Nitrourea: A Deep Dive
Historical Development
Discovery of nitrourea traces back to the energetic research boom of the mid-20th century, when chemists searched for compounds that could drive industrial and agricultural growth. Early papers describe its synthesis in the context of dye intermediates and chemical propellants. The practical uses stood out quickly: explosive research, pharmaceuticals, and even water treatment got a boost from this unique compound. Over the decades, scientists refined production, improved purity, and learned lessons from both success and mishap. As with any chemical discovery, handling protocols evolved alongside our growing understanding. Looking back, nitrourea forms a snapshot of scientific curiosity harnessed to solve pressing challenges, but it has never stayed just a laboratory curiosity. Its footprint stretches through several industries, igniting advances where traditional materials fell short.
Product Overview
Nitrourea, known to chemists and industry alike, brings a nitrogen-rich formula that sets it apart from typical urea-based products. The blend of nitro and urea functional groups leads to its distinct character. Manufacturing outfits use it as an energetic material—meaning it sometimes acts as a propellant or a blasting ingredient. At the same time, researchers examine it as an intermediate in pharmaceuticals for its unique reactivity. Production batches come in various grades, from technical for industrial uses to analytical for fine chemical research. Labeling always details composition, content, and recommended storage conditions, since even slight impurities or unintended decomposition products could spell trouble downstream. Safety data sheets remain glued to its containers, not as a legal exercise but because workers have learned, sometimes the hard way, that carelessness with nitrourea could trigger disastrous results.
Physical & Chemical Properties
Nitrourea typically appears as a white to pale yellow crystalline solid, and anyone who's opened a fresh bottle will remember the faint chemical odor that lingers in the air. It dissolves in water, though not as readily as standard urea; organic solvents like acetone or ethanol do a better job, reflecting its hybrid molecular design. Melting kicks in at relatively modest temperatures—often a sign of chemical instability in similar compounds. The dual nature shows up in its chemistry: the nitro group brings strong oxidizing potential, while the urea fragment moderates the molecule’s reactivity. This dance gives nitrourea a split personality, lending it to both construction and destruction, from rocket fuel preparation to pesticide creation. Its tendency to decompose on heating keeps every chemist cautious—runaway reactions have made more than one lab technician jump. Uncontrolled exposure to acids or bases can send the molecule off on unpredictable tangents, so inventory managers watch expiration dates with eagle eyes.
Technical Specifications & Labeling
Technical bulletins spell out the essentials: molecular weight, purity level, moisture percentage, and acceptable impurity thresholds. Packaging gets ruggedized, especially for large shipments—most handlers insist on inert lining, robust outer drums, and desiccant packs to beat back moisture during shipping. Labels plainly state hazard class, UN number, recommended PPE, storage temperature, and transport restrictions. For research batches, suppliers include spectral data or chromatography results, because minute impurity traces could alter chemical yields dramatically. Users should always verify date of manufacture, as degradation products can gather over time, affecting both safety and efficacy. Legal mandates around shipping and storage changed after several high-profile accidents, pushing for tighter chain-of-custody processes. Each of these steps came in response to gritty real-world lessons—nobody wants to risk uncontrolled decomposition, especially not in crowded warehouses.
Preparation Method
Synthesis starts from urea and nitrosyl chloride or other nitrosating agents. Early routes required careful temperature control, with even small thermal excursions causing byproduct formation or explosive decomposition. Modern processes favor batch reactors equipped with cooling jackets and active monitoring—not luxury, just necessity. Technicians set up feed tanks under negative pressure, ensuring no stray air or contaminants mix into reactions. Once nitro and urea moieties come together, intermediate purification steps begin: filtration, recrystallization, controlled drying, all under inert atmospheres. Sensitized sources of heat or friction knock loose the nitrourea from sticky reaction flasks. Even in commercial plants, tiny slips—a forgotten ground strap, a careless maintenance check, or a skipped gas purge—lead to contamination or serious safety breaches. After purification, careful packaging seals the product before shipment. More research teams reach out for alternative green synthesis routes, aiming to cut toxic byproducts and increase atom efficiency—primarily to reduce environmental and occupational hazards from legacy routes.
