Nitrotriazolone: A Deep Dive into an Energetic Material
Historical Development
Nitrotriazolone, also known as NTO, made its way onto the scene after decades of persistent work in finding alternatives to traditional explosives. Research gained traction in the late 20th century as experts looked for compounds that would provide better stability and reduced sensitivity to impact, heat, and friction. Early formulations of high-energy materials like RDX and HMX raised serious safety concerns, with incidents causing injuries and loss. Scientists and engineers focused on NTO because of its lower volatility and resistance to accidental detonation, providing a safer way to store, handle, and transport military-grade explosives. Testing during development at research labs, such as the ones at Los Alamos National Laboratory and similar facilities in Europe and Asia, showed promise not just on safety but practicality as well. Field use and continuous study over the last few decades built a solid reputation for NTO as a crucial high-energy ingredient for insensitive munitions.
Product Overview
NTO belongs to a group of heterocyclic nitro compounds, serving in formulations where both reliability and controlled power matter. NTO's chemical structure, 3-nitro-1,2,4-triazol-5-one, gives it a distinct performance profile. Manufacturers usually deliver NTO as a light yellow, crystalline solid, sometimes ground into a fine powder for use in pressed or cast munitions and composite explosives. Many military suppliers have migrated to using NTO not just for improved resistance to shock and environmental extremes, but also for compatibility with other energetic materials. Key products in the industry blend NTO with insensitive plasticizers and binders to form main charges, booster compositions, and rocket propellant grains, supporting safer large-scale use in complex weapon systems.
Physical & Chemical Properties
NTO's density lands in the range of 1.85-1.90 g/cm³, with a melting point near 270°C, higher than many legacy explosives. The thermal stability of the compound means it handles storage in hot and variable climates without decomposing or risking unintended ignition. Chemically, NTO resists changes from exposure to water and most organic solvents, letting it keep its energetic properties through rough logistics. Simple paper and digital material safety data sheets usually list NTO as much less sensitive than RDX or TNT, with a friction sensitivity threshold more than double that of conventional explosives. Such resilience has a direct effect on worker safety, where fewer accidents mean fewer injuries and more predictable manufacturing uptime.
Technical Specifications & Labeling
Suppliers mark NTO under its chemical name, 3-nitro-1,2,4-triazol-5-one, referencing a CAS number for clear identification in shipment and handling paperwork. Most batches leave factories with certificates of analysis showing purity above 98%, low moisture content, and particle sizes suitable for use in different manufacturing environments. Labels flag any contaminants or solvents from the production cycle, so downstream users avoid surprises when mixing NTO with oxidizers or plasticizers. Proper packaging keeps NTO dry, clear of sunlight, and sealed from contaminants that might reduce its utility or threaten safety.
Preparation Method
Commercial NTO comes from a multistep synthetic path. Chemists usually start with hydrazine and cyanic acid sources, reacting them to yield the core triazolone ring. Introducing nitric acid to this ring, under tightly controlled temperature ranges, pushes the nitration step to complete the molecule. Each stage relies on managing factors like acidity, temperature, and stirring to coax the highest yields from the raw inputs, with experienced operators watching for any side reactions. Purification comes through a sequence of cooling, crystallization, and washing, stripping out unwanted byproducts. Consistent attention to detail at every phase keeps what ends up in soldiers’ munitions or defense systems working as intended.
Chemical Reactions & Modifications
NTO reacts with reducing materials and some strong bases. Industrial labs have worked on ways to tailor NTO’s reactivity by introducing minor chemical tweaks—substituting atoms or functional groups to dial back or increase sensitivity, or to make composite explosives burn faster or slower. Some teams have tested adding stabilizers to extend shelf life or to further dull sensitivity. These modifications, trialed in real-world situations and through repeated lab tests, give defense planners new tools for controlling blast outcomes, shaping burn rates in propellants, and even safely demilitarizing old munitions.
