Nitrosyl Chloride: A Closer Look at an Unassuming Powerhouse
Historical Development
Nitrosyl chloride turned heads in European chemistry circles long before many of the flashy agents we see today came on the scene. As early as the 19th century, scientists learned that mixing nitric acid and hydrochloric acid didn’t just give you aqua regia for dissolving gold. It also led to a yellow gas—later called nitrosyl chloride—especially when the reaction ran under less-than-perfect conditions. That discovery slipped into industrial traditions by the late 1800s, particularly in England and Germany, where chemists looked for strong chlorinating and nitrosating reagents. I remember digging through old journals, discovering just how much early dye and explosives chemistry depended on clever, often dangerous, manipulations involving this pungent gas. Its unpredictable reaction and ease of formation made it both a blessing and a curse in laboratories itching for breakthroughs.
Product Overview
Nitrosyl chloride rarely gets the fame of its cousin phosgene, but it keeps cropping up wherever fine chemistry demands precision. It’s sold as a compressed gas or dissolved in solvents for easier handling. You’ll see NOCl used for making dyes, drugs, pesticides, and even polymers, though the quantities might look modest compared to big-ticket industrial chemicals. Cost always floats with purity and packaging, but most buyers pay extra for assurances around moisture content and stability, since the slightest impurity can mess up a whole downstream run. Smaller research labs often order it in lecture bottles, while bigger outfits might source tonnage under tight regulatory eyes. Over two decades of working in labs both academic and industrial, I’ve noticed you can always count on demand, even if the safety paperwork threatens to bury you.
Physical & Chemical Properties
Grab a bottle of nitrosyl chloride and you’ll spot a yellowish or sometimes reddish liquid under pressure, often shifting toward a dark yellow gas if vented. It boils at about 5.5°C and melts at -64°C—making it volatile and tricky in even mildly warm conditions. It carries a sharp, acrid odor you won’t soon forget, courtesy of its rapid hydrolysis to hydrochloric and nitrous acids. Solubility in cold water exists, but you’re asking for corrosion trouble. In organic solvents, it behaves predictably. With molecular weight just over 65, and a density about 1.36 g/cm³ at 0°C, NOCl doesn’t need high concentrations to show its effects. Handling this stuff without a chemical fume hood is tempting fate.
Technical Specifications & Labeling
Suppliers label nitrosyl chloride as a Class 2 poisonous, oxidizing compressed gas—DOT UN 1069, packing group I. Standard tanks must bear corrosion-resistant paint and etched labels to deal with drips and splashes. Product leaves warehouses packed with detailed hazard statements: extreme toxicity, corrosivity, and reactive hazards fill every sticker. Purity levels usually reach 98% or more, since even minor contamination hikes reaction risks. You’ll find certificates of analysis specifying moisture content, acid level, and sometimes photometric absorbance if destined for analytical work. Product data sheets mirror GHS labeling rules, and even the packaging and seals come in for routine inspection. In some regions, local agencies demand full traceability back to the lot number, including logbooks documenting every handover.
Preparation Method
On the bench, you mix concentrated nitric acid with dry hydrochloric acid gas or strong aqueous HCl, and a violent emission of yellow gas—NOCl—rushes out. Industrial processes lean on direct chlorination of nitrosylsulfuric acid, or by oxidizing hydrochloric acid with nitrogen dioxide under controlled pressure and temperature. Yields improve by drying reagents and running glass or Teflon reactors, since steel and moisture trigger side products and corrosion. From experience, glassware rarely lives long with repeated NOCl exposure, as fuming and leaks always find the tiniest seam or flaw. Producers often recover by-products—like chlorine and dinitrogen trioxide—selling them for secondary processes to squeeze every cent from each reaction.
