Nitrosylsulfuric Acid: A Closer Look at a Powerful Chemical
Historical Development
Before the rise of modern chemical engineering, manufacturers and researchers leaned into basic acids and salts to drive progress. Nitrosylsulfuric acid has turned up through the annals of industry, its value tied to the development of dyes, explosives, and fertilizers. Long before refined purification techniques, chemists stumbled upon its reactive punch in the course of fusing sulfur trioxide and nitrogen oxides. Propelled by the growing needs of explosive manufacturing, especially in Europe during the late 1800s, nitrosylsulfuric acid became a staple for anyone trying to harness or control potent nitration reactions. Industry kept circling back to it thanks to its role in munitions and synthetic organic reactions. Suppliers and labs cataloged the chemical under different names, which muddled things in early trade and regulation, but its hallmark—a sharp oxidizing strength—always gave it away.
Product Overview
This acid comes across as a dense, oily liquid with a yellowish to reddish tint. Workers who handle it pick up on its acrid, suffocating fumes and the feeling it gives on the skin—a tingle warning of burns on the way. In storage tanks and delivery jugs, the acid’s weight and tendency to react with moisture keep warehouse managers always on alert. Shipping regulations shine a bright light on containers, with strict tags and emergency sheets alerting transporters to the dangers of leaks or mishandling. Industrial users appreciate the chemical’s reliability in stripping away impurities and promoting key reactions, setting it apart from less aggressive acids.
Physical & Chemical Properties
With a molecular formula of NOHSO4, nitrosylsulfuric acid packs a punch both as an oxidizer and a dehydrating agent. Its boiling point hangs at around 180°C, but decomposition and gas emissions start far below that, which keeps plant operators wary during transfer operations. The compound dissolves in sulfuric acid with ease, giving manufacturers more flexibility in process design. Its fumes bring a harsh, nitric odor, breathing danger into any poorly ventilated lab. The orange-red color deepens as decomposition sets in, often betraying the presence of nitrogen dioxide. Hydrolysis—direct contact with water—leads to an eruption of NO and SO3, turning everyday spills into emergencies. These properties draw from my own time in the lab, watching seasoned chemists triple-check containers and fume hoods before opening anything marked with the nitrosylsulfuric warning label.
Technical Specifications & Labeling
Chemical plants set standard concentrations for nitrosylsulfuric acid, typically ranging from 20-40% in sulfuric acid, based on intended process or reaction. Safety data sheets set out the official hazard classification: corrosive, toxic by inhalation, and a clear player in producing poison gases on contact with many organics and metals. GHS pictograms declare the risks with skull-and-crossbones iconography, and all drums and containers need secondary labeling detailing emergency steps, specific gravity (around 1.75 at room temperature), and appropriate UN numbers, such as 3264 for corrosive liquids. The experience of reviewing paperwork next to nervous warehouse managers still fresh—the acid’s dangers demand paperwork squared and rechecked by every handler.
Preparation Method
Manufacturers mainly prepare nitrosylsulfuric acid by blending sulfuric acid with nitrogen oxides, especially nitrogen dioxide. Modern plants meter nitric oxide into a cooled mixture of sulfur trioxide and concentrated sulfuric acid, facilitating the capture of excess gases in scrubber towers. Temperatures must stay tightly controlled, edging high enough to encourage combination but well below levels where runaway reactions would kick in. Facilities route off-gasses through solid acid absorbers or bubbling units, keeping workplace safety up to par and minimizing environmental discharge. My early plant tours gave a first-hand glimpse at the dance of process controls—slight temperature spikes would trigger frantic valve checks and system lockdowns, every operator wary of the brittle line between stability and runaway emissions.
