O-Isopropylphenol: How A Simple Compound Shapes Science And Safety
Diving Into History: Origins Of O-Isopropylphenol
O-Isopropylphenol didn’t get much attention in its early days. Chemists started isolating and modifying phenols in the 1800s, and it wasn’t long before someone tinkered with isopropyl substitutions. Rather than being a household name, its journey mostly stuck to the lab until the petrochemical rush of the 20th century made large-scale aromatic chemistry possible. Petrochemical byproducts found new commercial uses, and niche compounds like o-isopropylphenol benefited from that curiosity. So, even though the name sounds clinical, this molecule grew out of practical exploration—a reminder that curiosity and useful accidents have nudged chemistry ahead for generations.
Getting To Know O-Isopropylphenol
This compound sits at the crossroads of simplicity and utility. It looks like a clear, oily liquid, releases a distinct medicinal odor, and doesn’t demand elaborate storage—just a good seal and a cool shelf. In industry circles, it often gets called “2-isopropylphenol,” a straightforward nod to its structure. Compare that to the family of cresols or xylenols, where branching leads to both confusion and opportunity. Even the minor change of swapping methyl for isopropyl on phenol changes how companies think about applications.
Physical And Chemical Footprint
O-Isopropylphenol doesn’t put up with water and prefers to hang out with organic solvents. With a boiling point modest enough to be manageable but high enough to avoid careless evaporation, it strikes a balance. Density settles slightly heavier than water, and a faintly yellow hue gives away age or contamination in undistilled lots. Chemically, it fits the pattern of activated aromatics: the isopropyl group draws electrons, boosting its reactivity compared to plain phenol. This trait helps explain why chemists reach for it in synthesis and why users want to handle it with care.
Technical Clarity, Not Just Labels
Chemical catalogs list its purity in percentage points and often flag water, residual solvents, or trace heavy metals. The standard test might lean on gas chromatography, which lays out impurities with unflinching honesty. Labeling follows local safety codes, all the way from GHS pictograms to safety phrases about wearing gloves and keeping your nose clear. Labels should matter—not as legal armor for companies, but as a straightforward warning for people who use these compounds in real labs. In my experience, ignoring the label invites mistakes, and those ripple out in unexpected ways.
How It Gets Made: Preparation In Practice
Most o-isopropylphenol comes from the alkylation of phenol under acidic conditions, usually by heating with propylene or isopropyl alcohol and a strong acid catalyst. Conditions matter—temperature controls product ratio, and excess acid means side reactions love to spring up. The selectivity isn’t perfect, so separating ortho, meta, and para products can get expensive. Sometimes, researchers use clever tricks: load the starting mixture onto a zeolite that favors ortho substitution, or tune the ratio of reagents. It’s a hands-on business, nothing like the high-throughput, hands-off vision of automated chemistry.
Ways To Tweak: Chemical Reactions And Modifications
Chemists like o-isopropylphenol because it’s a resilient platform for more complex molecules. Once in hand, you might nitrate, sulfonate, or halogenate the aromatic ring, setting the stage for products that go into medicines, colorants, or specialty rubbers. Sometimes, people oxidize the isopropyl group for flavor or fragrance precursors. Its role as an intermediate matters: one small tweak at the chemical level spirals out into value for dozens of end products. Rather than seeing it as just a destination, it’s a stop on a much longer, highly creative synthesis road.
Synonyms: The Many Faces Of One Molecule
O-Isopropylphenol pops up under more names than most realize. Besides 2-isopropylphenol, you’ll catch “ortho-isopropylphenol” or even trade designations in industrial supply lists. This confusion tempts mistakes, but consistency in naming brings order—a necessary habit, not just a bureaucratic hurdle. I’ve watched procurement confusion create headaches, forcing quality teams to double-check paperwork and raw materials. In regulated environments, these mix-ups erase profit and waste precious time.