Chemical Reactions & Modifications
Nitrosyl chloride acts as a dual-purpose agent. It tosses out both NO+ and Cl–, making it perfect for introducing nitroso and chloro groups into receptive aromatic rings or alkenes. In the textile industry, it shines in diazotizations, turning primary amines into diazonium salts that underpin dyes. In pharmaceuticals, formulators reach for NOCl to craft intermediates for antihistamines and dispersed pigments. Among organic chemists, chlorination of alcohols and nitrosoation of secondary amines create cleaner routes than older, more wasteful protocols. Run these transformations cold, as NOCl gas decomposes at high temperatures. Improper storage or exposure to sunlight sets off risks of rapid pressure build-up or explosions. I’ve seen research groups build custom reaction chambers with remote operation, simply to keep noses and lungs safe from unintended exposure.
Synonyms & Product Names
Industry and academic literature bounce among several names: nitrosyl chloride stands as the IUPAC favorite, but texts also call it Nitrosyl monochloride, Chloronitroso, or even Tilden’s gas. Chemical catalogues often use NOCl for brevity, especially in database searches. Some older references—and accident reports—mention “chloroxime” or “gaseous nitrosyl chloride,” though those designations fade with each regulatory update. In countries following European REACH, distributors and safety officers standardize around NOCl and the European inventory number EC 232-242-0. For old-timers in German or Russian factories, you may still hear shorthand from pre-war processes echoing through the workroom.
Safety & Operational Standards
Nothing about NOCl handling is casual. Laboratories must run certified fume hoods with laminar airflow at rated velocities, and operators wear full acid-resistant PPE, even during bottle swaps. Supplied air respirators remain the rule during cylinder changes, repairs, or in the event of leaks. I trained dozens of operators who all dreaded the quarterly fire marshal inspections, thanks to government guidelines treating nitrosyl chloride as potentially lethal by inhalation and corrosive to skin and mucous membranes. In industrial settings, detectors for nitrogen oxides double as early warning for accidental leaks—since NO₂ forms fast from breakdown. Processes integrate scrubbers using cold alkaline solutions to neutralize vented gas, and emergency plans require fast routes to showers, eyewash stations, and medical intervention. Safety data sheets stack thick with operating limits and spill containment protocols. Regulations force site owners into regular drills, chemical compatibility checks, and third-party audits.
Application Area
Few chemicals wear so many hats across fine chemistry. The best-known uses land in azo dye manufacture, where NOCl shaves time and waste from diazotization lines. In the pharmaceutical sector, it’s prized for introducing nitroso groups, key to making certain antihypertensives and expectorants. Agrochemical corporations reach for it during pesticide synthesis, especially to build halogenated intermediates that block pest metabolism. Polymer innovators use it to modify polyolefins, toughening plastics with controlled chlorination. Analytical chemists apply nitrosyl chloride in trace detection of specific amines or thiols, giving them sharper results than older colorimetric reagents. Every sector relying on NOCl has invested millions into safer handling and better training, since a mistake doesn’t give you a second try. I’ve spent late nights with process engineers fine-tuning vent controls, all to shave down exposure risks and meet ever-tightening emission norms.
Research & Development
Academic groups keep reimagining what nitrosyl chloride can accomplish, looking toward greener, more selective syntheses. People want non-aqueous routes, catalytic cycles to recover spent gas, or photochemical systems skipping the need for large gas cylinders entirely. In industry, R&D teams build tighter process controls, automating feeds to minimize manual intervention. Projects focus on selective chlorination of new pharmaceutical intermediates and on the runup to specialty pigments with tighter purity controls. Grants increasingly target reducing emissions, with funding tied to both safety and environmental goals. As I’ve watched project teams present at industry conferences, it’s clear most advances center on continuous flow reactors—small volumes, fast quench, improved safety, and easier scale-up. Though research dollars are hard-won, any group showing they can slice through energy and risk will earn a seat at the table.