Chemical Reactions & Modifications
Nitrourea’s unique setup unlocks some striking chemical pathways. Trans-nitrosation reactions draw the most interest, since nitrourea acts as a useful donor in preparing specialty nitrosamines. Reductions with metal hydrides break it down into amines and nitrites; oxidative stress (sometimes deliberate in water treatment) can push it further, forming reactive nitrogen species. Some chemists tweak the urea group, swapping in alkyl or aryl fragments to build up pesticide intermediates. Attempts to modify the nitro end shoot for more stable energetic derivatives, safer to handle or with tailored performance in explosives engineering. Industrial labs experiment with continuous-flow reactors to streamline reactions and push yields higher, but they keep a sharp eye on runaway scenarios—every gram can make a difference between routine success and a dangerous blowout. The research community continues testing catalysts and selective reagents, hoping to lower process energy requirements without sacrificing output or product integrity.
Synonyms & Product Names
The chemical world rarely sticks to one name for a compound. Nitrourea answers to N-nitrourea, nitrosocarbamide, 1-nitrosourea, and, in patents, sometimes NRU. Product datasheets also reference CAS Number 684-93-5, easing confusion across language barriers. Large manufacturers list it as “Nitrourea, technical,” while pharmaceutical firms may opt for “N-nitro urea, analytical grade.” In pesticide circles, it occasionally gets lumped under “nitrocarbamides.” Anyone searching patent archives or regulatory documents learns to keep these aliases close at hand—missing a trade name means missing important safety or hazard updates.
Safety & Operational Standards
Tough lessons from laboratory and factory floors shaped today’s safety standards for nitrourea. Eyes, lungs, and skin all need full protection, with fitted respirators and splash-proof goggles topping the list. Material safety training isn’t a checkbox; it’s the line between a typical shift and a trip to the emergency room. Grounded equipment stops static sparks, and storage rooms must stay cool, dry, and shaded from direct light. Emergency drills cover uncontrolled decompositions and fire scenarios, since nitrourea burns hot and leaves toxic oxides behind. Manufacturers and distributors follow both OSHA and REACH guidelines, logging lot numbers and customer chains in case of recall. Laboratories adopted detailed logbooks for inventory, waste tracking, and even incident reports, so that future teams learn from past mistakes. Disposal falls under strict hazardous waste regulations—no one dumps it down the sink. Most companies keep designated spill kits—complete with neutralizers and PPE—on every floor.
Application Area
Industry doesn’t pigeonhole nitrourea. Explosives manufacturers look for its performance as a low-melting energetic material, fitting specialty demolition and mining needs. Agricultural scientists once explored it as a controlled-release nitrogen source, although toxicity pulled back some of these efforts. Fine chemical labs use nitrourea to synthesize intermediates for dyes, drugs, and herbicides, taking advantage of its potent nitrogen content and unique reactivity. Water treatment labs study it during advanced oxidation tests, tracing breakdown routes and watching for secondary pollution. Analytical labs sometimes use it in calibration routines or as a nitrosation standard. In every application, end-users tweak batch recipes and process flows, looking for an edge in performance or safety. Regulations now steer most consumer-facing uses away from nitrourea, especially after international agencies tightened restrictions.
Research & Development
Scientists keep pushing on several fronts. Green chemistry gets attention—can factories make nitrourea without chlorine or heavy metal catalysts? University teams tackle molecular redesign, aiming for compounds that deliver the same utility without destabilizing shelf lives or high toxicity. Analytical chemists draft better detection and disposal methods, trying to close the loop on process waste. Some pharmaceutical research groups tinker with minor modifications, exploring roles for nitrourea derivatives in cancer therapies and prodrug design. Funding goes to safer reaction monitoring systems, hoping to catch process upsets long before they turn into disasters. Computer modeling—the darling of the last decade—helps predict new reaction pathways and flag dangerous combinations in silico before anyone needs to crack a bottle open in the lab.