Chemical Reactions & Modifications
In chemical syntheses, nitrosylsulfuric acid stands out for its role as a nitrating agent. It contributes NO+ ions, which become essential for manufacturing explosives, dyestuffs, and some pharmaceuticals. On contact with anilines or aromatic compounds, it injects nitroso groups, driving yields higher than less reactive acids could achieve. Once mixed into process streams, side reactions often generate more complex mixtures—industry chemists develop targeted cooling and monitoring routines to prevent runaway exotherms. More advanced research explores selective modification of the acid for green chemistry, swapping out traditional precursors for less polluting alternatives. The strength and flexibility of the acid have always made it a favorite among those eager to tweak, modify, and optimize reactions for specific yields.
Synonyms & Product Names
This acid picks up labels depending on manufacturer, region, and application. Commercial literature boasts names like nitrosyl hydrogen sulfate, NOHSO4, or more generically, “oleum with nitrosyl.” Older texts use variations like “chamber crystal” or “nitrating acid D,” especially in explosives industries. Potential buyers and regulators keep tabs on these names to cut down on confusion and ensure consistent records across borders and suppliers. Experiences at regulatory workshops showed how quickly an unlabeled barrel featuring only a synonym caused confusion—even grizzled safety staff had to double-check before signing off on shipments or waste declarations, highlighting the importance of clear and universal labeling.
Safety & Operational Standards
Without strict safety, nitrosylsulfuric acid can turn everyday work into potential disaster. Even a small splash eats away at metal fittings and human tissue fast—the acid commands respect from all who work with it. Facilities maintain extensive personal protective gear requirements, including full-face shields, acid-resistant gloves, boots, and specialized spill kits. Drums and pipes undergo regular inspection for pitting and corrosion; ventilation must draw off escaping fumes before workers set foot in storage zones. Emergency showers, eyewash stations, and leak detectors fill every corner. Regulatory agencies enforce detailed training for all handlers, including rescue and decontamination drills. It takes discipline to maintain high standards, but stories of near-misses underline why every shortcut can have lasting consequences.
Application Area
Heavy industry depends on nitrosylsulfuric acid for key chemical transformations. In dye manufacture, it helps produce bright, stable colorants by efficiently introducing nitro and nitroso groups into aromatic rings. The explosive sector leans heavily on the acid’s nitrating potential, turning out vital ingredients for dynamite and propellants. Water treatment operations have found ways to use it for removing stubborn organics and breaking down toxic residues, though the danger of accidental releases has driven several operators to rethink its use. Academic and commercial labs keep it on hand for synthetic routes where less aggressive acids disappoint. Over decades, industries have extracted remarkable value from the acid, relying on its unique chemistry to power growth and innovation.
Research & Development
The investigative work around nitrosylsulfuric acid focuses on harnessing its potential with more safety and less waste. Chemists push for greener synthesis, trying to cut the acid’s nitrogen oxide emissions and reduce the volume of hazardous waste during disposal. Some research teams experiment with catalysts or process intensification—speeding up reactions while cutting down on unreacted byproducts. Other labs evaluate alternative reagents that could replace the acid in nitration, balancing cost and performance with new safety benchmarks. As someone who’s watched R&D in both big chemical plants and university labs, the search for more efficient, less destructive use continues to shape how the acid appears in future applications.
Toxicity Research
Toxicologists flag nitrosylsulfuric acid as a top-tier health hazard. Exposure to its fumes leads swiftly to burns of the skin, eyes, and respiratory tract. Inhalation episodes cause pulmonary edema, and direct contact scars tissue in breathtakingly brief timeframes. Chronic exposure, even at lower levels, increases risk for respiratory disease and possibly long-term organ damage. Animal tests lend data to regulators, underlining severe acute toxicity and prompting strict workplace exposure limits in most industrialized countries. Safety officers never downplay the risks, demanding not just gear but routine health monitoring for exposed workers. It’s hard to overstate the dangers—stories abound in chemical circles of colleagues hospitalized or burned from a brief lapse in vigilance, hammering home why training, preparedness, and medical readiness shape every aspect of handling and disposal.