Toxicity Research
Toxicologists describe nitrosyl chloride as a fierce respiratory hazard. Inhalation at low concentrations can prompt severe irritation, leading to coughing, chest pain, pulmonary edema, and sometimes delayed onset of symptoms that spiral into a medical emergency. Chronic exposure, especially among operators in poorly ventilated spaces, risks bronchitis and even chemical pneumonia. Animal studies confirm both acute and chronic damage with exposure routes extending through inhalation, dermal contact, or eye splashes. Epidemiological data in older dye and chemical plants hint at elevated respiratory illnesses where NOCl ran without modern fume controls. Some countries regulate permissible exposure limits at fractions of a ppm. Medical guidelines stress decontamination, oxygen therapy, and in rare cases, corticosteroid administration. Every plant I’ve visited in the last decade, from Shanghai to Houston, runs medical drills and keeps antidote kits stocked, even if they rarely see serious incidents. That investment pays off every time something unexpected happens.
Future Prospects
New demands for tighter control and lower waste have forced manufacturers to reevaluate every step of nitrosyl chloride production and use. Regulatory agencies press for closed systems and real-time monitoring, pushing the industry away from open-batch operations. As green chemistry grows, researchers knock at the door for safer substitutes or new ways to recover and recycle spent gases. Specialty applications in pharmaceuticals and materials science still rely on the unique dual-functionality of NOCl, so total transition away will take time. Digitization of plant operations, along with AI-driven safety monitoring, offers hope for minimizing risk. In the next ten years, the companies investing in hazard-reduction technology, automated reactor systems, and circular chemical logistics will enjoy a competitive edge. The push toward zero-accident workplaces and near-zero emissions has already sparked a new generation of process chemists to design smarter, cleaner synthesis routes for this old gas.
What is Nitrosyl Chloride used for?
What’s Nitrosyl Chloride All About?
Nitrosyl chloride doesn’t pop up in everyday conversation, but its bright yellow, sharply pungent gas plays a big role in certain industries. Some chemists even call it “NOCl.” This compound comes with some tricky handling requirements, yet it remains important in labs and factories around the globe. Its main applications touch chemical manufacturing, pharmaceuticals, and sometimes research.
Why Factories Use Nitrosyl Chloride
I worked in a chemical plant that occasionally bought small tanks of nitrosyl chloride. It helps transform raw materials into more finished chemicals—kind of like passing a simple drawing through a copier with extra features attached. Factories rely on nitrosyl chloride for things like making nitroso compounds and as an agent in organic synthesis. Without this chemical, some dyes and pesticides would take much longer and cost more to produce. Companies use it because it often reacts quickly and gives a high yield of the end product. In the world of production, speed and reliability mean profit and fewer headaches.
Pharmaceuticals and Nitrosyl Chloride
Medicines sometimes come from surprising chemical processes. Nitrosyl chloride acts as a kind of helper in making specific drugs, combining with other basic ingredients during the synthesis process. Skilled chemists know how to use it safely to steer those reactions and make the process shorter than other available methods. By saving time and energy, companies also cut overall expenses—a crucial factor when healthcare already eats up so much of household budgets.
Challenges of Using Nitrosyl Chloride
Let’s not gloss over the dangers just because it’s useful. Nitrosyl chloride is corrosive and highly toxic. A whiff of it would send most people running for the emergency shower. One slip—one cracked valve or unsecured hose—and the gas escapes, endangering workers and possibly the neighborhood. In my experience, safety teams drill procedures for accidental leaks every few months. Strict rules make it safer, but risk never goes away fully.
Environmental Impact and Community Safety
Chemical spills involving nitrosyl chloride can pollute air and water. If it lands in a stream, it quickly reacts with the water. Resulting byproducts can irritate animals and plants, sometimes even harming crops nearby. Communities near production plants rightly ask for transparency. As someone living near an industrial park, I’ve seen how local councils press companies to invest in better leak detection and response plans. Upgraded ventilation, automatic shutoff systems, and clear communication all help. The companies that cut corners often end up facing steep fines and lasting reputational damage.
Responsible Use and New Solutions
Many researchers now hunt for safer substitutes to nitrosyl chloride. Some universities test other chemicals that get the same results without as much danger. But industry rarely changes overnight. Until a real alternative comes along, training and equipment upgrades stay important. I’ve met people who turn down job offers in plants that handle nitrosyl chloride, and I understand why. It’s one of those chemicals that demands respect—and sometimes, just extra caution isn’t enough.
