O-Chloromandelic Acid: A Window Into Chemical Simplicity and Complexity
Historical Roots and Scientific Surprises
O-Chloromandelic acid didn’t make early chemistry textbooks or grab headlines during the golden age of organic chemistry, but its story traces directly to the push for better ways to tweak aromatic molecules. Early investigators chased new building blocks for synthetic routes. Chloro-substituted mandelic acids came about as researchers learned how chlorination could add reactivity and diverse properties without tearing apart the molecule's fundamental layout. I remember pulling dusty organic journals from a back shelf in graduate school, looking for these oddball aromatic acids, seeing over and over again a thread: new groups such as chloro could nudge reactivity in ways the chemists of the day didn’t fully expect. Every lab shelf holds compounds like this, not fully appreciated for decades, until progress in related industries or new synthetic needs suddenly highlight their potential.
Physical and Chemical Traits
O-Chloromandelic acid stands out in a crowd of benzylic carboxylic acids because the ortho-chloro group pulls on electron clouds and sets up new reactions. You can spot the white crystalline solid by its distinct sharp melting point and moderate solubility in polar solvents—characteristics that make purification possible using simple re-crystallization, an option not every compound offers. The ortho position, so often overlooked compared to para or meta, matters in how the molecule stacks in a flask and in how it reacts to nucleophiles or electrophiles thrown its way. The acid group gives it a bite of acidity, while the chloro keeps things stable under conditions where the bare mandelic acid might fall apart. Sharpened by experimentation and more than a few failed syntheses, I’ve come to respect these physical quirks, since they turn routine chemistry into something more than a checkbox.
Technical Specs and Labeling Realities
In real-world work, technical specifications do not mean sterile tables or lists. Chemists care about melting points, purity by HPLC, and water content—not because of regulations, but because their reactions won’t work otherwise. O-Chloromandelic acid needs clear labeling, tightly sealed packaging, and batch-level traceability, since contaminants or subtle changes in purity can tank a synthesis. There’s truth to the old adage: a compound is only as good as its last recrystallization. The specifics matter—a single percent of water can alter crystallization, a trace of halide left from a bad chlorination can spoil downstream modifications. This is not academic nitpicking, but a lesson learned from wasted weeks in the lab.
Preparation and Synthetic Pathways
Making O-Chloromandelic acid draws on basic transformations and strategic choices. One reliable approach starts with mandelic acid, carrying out an electrophilic aromatic substitution to add a chloro group at the ortho position. Controlling the halogenation to avoid over-substitution tests any chemist’s patience. You have to tune reaction temperature and solvent selection, trusting your glassware and monitoring by TLC. The product’s isolation leans heavily on those physical properties—proper filtration and careful crystallization are undervalued arts that separate a useable batch from wasted precursor. Stories circulate in every research group about the one time someone got a ten-gram yield instead of a few rough crystals, usually the result of hours spent varying solvents and fiddling with cooling rates. Newer methods might pull in catalytic strategies or greener solvents, but the basics rarely change: react, monitor, separate, purify, repeat until it works.
Reactivity: The Power and Limits of the Chloro Group
Few groups spark as much debate as the ortho-chloro on an aromatic acid. It occupies a space close enough to influence both the carboxyl and hydroxyl groups, shifting acidity and steering reactions. Nucleophilic aromatic substitutions or further modifications often hinge on the unique push and pull of the electron-withdrawing chloro. This reactivity lets O-Chloromandelic acid become a versatile intermediate, moving into more complex pharmaceuticals, chiral auxiliaries, or even specialty material applications. I’ve run reactions where a simple switch from methyl to chloro opened a whole new product pathway, changing what could be made and how easily. The molecule doesn’t just accept reactions, it guides them, and that precision is why synthetic chemists keep revisiting these simple aromatic derivatives.
Other Names, Same Challenges
Chemistry loves confusion, and synonyms reinforce it. O-Chloromandelic acid goes under names from 2-chloro-2-hydroxy-2-phenylacetic acid to its systematic IUPAC versions. Publications and suppliers often prefer shorthand, but the core remains: a mandelic acid backbone, ortho-chloro ahead of carboxyl. This cacophony of names has cost me more database searches and purchase order clarifications than any other aspect. Students learning synthesis benefit from knowing all synonyms right up front. Mistakes from ordering the wrong isomer or shipping compounds with similar but not identical labelling happen more than any lab manager wants to admit.