Toxicity Research
Toxicology results hang over nitrourea like a storm cloud. Animal studies from the 1970s painted a clear picture: acute exposure damages liver, kidney, and central nervous tissues. Chronic low-dose studies linked the molecule to increased risks of mutagenicity and carcinogenicity. Regulatory agencies flagged nitrourea-containing pesticides as high risk, sparking bans and recalls. Water quality labs track nitrourea and byproducts once they escape into rivers—nitrosamines in particular signal trouble, since they rank as potent carcinogens. Workers exposed to airborne or skin-contact nitrourea report headaches, respiratory distress, and skin irritation. Even indirect exposure (from contaminated surfaces or gear) sometimes pushes levels above occupational limits, depending on job site ventilation and hygiene. Ongoing studies test cleanup techniques and medical countermeasures, but prevention remains the best tool—a lesson every safety officer drills into new hires.
Future Prospects
Nitrourea faces pressure from every side today. Tighter regulations mean chemical plants evaluate their safety and waste flows constantly. Environmental watchdogs watch for spills and air emissions. Persistent health risks keep most consumer uses on the sidelines. Yet research sticks with nitrourea, searching for ways to harness its potent chemistry responsibly. Engineers build smarter reactors, giving operators more control and visibility. Analytical breakthroughs flag contaminated sites and guide remediation. Green chemistry tries to dial down toxicity without losing performance. For all its challenges, nitrourea still tempts innovators—anyone who can tame its dark side and boost safety could unlock new fields, especially in specialty materials or controlled-release agricultural applications. Until then, caution and evidence drive every decision linked to this complex nitrogen compound.
What is Nitrourea used for?
Understanding Nitrourea’s Role
Nitrourea belongs to a group of compounds known as nitrosoureas. It often finds use in the world of medicine, especially for its cancer-fighting properties. Doctors rely on medications like carmustine and lomustine, both based on nitrourea, to treat certain types of tumors. These agents show results against brain cancers and lymphomas—areas where other therapies fall short.
Cancer Treatment and Patient Impact
Treating brain tumors comes with a unique set of barriers. The brain protects itself with a filter called the blood-brain barrier, which blocks out many drugs. Agents built from nitrourea cross this barrier and reach the tumors. My uncle, who battled a glioblastoma years ago, received carmustine. The relief in my family’s eyes after hearing about a treatment option that actually entered the brain felt unforgettable. While the chances were never high, having a shot mattered.
Statistics offer hope and caution. Drugs containing nitrourea shrink tumors and help lengthen survival for patients with specific cancers. Of course, side effects can be harsh: low blood counts, nausea, or lung issues. Every patient balances what the medicine fights and what the medicine brings. Facing cancer strips away the luxury of easy choices.
Research and Development
Doctors and researchers continue to ask tough questions about nitrourea-based drugs. Some studies examine whether these agents boost the power of radiation therapies or combine well with other medicines. Efforts over the years have moved from simple lab studies to more personalized approaches. Genetics, patient age, and tumor specifics help doctors pick smarter treatments.
Laboratories also test new nitrourea derivatives. The goal isn’t just to make a stronger cancer fighter. Scientists look for ways to limit harmful side effects and deliver the medicine more accurately to sick tissue. The entire process takes years. Research grants and patient volunteers push the science further along.
Risks, Challenges, and Solutions
No medicine solves every problem. People using nitrourea drugs face risks. Their bone marrow, which makes blood cells, can slow down production. Doctors check blood levels often and wait before giving the next treatment. Some patients experience changes in lung function. Nobody should face these drugs without close medical supervision.
Health care teams, including pharmacists, watch for drug interactions. Education helps families prepare for rough days. My experience volunteering at a cancer center showed me how pharmacists explain risks in plain words. Patients ask better questions when they trust their care team. Time spent on education saves lives and reduces mistakes.
