industry isn’t only about following recipes. Often, the job calls for breaking a stubborn bond or replacing parts of a molecule with new ones. O-Nitrobenzenesulfonic acid acts as a scaffolding material during nucleophilic aromatic substitution and related reactions. It lets chemists swap, remove, or install functional groups across complex frameworks.
This versatility goes far beyond the lab. The expertise needed to handle these tools safely calls for strong oversight, with attention to workplace procedures and environmental standards. Handling nitro and sulfonic acid groups can raise hazards—personal protective equipment and waste treatment systems must stay up to date. A spill or misstep during use could pose dangers to workers or local ecosystems.
Environmental and Safety Challenges
Regulations around chemicals like O-Nitrobenzenesulfonic acid grow stricter each year, for good reason. I’ve seen smaller labs trip up on documentation and storage. Larger suppliers invest heavily in proper fume exhaust, neutralization tanks, and safety training. Real-world improvements in green chemistry sometimes start with reducing reliance on older, more toxic reagents or designing pathways with fewer hazardous by-products.
In my own experience, talking with process engineers reveals that seeking substitutes or refining waste handling always gets a seat at the table in product planning meetings. Some companies lead by innovating chemistry that cuts down environmental risks, using catalytic or enzymatic approaches as replacements where possible. Others push for better recycling of by-products from O-Nitrobenzenesulfonic acid reactions. It takes a blend of strong scientific knowledge, practical safety habits, and a willingness to invest in new technology.
What are the safety precautions when handling O-Nitrobenzenesulfonic Acid?
Why Attention Matters
O-Nitrobenzenesulfonic acid offers real utility in chemical labs, especially for introducing sulfonic groups and other advanced synthesis steps. Yet this usefulness comes packaged with danger. I remember my first encounter with it in a research setting—our safety training instructor didn’t mince words, making sure we all understood that missteps weren’t an option.
The compound hits you with a mix of reactivity and corrosiveness. Skin and eye contact triggers burns. Dust irritates lungs. Anyone handling materials like this has probably had those nervous moments, double-checking gloves and goggles. Carelessness leads to prompt reminders that chemical protection isn’t just a box on a checklist.
Basic Barriers: Gloves, Goggles, Lab Coats
Nitrile or butyl rubber gloves block most splashes and spills. Thin latex just doesn’t cut it. Safety goggles should wrap around, keeping fumes and droplets away from eyes. I once saw a peer skip sturdy goggles, only to rush for the eyewash after a splash—thankfully, he got help quickly, and nothing permanent happened. Lab coats should be buttoned up, not hanging open like a borrowed jacket. Avoid synthetic fibers—cotton resists melting if things go sideways.
Managing Air and Workspace
Ventilation counts for a lot. Fume hoods aren’t just furniture for storing glassware—they pull away acidic vapor and prevent buildup. Working in open air adds risk, since volatile droplets drift easily. Surfaces need lining with absorbent pads. In my experience, wiping up with paper towels won’t cut it if powder lands on a benchtop; it spreads invisible residue. Dedicated area signage keeps folks from wandering through where they shouldn’t.
Eye Wash and Emergency Showers: Not Optional
Quick response systems make the difference. I once asked if anyone had actually used the safety shower during an emergency—two senior researchers spoke up, both saying quick access prevented disaster after an accidental spill. Bottled eyewash stashed near the workbench saves crucial seconds. Knowing exactly where to find showers or eyewash fountains means less fumbling if a colleague needs help fast.
Storage and Spill Plans
Short-term storage works best inside dedicated acid cabinets, away from direct sun or heat. Containers shouldn’t touch incompatible chemicals like bases or strong oxidizers. Labels must match contents; hand-lettered tape leads to confusion. Everyone in the lab used to do a double-check at the end of each week—inspections spotted leaks or weak seals before they turned into safety issues.
If a spill happens, absorbent powder like sodium bicarbonate neutralizes acid. Never touch a spill with bare hands. Waste goes in acid-resistant bins, not standard plastic bags. I’ve seen what happens when these bins fill up too quickly—overflow strengthens the argument for disciplined waste removal, logged and sent to appropriate disposal teams.