What Can Improve the Situation?
Building a culture where workers can speak up and stop processes over safety concerns helps a lot. I’ve seen shifts where one nervous worker’s question stopped a major accident. Routine inspections, better emergency gear, and relentless focus on details protect those who show up every day. Investment in research for less hazardous chemicals deserves bigger budgets, too. Residents near plants need real-time alerts and frequent updates, not just press releases after the fact. The more people talk about risks like nitrosyl chloride—not just the benefits—the safer communities and workers become.
What is the chemical formula of Nitrosyl Chloride?
The Essentials: NOCl
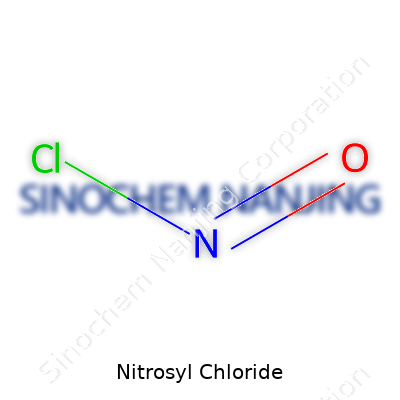
Nitrosyl chloride carries the chemical formula NOCl. It comes together from nitrogen, oxygen, and chlorine—nothing complicated in how it’s written, but there’s a lot more going on beneath the surface. In my college days, I saw plenty of students slip up on its formula during organic chemistry exams. Trust me, that one little “O” sandwiched between nitrogen and chlorine throws folks off more than you might think.
Behind the Elements: How NOCl Comes Together
NOCl doesn’t just pop up in a test tube by accident. Factories often generate it by reacting nitric oxide (NO) with chlorine gas (Cl2). That reaction is no chemistry class demonstration either—handled with the right respect, since both reactants can cause serious harm. Even so, the value NOCl brings to the table makes that risk worthwhile under controlled conditions. The product appears as a yellow gas under room temperature and has a strong, choking odor; working with it means paying attention to safety gear and proper ventilation at all times.
Why Even Care About NOCl?
Seeing a chemical formula in a textbook is one thing. Seeing what that compound does in the real world feels like a whole other story. Chemistry has an everyday side, and nitrosyl chloride isn’t locked away in a lab. It’s out there in industry, mostly used to make other chemicals—think of it as a stepping stone in larger chemical syntheses. Manufacturers rely on its abilities for making products like dyes, pharmaceuticals, and even some kinds of explosives. Errant handling risks environmental pollution and poses real threats to workers’ health, so every bit of its use deserves attention.
The Knowledge Gap and Public Safety
I’ve seen news stories spin chemical names as scary jargon because people don’t see how formulas reflect everyday products. Yet knowing the simple structure—NOCl—grants a foothold to dig deeper. Nitrosyl chloride has a long-standing spot in industrial chemistry, and accidents like leaks or improper disposal pack the potential to damage ecosystems or air quality near plants.
Regulations exist, but paperwork doesn’t stop leaks. The difference maker has always been professionals who treat chemicals with respect and the public’s understanding of what roles substances like NOCl play. Chemical plants cannot expect the community to trust them if secrecy or carelessness rule the day. All it takes is a minor slip for an incident to make headlines and erode that trust.
Building Better Habits and Solutions
Real progress starts with education. Every science teacher who draws NOCl on the chalkboard lays the foundation for safer chemical use down the line. Beyond the classroom, industry leaders must keep investing in real-time leak detection and transparent reporting. I’ve met workers whose pride in their safety records came through in conversation, and their efforts drive improvements year after year. More robust emergency plans and public education campaigns help too. In cities with large chemical sectors, knowing what to do if an accident happens can mean everything for workers and neighbors alike.