Staying Safe With O-Isopropylphenol
It only takes one exposure to remember why chemical safety isn’t optional. Vapor from o-isopropylphenol can irritate the nose, eyes, and skin. Spills feel greasy and soak in fast. Labs and factory workers deserve more than a token nod to gloves and goggles—they benefit from a culture where supervisors lead by example, and open conversation about incidents is never off-limits. Good ventilation, routine training, and straightforward spill protocols limit harm before it ever crops up. Chemical safety won’t win headlines, but it keeps people coming back to do good work, year after year.
Where It Fits: Application And Real-World Use
Most o-isopropylphenol heads for the synthesis of larger molecules. Pharmaceutical companies, flavor and fragrance labs, and makers of custom resins all rely on it somewhere in the chain. Small changes in its structure tune odor, solubility, and reactivity, making it a testing ground for new ideas. Outside the lab, it occasionally crops up in antiseptics or cleaning agents, when the balance of germ-killing power and manageable toxicity fits regulatory limits. As regulations cut down on harsh or legacy chemicals, o-isopropylphenol sometimes gets a second look—not because it’s flawless, but because familiar compounds remain easier to manage and predict.
Chasing Better: Research, Toxicity, And Rethinking Risk
Research into o-isopropylphenol never slows entirely, because toxicology tests continue revealing surprises. Chronic exposure—by inhalation or skin absorption—doesn’t just bring short-term discomfort. Some studies link long-term handling to organ effects, and environmental scientists track residues in water or soil, since phenolics resist breaking down. Regulators and product engineers dig through data before approving new uses. Sometimes, they pause research, unsure if the benefits outweigh the unknowns, especially when more benign alternatives hover on the horizon. I remember a time when everyone in a lab trusted experience more than safety data; the culture’s shifted, and words like “risk assessment” don’t just sit on checklists—they thread through day-to-day choices.
Looking Ahead: Possibilities And Limits
Future prospects for o-isopropylphenol look like a balancing act—innovation, practicality, caution. Pharmaceutical and chemical industries keep it on the bench because it opens doors for new drugs and specialty chemicals that need fine structure control. Green chemistry promises milder catalysts and less waste in its preparation, but upfront capital and regulatory pushback bring their own set of challenges. Digital inventory and advanced analytics hold promise for tracking hazardous materials, catching mistakes before they scale. Academic labs may lean into “design out hazard” models, minimizing the footprint of traditional phenolics. Whether through smart synthesis, clever waste management, or persistent toxicology screening, future research targets both smarter use and honest reckoning with risk.
What are the main uses of O-Isopropylphenol?
Behind the Label: Everyday Connections
O-Isopropylphenol shows up in more places than most people realize. It carries a hint of clove-like aroma and finds its way into industries that touch everyday routines. I remember reading labels on household cleaners and wondering about the strange-sounding ingredients. This one stood out because it does more than sit on a shelf; it keeps bacteria and fungi from growing, cutting down on the risk of illness in homes, hospitals, and offices.
Barrier Against Germs
Disinfectants need muscle to do their job. O-Isopropylphenol brings that punch to antibacterial sprays, surface wipes, even some medical soaps. It disrupts bacteria and stops mold from spreading—peace of mind for anyone scrubbing a countertop after preparing raw chicken. Hospital workers rely on compounds like this every day, knowing the risk if bacteria slip through.
Supporting Personal Care and Wellness
Beyond cleaning, this chemical also appears in personal care. It winds up in toothpastes and mouthwashes, helping control bad breath and oral bacteria. When I dealt with frequent canker sores in college, my dentist pointed out ingredients like O-Isopropylphenol for keeping germs at bay. It also pops up in medicated soaps and shampoos, especially those targeting dandruff or irritated skin.
Stepping Into Chemical Manufacturing
Factories and labs use this compound as a starting point. Here, it helps shape other products, such as dyes, perfumes, or more complex chemicals. In the world of fragrances, its subtle spicy scent helps craft perfumes and air fresheners with a little more warmth. Manufacturers count on its stability during tough industrial processes.
