Nitroguanidine: A Closer Look at Its Journey, Properties, and Future
Historical Development
Nitroguanidine first emerged in the late 19th century, with scientists in Europe experimenting with new energetic materials for military and agricultural uses. DuPont and other chemical pioneers dove into the study of nitroguanidine as early as the World War I era, searching for less sensitive alternatives to TNT and picric acid. Nitroguanidine’s intrigue came in part from its ease of handling compared to other explosives of its generation. Early patents highlight methods for making nitroguanidine safer than earlier energetic compounds. Its reduced brisance made it less likely to detonate unintentionally, opening the door for widespread adoption in artillery and munitions research by the 1930s. Over the years, as chemistry labs across Europe, Russia, and the United States kept tweaking synthesis routes, the process grew more reliable, but nitroguanidine’s basic formula stayed consistent.
Product Overview
Nitroguanidine, a white, crystalline solid, carries a reputation as a low-sensitivity high explosive and a key intermediate for fine chemicals. Industries such as defense, agriculture, and pharmaceuticals all pull from stocks of nitroguanidine, recognizing its versatility for propellants, insecticides, and specialty chemicals. Factories usually supply the material in bulk, shipping the product in moisture-proof bags to prevent caking. It’s not uncommon to find it under other names in commercial labs, and chemists have used it for decades to prepare tetrazole and guanidine-based compounds for various industrial uses.
Physical & Chemical Properties
Nitroguanidine has a melting point close to 257 °C, but it decomposes rather than melts cleanly, giving off nitrogenous gases. Its density runs around 1.44 g/cm³, and the compound registers as almost insoluble in water at room temperature but picks up more solubility in hot water. It has a notoriously low sensitivity to impact and friction, a quality appreciated in munitions production. The solid’s crystal structure brings a unique stability, which helps explain its long shelf life in controlled storage conditions. Chemically, nitroguanidine presents a pretty straightforward formula, CH4N4O2, and stores ample chemical energy in its nitro and guanidine groups, both critical for explosive function and chemical syntheses.
Technical Specifications & Labeling
Suppliers of nitroguanidine must print technical grades, purity levels, and hazard information clearly on packaging. Standard grades range from 99% purity for laboratory use down to 95% for bulk industrial applications. Labels show UN identification and reminders for dry and cool storage. Transport regulations classify it under UN 0282 due to explosive risk. Conscientious handlers keep it away from ignition sources, acids, and strong oxidizers. Manufacturers highlight batch numbers, handling guidelines, combustion byproduct details, and expiration dates, all responding to global hazmat standards and facilitating traceability from factory to end user.
Preparation Method
Factories synthesize nitroguanidine by treating guanidine salts with concentrated nitric acid. Most contemporary routes use guanidine nitrate, adding it to chilled nitric acid under vigorous stirring. Temperature control during reaction stops runaway decomposition, so refrigerated reactors or ice-salt baths see use in nearly every commercial setup. Operators isolate the crude nitroguanidine by precipitation, followed by washing and neutralization steps to flush out acidic byproducts. Purifying the product benefits from charcoal filtration and careful recrystallization to guarantee high yields and low contaminant levels. I remember seeing pilot setups where a loss of temperature control spoiled whole batches, turning a straightforward process into a hazardous mess.
Chemical Reactions & Modifications
Nitroguanidine acts as both a nucleophile and an electrophile, depending on the reaction partner. Its structure supports the preparation of heterocyclic compounds like tetrazoles and triazoles, staples in explosives chemistry and pharmaceutical intermediates. Heating with alkali metal hydroxides transforms it into guanidine or urea derivatives, while condensation with aldehydes sparks ring-forming reactions that feed synthetic drug pathways. Researchers have even explored catalytic hydrogenation of nitroguanidine to create reduced guanidine forms with different properties. For propellants, nitration or acetylation can tune burn rates, and some labs tinker with doped nitroguanidine to suppress smoke or reduce sensitivity further.