Synonyms & Product Names
NTO, the shorthand common in defense contractor circles, often appears alongside 3-nitro-1,2,4-triazol-5-one in paperwork and catalogs. Other less frequent labels include TATB-Nitro or simply Nitrotriazolone, though those cause confusion with similar-sounding compounds. Exporters sometimes assign proprietary names when bundling NTO in pre-mixed charges, labeling products like IMX-101 and PBXN-7 depending on the application. Familiarizing all users—technicians, suppliers, and buyers—with these alternate names helps avoid costly mix-ups.
Safety & Operational Standards
Factories treat NTO with respect, but established protocols keep things manageable. Unlike historical explosives that sparked with the smallest static shock, NTO shrugs off vibration and ordinary bumps in armored transport or field supply chains. Staff follow standard practices: anti-static work clothes, sealed containers, dedicated explosion-proof mixing rooms, and clearly marked storage. Safety audits keep everyone honest, checking records and equipment on a regular schedule. Over the years, adherence to these standards cut incident rates, teaching hard lessons not found in the average chemistry textbook.
Application Area
NTO runs across a broad set of uses, mostly military-focused, but researchers have eyed limited civil applications. In modern munitions, NTO holds center stage as part of insensitive high-explosive formulations—artillery shells, bomb casings, missile warheads that need to survive rough handling before seeing combat. Aircraft bombs like the US Air Force’s Joint Direct Attack Munition use NTO blends, aiming for high power with minimal risk of premature detonation. Mines and demolition charges rely on the same properties, letting sappers and technicians operate with less risk than older, touchier materials. Some studies have discussed niche uses in controlled mining or satellite launch boosters, but government oversight and regulatory burdens slow any real shift into these fields compared to more traditional explosives.
Research & Development
Research teams continue to poke and prod at NTO, seeking fresh ways to stretch its capabilities. One branch looks at synthesizing NTO more efficiently, slashing production costs and waste streams so factories run leaner. Another push focuses on greener chemistry—finding ways to reduce nitric acid and solvents, or to recycle reaction byproducts. Military scientists have drilled into blending NTO with nano-materials, tweaking the power curve or adjust the shattering effects during a blast. Each new publication, patent, or conference talk points to NTO’s staying power as a foundation for the next era of safer, smarter energetic materials.
Toxicity Research
Animal and environmental studies suggest NTO ranks below many common explosives for toxicity. Water solubility rates stay low, limiting runoff danger when rounds break or shells degrade after long-term storage. Toxicologists pay close attention to nitrosamine byproducts and breakdown components, screening for anything that might harm wildlife, water sources, or factory workers. So far, regular monitoring hasn’t turned up cause for alarm, but periodic reviews keep everyone on alert, sharing results between militaries, research labs, and environmental agencies. Committees update guidelines for PPE, waste management, and emergency cleanup based on the latest toxicological findings.
Future Prospects
NTO stands as the backbone of many modern insensitive munitions, but the story keeps moving. Defense programs budget multi-year plans around new NTO-based mixtures, banking on improvements in performance without sacrificing safety. Civilian researchers keep hunting for applications in demolition, mining, or controlled access where public safety standards demand nontraditional materials. Advances in synthesis, automation, and recycling may cut costs, taking NTO beyond the limited circle of elite munitions. My own experience—watching industry shifts over decades—makes one thing clear: Reliable energetic materials, safer by design, will shape not only how we fight future wars, but how we manage risks in every setting where controlled energy release matters.
What is Nitrotriazolone used for?
The Role of Nitrotriazolone in Modern Munitions
Nitrotriazolone, often called NTO, pops up in conversations about explosives and military materials. It’s not just another chemical with a tongue-twisting name. NTO has carved out a spot in military technology because of a simple idea—building safer and more reliable ammunition. In the late twentieth century, concerns about accidental detonations and the safety of storing and handling traditional explosives led researchers down a new path. Out of that journey grew NTO, a substance with some real strengths where it matters most.
Why Safety Matters in Explosives
Old-school explosives often bring a hidden danger. Drop them, heat them up, or give them a sharp bang, and disaster strikes. Life in demolitions or the military means enough risk already. NTO changes the equation. Its molecular structure gives it what chemists call “insensitivity.” In plain talk, NTO shrugs off accidental shocks and burns far better than older materials like TNT or RDX. The U.S. Army and several NATO partners have clearly shown interest in this advantage, for good reason. An insensitive explosive cuts down on accidental injuries and protects the people involved from unexpected blasts on and off the battlefield.