Tackling Health and Safety Concerns
O-Isopropylphenol does its job well, but all chemicals have risks if handled incorrectly. Exposure, especially in concentrated forms, can create problems for workers—skin irritation, eye discomfort, breathing trouble. I learned early in my work with cleaning products to pay attention to gloves and proper ventilation. Regulatory agencies keep tabs on safe levels for products that reach the public. It’s important that companies not cut corners and that clear labeling stays standard on store shelves.
Looking for Responsible Production
The world doesn’t want more pollution or unsafe working conditions. Sustainable production matters. Green chemistry methods keep emissions lower and relieve pressure on disposal systems. Moving toward biodegradable forms or reusing waste from chemical processes can make a difference, even if it means higher up-front costs. Businesses already face pressure from environmentally aware customers who pay close attention to sourcing and transparency.
Thinking Ahead
Communities stand to benefit when chemicals like O-Isopropylphenol remain both effective and safe. Strong research into health impacts and safer alternatives deserves support, especially as consumers ask for cleaner, greener options. It’s possible to address modern needs—sanitation, affordable manufacturing—without cutting out safety or long-term health. Smart choices around chemicals often start at the factory, but they reach right into homes and daily routines.
What is the chemical formula of O-Isopropylphenol?
Understanding O-Isopropylphenol
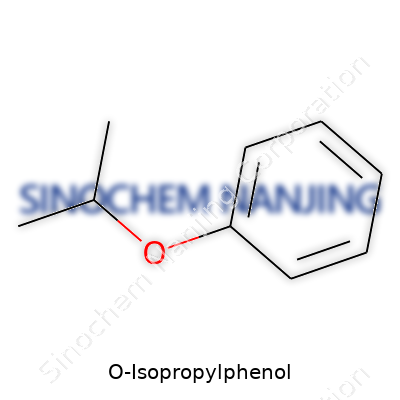
O-Isopropylphenol, often called 2-isopropylphenol or o-cumenol, comes with the chemical formula C9H12O. On paper, it looks straightforward—nine carbons, twelve hydrogens, and one oxygen. Beyond those letters and numbers, it tells a story about how chemistry shapes the products we use every day. As a phenol with an extra isopropyl group tagged at the ortho position, this compound stands out in more places than people realize.
A Place in Everyday Life
My first encounter with o-isopropylphenol wasn’t in a lab textbook but through stories of the old days at my local pharmacy. Back then, folks used products with phenols to disinfect wounds and keep surfaces clean. O-isopropylphenol plays a role not just as an antiseptic, but also as a crucial building block for fragrances and other chemical processes. Factories rely on it for certain reactions that help create flavors and synthetic resins.
Many cleaning products, especially those with a lingering medicinal scent, often owe their distinctive aroma to phenolic compounds like o-isopropylphenol. Its molecular setup lets it interact with bacteria in ways that slow down microbial growth. This has made it valuable for manufacturers looking for alternatives to harsher chemicals—since o-isopropylphenol tends to be less aggressive while still delivering results.
Questions of Safety and Responsibility
Efficiency means nothing if something harms people or the environment. This is where experience comes in. I’ve seen discussions grow over what small exposures mean over time. O-isopropylphenol isn’t as hazardous as some of its cousins, but it’s not risk-free. Touching or inhaling concentrated phenols can irritate skin and airways, and there’s always the matter of accidental spills polluting water or soil.
Studies point out these dangers. The International Agency for Research on Cancer hasn’t flagged o-isopropylphenol as a major health threat, but even less-threatening chemicals deserve respect. Factories handling it use ventilation and protective gear, and many have switched to green chemistry practices when possible. Even small adjustments—like recycling solvents during o-isopropylphenol extraction—go a long way toward safer workplaces.
Finding Better Ways Forward
The future depends on both smarts and care. Students today learn about C9H12O in textbooks, but real progress comes from how we manage the stuff outside of class. Cleaner disposal methods, stricter workplace rules, and smart research into alternative chemicals matter much more than memorizing the formula. Companies have started investing in new ways to produce similar effects using plant-based compounds, reducing the environmental impact.