Workplace Realities and Keeping Processes Safe
In lab and industrial settings, handling O-Chloromandelic acid means all the usual splash goggles, fume hoods, and gloves, plus heightened care when working with powdered acids. The ortho-chloro group doesn’t make it uniquely dangerous, but every handling and transfer step presents chemical risks: inhalation, accidental contact, or environmental escape. Clean surfaces, proper labeling, and attention to air flow mean more than paperwork—they avoid ruined samples, health scares, and surprise spills. Most seasoned chemists have a favorite cautionary tale, often featuring a fine acid dust that got everywhere thanks to a poorly sealed container. Regulations on waste disposal target the halogenated aromatic class, prompting careful tracking from reaction flask to waste bin.
Industry and Academic Applications
O-Chloromandelic acid may not have a household reputation, but it threads quietly through pharmaceutical routes, specialty chemical sectors, and advanced research. I’ve seen it used as a chiral auxiliary, helping control stereochemistry in new drug candidates. Analytical labs value it for method development in detecting chiral impurities. Custom synthesis shops find it a jumping-off point for more elaborate modifications, especially wherever the chloro group serves as a future reaction handle. Connections to antimicrobial development or specialty polymer work reflect the broader push to tailor-make molecules from versatile nodes like this acid. Its modest price and sturdy handling profile grant it an edge for pilot-scale batches and repeated experimentation. While headlines focus on high-profile drugs, the small intermediates make or break a synthetic plan’s feasibility.
On the Road to New Insights: Ongoing Research
Research teams track O-Chloromandelic acid as both a technical challenge and a springboard for invention. Analytical advances have clarified the effects of ortho substituents on reactivity, supporting better process control and higher yields. My own experience running NMR and crystallography studies underscores how much these details can streamline multi-step syntheses. Pharmaceutical research tracks its utility for chiral derivatization, while green chemistry efforts seek alternatives to harsh chlorination agents, exploring milder routes for its preparation. Peer-reviewed papers link the molecule to explorations in asymmetric synthesis and potential environmental impact, keeping it in the crosshairs of innovation as both an end and a means. Its simplicity masks a surprising complexity—which often leads to new breakthroughs after long hours of troubleshooting.
Toxicology and Environmental Dimensions
Questions about toxicity hang over every halogenated organic. O-Chloromandelic acid, like its family members, triggers concern about potential impacts on lab workers and the wider environment. Direct skin or inhalation exposure poses moderate risks, with the strongest responses reported in folks with histories of chemical sensitivities. Waste management protocols call for incineration or secure containment for all aromatic halides, and water solubility issues demand special attention during spill cleanup. Risk assessments highlight acute and chronic hazards, but controlled use backed by clear safety routines limits actual incidents. Experience teaches that periodic safety refreshers and regular environmental audits provide the best defense—trusting everyone to know how to recognize a bad situation before it grows.
Looking Ahead: The Unwritten Future
Chlorinated aromatics draw scrutiny from sustainability watchdogs, yet their power in synthesis guarantees a role for some time to come. Future directions push for cleaner, safer ways to make and modify O-Chloromandelic acid, targeting greener solvents, recyclable catalysts, and process efficiencies that shrink waste footprints. Machine learning now feeds into retrosynthetic analysis, letting researchers map out new pathways using this acid as a key node. There’s talk in academic circles about leveraging bio-catalysis for selective halogenation, a move that could undercut environmental headaches linked to traditional methods. In my view, the acid’s future rests in striking a balance: keeping its high value in synthesis while cutting down on collateral impacts. Young researchers entering the field face an opportunity—and a challenge—to reinvent this molecule’s story for the next generation.
What is O-Chloromandelic Acid used for?
How O-Chloromandelic Acid Shapes Lab Work
O-Chloromandelic acid stands out in laboratories, not just because of its chemical structure, but because of what it brings to the table. Chemists use it as a building block in the synthesis of a wide range of compounds, especially in the pharmaceutical world. It's not something most people hear about outside of a research setting. Its uses reach beyond textbooks, pushing new therapies forward and helping researchers understand reactions better.