Future Prospects
Industry attention now turns toward greater sustainability and safety. The acid’s strong performance as a nitrating and oxidizing agent attracts researchers hoping to engineer safer substitutes or less hazardous manufacturing routes. Innovations in closed-system handling promise to limit exposure and minimize environmental leakage, but cost and technical hurdles still bog down widespread adoption. Regulatory landscapes tighten, punishing polluters and those who sidestep robust controls, which pushes manufacturers to examine every leak, vent, and waste stream. As environmental expectations mount and markets evolve, the pressure rises to rethink the chemistry, improve process safety, and drive down accidents. In this transition, scientists, engineers, and policymakers keep weighing the benefits against the health costs, always striving to shift nitrosylsulfuric acid’s legacy toward safer and more responsible stewardship.
What is Nitrosylsulfuric Acid used for?
The Role Nitrosylsulfuric Acid Plays in Chemical Processes
The factory floor runs on more than sweat and steel. It relies on chemicals designed to kickstart reactions or strip impurities. Nitrosylsulfuric acid deserves attention because it does a job few other substances pull off—transforming raw materials in some of the world’s most important manufacturing chains. This compound, with its sharp, corrosive punch, takes center stage in nitration and chemical synthesis.
Unlocking Dyes and Pharmaceuticals
Growing up near an older industrial town, I could always spot the clusters of chemical plants—tall stacks, clouds of vapor. The products those plants churn out wind up in a thousand everyday goods, especially dyes and pharmaceuticals. Here’s the thing about nitrosylsulfuric acid. It acts as a key intermediate for making nitro compounds. The most familiar one? Nitrobenzene, the stepping stone for aniline, which forms the backbone of synthetic dyes and medicines. Factory operators combine sulfuric acid with sodium nitrite to get their dose of nitrosylsulfuric acid, then use it to push aromatic compounds through nitration. Without this acid, paint colors would look faded and drugmakers would face bottlenecks in production.
Metal Cleaning and Pickling
Anyone who has spent time working with metal knows rust and scale. Before steel can get shaped into bridges, cars, or machinery, it needs a clean slate. Nitrosylsulfuric acid provides a strong oxidizing blend that tackles stubborn deposits. With proper handling, it strips metals and preps them for further treatment or galvanization. Building safe infrastructure and durable goods relies on these critical cleaning steps. Cutting corners on the cleaning process leads to weak welds and early corrosion, which nobody wants in a car frame or elevator shaft.
Environmental Impact: Handling With Care
Toxic releases from chemical plants have sparked concern in many communities, and nitrosylsulfuric acid demands respect. It can break down into nitrogen oxides, contributing to air pollution. Nobody wants that kind of problem next door. Operators must keep a close eye on storage, handling, and disposal. Regulations guide plant managers to install scrubbers, train workers, and monitor for leaks. Safety data shows serious injuries can result from direct contact or inhalation, so gloves, masks, and strict protocols aren’t optional—they’re life-saving. Seeing the effects of poor safety in some older sites makes you grateful for today’s higher standards, but constant vigilance stays necessary.
Safer Alternatives? A Work in Progress
Green chemistry teams want to reduce reliance on toxic compounds like nitrosylsulfuric acid. Research pushes toward using milder reagents or closed-loop systems to trap emissions before they escape. Some new processes skip traditional nitration in favor of greener solutions. Implementation takes time and investment, but it keeps moving forward. The shift doesn’t happen overnight, especially for entrenched production lines, but gradual adoption of less hazardous alternatives builds a safer, cleaner practice for industry and community alike.
Balancing Progress With Safety
Nitrosylsulfuric acid makes things possible—from vibrant dyes to cleaner metals. Industry turns it into value, but every time a company measures out a batch, it steps into a pact with workers and neighbors: keep it contained, keep it safe, look for better ways in the future. Recognizing and managing the risks tied to this substance protects both people and progress.
How should Nitrosylsulfuric Acid be stored safely?