For me, this topic always comes back to the basics: knowing the formula has its place, but what society does with that knowledge matters more. O-isopropylphenol acts as a good reminder that chemistry shapes daily life, from cleaning supplies to the fragrances we notice in the air. Good science combined with careful habits keeps everyone safer, and that’s worth more than any single chemical structure.
What safety precautions should be taken when handling O-Isopropylphenol?
What’s the Deal with O-Isopropylphenol?
O-Isopropylphenol, better known as 2-isopropylphenol or o-cumenol, finds its way into all kinds of labs and industries—cleaners, chemical syntheses, and even some niche formulations. Anyone who’s worked with chemicals like this knows how quickly things can get risky if basic safety steps get skipped.
Why Respecting O-Isopropylphenol Matters
After a few years working with all sorts of lab reagents, I’ve seen what a little forgetfulness around organic solvents can do. Just because something sits on a shelf doesn’t mean it’s harmless. Science backs up that O-Isopropylphenol can cause nasty burns, eye injury, and trouble breathing if there’s a spill or poor ventilation. The Globally Harmonized System flags it as an irritant, and with enough exposure, it could damage organs.
Gloves and Goggles: Not Optional
I put gloves and safety goggles front and center every time O-Isopropylphenol comes out. It doesn’t belong on bare skin, ever. Lab coats with sleeves protect arms and street clothes, and a splash to the eye can land someone in the ER. That’s not an exaggeration—I’ve seen colleagues spend hours flushing their eyes because safety specs stayed in the drawer.
Ventilation: Forget the Window, Think Local Exhaust
Breathing in fumes from O-Isopropylphenol leaves people coughing and wheezing fast. It’s flammable, so mixing with air in a confined space just invites explosions. Open windows won’t cut it. Use a fume hood or, at minimum, strong local exhaust. A sniff of phenolic fumes can leave your head pounding all day, and more serious effects can follow over time.
Be Ready for Spills: No One Remembers Until It’s Too Late
All it takes is a knocked-over beaker or a cracked bottle. Spill kits—absorbent pads, neutralizing agents—should stay close. Wearing only latex gloves leads to trouble; nitrile or neoprene stand up far better. Containing the area, ventilating, and scooping up residues with the right sorbent saves clean-up crews from long-term health trouble. Wash skin with soap and lots of water if contact happens.
Storage Smarts
O-Isopropylphenol belongs in tight-sealed containers, marked clearly with hazard labels. Keep it away from direct sunlight, incompatible reagents, or sources of heat. Flammable cabinets work well, and using secondary containment (like a tray beneath bottles) stops drips from spreading through storage areas.
Train New Folks: Sweat the Small Stuff
I’ve watched new lab workers make small mistakes—mixing up containers, leaving caps loose, and skipping the MSDS review. One way to dodge accidents is to walk through procedures, show how to read safety labels, and insist on reviewing gloves and goggles. Walking the walk matters more than just handing over paperwork.
Looking Ahead: Upgraded Tech and Safety Culture
Ferris State University’s labs invested in better chemical sensors and online reporting, and the number of accidents dropped. Employers choosing to pay for on-site eyewash stations, spill kits, and basic training don’t just meet regulations—they cut down on days lost and ruined experiments.
Final Takeaway: Keep Safety Personal
Complacency breeds disaster. Understanding the risks, using the right gear, focusing on simple ventilation, and teaching others means going home healthy at the end of a long day. Safety demands more than rules—it needs a sense of responsibility every time someone grabs that bottle.
What is the shelf life and recommended storage conditions for O-Isopropylphenol?
The Essentials Every User Should Know
O-Isopropylphenol, a pale yellow liquid known in labs and industries, sparks plenty of curiosity once it arrives on the workbench. The act of storing this compound brings its own questions, especially about shelf life. Staying careless risks unwanted degradation, and no one wants an unreliable reagent when accuracy and safety can suffer.
O-Isopropylphenol: How Long Can You Keep It?