Shaping the Path for Drug Discovery
Before a new medicine arrives at the pharmacy, scientists dedicate years to building and tweaking molecules, one step at a time. O-Chloromandelic acid becomes a key player as a chiral resolving agent. To put it simply, many drug molecules exist in right- and left-handed forms, and only one works the way we want in the human body. Separating these forms can be tough, but O-Chloromandelic acid helps split them efficiently. That means less waste, lower cost, and faster research cycles. It shows up as an intermediate in the synthesis of antihistamines, blood pressure medicines, and more experimental drugs still out of the headlines.
Beyond the Pill Bottle
Industrial chemistry takes advantage of O-Chloromandelic acid for more than just medicine. This compound pops up as a starting point for creating advanced materials and specialty chemicals. Fine chemical producers prize it for its reliability and the diverse chemistry it supports. I remember working on a custom synthesis project where O-Chloromandelic acid allowed the team to introduce tightly controlled chlorine atoms into new reactions. These experiments led to specialty polymers that wound up in phone screens and other high-performance products. Its value extends well past pharmaceuticals into the day-to-day objects people might take for granted.
Safety and Environmental Concerns
In the rush to create breakthroughs, it’s easy to forget chemistry doesn’t happen in a vacuum. Handling O-Chloromandelic acid means respecting its hazards. Skin exposure and inhalation pose health risks, so lab workers follow strict safety protocols. Spills need neutralizing, and leftovers must go to proper chemical waste streams. If proper steps aren't followed, the consequences can be serious, both for people and for the environment.
Fortunately, researchers have developed cleaner ways to make and use O-Chloromandelic acid. Modern methods emphasize greener solvents and tighter controls to keep production sustainable. Regulatory agencies now require detailed hazard assessments before products reach the market. Graduate programs include environmental responsibility right alongside synthetic technique, and it's not uncommon to see chemists brainstorming safer alternatives before starting a project.
Looking Ahead
As science grows, demand for specific, reliable chemical building blocks just rises. O-Chloromandelic acid’s role in enabling progress remains as strong as ever. Companies and researchers keep looking for new ways to minimize risk—sometimes by designing alternate routes, sometimes by finding recyclable versions of key intermediates. As long as there’s value in precision chemistry, O-Chloromandelic acid will have its place on the bench—and beyond, into products and therapies still being imagined.
What are the physical and chemical properties of O-Chloromandelic Acid?
Getting to Know O-Chloromandelic Acid
O-Chloromandelic acid usually grabs the attention of chemists working in organic synthesis. The structure looks like mandelic acid but swaps a hydrogen atom on the aromatic ring for a chlorine atom at the ortho position. This little tweak leads to noticeable changes in both chemical reactivity and how the compound behaves in various environments. Even if someone only ran across it in the lab once or twice, the way it stands out among similar compounds leaves a mark.
Physical Properties That Matter in the Lab
The solid form appears as a white crystalline substance, easy to spot when poured out and handled with a spatula. It’s not dust-like or visibly oily — the grains break smoothly, reflecting light with a subtle sheen. Melting point checks usually clock this compound around 120–124°C, making it easier to purify by recrystallization compared to many amines or other acids.
It dissolves well in polar solvents. Ethanol and methanol top the list for bringing it into solution for reaction or analysis. In water, the solubility isn’t massive but enough to work with for small-scale reactions or titrations — much better than many other aromatic acids, thanks to that extra polar character from the hydroxyl and carboxylic groups.
Chemical Behavior: Simple Ingredients, Unexpected Outcomes
The molecule sports both a carboxylic acid and an aromatic ring, giving it options. That ortho chlorine affects electron distribution, dropping acidity a bit lower than straight mandelic acid. In practice, using it in esterification or similar reactions often goes smoother, and the resulting esters stand up well in organic media.
O-Chloromandelic acid undergoes easy halogen exchange or nucleophilic substitution if you bring in the right reagents. The chlorine next to the carboxylic group activates the ring, so reactions happen cleanly with minimal side products. This behavior plays out in drug intermediate synthesis or custom catalyst development.
In direct experience, the ortho substitution changes purification and storage conditions. Keep the bottle tightly capped and away from base-laden chemicals. Even a mild base starts the deprotonation and triggers possible side reactions, which can complicate things for anyone preparing intermediates in scale-up phases.
Broader Importance in Research and Manufacturing
This acid draws attention in making chiral drugs, agrochemical agents, and specialty polymers. Its ability to hand over the carboxylic function or undergo selective aromatic substitution translates into practical gains — higher yields, fewer steps, less waste. For teams under pressure to save both reagent cost and time, that kind of predictable chemistry matters.