Applications in Insensitive Munitions
You’ll find NTO pulled into the mix for new artillery shells, bombs, and warheads, especially in weapon systems that demand higher safety. One good example is the U.S. Army’s EXCALIBUR artillery round, which uses an NTO-containing explosive called PBX-109. This kind of shell can land rough and still avoid going off unless triggered the right way. That gives soldiers much-needed peace of mind. The design of these insensitive munitions means less worry about storage or transport in tough conditions, where unwanted jolts might cause big problems with older explosives.
Environmental Footprint and Health Considerations
Concern about explosives always brings up the question of environmental and health impact. Traditionally, energetics like TNT have left behind toxins that linger in soil and water. NTO’s breakdown and persistence in the environment haven’t shown the same scale of risk so far, according to studies published since 2010. Researchers with the U.S. Army Corps of Engineers have tracked NTO and its byproducts and found lower levels of toxicity for fish and other aquatic life compared to old explosives. That said, more independent work needs to expand on these early indications.
Pushing for Better Solutions
NTO doesn’t fix every problem in military energetics. Critics point out that making it in large amounts demands strong chemical controls and protective gear. A few studies have raised concerns about possible contamination if mishandled or dumped in large amounts. Ongoing oversight, clear regulations, and better recycling or decontamination methods will make NTO’s benefits reach wider. At the same time, funding for further research into greener manufacturing and faster detection when leaks occur could pay off in both environmental and human health.
Looking Ahead
In the long run, the use of NTO in explosives reflects a shift in values alongside technology. No chemical choice comes without trade-offs, but in places where lives and safety are on the line, pushing for progress always matters. If research keeps moving ahead and smarter regulations shape its use, NTO will likely stay part of the modern explosives toolkit for years to come.
What are the safety precautions when handling Nitrotriazolone?
If You Work with NTO, Take It Seriously
Nitrotriazolone, or NTO, finds its way into explosives used in the defense industry. People on the shop floor or in the lab share a common concern—safety. I remember walking through a military testing lab, and the smell of chemicals always made my hands sweat. I’ve learned it’s better to feel nervous and cautious than to assume everything will work out fine.
Understanding the Hazards
NTO delivers lower sensitivity compared to legacy explosives like TNT, but it still packs a punch. Any lapse in basic handling can mean serious consequences. Static electricity, friction, and impact can set off this compound. A drop on a hard floor won’t just leave a dent—it could leave a crater. Even cleaning up a spilled pinch means that you need to stop, think, and protect yourself.
Personal Protective Equipment Is Non-Negotiable
I’ve seen co-workers roll their eyes at goggles and gloves. That attitude disappears after hearing an explosion in another room or reading injury reports in safety briefings. NTO dust can irritate skin, affect breathing, or hurt the eyes. Wear lab coats, gloves, and safety goggles every time. If you’re working with a big batch or in a tight room, use a face shield and a proper respirator. NTO sneaks into the air if given the chance.
Respect the Workplace
Work areas dealing with NTO need strict control. Grounding equipment, anti-static mats, and spark-proof tools all play a role. I’ve tripped over clutter before and realized a clean work area is more than just tidy—it keeps everyone alive. Hand tools need to be made from plastics or non-sparking metals. Floors can’t have spots where dust piles up. Designated zones for NTO storage cut the risk of hazardous cross-contamination with incompatible chemicals, especially things like metals that trigger messy reactions.
Handle Spills and Waste Thoughtfully
Nobody likes cleanup duty, yet spills happen. Take time to isolate the area. Wear all your protective gear. Carefully sweep up small spills with non-sparking tools into safe, labeled containers. Treat the cleanup as hazardous waste—no dumping it down a drain or ordinary trash. I’ve watched people double-bag waste, and I’ve done it myself, knowing it slows down the work but keeps the danger out of our everyday lives. Chemical neutralizers can also help, but any cleanup method needs to match strict company policy.