Manufacturers typically print a shelf life of about two years for unopened containers. The real story unfolds in storage habits. Open bottles don't always last as long. Moisture, oxygen, and sunlight all play their part in shortening the lifespan. The bottle lid makes a difference—a tightly sealed cap fends off water vapor and air, which help prevent slow breakdown into unwanted byproducts. The nose knows too: if it starts to smell off or darken, that bottle probably overstayed its welcome.
People who work with phenolic compounds, including myself, can tell when a mishap in storage shifts a lab’s rhythm. You might measure a reagent, and things just won't react like the textbooks describe. It’s tempting to overlook old stock, but every missed reaction wastes time and money, not to mention possible safety concerns from degraded material.
Temperature and Its Impact
O-Isopropylphenol holds up best at room temperature, between 15°C and 25°C. Consistency brings the most benefit here. Heat kicks off spoilage. I remember a small bottle left near a sunny window; it didn't last two months before forming a brown layer on the glass. Cold storage, like placing it in a regular fridge, reduces reaction rates with air, but going below 0°C can thicken the liquid or turn it cloudy. Fluctuations in temperature encourage condensation, another easy path for contamination.
Protecting from Light and Air
Direct sunlight speeds up the breakdown. Clear glass containers only make things worse. An amber glass bottle works far better, blocking most UV rays and shielding the contents. In practice, simply storing the bottle in a dark cupboard or a closed chemical cabinet serves the same purpose. Regular container checks prevent surprises. Any sudden pressure when opening suggests unwanted reactions.
Sealing containers tight matters as much as temperature. Air introduces both moisture and oxygen. Labs using desiccators, either with silica gel or activated alumina, cut down exposure. I’ve seen big improvements in shelf life with even this simple step—something small labs miss far too often.
Labeling and Rotation Make a Difference
Good housekeeping begins with clear labeling. Write down the date of opening and pay attention to every new stock. Using up the oldest bottles before breaking into a fresh one helps avoid reliance on chemicals that may already be past their best.
Most importantly, build a habit of regular inspection. Look for any cloudiness, unexpected colors, or strange smells. Moving suspect bottles straight to hazardous waste is safer than risking a ruined experiment or even an unsafe reaction.
Solutions for Safe Storage
Keep O-Isopropylphenol at a stable, cool temperature. Use amber glass, store it away from direct light, seal every bottle tight, and fully record the opening date. Everyone can improve storage habits, and these small changes reward you with longer product life, more reliable results, and a much safer lab or storeroom.
Is O-Isopropylphenol hazardous to health or the environment?
Understanding What O-Isopropylphenol Is
O-Isopropylphenol, also called 2-isopropylphenol, shows up in labs and industry settings as a chemical building block. It looks like a clear, oily liquid with a sharp aroma. You’ll find it cropping up in the production of things like disinfectants, fragrance ingredients, and sometimes even pesticides. This stuff doesn’t come from your average household item, but it can make its way into the environment through industrial waste or improper disposal.
Health Concerns from Exposure
The science around O-Isopropylphenol focuses on what it does to people who touch, breathe, or swallow it. Breathing in high levels may irritate airways and cause headaches or dizziness. I remember walking past a chemical plant in summer, getting hit by fumes, and feeling a scratch in my throat—volatile phenols like this one tend to have that effect. Skin contact won’t just result in a foul odor; it burns and dries out tissue, and over time, repeated exposure could bring on further irritation or even allergic reactions.
If someone swallows the chemical, the toxic effects ramp up. Animal studies point toward possible liver damage and changes in kidney function at high doses. The European Chemicals Agency and similar organizations flag O-Isopropylphenol for acute toxicity and categorize it as hazardous for both short and long-term exposure. Workers need real protections: gloves, goggles, and solid exhaust systems cut risks. People outside of the industry rarely meet this compound directly, but accidents and leaks don’t always stay contained.