NOCl stands as more than a collection of atoms. Understanding its formula opens the door to conversations about chemical safety, environmental care, and public trust. With that knowledge, conversations shift from fear toward responsibility and solutions that stick.
Is Nitrosyl Chloride hazardous to handle?
What Working With Nitrosyl Chloride Looks Like
Nitrosyl chloride draws a lot of caution in chemical plants, and for good reason. People who work with this compound cannot ignore the sharp smell or eye-watering yellow fumes, even from a distance. I remember my early days on the factory floor, watching as seasoned technicians prepped their gear—the extra care, the double-checks, the tight seals. It's not just about procedure. It’s about knowing you’re up against a substance that fights back if you get complacent.
Why Some Chemicals Command Respect
Every chemical carries risk, but nitrosyl chloride reaches a different level. Breathing it in has immediate effects—burning sensation in the eyes, nose, and throat. Long-term exposure means coughing fits and chest pain that nobody wants to experience. Health agencies, like the CDC and OSHA, include nitrosyl chloride in their lists of strongly hazardous chemicals. Contact causes severe skin burns; even splashes mean a dash to the safety shower.
Besides direct harm to people, nitrosyl chloride doesn’t mix well with water or organic materials. It reacts fast and releases more dangerous fumes. Accidental leaks can fill a lab or workspace with choking smoke, leading to full evacuations and emergency gear. I’ve seen cleanup teams working in full respiratory protection after a valve failure. Their work took hours and cost the company dearly in downtime and equipment checks.
Hazards That Don’t Stay in the Laboratory
Nitrosyl chloride doesn’t just threaten those handling it at work; it also presents a wider environmental issue. Accidental releases contaminate air and water. Its strong oxidizing power wrecks waste systems and leads to other pollutants, including nitrogen oxides and hydrochloric acid. Local communities get nervous each time a plant handles or transports this product.
Reduction Starts With Training and Mindset
Reducing danger starts well before the lid is unscrewed. Worker training drills need to get specific, skipping generic slides and getting into realistic first-hand scenarios. Routine and boredom create blind spots. New hires need more than a quick rundown—they should shadow skilled operators and run through worst-case drills. Where I worked, seasoned supervisors ran weekly checks, looking for forgotten gloves or poorly latched chemical hoods.
Engineering Controls and Smart Facility Design
Good ventilation matters. Well-designed extraction systems work non-stop at every bench and storage point. Monitoring equipment doesn’t just sit on walls—it gets checked every day and replaced at the first sign of trouble. Spill kits and emergency showers should always be ready—no excuses about cost-cuts.
Facilities need locked-down protocols for shipping and storage. Regular maintenance on containers and transfer piping goes a long way. Labels, hazard lights, and strict checkpoints catch mistakes before they harm real people.
Forward-Looking Safety
Smarter chemical substitutes deserve a closer look for those who still depend on nitrosyl chloride. Teams and safety managers talk openly about what works and what nearly went wrong in the past. Regulatory audits push for more than just paperwork; they dig into daily habits and honestly ask about near-misses.
Companies that treat nitrosyl chloride as something routine usually run into problems. Respect, hands-on skill, and honest talks keep everyone a little safer.
How should Nitrosyl Chloride be stored?
The Risks We Face When Mishandling Nitrosyl Chloride
Nitrosyl chloride sits among the nastier chemicals out there. It’s toxic, corrosive, quick to react with air and water. Not something you want to cut corners with. I’ve seen what happens when people ignore these warnings: headaches, coughing fits, even permanent lung issues for those caught unprepared. Let’s not forget the chemical burns, damage to storage rooms, and the headaches for emergency responders. This isn’t one of those compounds you tuck away like an old can of paint—it demands real respect.
What’s Required for Safe Storage?
A steel drum in a corner or a plastic bottle on a shelf isn’t good enough. Nitrosyl chloride reacts with moisture, so humidity becomes the enemy. It gives off nitrogen oxides and hydrochloric acid. In my early days at the plant, one slip-up with a leaky container chewed through the shelving and set off the fire alarm before anyone realized what happened. Simple mistakes, big consequences.