The Reality of Strong Acids in the Workplace
Strong acids stop anyone in their tracks, especially ones like nitrosylsulfuric acid. This compound carries more risks than common industrial chemicals thanks to its unique makeup. It has a striking yellow to reddish color and fumes aggressively. Breathing in those fumes or coming in direct contact can land someone in real trouble fast. Because of this, every move involving nitrosylsulfuric acid calls for careful planning, real training, and the right storage methods from the start.
Never Treat Storage as an Afterthought
Having worked in chemical storage facilities and handled concentrated acids, I learned that details matter more than rules on paper. Nitrosylsulfuric acid reacts ferociously with water and most metals. Just the tiniest leak or a hint of moisture can lead to fumes, risk of fire, or even violent explosions. Only certain containers can withstand this assault. Most commonly, thick, qualified glass and special grades of plastics hold up for the long haul. Ordinary steel and aluminum corrode quickly, leading to leaks that only get bigger if unnoticed.
Keep Moisture Out, Always
This acid chases after water. Leave a cap loose or lid unsealed, moisture will seep in from surrounding air almost instantly. The reaction doesn’t just fizzle—it releases dangerous gases including nitrogen oxides and sulfur dioxide. For that reason, every container needs to close airtight. Storage areas should stay dry and cool, with humidity kept below 50%. I’ve seen what happens when a warehouse gets too damp. Even a hairline crack in the bottle turns into a plume of fumes, setting off alarms and putting people nearby in immediate danger.
Ventilation and Segregation Save Lives
Never underestimate fume buildup. An enclosed room without good ventilation becomes a hazard zone. Strong local exhaust and general ventilation are critical—hood systems work well in storage rooms, giving fumes nowhere to settle. No acid sits next to incompatible chemicals. Bases, combustibles, organics, and water-reactive materials belong somewhere far away. One small spill can turn into a toxic cloud or even a fireball if nil attention gets paid to what shares the shelf.
Training Makes the Difference
People, not policy manuals, prevent accidents. Every employee handling nitrosylsulfuric acid should know its dangers backwards and forwards. Spills happen despite best efforts. Without spill kits and neutralizing agents at arm’s reach, panic replaces calm decisions. Lab coats, goggles, and gloves come standard. For messier jobs, face shields and acid-resistant suits turn a risky situation into a controlled one. Regular drills, ongoing hazard communications, and honest conversations about past incidents breed a culture of respect for what can go wrong.
Proper Labeling and Inventory Controls
Every bottle, canister, and jug needs a clear label visible from a distance. The label means more than just a name. It shouts out hazards, provides a quick HAZMAT reference, and even includes emergency contact info for good measure. Digital inventory tracking, updated in real-time, lets supervisors know what’s on hand, where it sits, and how much remains. Nothing gets lost, misplaced, or overlooked. Regulatory checks become straightforward instead of headaches.
Preparedness—Not Just a Buzzword
Fire extinguishers suited for chemical fires and proper evacuation plans make the real difference in high-stakes situations. Emergency showers and eyewash stations remain close by—not hidden in a hallway. I’ve seen places that put drills on the calendar and treat preparedness as a living thing, not a box to check. Quick response can turn a disaster-in-the-making into a story with a much better ending.
Safe storage of nitrosylsulfuric acid doesn’t just protect products—it keeps people out of harm’s way, saves money in the long run, and shows real respect for the power of chemistry.
What are the hazards of handling Nitrosylsulfuric Acid?
Real Dangers Hidden in Clear Solutions
Most people don’t picture a colorless or yellowish liquid made in the lab when they think of a dangerous job. I had no clue how real the risks could be until faced with nitrosylsulfuric acid myself. It’s not a chemical that makes headlines, but one mistake and you wish it stayed on the shelf.