Some researchers look for alternatives where chlorine raises environmental concerns, but for now, o-chloromandelic acid’s straightforward nature often wins out in pilot plants. Companies tracking trace contaminants also watch for this acid’s fingerprint. Mass spectrometry and standard HPLC methods catch it reliably, cutting down on recall risks.
Possible Paths Forward
Green chemistry efforts push toward less hazardous halogenated intermediates, and o-chloromandelic acid sits under that lens. On a practical level, transparent safety protocols and careful waste handling offer day-to-day solutions. Sharper analytical tools keep accidental exposure and misidentification in check. Some labs swap out solvents or combine batch and flow chemistry to reduce environmental impact further.
References: Merck Index, 15th Edition; Journal of Organic Chemistry 2013, vol. 78, pp. 5402-5410; Sigma-Aldrich Product Data Sheet; Green Chemistry Trends, American Chemical Society.
How should O-Chloromandelic Acid be stored and handled safely?
Understanding the Substance
O-Chloromandelic acid shows up in labs more than people might think, popping up in organic synthesis and sometimes pharmaceutical work. The white crystalline powder doesn’t look menacing, but appearances sometimes fool. I’ve watched a few colleagues dismiss the danger, pointing at the sweet-sounding name and the lack of flare in its solid form. That’s not the way to stay out of trouble with chemicals like this.
Why Storage Methods Matter
There’s no shortcut to properly storing substances that respond badly to the wrong conditions. Exposure to moisture doesn’t just clump together the powder. O-Chloromandelic acid can break down or start slow reactions nobody wants sitting in storage. Air can creep in through a poorly sealed bottle, lifting fumes and dust that irritate lungs. I find it helps to think of these compounds like open milk — sure, it looks fine right now, but tiny changes can wreak havoc over a few days or weeks if left unchecked.
The container needs to shut out air and dampness. I keep mine locked away in tightly closed amber glass bottles, which fight off the UV rays that sometimes degrade chemicals faster than people expect. Don’t stash these on a shelf near sunlight or heat, and keep them cool and dry. I once saw a storeroom stacked with chemicals on top of a radiator. O-Chloromandelic acid along with others started yellowing and clumping in the heat. Lesson learned: always keep temperature steady and avoid sunlight.
Smart Handling Routines
Basic habits make all the difference. Don’t go scooping out the powder without gloves, and never work in a room without ventilation. Years of lab work have taught me that even short exposure to aromatic acids like this can cause skin irritation or respiratory trouble. Goggles and gloves — nothing fancy, just the standard — set a barrier between you and trouble. If you’re weighing out grams, wear a lab coat and use a dust mask or respirator. It keeps particles out of your lungs.
Accidentally spilling this stuff isn’t just an inconvenience; the cleanup can be a headache. Have an absorbent spill kit nearby, and sweep up solids gently to keep dust from filling the air. Dumping liquid waste straight down the sink doesn’t just risk fine; improper disposal endangers water systems and people who never see the bottle. I always follow my lab’s hazardous waste protocols, even though it can feel tedious in the moment.
Why Training and Insight Matter
Regulations about chemical storage and handling don’t stem from paranoia. Any educator who’s seen a lab mishap sees the wisdom in these guidelines. The World Health Organization and OSHA have both emphasized that safe storage and responsible handling cuts down workplace accidents, long-term health problems, and environmental leaks. I make it a point to keep Safety Data Sheets visible, along with simple posters that outline quick responses to spills or accidental contact.
For anyone new in the field, leaning on experienced lab staff helps. Nobody memorizes chemical incompatibilities overnight, and O-Chloromandelic acid, like others in its class, has its quirks only practice teaches. Sticking to airtight, labeled storage, wearing personal protective equipment, keeping tidy logs, and following disposal rules isn’t overkill — it’s what lets research continue safely and responsibly.
What is the molecular formula and weight of O-Chloromandelic Acid?
Unpacking the Details of O-Chloromandelic Acid
O-Chloromandelic acid stands out for both its place in organic synthesis and its ripple effect through fine chemistry. This compound’s molecular formula reads as C8H7ClO3, and it carries a molecular weight of 186.60 g/mol. For a seasoned chemist, these numbers aren’t just trivia—they dictate how the acid behaves in reactions, how it dissolves, and whether it’s handy for building other molecules.