Synonyms & Product Names
Across textbooks and safety data sheets, you’ll encounter nitroguanidine called “NQ,” “Picrite,” or “N-Guanylurea nitrate.” Chemtrade, BASF, and other suppliers occasionally brand it under their own proprietary names, but chemical registries list it under CAS 556-88-7 and EC No. 209-132-6. Some legacy texts refer to it by obsolete synonyms, but professional circles now stick to the straightforward nitroguanidine or its abbreviation for clarity in international commerce.
Safety & Operational Standards
Shippers and operators treat nitroguanidine with firm respect since it can explode under intense heating or strong shock. In my early years at a chemical plant, I witnessed mishandling of transfer lines that could have gone badly—it was only discipline and thorough training that kept things safe. Facilities keep humidity low and forbid open flames and metallic tools near open batches. Safety goggles, antistatic gear, and respirators form the basic field kit, reflecting the dust’s mild toxicity and irritation risk. Disposal calls for slow dilution and wet incineration with full atmospheric controls to neutralize fumes. Regulations demand thorough risk assessments, and smart teams run regular emergency drills. Most incidents stem from complacency, especially during transfer, emphasizing the value of hard-earned habits.
Application Area
Defense sectors found the greatest use for nitroguanidine in propellants for tank shells and medium-caliber rounds. Its low flame temperature cuts barrel erosion, which means fewer maintenance stops and longer service lives—an underrated but critical detail on the battlefield. In agriculture, chemists convert nitroguanidine into neonicotinoid insecticides, giving farmers tools for pest management, though new regulatory limits raise questions about environmental impact. In pharmaceuticals, nitroguanidine’s guanidino structure spins out new antihypertensive agents, antiviral drugs, and research probes. Pyrotechnics makers like the material for cool-flame colored flames with reliable burn rates, finding a place at public events and military exercises alike.
Research & Development
Laboratories worldwide keep exploring tweaks to nitroguanidine synthesis, eyeing greener processes and higher yields. There’s a constant cycle of engineering work done to reduce waste acid and energy use in industrial plants, sometimes with catalysts or continuous-flow systems. In defense R&D, scientists chase new propellant blends that maximize range and minimize fouling, using nitroguanidine as a backbone for further chemical modification. I’ve seen collaborations between university teams and private contractors focus on encapsulating nitroguanidine in polymers, creating new composite explosives with reduced sensitivity to impact and temperature swings. Environmental analysts test breakdown products in soil and water, feeding data back to product designers for safer next-generation formulations.
Toxicity Research
Toxicologists report that nitroguanidine exerts low acute toxicity in mammals. Studies on lab animals show only minor changes in liver enzymes and occasional irritation from chronic exposure, with no evidence of major organ damage at doses used in industry. Environmental research points to the molecule’s stability, which cuts both ways: it doesn’t break down quickly in soil or water, so factories need to manage run-off sharply. There’s more concern for aquatic systems, where build-up could stress invertebrates or upset microbial communities, especially near production plants. New monitoring programs and waste treatment developments focus on catching problems before they escalate. Tight workplace controls and ongoing medical surveillance cut occupational risk to a minimum.
Future Prospects
As regulation tightens around energetic materials and environmental release, the future for nitroguanidine will hinge on sustainable manufacturing and stronger ecological safeguards. Chemists focus on recycling acids and solvents, plus capturing byproducts for reuse, moving towards closed-loop systems. In specialty propellants and advanced munitions, nitroguanidine’s value endures, and refinements like nanostructured forms or smart composites could extend shelf lives and safety margins even further. In agriculture, new studies on environmental breakdown could spur tweaks to molecular structure, favoring biodegradability without sacrificing effect. In pharmaceuticals, ongoing research into guanidine derivatives points to possible breakthroughs in antiviral and antihypertensive therapies. Overall, nitroguanidine rides at the intersection of tradition and innovation, holding relevance for as long as the balance between performance and safety continues to matter in chemistry, defense, and public health.
What is Nitroguanidine used for?
A Closer Look at Nitroguanidine
Nitroguanidine grabs attention for reasons that stretch from the chemistry bench right into fields and factories. With its pale crystalline nature, most people wouldn’t guess how crucial it has become in a world that thinks it already knows all about explosives and propellants.
What Really Drives Its Use?