Corrosive Power That Burns Deep
Touching nitrosylsulfuric acid feels like a slow-motion disaster. I’ve seen gloves melt and skin turn white even through layers of nitrile. This acid doesn’t just sting; it eats away at tissue, destroying cell walls and leaving wounds that don’t heal quickly. Eyes are even more vulnerable. One drop can blind. I learned this lesson after a lab accident that sent a colleague to urgent care. He stood half-blind for days, desperate for relief, after the tiniest splash. The risk is not just personal; even fume hoods can corrode if they’re not handled with respect.
Breathing Fumes Brings Its Own Risks
I used to think the worst chemicals gave off smells as a warning. Nitrosylsulfuric acid drifts in with invisible fumes. These gases irritate the respiratory tract and lungs, and once inhaled, you feel it almost immediately—a scratchy throat, then burning deep in the chest. Long exposure has even bigger consequences. Some researchers have developed lifelong asthma, and there’s literature on risks of pulmonary edema, which fills your lungs with fluid. Nitrogen oxides released can deepen the hazard, causing headaches and confusion before you even notice the leak.
Reactivity That Surprises Even the Seasoned Chemist
One problem: water and nitrosylsulfuric acid don’t mix well, and people forget. Add a bit of moisture, and things start to bubble, spill, or splatter. That’s how I learned what a chemical burn feels like. The acid reacts with organic materials, metals, and even paper—releasing heat, fumes, and sometimes flames. Stories of small explosions are common gossip among lab techs. It cost one lab team thousands in repairs, and nobody wants to be the person who cleans up that mess.
Simple Steps Make a Difference
Over the years, I’ve learned that good habits stop accidents before they start. Forgetting safety goggles or ignoring a spill—it only happens once before you never forget again. Anyone working with nitrosylsulfuric acid keeps running water nearby, checks fume hoods every time, and double-gloves with silicone or equivalent. No shortcuts ever pay off. Spills demand quick response with neutralizing agents like sodium bicarbonate, since water can make things worse. Stocking antidotes may sound old-fashioned, but access to fresh eyewash and shower stations can mean the difference between a bad scare and a life-changing injury.
Staying Ready, Staying Alert
Nitrosylsulfuric acid doesn’t forgive carelessness. Training never ends; staff rotate through drills, and everyone knows where the safety sheet is. Sometimes new faces ignore these lessons, so old hands step up to show the scars and tell the stories. Forgetting never helps anyone, and staying alert isn’t just a rule—it’s a habit that spills over to every part of life, at work and home.
What is the chemical formula of Nitrosylsulfuric Acid?
The Formula in the Spotlight
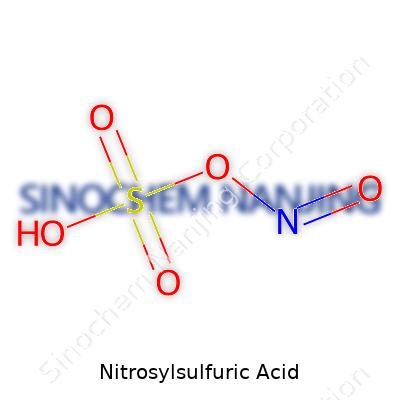
Anyone who’s mixed chemicals in a school lab or handled industrial processes will probably remember bitter smells, steamy glass, and lab coats stained yellow after careless moments. Nitrosylsulfuric acid, known for its striking yellow hue, isn’t the compound you stumble upon in a kitchen experiment. Its formula—NOHSO4—describes its makeup: one nitrogen, one oxygen, one hydrogen, one sulfur, and four oxygens arranged in a way that keeps professionals alert.
Where You Meet It
Factories don’t shout about nitrosylsulfuric acid, but it’s a familiar name in industries working with dyes, explosives, and pharmaceuticals. The chemical steps it enables run deep in dye manufacturing—especially the diazotization process. Every time you see brightly dyed fabric or specialized diagnostic reagents in a lab, there’s a fair chance nitrosylsulfuric acid played a backstage role.