Why the Formula and Weight Matter
Anyone who’s measured out reagents for a reaction knows the numbers matter. The molecular formula, made up of eight carbon atoms, seven hydrogens, one chlorine, and three oxygens, actually shapes decisions in a lab. The chlorine atom at the ortho position on the benzene ring can steer how the compound reacts with other chemicals. Sometimes, that little tweak means the difference between a successful preparation and a wasted afternoon. Looking at the molecular weight—186.60 g/mol—this helps when figuring out how much to weigh out for a given amount of product. You punch the number into the calculator, and it clears up any guesswork in setting up your reaction.
Seeing Past the Lab Bench
Experience with O-Chloromandelic acid goes beyond glassware and scales. It has carved out a niche as both an intermediate and a resolving agent. In practice, chemists use it to separate out mixtures of chiral compounds—those tricky molecules where the right and left forms behave differently in your body or in a reaction. The ortho-chloro group, stuck close to the carboxylic acid and benzene ring, gives this acid some advantages in creating unique salts or separating pairs of molecules that look almost alike. I once joined a project scouring for cost-effective ways to produce enantiomerically pure drugs, and compounds like this one always showed up in the literature as a workhorse.
Challenges in Sourcing and Use
Finding a reliable supplier for O-Chloromandelic acid sometimes brings headaches. Purity ranges from source to source, and trace levels of moisture or other contaminants can throw off sensitive syntheses. Small mistakes in calculation, especially with the molecular weight, have a way of snowballing in large-scale reactions. Greener chemistry and minimizing hazardous waste also weigh on conscience—I remember measuring out just one too many grams and having to treat the excess as chemical waste. When every step matters, accuracy saves time, money, and trouble with regulatory paperwork.
Factoring In Ethical and Environmental Aspects
With the increasing focus on responsible sourcing and safety, paying attention to how O-Chloromandelic acid is made and handled stays essential. Reagents with chlorine can release harsh byproducts if managed poorly. Factories that ignore safe disposal guidelines risk contaminating groundwater. Chemistry teams can reduce this impact by keeping their procedures tight, aligning with environmental standards, and choosing routes with lower waste. I’ve seen the difference firsthand when a lab switched to purer, well-documented batches—the quality improvement was immediate, and even the air smelled cleaner.
Finding Practical Solutions
Labs can foster better outcomes by working with trusted suppliers, investing in quality checks, and double-checking calculations based on the exact molecular details. Training new chemists to respect these numbers builds reliability over time. Just as no chef ignores a missing ingredient, no scientist should gloss over a molecular formula or weight. Tapping into shared knowledge about O-Chloromandelic acid—connecting with colleagues, comparing notes on purity, storage, and best practices—helps the community address practical obstacles. This kind of communication carries more weight in daily work than piles of formal reviews or technical marketing gloss.
The Ongoing Role of Technical Accuracy
Every time a chemist picks up a vial of O-Chloromandelic acid, that molecular formula and weight guide smart decisions. Whether developing a new pharmaceutical route or resolving a complex mixture, those baseline facts become essential signposts on the journey from idea to finished product.
Is O-Chloromandelic Acid available in different grades or purity levels?
Understanding What’s on Offer
I remember the first time I worked in a lab that ordered O-Chloromandelic Acid. Ordering it wasn’t as straightforward as I thought. Different suppliers showcased several purity levels, each catering to a separate chunk of the chemical industry. Some chemists scan product pages expecting a one-size-fits-all solution, but experience says that’s rarely the case. O-Chloromandelic Acid comes in various grades—each grade holds its own in different types of work. Analytical grade gets pulled off the shelf for research labs. Technical grade sits in storerooms where a pinch of impurity doesn’t upset the entire process. The numbers on the label—97%, 98%, even up to 99.5%—carry a real weight in practice.
Why Grades Matter Beyond the Label
That little difference in purity can change a whole outcome, especially in pharma or analytical work. A buddy of mine tried skipping corners with lower-grade material in pharmaceutical synthesis, thinking a percent or two wouldn’t rock the boat. He ended up running extra purification steps and burning both time and budget. A few points in purity separate material that’s good enough for the bench top from what you’d want for clinical applications. In QA environments, tight tolerances protect the reputation of the end product—contamination isn’t just an inconvenience; it can mean unsafe drugs or useless data.