Environmental Impact: More Than a Passing Concern
Dumping chemicals like O-Isopropylphenol into wastewater or soil spells trouble for wildlife. The compound doesn’t vanish overnight; bacteria and sunlight can break it down, but the process drags on. Fish and water invertebrates exposed to low concentrations show signs of stress and even death, according to toxicity tests. Ecosystems rely on a careful balance, and the bioaccumulation of phenolic substances pushes out the weakest links first: insects, then amphibians, then fish.
My own town sits near a creek that used to run clear but turned murky after a spill from an upstream facility. Local biologists measured phenol derivatives—including O-Isopropylphenol—in the water for weeks. Fish kills left gaps in the food chain. Results like those shouldn’t be written off as flukes. Every spill, intentional or not, builds up consequences.
Regulation and Solutions on the Table
National agencies like the EPA track phenolic discharges and classify O-Isopropylphenol as a priority pollutant in industrial wastewater. France’s ANSES and Germany’s BAuA recommend strict limits for workplace air and suggest quick cleanup protocols for any accidental release. These guidelines protect workers and neighborhoods living with big chemical facilities.
Switching to safer alternatives in manufacturing could shoulder some risk away from this compound. Closed-loop processes and on-site waste treatment systems limit how much ends up in rivers or landfills. Educating the next group of workers about handling and protecting against this chemical cuts down on hospital visits and environmental fines. If companies commit to regular health monitoring and keep chemical inventories honest, the odds of a disaster shrink even further.
People deserve clean air and safe jobs. Anyone with a hand in managing O-Isopropylphenol—from the lab tech to the plant manager—shares a duty. Facts show that hazards can be pinpointed, tackled, and reduced, if we pay attention and speak up when seeing mistakes. That is what keeps families and communities out of harm’s way.
| Names | |
| Preferred IUPAC name | 2-(Propan-2-yl)phenol |
| Other names |
2-Isopropylphenol Isopropylresorcinol o-Cumenol o-Isopropylphenol |
| Pronunciation | /ˌoʊˌaɪ.səˈproʊ.pɪlˌfiː.nɒl/ |
| Identifiers | |
| CAS Number | 88-69-7 |
| Beilstein Reference | 4098733 |
| ChEBI | CHEBI:77593 |
| ChEMBL | CHEMBL165986 |
| ChemSpider | 20719 |
| DrugBank | DB08313 |
| ECHA InfoCard | 100.063.116 |
| EC Number | 202-753-4 |
| Gmelin Reference | 8791 |
| KEGG | C06559 |
| MeSH | D010383 |
| PubChem CID | 7604 |
| RTECS number | UE5950000 |
| UNII | XH5D6UKA4X |
| UN number | 2810 |
| CompTox Dashboard (EPA) | O-Isopropylphenol CompTox Dashboard (EPA) string: **DTXSID8044366** |
| Properties | |
| Chemical formula | C9H12O |
| Molar mass | 136.19 g/mol |
| Appearance | colorless to pale yellow liquid |
| Odor | phenolic |
| Density | 0.969 g/mL at 25 °C |
| Solubility in water | Slightly soluble |
| log P | 2.7 |
| Vapor pressure | 0.4 mmHg (25°C) |
| Acidity (pKa) | 9.98 |
| Basicity (pKb) | 10.39 |
| Magnetic susceptibility (χ) | -74.5e-6 cm³/mol |
| Refractive index (nD) | 1.543 |
| Viscosity | 7.8 mPa·s (25 °C) |
| Dipole moment | 2.19 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 143.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -217.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3502.9 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H314, H410 |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338, P370+P378 |
| Flash point | 88 °C (closed cup) |
| Autoignition temperature | 410°C |
| Explosive limits | 1.3% (LEL) – 3% (UEL) |
| Lethal dose or concentration | LD50 oral rat 300 mg/kg |
| LD50 (median dose) | LD50 (median dose): 270 mg/kg (rat, oral) |
| NIOSH | NN3675000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of O-Isopropylphenol: "5 ppm (19 mg/m3) TWA |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | IDLH: 175 ppm |
| Related compounds | |
| Related compounds |
Phenol Cresols Thymol Carvacrol |