Dry, well-ventilated rooms are essential. A tightly controlled climate—preferably with an inert atmosphere like dry nitrogen—keeps it stable. Don’t count on your standard warehouse either; materials like wood or certain plastics won’t hold up if vapor escapes. Stainless steel or glass with Teflon seals work best. If you’re working near water lines, move—nitrosyl chloride doesn’t forgive contact with even tiny amounts of water.
The Importance of Training and Planning
Labels and folders full of procedures don’t do the heavy lifting here—people and training matter most. I remember a colleague who skipped his mask to “just check a drum.” It landed him in the ER. Even with the right storage, you need protocols: double-check seals, ensure ventilation, wear protective suits and masks. Remember, a splash on skin isn’t just a quick wash-and-go. You’ll want calcium gluconate gel handy to stop deeper chemical wounds. Personal experience—having emergency supplies within arm’s reach saved a friend from losing function in his hand after a sudden spill.
Regulatory Standards and Common Sense
Government safety rules back up all this caution. OSHA sets clear exposure limits. Environmental bodies keep tabs on air handling and accidental releases. Skipping secondary containment leaves you open to big fines, sure—but it also increases the chance you, your team, or even your neighborhood ends up in harm’s way. Leaks don’t stay contained for long.
Not every solution comes from a rulebook. Staff should walk the storage area frequently. Automated monitors for leaks and moisture detect issues humans might miss. Back-up power for your climate controls means storms and outages don’t turn storage into a hazard zone overnight.
Building A Responsible Chemical Culture
Plenty of labs and warehouses handle their jobs with care, but complacency creeps in. Inspect records, dig into which materials line containers, and don’t rely on hope. A team that values vigilance stops problems before they start. Every spill I’ve seen started with a shortcut—an assumption someone else checked everything. Nitrosyl chloride punishes these lapses much harder than most.
Smart storage and a bit of humility around dangerous compounds protect workers, neighbors, and the business itself. Safe practices pay off, and nobody has to learn the hard way how unforgiving this chemical can be.
What are the safety precautions for Nitrosyl Chloride exposure?
What Makes Nitrosyl Chloride So Dangerous?
Nitrosyl chloride isn’t a chemical you can casually brush aside. Inhaling this compound, even just a small amount, can hit your respiratory system like a wall. Rapid coughing, throat irritation, and chest pain often follow. A friend of mine, a lab technician, once shared his experience during a minor lab leak. He realized in seconds how quickly symptoms develop when basic protection slips.
On top of breathing hazards, skin or eye contact leads to severe irritation and potential burns. A splash to the eye or skin means pain, redness, and potentially long-term damage. The real kicker is that nitrosyl chloride doesn’t give you chances for mistakes. Its effects come fast and don’t cut corners.
Protecting Yourself in Labs and Industry
Every safety officer I’ve met agrees on one thing: no one steps into a nitrosyl chloride environment without proper gear. A full-face respirator or supplied-air system gives lungs the best chance. Standard dust masks or simple cotton cloths won’t cut it. You need gloves resistant to strong acids—nitrile, neoprene, or butyl rubber offer effective barriers.
Splash-proof goggles and a chemically resistant face shield become essential, not an afterthought. One mistake, like rubbing your eye with a contaminated glove, can land you in the emergency room. Long-sleeved protective clothing—typically a chemical-resistant suit—keeps the compound off your skin and out of contact with street clothes that could carry residues home.
Poor Ventilation Equals Trouble
I’ve walked into plenty of labs with questionable ventilation, and you can feel the discomfort pretty quickly. Nitrosyl chloride forms dense vapors that hang low. The only way to avoid dangerous buildup means using local exhaust ventilation where the chemical gets handled. Fume hoods, if certified and unblocked, provide the highest protection. Never work with nitrosyl chloride in tight, closed rooms.