Beyond Cancer: Other Uses
Nitrourea doesn’t only serve cancer care. Industrial research uses nitrourea-derived chemicals for certain plastics and agricultural products. Strict regulations keep these substances out of untrained hands. Experts monitor workplace exposure and waste disposal to protect the environment and community health.
Looking Forward
More cancer cases will ask for better answers. Nitrourea and its relatives help keep hope alive for patients and families. Constant research, tight regulation, and honest talk between doctors and patients shape the future of these treatments. My personal take—having witnessed both results and struggles—underlines the need for balanced progress. People want cures, but they deserve safety and dignity just as much.
Is Nitrourea safe to handle?
The Reality of Nitrourea Handling
Every so often an unfamiliar chemical pops up in a lab or worksite, and people start asking the same question: Is this stuff safe to handle? Nitrourea is one of those chemicals where the answer is anything but simple. Common sense and a little bit of digging into safety sheets will tell you straight up — nitrourea isn’t in the same category as table salt.
Danger Hides in Details
Nitrourea has made its way into specialty chemical synthesis, sometimes considered for use in energetic materials. It’s not something you’ll find on a hardware store shelf, or in any setting outside of controlled environments. Safety data from reputable agencies puts nitrourea among the compounds that demand strong respect. If it comes into contact with moisture, it can decompose or even form nitrogen oxides — and those are anything but harmless. Exposure risks include skin burns, eye damage, and respiratory system irritation. One wrong move can hurt someone for life, and I’ve seen colleagues return with red eyes or rashes even after brief, accidental contact with reactive chemicals.
Fact and Hands-On Experience
I’ve spent enough years in research labs to know that guidelines aren’t just paperwork written for insurance. Wearing gloves, goggles, and a properly rated mask makes a difference. The polymer gloves most folks trust won’t always cut it if nitrourea spills. It eats straight through nitrile or latex over time. Chemical-resistant gear, a real lab coat, even sealed shoes — these aren’t options, they’re necessities. Most safety protocols call for fume hoods. At one plant, a minor nitrourea spill cost hours in cleanup and several pairs of ruined shoes.
Known Health Risks Backed By Data
The National Center for Biotechnology Information and European Chemicals Agency both flag nitrourea as potentially toxic by inhalation, skin absorption, or ingestion. Carcinogenicity isn’t fully mapped, but repeated exposure in animal studies hints at chronic risks. One poison center case I read involved lab techs needing hospital treatment after a poorly ventilated job. Sometimes, danger doesn’t announce itself right away. Nausea, dizziness, trouble breathing, or lasting skin redness have shown up from indirect exposure, even when everything looked clean on the bench.
Solutions for Safer Handling
Sitting through dry safety meetings as a student used to seem tedious to me. Now, I look back and wish more people would pay attention. Standard operating procedure for nitrourea goes well beyond what most chemicals demand. Good air flow matters most. Never work alone. Keep a prepared spill kit nearby with absorbents and neutralizers designed for corrosive organics. The smallest sample should get treated with the maximum caution. Waste needs special containers labelled for hazardous energetic materials, not just tossed in the nearest trash can.
Training isn’t a one-and-done event. Chemical safety works best as a regular habit. Cross-check safety sheets before any handling, and don’t improvise protocols. If equipment fails, or a spill occurs, don’t keep quiet hoping nobody will notice. Report, evacuate, document, and call the right emergency contacts. These steps save fingers, lungs, and sometimes lives.
Looking Forward
Making nitrourea safer to handle means more investment in training, better safety gear, and stricter inventory controls. Every lab and facility needs to treat unfamiliar chemicals with more suspicion than trust. My experience says technology only helps so much; people with good habits keep each other safe. It’s not about fear, but about respect for what could go wrong. When folks put in that work, even risky compounds like nitrourea cause fewer bad days overall.
What are the storage requirements for Nitrourea?