Handling Real-World Risks
NOHSO4 isn’t exactly warm and welcoming; it’s highly corrosive and unleashes toxic fumes. Years spent in labs have taught me that strong acids reward care. Gloves, protective goggles, and fume hoods aren’t just for show—they’re essential for safety. Even the most seasoned chemist has stories about near-misses. Once, an improperly capped bottle threatened to eat through a bench, sending up orange fumes that could choke the back of your throat. Immediate cleanup, good ventilation, and strict storage rules kept disaster at bay.
People in surrounding communities rely on strict chemical management to protect local air and water from substances like nitrosylsulfuric acid. Local news sometimes covers spill incidents, and I’ve seen real anxiety in these stories from residents who live near chemical plants. Environmental teams move quickly: neutralize with lime, contain runoff, prevent exposure. Regulations like OSHA’s chemical safety standards and the EPA’s reporting requirements didn’t appear out of thin air—they respond to tragedies from the past.
Learning From Experience
Many processes still use NOHSO4 because it works efficiently where milder chemicals fail. At the same time, manufacturers have started seeking alternatives. Personally, I’ve seen projects pivot to less hazardous routes, trading off a bit of efficiency for greater peace of mind. Investments in proper containment systems, chemical detection, and staff training go a long way to reduce risk.
Safer handling starts with awareness. Clear labeling, regular drills, and honest reporting of leaks or mistakes build the safest workspaces. Several chemical companies now require cross-checks and “buddy systems” for tasks involving strong acids. There’s wisdom in teamwork—nobody’s left guessing whether the air’s still safe to breathe.
What’s Next
Chemistry keeps evolving, but some ingredients of safety and responsibility never go out of style. As science moves forward, the chemical formula of nitrosylsulfuric acid reminds us how much is possible when knowledge meets real-world caution. With each new use case, the lessons learned in labs and on factory floors keep shaping better, safer chemistry for everyone.
How should spills or exposure to Nitrosylsulfuric Acid be handled?
Why Care About Nitrosylsulfuric Acid?
Nitrosylsulfuric Acid stands in a category few would want to confront outside of a controlled lab. This highly corrosive, reactive substance can cause severe injuries on contact and generate toxic gases. For those of us who have worked in process chemistry or chemical manufacturing, this isn’t just something buried in an old MSDS. It’s a real-world risk found in dye, pigment, and explosive production. Stories of complacency come up once in a while: a drum leaks, a line fails, or a worker mistakes one barrel for another. Learning never feels as urgent as the moment you’re standing near a leaking pipe, wondering if the drill worked.
Immediate Steps to Take
Bagging the right personal protective equipment always comes first. Gloves rated for strong acids, chemical splash goggles, a face shield, and acid-resistant suits do more than make you sweat—they save skin and eyesight. The vapor risk means a full-face respirator or self-contained breathing gear isn’t just for show. I’ve seen colleagues brush off “extra” PPE on hot days—later, regretting it.
Isolate the area fast. Anyone who doesn’t work with corrosives should be somewhere else. No heroics or trying to “clean a little” before sounding an alarm. Doors close and warning signs go up. Every facility worth its salt lays out a clear “who to call” and “who leads” plan for this purpose, and it works only if folks actually use it.
Tackling the Spill
Small spills often start with containment. Neutralizing agents—like sodium carbonate—should be ready nearby. Dumping water isn’t just a basic mistake; it can send toxic nitrogen oxides into the air or trigger fires. Spill kits designed for strong acids usually feature absorbent pads and neutralizing granules. Proper kit selection matters: generic absorbents don’t cut it for acids like Nitrosylsulfuric.
Attack the problem from the outside in, not the center out. Working from edges toward the middle contains the liquid and curbs gas release, protecting lungs and colleagues. Real training drills demand practice on this step because panicking with a mop isn’t just ineffective—it's dangerous.