Always Double-Check Training and Policies
People skip over procedure updates or brush off training for the sake of speed. No shortcut is worth the risk. The military and manufacturers update handling techniques when new research finds a safer way. Making time for drills and refreshers makes a real difference. If you see a co-worker skipping steps, speak up. It’s not about calling them out—it’s about sending everyone home safe.
Potential Solutions for Better Safety
Modern labs invest in real-time environmental sensors, so workers get immediate alerts if dangerous concentrations get released. Automatic grounding systems can zap away static before it builds up. Digital checklists on tablets force teams to stick to safety protocols before they start anything. More companies now rotate assignments so no one person spends too much time around hazardous materials, keeping everyone focused and careful. Research also continues on creating even less sensitive explosives to reduce danger at every step.
What is the chemical structure of Nitrotriazolone?
Understanding Nitrotriazolone at the Molecular Level
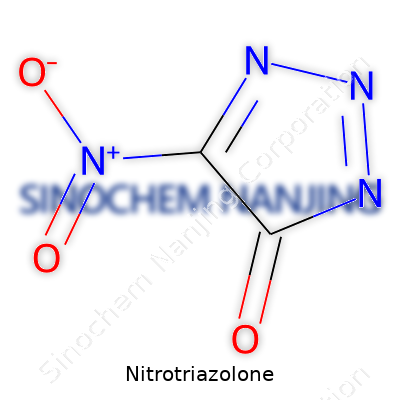
Nitrotriazolone, known by its chemical formula C2H2N4O3, stands out in the field of energetic materials. Chemists who have handled it recognize its stability compared to older explosives and appreciate its unique structure. Looking closely, the molecule forms a five-membered ring—officially called a triazolone ring. Imagine three nitrogen atoms lined up in a ring with two carbon atoms, one of which carries a nitro group (-NO2). This nitro group forms the core site of the compound, lending much of its stability and performance profile.
Bonds in Nitrotriazolone create a tapestry of tight angles and electronic interactions. The nitro group connects to the carbon at position five on the ring. An oxygen atom double-bonded to another carbon forms the "lactam" (ketone-like) site. The remaining positions around the ring fill in with hydrogens, balancing the molecule’s charge and contributing to its unique thermal properties. Chemists often marvel at the electron delocalization over the ring, which promotes both resilience to accidental detonation and a finely-tuned burn rate once triggered.
Why Structure Shapes Application
Veterans in laboratories know that a molecule’s structure determines its day-to-day behavior. Nitrotriazolone’s ring looks simple at first but the balance between its nitro and lactam groups creates a sweet spot. Explosives based on this molecule offer a step forward—safe to store and handle, yet effective in use. Factories producing propellants or advanced munitions now keep Nitrotriazolone on their radars precisely because its compact, nitrogen-rich ring weighs against instability. Patents and military research publications consistently mention its low sensitivity to impact and friction. For people living near production facilities or ammunition depots, these actual safety margins matter more than technical jargon.
Research studies published over the past two decades confirm its thermal stability and low risk of accidental ignition. Beyond safety, the ring system easily bonds with plastics or other stabilizers, broadening its usefulness for main charges, boosters, and propellants. Modern research points to Nitrotriazolone as a backbone for new, “insensitive” munitions, which resist accidental detonation from fire or shock.
What Makes It Important Today
Conflicts worldwide still demand safer yet reliable explosives. Nitrotriazolone’s chemistry gives it a real-world edge, especially as engineers seek to design greener or less polluting energetic materials. Environmental impact cannot take a backseat; the ring structure offers a way to break down more cleanly in the environment. Research from military contractors and academic labs supports this claim. Waste byproducts tend to be less harmful compared to traditional explosives.
Efforts to improve energetic compounds now often focus on tweaking the triazolone ring, hoping to find versions that work at lower cost or greater efficiency. Chemists sometimes substitute different atoms or groups on this ring to modulate performance, replacing pieces of the nitro group, or adding other stabilizers. The field can move quickly when theory meets practical needs, and Nitrotriazolone’s structure provides just the right blend of robustness and adaptability.
Moving Toward Better Solutions
Safer production lines remain a must for industry. By using Nitrotriazolone, plants see fewer unplanned incidents—saving lives and lowering insurance costs. Training staff on handling procedures grows simpler too, since the material resists shock and friction better. Even disposal becomes easier, cutting risks for communities near storage sites.