Training: A Habit, Not a One-Off
Even veterans need refreshers. The minute things get rushed, shortcuts tempt, and someone forgets a step. Annual safety drills give everyone a chance to practice with gear, review protocol, and ask practical questions. Real-world experience—troubleshooting, responding to unexpected issues—matters more than perfect recall of written procedures.
Every accident prevented saves time, money, and trust within a team. Respect for chemicals like O-Nitrobenzenesulfonic acid doesn’t happen by accident; it grows from careful routine, planning, and sharing lessons learned.
What is the chemical formula and structure of O-Nitrobenzenesulfonic Acid?
Chemical Formula and Structure
O-Nitrobenzenesulfonic acid has the chemical formula C6H5NO5S. The structure comes out of a benzene ring with two key features: a nitro group (-NO2) linked to one carbon and a sulfonic acid group (-SO3H) tied to the carbon directly next to it. These groups line up in the ortho position, which means the nitro and sulfonic acid groups are neighbors on the benzene ring. Chemists often draw this molecule showing the benzene ring as a hexagon, with the -NO2 and -SO3H labels sticking out at adjacent corners.
This specific arrangement brings out unique chemical behaviors. The electron-withdrawing power of both groups makes the aromatic ring less reactive toward some typical benzene reactions, like electrophilic substitution. Getting the exact positions right is crucial in lab work, or you’ll end up with a mixture of isomers that give entirely different results.
Why Its Structure Matters in Real Life
In day-to-day lab practice, knowing a compound's structure helps in more ways than just drawing it on paper. O-Nitrobenzenesulfonic acid gets used regularly as a starting point for synthesizing dyes, pharmaceuticals, and specialty chemicals. Its sulfonic acid group boosts the water solubility of many products, making them easier to handle in industrial processes or medical applications. The nitro group, meanwhile, allows for further chemical tinkering—catalysts or reducing agents can turn this group into an amine, opening doors to entirely new families of compounds.
I’ve seen the convenience of these features play out on the lab bench. During grad school, adding the sulfonic acid group to a molecule often meant fewer headaches about limited solubility, especially in reactions needing water as a solvent. The nitro group, when sitting close to the sulfonic acid, changed the course of reduction steps, requiring milder conditions and giving better selectivity. These small changes matter a lot when scaling up reactions for industrial production or pharmaceutical research.
Key Challenges and Solutions
One issue with O-Nitrobenzenesulfonic acid centers on handling and storage. The acid is pretty strong and tends to attract moisture from the air. This property can make it clump up or start reacting in ways you didn’t intend. Clear labeling and storage in airtight containers solve most of these problems. The strong acid also means you’ve got to be careful mixing it with other chemicals—trying to neutralize the sulfonic acid without recognizing the powerful nitro group can lead to hazardous situations.
There’s also the matter of contamination. Purity counts in applications like pharmaceuticals or electrical dyes. Analytical testing like NMR or HPLC should be routine, not just a box to tick. Instrument access has become easier, so there’s no excuse for skipping basic purity checks.
Impact Beyond the Lab
Outside the bench, the dual nature of O-Nitrobenzenesulfonic acid shapes more than just chemistry. Its high reactivity has driven safer manufacturing protocols in factories and new waste treatment methods for sulfonic acid and nitro byproducts. Strict safety regulations now demand better training for people working with reactive organics. The real progress appears when companies share best practices—moving the industry forward one molecule at a time.
How should O-Nitrobenzenesulfonic Acid be stored?
Why Careful Storage Matters
O-Nitrobenzenesulfonic acid can stir up plenty of concern among chemists and lab managers for a simple reason: it’s both corrosive and reactive. Mishandling can lead to damaged equipment, injured workers, and contaminated environments. In daily life at a chemical facility, one spill often means lost time, an expensive clean-up, and risk to personal safety. My own experience with storing chemicals taught me quickly how even straightforward procedures prevent long-term headaches.
Common Risks with O-Nitrobenzenesulfonic Acid
This acid, with its sharp properties, will eat away at unprotected skin and even some plastics. Unsealed containers often result in fumes or leaks, posing not just a workplace hazard but also health problems for those nearby. If you’ve ever had to scrub down a spill, you know tight storage rules keep everyone safer.