Why Storage Matters For Nitrourea
Nitrourea isn’t a compound you can stash anywhere. I’ve worked in labs where strict chemical handling rules saved the day, and Nitrourea stands out for good reason. The main concern comes down to how reactive it can get, especially if left in the wrong environment. This isn’t about paranoia — too many stories exist where negligence led to lab accidents, not just ruined experiments but property damage or worse.
This compound’s got a reputation: even a small spark or a build-up of heat can set off a chain reaction. That means keeping it away from direct sunlight and sources of ignition isn’t just a suggestion; it’s the difference between a secure lab and a headline in tomorrow’s news. Occupational health data makes it clear: storage rules have real-world impact.
Storing Nitrourea Step By Step
Let’s get specific. Nitrourea should stay in a cool, well-ventilated spot. I’ve seen it go wrong when stored near heat or in a closed space without airflow; the heat builds up, and so do the risks. Air conditioning beats an emergency phone call every time.
Moisture spells trouble here. That means storing Nitrourea in tightly sealed containers. I’ve always trusted glass jars with solid lids, not just shrink-wrapped packaging tossed in a corner. If any moisture sneaks in, degradation kicks off, which can undermine research quality and introduce uncontrolled hazards. Keeping containers off the floor, in dry cupboards or on chemical storage shelving, sets a strong line of defense.
Separation stands just as tall in importance. Nitrourea should not sit next to acids, reducing agents, or anything flammable. Mixing these chemicals, even by accident, raises the chance of a dangerous reaction, and chemical burns or fires aren’t experiences anyone forgets in a hurry. Labels need to stay clear and updated—too many incidents crop up because someone thought they grabbed something else.
Supporting Facts And Solutions
Scientific literature and the OSHA Hazard Communication Standard back up these points. Anyone curious can pull up incident reports — combining oxidizers with organic materials or other incompatible chemicals produces explosive results. Trained staff and tight inventory controls beat cost-cutting or shortcuts every day of the week.
Installing distinct, well-marked chemical storage cabinets for oxidizers (with clear signage) will drop risk dramatically. Temperature monitoring strips and humidity indicators give peace of mind. Larger sites go a step further, adding small alarms that sound if temperature or moisture creeps up inside the storage area. These actions aren’t just for large-scale research centers — even high school labs see the benefit.
Regulations often set the baseline, but experience talks. I’ve seen labs lose grant funding after unannounced audits because of a minor slip in chemical storage practices. Direct supervision, regular refresher training, and detailed logbooks tie all these safety habits together.
The Human Cost Of Mistakes
Reading about an explosion in a report feels disconnected until you hear from someone involved. In my own work, I met a technician who suffered burns after a storage cupboard heated up over a weekend. One lapse in following basic storage protocol changed his career path. It’s stories like these that stick in your memory and keep you following best practice, even if it sometimes feels excessive.
In short, storage isn’t an afterthought. By keeping trustworthy records, maintaining dry and cool storage spots, and making safety gear available and visible, labs and businesses dealing with Nitrourea can avoid costly, life-changing mistakes. Smart, disciplined storage habits truly pay off in the long run.
What are the potential health hazards of Nitrourea?
The Real Dangers Lurking in Laboratories and Workplaces
Nitrourea isn’t something most folks run into daily, but for people working in chemical labs or certain industries, the risks can add up fast. As someone who’s spent years with safety goggles on, there’s a real difference between reading about toxic chemicals and actually seeing their effects. Nitrourea, a compound known for its reactivity and toxic traits, deserves much more caution than it tends to get.
How Nitrourea Enters the Picture
Workers come across nitrourea mostly during research or in the manufacturing of specialized chemicals. It’s a white, crystalline solid, looking harmless enough, but don’t let appearances fool you. Nitrourea’s molecular structure means it can break down into dangerous byproducts. There’s a reason regulatory agencies sound the alarm about personal protective gear when handling it.