Handling and Storage Matters
People sometimes underestimate the threat until something goes wrong. I once saw the aftermath of storing it in a non-inert container. Nitrosyl chloride reacts with water—even humidity can trigger dangerous gas releases. Always store it in tightly sealed glass or PTFE-lined containers, away from sunlight and moisture. Double-checking the seal matters just as much as wearing gloves.
Label the container with bold warnings and keep an updated log of amounts on hand. Emergency procedures should be visible, and everyone needs to understand their role if a leak occurs. Integrity of the storage area should get checked routinely—cracked seals or damaged containers should be replaced immediately.
Quick Action for Spills or Exposure
If nitrosyl chloride spills, trained staff need to respond fast. Evacuate bystanders, and only trained responders with full protective suits and respirators should handle cleanup. Neutralize with a suitable agent if possible, then ventilate the area until air monitoring confirms it’s clear.
If someone breathes in vapors, move them to fresh air. Have them rinse their eyes with water for at least 15 minutes if contact occurs, and seek urgent medical attention. Every minute counts. In my experience, clear communication and practiced drills make a bigger difference than any single piece of equipment.
The Real Safety Net: Preparedness and Respect
Learning to handle nitrosyl chloride means respecting what it can do. No shortcut or half-measure replaces awareness and careful procedure. Relying on protective equipment, regular training, checked ventilation, and solid teamwork protects everyone who works around this reactive gas.
| Names | |
| Preferred IUPAC name | Chloronitrosyl |
| Other names |
Nitrosyl oxychloride Chloronitroso Chloronitric oxide |
| Pronunciation | /naɪˈtrəʊsɪl ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 7758-05-6 |
| Beilstein Reference | 1366516 |
| ChEBI | CHEBI:48150 |
| ChEMBL | CHEMBL1231110 |
| ChemSpider | 10085711 |
| DrugBank | DB09345 |
| ECHA InfoCard | 100.003.049 |
| EC Number | 233-045-4 |
| Gmelin Reference | Gmelin Reference: 1408 |
| KEGG | C14162 |
| MeSH | D009595 |
| PubChem CID | 24545 |
| RTECS number | QG9625000 |
| UNII | 7906O3V8KM |
| UN number | 1051 |
| Properties | |
| Chemical formula | NOCl |
| Molar mass | 65.46 g/mol |
| Appearance | Yellow gas |
| Odor | Pungent |
| Density | D:1.41 g/cm³ |
| Solubility in water | Reacts with water |
| log P | -0.1 |
| Vapor pressure | 24.5 atm (20 °C) |
| Acidity (pKa) | 0.3 |
| Basicity (pKb) | 10.52 |
| Magnetic susceptibility (χ) | '−25.5·10⁻⁶ cm³/mol' |
| Refractive index (nD) | 1.437 |
| Viscosity | 0.244 cP (25 °C) |
| Dipole moment | 1.90 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 261.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | +51.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -80.7 kJ/mol |
| Hazards | |
| Main hazards | Toxic if inhaled, causes severe skin burns and eye damage, may cause respiratory irritation, corrosive to metals. |
| GHS labelling | GHS02, GHS06, GHS05 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H331, H314, H270 |
| Precautionary statements | P261, P271, P280, P301+P310, P304+P340, P305+P351+P338, P308+P311, P403+P233 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Autoignition temperature | > 120°C (248°F) |
| Explosive limits | Explosive limits: 8-23% |
| Lethal dose or concentration | Lethal dose or concentration (Nitrosyl Chloride): "LDLo (inhalation, human): 17 ppm/30 min |
| LD50 (median dose) | LD50 (median dose): 150 mg/kg (rat, inhalation) |
| NIOSH | SN 0830 |
| PEL (Permissible) | PEL = "Nitrosyl Chloride: 2 ppm (5 mg/m3) as ceiling |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | IDLH: 20 ppm |
| Related compounds | |
| Related compounds |
Nitrosylsulfuric acid Nitrosylsulfuric acid Nitrogen dioxide Nitrous oxide Dinitrogen tetroxide Nitric oxide |