Smart Storage Techniques in Real-World Labs
Use Sealable, Acid-Resistant ContainersStrong glass or certified plastic containers generally hold up best against corrosive acids like this one. Closures must fit tightly—replace a warped lid on sight, since even a tiny gap turns into a major headache overnight.
Label Everything ClearlyA smudged or missing label only invites confusion. Bringing a new technician onto a shift always goes smoother if every container is readable and unmistakable. Labels must show contents, hazard class, and a clear date.
Keep Chemicals ApartAcids shouldn’t sit near bases or other reactive materials. In shared lab environments, a dedicated acid cabinet shields other supplies from potential contamination. I learned to arrange these cupboards away from work tables or high-traffic spots after seeing how a misplaced beaker led to an after-hours evacuation.
Maintain Stable Environment ConditionsCool, dry, and well-ventilated spots slow down degradation and fumes. No one wants surprises caused by high humidity or heat. In one instance, a storeroom without air conditioning on a hot day led to pressure buildup inside an old bottle—luckily, it held, but few want to roll those dice.
Health and Safety: The Human Factor
Storing O-nitrobenzenesulfonic acid isn’t just about chemical compatibility—it’s about the people lucky enough to avoid incidents. Gloves, eye protection, and spill kits should always stay nearby. Most injuries happen not in the chemistry itself, but while carting materials from shelf to bench. Workers get tired, routines slip, and someone ends up dealing with chemical burns.
A responsible storage routine protects both assets and people. Conducting a walk-through with new staff pays off much more than sending out reminders by email. Regular training saves real injuries and real money.
Building a Safer System
Storing hazardous acids like O-nitrobenzenesulfonic acid demands a culture of care. Quick access to safety showers and neutralizing agents counts for as much as locking supplies up. Documentation shouldn’t pile up in a drawer—it belongs right with storage records, up-to-date and easy to find.
A minor investment in better storage delivers far more than ticking boxes for compliance. From direct experience in more than one lab, cutting corners always leads to some sort of trouble. With transparent, reliable storage practices, risks stay low and work stays productive.
What are the physical and chemical properties of O-Nitrobenzenesulfonic Acid?
Everyday Chemistry, Real Impact
O-Nitrobenzenesulfonic acid isn’t something most people have sitting around in the garage. In the lab, though, this compound means business. Imagine a yellowish solid—nothing flashy, not dangerous to touch for a moment—but it holds potential that stretches out into the world of dyes, drugs, even analytical chemistry.
Physical Properties: Tangible Signs
Visually, O-nitrobenzenesulfonic acid stands out thanks to its color. It shows up as pale yellow, and feels solid at room temperature. This stuff pulls moisture from the air, a trait that makes storage a key concern for anyone working with it. Nobody wants a brick of clumped-up chemical. It dissolves well in water, which makes sense for a compound built for reactivity. That solubility makes it useful in watery environments—think tests and analyses, or chemical reactions where water carries other needed ingredients.
You would not want O-nitrobenzenesulfonic acid near strong heat, since it breaks down easily at higher temperatures, releasing toxic gases. Those vapors could mean danger in confined spaces. Good ventilation matters, and decades of workplace safety guidelines around chemical handling back this up. This isn’t just science talk—ignoring these facts leads to mistakes nobody wants to relive.
Chemical Profile: Why Chemists Care
This compound carries both nitro and sulfonic acid groups on a benzene ring. That combination isn’t like tossing just anything into a beaker. Structurally, the nitro group pulls electrons from the aromatic ring, while the sulfonic acid pushes the molecule further toward water-loving reactions. Chemists notice this right away, because it opens doors to several reactions.
In practice, those chemical groups mean o-nitrobenzenesulfonic acid ends up acting as a strong acid. It can trigger further chemical reactions that build new dyes, certain medications, or help with tests to spot specific ions in solution. I remember watching a lab demonstration years ago where just a pinch of this compound turned clear water a surprising color after an electrical current was applied—proof that lab chemistry sometimes feels like magic.