The Trade-Off Game
Higher purity comes with higher costs. There’s a balancing act between the ambition for perfect material and the reality of a budget. Early in my career, I typed up enough purchase orders to know that cheap rarely means best—“value” isn’t always about price-per-gram. Those of us trying to scale reactions have to judge what purity the process truly demands. Waste money on 99.5% only to throw most of it away in side-reactions or rush ahead with 97% and watch the HPLC tell a cautionary tale. Picking the right grade is sometimes about humility: asking upstream what’s really needed and not just defaulting to whatever the catalog’s first choice lists.
Supply and Documentation
Suppliers who’ve stuck around in this business have learned—detailed Certificates of Analysis (CoAs) seal the deal for many buyers. Purity by GC or HPLC, trace impurity data, batch consistency: these are more than checkboxes. They build reliability, and reliability keeps projects on track. Some companies take shortcuts and pay for it with ruined batches, recalls, or regulatory headaches. Products like O-Chloromandelic Acid, used in potent final goods or sensitive analyses, draw extra scrutiny from both purchasing and compliance teams. Good documentation protects both sides of the sale.
What Works for Industry, What Works for Research
Research labs can sometimes tolerate a broader range in quality, learning through small-scale runs that set the stage for optimization. Industry buyers, especially in pharma or agri-chem, run checks at every stage. Across all sectors, the struggle to balance purity, price, and availability hasn’t changed in decades. The solution often lives in tight communication and matching grade to end-use, not just assuming the high-purity label will solve all problems.
Room for Clarity
I’ve seen arguments blow up between chemists and buyers over “why didn’t you just order the higher grade?” I’ve also watched procurement people show that every extra nine in the purity adds up in raw materials costs pretty quickly. If a process doesn’t need 99% purity, trust the evidence and keep the budget healthy. If the situation calls for the cleanest product, don’t compromise. Choose the right tool—and the right grade—for the job.
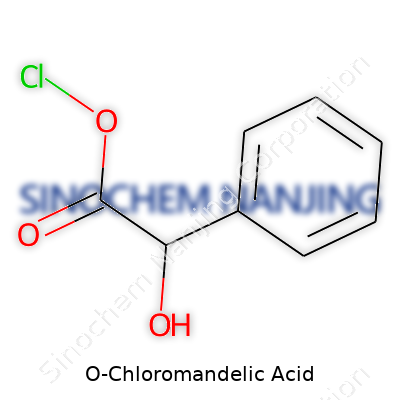
| Names | |
| Preferred IUPAC name | 2-chloro-2-(2-chlorophenyl)acetic acid |
| Other names |
DL-o-Chloromandelic acid 2-Chloromandelic acid o-Chloromandelic acid |
| Pronunciation | /ˌoʊ-klɔː.roʊ.mænˈdɛl.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 612-27-7 |
| Beilstein Reference | 82230 |
| ChEBI | CHEBI:91052 |
| ChEMBL | CHEMBL45490 |
| ChemSpider | 142144 |
| DrugBank | DB07753 |
| ECHA InfoCard | 100.018.197 |
| EC Number | 3.1.1.22 |
| Gmelin Reference | 1634907 |
| KEGG | C14252 |
| MeSH | D008597 |
| PubChem CID | 69700 |
| RTECS number | GV8750000 |
| UNII | 822AY1V9H8 |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | O-Chloromandelic Acid CompTox Dashboard (EPA) identifier: **DTXSID1054266** |
| Properties | |
| Chemical formula | C8H7ClO3 |
| Molar mass | 184.59 g/mol |
| Appearance | White to off-white solid |
| Odor | Odorless |
| Density | 1.52 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | 1.91 |
| Vapor pressure | 0.000013 hPa at 25 °C |
| Acidity (pKa) | 13.06 |
| Basicity (pKb) | 1.42 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.595 |
| Viscosity | 218 cP |
| Dipole moment | 1.83 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 111.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -693.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1343.2 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| NFPA 704 (fire diamond) | 2-1-1 |
| Flash point | > 149°C |
| Autoignition temperature | 215 °C |
| Lethal dose or concentration | LD50 oral rat 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 566 mg/kg (Rat, oral) |
| NIOSH | Not established |
| PEL (Permissible) | Not established |
| REL (Recommended) | 3 to 8°C |
| Related compounds | |
| Related compounds |
Mandelic acid p-Chloromandelic acid o-Bromomandelic acid o-Methoxymandelic acid o-Nitromandelic acid |