Short-Term Health Hazards
Breathing in dust or vapors from nitrourea leads to immediate irritation in the nose, throat, and sometimes even the lungs. Eyes become red and watery. Skin contact causes rashes or burns. The compound quickly absorbs through the skin, so gloves and face shields aren't just recommended—they’re essential. During my own lab days, I saw a co-worker forget this rule just once. His red, blistered hands were a tough reminder.
Long-Term Exposure: The Hidden Threat
Nitrourea packs a more serious punch over the long haul. Chronic exposure links up with damaging effects on organs like the liver and kidneys. Some studies tie lengthy contact with certain nitrourea compounds to an increased risk of cancer. Animal research shows these chemicals messing up blood cell formation, leading to anemia and immune system slowdowns. While one bad spill is scary, letting workplace safety standards slide over weeks or years can lead to health disasters down the line.
Dangerous if Unchecked
Sometimes labs or plants cut corners, skipping regular air monitoring or ignoring cracked storage containers. The catch is, nitrourea doesn’t forgive sloppiness. It easily finds its way into the air or water, then straight into workers’ bodies. No chemical hood or sturdy gloves? Sooner or later, someone in that building pays the price.
What Actually Protects People?
Real protection goes way beyond printed safety data sheets. Reliable personal protective equipment only works when it fits well and gets checked all the time. Training should mean more than a signed form—it needs to sink in, be repeated, updated with every safety bulletin. Supervisors have to make air quality tests a regular thing, not just a yearly show-and-tell. Leaky hoods and poor glove quality already caused preventable injuries in factories I’ve visited.
Chemical Management Shouldn’t Be Rare
Safe storage and rapid spill response make the difference between a near miss and a serious poisoning. Nitrourea needs a well-marked, secure container and ventilation that does its job—every hour of every shift. Staff deserve open access to poison control info and real drills. Reporting problems shouldn’t put jobs at risk. Healthy workplaces grow from a culture that admits risk and plans for it, every single day. Keeping nitrourea under control is possible, but only with steady focus and the right investment in safety.
How should Nitrourea be disposed of?
Real Hazards, Not Just Numbers on a Label
Nitrourea stands out in chemical labs as anything but ordinary. Its use in research and explosives means its disposal carries real consequences for people and the environment. Folks who’ve handled even small quantities know the sharp, acidic smell; some learned the hard way how quickly it can cause skin or eye irritation. I remember once seeing a bottle with crystals forming around the cap—nobody wanted to touch it. Negligence isn’t just risky, it’s irresponsible. One misplaced container or a tossed rag soiled with nitrourea residue can ruin far more than a day. Hospitals and emergency rooms aren’t equipped to treat accidental chemical burns or inhalation from this kind of compound on a routine basis. Chemical exposure can cause more trouble down the road, too, even when someone feels fine right after an incident.
Why the Usual Waste Bin Won’t Work
This isn’t kitchen trash. Nitrourea is both toxic and unstable under the wrong storage conditions. Dumping it in a regular landfill or pouring it down the sink risks unforeseen reactions, leaks or contaminating groundwater. Fishing communities and people living near waterways can end up paying the price. I grew up near a river tainted by improper disposal of industrial chemicals. The fish population tanked, and the clean-up took decades. Discarded nitrourea can have the same ripple effect—I’ve seen those impacts up close, and it’s not just the professionals who suffer.
Following Science, Not Shortcuts
Regulations on chemical handling exist for a reason. Nitrourea belongs in a sealed, clearly labeled hazardous waste container and never mixed with other waste. Most labs contract with licensed hazardous waste firms, which collect, document and transfer the material safely. Chemicals like nitrourea usually get neutralized by experts, who follow strict protocols: they control pH, temperature, and ventilation, reducing the risks of toxic fumes or spontaneous decomposition. Homegrown solutions or shortcuts, such as covering waste with sawdust or dumping it in regular bins, multiply the hazards. I’ve seen attempts to mask hazardous waste fail, ending with emergency evacuations and stiff penalties from local authorities.