Collected residue and cleanup tools go into corrosion-resistant, clearly labeled containers. Someone with real chemical waste disposal experience takes over next. Tossing anything into the regular trash or a random drum has led to long-lasting consequences and environmental violations.
Exposure: Fast, Smart Action
Humans always matter more than property. Direct skin contact needs a proper drench under the safety shower, not a quick rinse. Ten solid minutes. Skip that and risk burns that won’t heal well. Contaminated clothes come off immediately. Eye exposures take precedence: emergency eyewash stations stay in top shape for a reason—fifteen minutes minimum, lids forced open. Breathing problems after gas exposure require medical attention on the spot, so call for a medic, not just a supervisor.
Building a Safer Culture
Plastering safety signs on every wall cannot replace genuine education and respect for hazardous chemicals. Managers, techs, and new hires need reminders that shortcuts deliver accidents. Rehearsing spill drills, testing alarms, and reviewing protocols can get repetitive, but ingraining vigilance is non-negotiable. Industrial disasters often track back to routine neglect: open waste containers, absent PPE, or guessing at ingredients.
Across industries, fostering a culture where questions, alerts, and hesitation face encouragement—not annoyance—has proven most effective. Bad habits fade with strong, supportive teams and regular reminders that every drop spilled can ripple beyond the lab or loading dock.
Local emergency support—firefighters, medics—benefit from plant tours and sharing process diagrams. Giving them time to familiarize themselves with chemicals like Nitrosylsulfuric Acid transforms responses from chaotic to competent if something goes sideways.
| Names | |
| Preferred IUPAC name | Nitrosylsulfonic acid |
| Other names |
Nitrosyl hydrogen sulfate Sulfuric acid, nitrosyl Nitrosulfuric acid Oniumhydrogensulfat Nitrosylsulphuric acid |
| Pronunciation | /naɪˌtrəʊsɪl.sʌlˈfjʊər.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 7782-78-7 |
| Beilstein Reference | 1639618 |
| ChEBI | CHEBI:29377 |
| ChEMBL | CHEMBL1201586 |
| ChemSpider | 154204 |
| DrugBank | DB14511 |
| ECHA InfoCard | ECHA InfoCard: 03-2119950490-45-0000 |
| EC Number | 232-300-2 |
| Gmelin Reference | 7869 |
| KEGG | C14133 |
| MeSH | D009617 |
| PubChem CID | 139604 |
| RTECS number | QS9600000 |
| UNII | 58S36X038T |
| UN number | 1886 |
| CompTox Dashboard (EPA) | Q419495 |
| Properties | |
| Chemical formula | HSO₄NO |
| Molar mass | 115.08 g/mol |
| Appearance | colorless to yellowish oily liquid |
| Odor | Pungent odor |
| Density | 1.898 g/cm³ |
| Solubility in water | Decomposes in water |
| log P | -4.35 |
| Vapor pressure | < 0.1 hPa (20 °C) |
| Acidity (pKa) | -4.2 |
| Basicity (pKb) | pKb ≈ 3.0 |
| Magnetic susceptibility (χ) | −46.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.433 |
| Viscosity | 47.6 mPa·s (20 °C) |
| Dipole moment | 3.46 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 162 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -444.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -784.9 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB33 |
| Hazards | |
| Main hazards | Corrosive, causes severe burns, releases toxic nitrogen oxides and sulfur oxides, reacts violently with water. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | Hazard statements: Causes severe skin burns and eye damage. May cause respiratory irritation. |
| Precautionary statements | P260, P264, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Lethal dose or concentration | LD50 (oral, rat): 2140 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2140 mg/kg (rat, oral) |
| NIOSH | SC8925000 |
| REL (Recommended) | REL: 0.1 mg/m3 |
| IDLH (Immediate danger) | IDLH: 6 mg/m³ |
| Related compounds | |
| Related compounds |
Nitric acid Sulfuric acid Dinitrogen tetroxide Nitrosyl chloride Oleum |