The pace at which new applications pop up shows how valuable this molecule has become. Next-generation explosives built around Nitrotriazolone point to a future where technical innovation goes hand-in-hand with environmental and public safety. The chemistry inside that five-membered ring changes the story, not just on paper, but everywhere these energetic materials play a part.
How should Nitrotriazolone be stored?
Understanding What’s at Stake
Nitrotriazolone (NTO) gives explosives their punch—without the volatility of some traditional compounds. Its value to military and mining industries is clear, but few outside these fields ever hear about the care it demands behind closed doors. Handling NTO well isn’t just about ticking boxes for compliance; it’s about making sure workplaces and communities stay safe from harm.
Why Pay Attention to Storage?
From my years working near industrial materials, it’s easy to see that treating energetic chemicals with casualness can spell disaster. NTO brings lower sensitivity than many alternatives, yet nobody should treat it like an everyday chemical. Reports from the U.S. Army and the Environmental Protection Agency highlight that NTO, while safer in some regards, can still present risks linked to fire, explosion, and environmental contamination. Incidents from sloppy storage practices—mistakes with humidity, wrong packaging, poor tracking—have caused injuries and led to long clean-ups.
Getting Conditions Right
One mistake I’ve seen is assuming NTO can handle temperature swings or moisture. In fact, NTO’s stability depends on steady, dry, and cool surroundings. Chemical safety references like those from OSHA make one thing clear: low humidity and constant temperatures keep instability at bay. Materials need solid, airtight containers—preferably made of plastic or lined metal that won’t corrode or react over time.
Shelves should be sturdy and away from any source of heat (boilers, sunlight, or machinery), keeping ambient temperature below 25°C if possible. Some crews use desiccants in storage rooms to reduce the risk of clumping or accidental reaction. These steps protect not just the product, but those who handle it each day.
Separating and Securing the Stock
Safe chemical management often boils down to avoiding bad combinations. Don’t keep NTO close to oxidizers, fuels, acids, or anything else that could start an unintended reaction. Having spent years observing chemical stockrooms, I’ve seen how sharp labeling and detailed logs help prevent mix-ups. Access needs controlling: workers should have training, protective gear, and understand emergency routines.
Long-Term Stewardship and Responsibility
Storage rules shouldn’t gather dust in a binder. Facilities handle audits, sometimes unannounced, and tracking every batch, every container, means nobody’s left guessing about what’s on the shelf. In the years after bigger incidents, insurance companies and regulators started asking about surveillance, alarms, and even staff background checks—not just locks on doors. Responsible users draw up spill and fire response plans and rehearse them, treating preparedness as a daily discipline, not a formality.
There’s an environmental angle here too. Spills or leaks from neglected NTO stockpiles can seep into soil or water, harming local ecosystems and public health. Site managers rely on secondary containment—pans, sealed flooring, rapid drain covers—to make sure a single mistake doesn’t spread beyond the warehouse.
Improving the Culture Around Storage
Industry experts and workers agree: real safety comes from a culture where people ask questions and point out concerns, not just follow orders. Sometimes it’s the veteran who spots a shelf sagging or a rookie who notices a leaky seal. Pointing these out shouldn’t earn pushback—it should be rewarded. Regular review and open conversation go further than any written policy in keeping materials like NTO contained and communities protected.
What are the potential hazards of Nitrotriazolone?
Understanding Nitrotriazolone’s Role and Risks
Nitrotriazolone pops up most often in conversations about military explosives. Chemists count on it for making safer, less sensitive explosive materials compared to old-school compounds. This sounds good on the surface, though experience has taught me to take a second look at “safer” labels, especially with chemicals that pack a punch like this one. Anytime you’re dealing with something used in munitions, potential hazards come with the territory.
Chemical Properties: What Lurks Under the Hood
This stuff doesn’t just sit quietly on a shelf. Nitrotriazolone offers relative stability against heat and physical shock, but its decomposition can kick off quickly at higher temperatures. Heavy handling, or storage near open flames or electrical sparks, will push it past its limits. If stored improperly, its degradation releases gases that raise the risk of fire or explosion.