The sulfonic acid group lets this molecule attach to other chemicals in precise spots, a trait any synthetic chemist values. The nitro group also matters because it can be changed into an amino group, unlocking a new world of molecules. Transformation like this helps pharmaceutical chemists create unique drugs that fight off infection or help diagnose conditions in the body.
Environmental and Safety Concerns
Handling o-nitrobenzenesulfonic acid safely takes more than gloves. Its strong acidity means it can damage skin or eyes. I’ve watched a spill etch a faint mark on a stainless-steel bench, reminding me how some mistakes linger. Toxicity worries do not end at the lab bench. If it leaks into the environment, water-loving properties mean it can move quickly through water and soil, possibly harming plants and small aquatic life. Strong rules for waste disposal grow from this reality, sometimes slowing down work, but keeping people and the local environment safer over the long run.
Building Better Practice
Safer chemical storage—sealed containers, clearly labeled—goes a long way. Regular air checks and protective equipment take care of human health risks. Industry now leans on closed systems and regular audits to spot problems before actual harm happens. In research, green chemistry shows promise by looking for less dangerous alternatives that get the same job done. Chemists both old and new carry a responsibility to understand these risks and work with a sense of care and skill. Living up to that standard is just part of what it means to really know your chemistry.
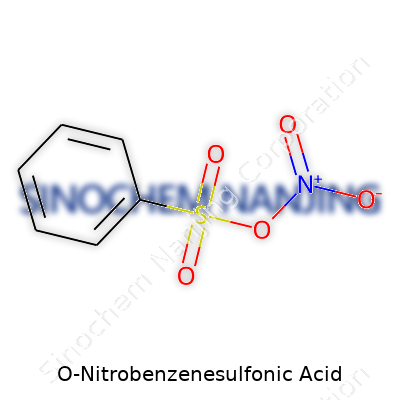
| Names | |
| Preferred IUPAC name | 4-nitrobenzenesulfonic acid |
| Other names |
2-Nitrobenzenesulfonic acid O-Nitrobenzenesulphonic acid Benzenesulfonic acid, 2-nitro- |
| Pronunciation | /ˌoʊˌnaɪtroʊ.bɛnˈziːn.sʌlˌfɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 98-47-5 |
| Beilstein Reference | 358080 |
| ChEBI | CHEBI:76158 |
| ChEMBL | CHEMBL4233668 |
| ChemSpider | 2010153 |
| DrugBank | DB03827 |
| ECHA InfoCard | 13c4db67-605e-41b5-a490-efb6fe2e5f57 |
| EC Number | 248-276-0 |
| Gmelin Reference | 114187 |
| KEGG | C14162 |
| MeSH | D009818 |
| PubChem CID | 7437 |
| RTECS number | DA6475000 |
| UNII | SE3V14ME4D |
| UN number | UN3261 |
| CompTox Dashboard (EPA) | O-nitrobenzenesulfonic acid CompTox Dashboard (EPA) ID: **DTXSID0021586** |
| Properties | |
| Chemical formula | C6H5NO5S |
| Molar mass | 265.17 g/mol |
| Appearance | Yellow crystals |
| Odor | Odorless |
| Density | 1.573 g/cm³ |
| Solubility in water | Soluble |
| log P | -0.89 |
| Vapor pressure | <1 mmHg (20°C) |
| Acidity (pKa) | -0.7 |
| Basicity (pKb) | pKb: 10.7 |
| Magnetic susceptibility (χ) | -76.7·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.595 |
| Viscosity | 1.64 mPa·s (20 °C) |
| Dipole moment | 3.6 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -186.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1297.8 kJ/mol |
| Hazards | |
| Main hazards | Corrosive, causes severe skin burns and eye damage, may cause respiratory irritation |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H301 + H314 |
| Precautionary statements | P234, P261, P280, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Flash point | 187°C |
| Lethal dose or concentration | LD50 oral rat 2480 mg/kg |
| LD50 (median dose) | LD50 (median dose) Rabbit (oral) 1000 mg/kg |
| NIOSH | SF8750000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Benzenesulfonic acid p-Nitrobenzenesulfonic acid o-Nitrobenzenesulfonyl chloride o-Nitrophenol Nitrobenzene |