Accountability Starts With Each User
Better disposal habits start with proper training and honest record-keeping. Every container needs dating and precise labeling. Staff must know what emergency steps to follow if something spills or leaks. I’ve always believed in double-checking—confirming disposal logistics with a supervisor or environmental health office, no matter how rushed things feel. Giving in to shortcuts brings more headaches later. Through frank conversations and consistent reminders, safer habits take root, and new staff catch on quickly.
Progress in Green Chemistry
Researchers today look for safer alternatives wherever possible. Universities and industries now screen substances for toxicity long before ordering bulk materials. One solution includes switching from nitrourea-based reagents to milder, less hazardous options, even if they cost a little more. Companies can reduce demand for high-risk substances by investing in green chemistry solutions and stricter purchasing controls. This shift protects not just employees, but whole communities downwind or downstream from research centers and manufacturing plants.
A Shared Responsibility
Safe disposal isn’t just a checkbox on a lab inspection form. It’s a long chain, linking every user to the world outside the lab walls. Getting it right means relying on science-backed protocols and embracing newer, less toxic materials. Our neighborhood rivers, the air we breathe, and future researchers all benefit from thoughtful stewardship today. People who work with chemicals shape their environment far beyond the bounds of the workplace—one decision at a time.
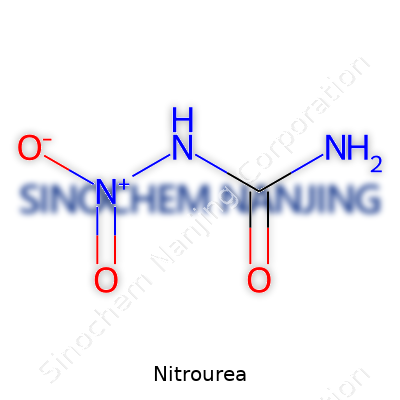
| Names | |
| Preferred IUPAC name | N-nitrocarbamide |
| Other names |
N-Nitrourea Ureanitrone Nitrocarbamide |
| Pronunciation | /naɪˈtroʊ.jʊˌriː.ə/ |
| Identifiers | |
| CAS Number | 556-89-8 |
| Beilstein Reference | 146207 |
| ChEBI | CHEBI:75364 |
| ChEMBL | CHEMBL15610 |
| ChemSpider | 56496 |
| DrugBank | DB00268 |
| ECHA InfoCard | 03dc251f-d19b-4dd1-a7cd-c3b504f7fe2d |
| EC Number | 206-174-3 |
| Gmelin Reference | 8220 |
| KEGG | C07324 |
| MeSH | D009585 |
| PubChem CID | 10477 |
| RTECS number | NL2975000 |
| UNII | 307S086S38 |
| UN number | 1330 |
| Properties | |
| Chemical formula | CH4N2O2 |
| Molar mass | 104.054 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 1.622 g/cm³ |
| Solubility in water | Soluble |
| log P | -0.6 |
| Vapor pressure | 0.0014 mmHg (25°C) |
| Acidity (pKa) | 15.5 |
| Basicity (pKb) | 13.39 |
| Magnetic susceptibility (χ) | -62.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.461 |
| Viscosity | 2.3 cP |
| Dipole moment | 4.06 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 119.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -219 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -631 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | L01AD01 |
| Hazards | |
| Main hazards | May cause fire or explosion; strong oxidizer. Toxic if swallowed, inhaled, or in contact with skin. Causes damage to organs. |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS06,GHS03 |
| Signal word | Danger |
| Hazard statements | H272, H302, H315, H319, H335 |
| Precautionary statements | P201, P261, P280, P308+P313, P405, P501 |
| NFPA 704 (fire diamond) | 3-1-2-OX |
| Autoignition temperature | 220 °C (428 °F; 493 K) |
| Explosive limits | 7-100% (in air) |
| Lethal dose or concentration | LD50 oral rat 83 mg/kg |
| LD50 (median dose) | LD50 (median dose): 83 mg/kg (rat, oral) |
| NIOSH | NA |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.05 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Nitroguanidine Nitrosourea Urea |