Beyond its reactive side, this compound mixes poorly with several other chemicals used in similar applications. Unplanned reactions with strong acids, bases, or specific metals can start a chain of events that puts workers in danger fast.
Health Effects: Taking the Human Angle
Lab techs and cleanup crews know what an accidental spill means. Direct skin exposure to nitrotriazolone – even grainy dust floating in the air – could irritate eyes and lungs. There’s also evidence showing certain breakdown products might affect the nervous system. These small, repeated hits take a toll, and older research has suggested possible connections to longer-term organ damage. This isn’t just something I picked up from safety briefings; stories from people working in production plants always sound the same. Folks end up at the clinic with headaches and breathing trouble.
There’s less research on what happens if it seeps into water or sticks to soil. What we do know points to a compound that doesn’t disappear overnight. Local streams could carry it far beyond the original site, putting fish, birds, and groundwater at risk for longer stretches than anyone would like.
Dealing With Nitrotriazolone: Safer Practices Matter
Using this compound safely starts with paying attention during transport and storage. Keeping containers cool, dry, and tightly sealed keeps accidents at bay. Specialized training should be regular—refresher drills, hands-on practice with respirators and chemical suits, and clear labeling all the way down the supply chain help keep things running smoothly.
Facility managers have a responsibility to set up systems for rapid spill response. That means well-marked showers, eyewash stations, and the right kind of absorbent materials ready within arm’s reach. Waste products need disposal under strict rules; chemical incineration—done offsite—usually gets the green light from environmental authorities.
Better Testing and Community Awareness
Manufacturing giants and military contractors need to invest in better monitoring for leaks or spills. I’ve seen smaller shops cut corners on regular air and soil testing, and that almost always bites them later. Local communities also benefit from transparency—knowing which chemicals move through nearby railways or what steps factories take if an accident happens.
Anyone working around nitrotriazolone gains from a healthy respect for its risks. If handled with care and solid planning, it plays its part without causing unplanned disasters. Shortcuts rarely pay off with materials as energetic as this one.
| Names | |
| Preferred IUPAC name | 5-nitro-2-oxo-1,2-dihydro-1,2,4-triazol-3-one |
| Other names |
NTO 3-nitro-1,2,4-triazol-5-one 3-Nitrotriazol-5-one |
| Pronunciation | /ˌnaɪtroʊˌtraɪ.əˈzoʊ.loʊn/ |
| Identifiers | |
| CAS Number | 932-64-9 |
| Beilstein Reference | 147366 |
| ChEBI | CHEBI:76272 |
| ChEMBL | CHEMBL2104741 |
| ChemSpider | 21590200 |
| DrugBank | DB11638 |
| ECHA InfoCard | 100.112.011 |
| EC Number | 208-732-1 |
| Gmelin Reference | 67668 |
| KEGG | C18704 |
| MeSH | D020123 |
| PubChem CID | 135398 |
| RTECS number | XN8575000 |
| UNII | S4TFG8YK4A |
| UN number | UN0237 |
| Properties | |
| Chemical formula | C2H2N4O3 |
| Molar mass | 129.07 g/mol |
| Appearance | Yellow crystalline solid |
| Odor | Odorless |
| Density | 1.93 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -0.7 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 3.44 |
| Basicity (pKb) | 6.41 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.809 |
| Dipole moment | 5.4892 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 216.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –82 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2952 kJ/mol |
| Pharmacology | |
| ATC code | N03AX13 |
| Hazards | |
| Main hazards | Explosive; toxic if inhaled, ingested, or absorbed through skin; causes irritation to skin, eyes, and respiratory tract. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS01,GHS03,GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H317: May cause an allergic skin reaction. |
| Precautionary statements | P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-2-4-OX |
| Autoignition temperature | > 240 °C |
| Explosive limits | Lower: 0.8% Upper: 70% |
| Lethal dose or concentration | LD50 oral rat 810 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1320 mg/kg (oral, rat) |
| NIOSH | NA Delegation |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 40 mg/m³ |
| Related compounds | |
| Related compounds |
TATB DATB TNAZ ONTA RDX HMX |