Walking through the story of nitroguanidine, you find one thing that sets it apart: stability under pressure. In the heavy business of explosives, this matters. Traditional explosives have a habit of being a little too eager to react. Nitroguanidine provides a safer alternative because it resists heat and shock better. You won’t find the same level of detonation risk transporting or storing it. This means fewer workplace accidents, fewer tragic headlines, more trust from workers handling dangerous materials.
While working near defense manufacturing in my early twenties, I came to realize how much nitroguanidine shaped safety protocols. Unlike old-fashioned gunpowders or TNT, plant operators had fewer emergency drills sparked by volatility. People sometimes overlook small improvements like this. Fewer scares at work make a real difference for anybody who brings a lunch pail and hopes to get home safely.
In the Field and in the Factory
On the battlefield, nitroguanidine steps in as a core ingredient for some propellants. Its low flame temperature means less flash and less barrel wear for artillery, rifles, and tank cannons. Anybody who has ever listened to complaints about frequent barrel replacements or wild muzzle flashes on direct-fire weapons knows this isn’t a detail to brush off. Gun tubes and launchers financed by taxpayers run longer and perform better.
Agriculture sees a different side of nitroguanidine. While not talked about much, derivatives play a role in pesticide formulations. Here, it helps target bugs more precisely and with less collateral damage. Farmers around the world struggle to keep yields up without hammering the land with harsh chemicals. Smarter use of compounds like nitroguanidine brings the opportunity for better crop management and less environmental runoff.
Keeping an Eye on Risks
Nitroguanidine’s upside comes with strings attached. Regulations exist for handling, shipping, and storage, but some companies cut corners to squeeze budgets or timelines. History has taught tough lessons, especially following fires or leaks. As a writer who has spent time around legacy manufacturing towns, the damage from one poorly handled shipment can ripple out into local rivers and air, leaving a stain on more than the factory floor.
Today’s watchdogs—regulatory agencies, local emergency response teams—need to keep close tabs on how nitroguanidine travels and gets stored. Experienced chemists and engineers recommend better sensors and training so employees spot fire hazards in time. Community briefings, up-to-date chemical reporting, and quick access to first-responder guides put real power in residents’ hands.
The Path Forward
The right answer for the challenges around nitroguanidine doesn’t always sit in a corporate memo or policy manual. People working day to day with it—engineers, line workers, regulators—build real safety by sharing their practical knowledge. Tech upgrades should include workplace learning, not just fancier equipment. Incentives could reward companies for transparency instead of just ticking off compliance boxes.
Trust in chemistry, like trust in any field, builds with candor and lived experience. Nitroguanidine stands as proof that thoughtful scientific work, rolled out with care and oversight, can deliver broad benefits—from military readiness to more sustainable farming. At the same time, it takes eyes open, honest reporting, and community involvement to keep this compound making good headlines instead of tragic ones.
Is Nitroguanidine explosive or hazardous?
Strong Chemical, Strong Responsibility
Nitroguanidine catches attention for good reason. This pale, crystalline substance serves as a key part of many energetic formulations, like certain propellants or explosives. Folks with backgrounds in chemistry labs or the defense industry often recognize its formula—CH4N4O. Even if you’ve never seen it in a pure state, nitroguanidine makes its way into products and research that demand serious respect for risk.
Is Nitroguanidine Actually Explosive?
Nitroguanidine offers high energy, sitting up there with other well-known explosives. It doesn’t explode at the slightest touch, but its nature gives reason to pause. Drop a block on concrete and you probably won’t see fireworks, but subject it to shock, high heat, or open flame, and you’re asking for trouble. I remember a college demonstration where the tech broke down potential hazard scenarios—nitroguanidine doesn’t look dangerous, but given the right push, it reacts with real force.
Practical lab guides list nitroguanidine as a relatively insensitive explosive compared to nitroglycerin or RDX, so its threshold for detonation is higher. That’s not a permission slip to treat it casually. House it alongside strong acids or oxidizers, and its risk factor rises. In a poorly ventilated environment or with static build-up, it can become a serious danger.
Physical and Health Risks
The chemical structure of nitroguanidine gives off toxic gases if burned or detonated—think nitrogen oxides and cyanide compounds. Short-term exposure during accidents can cause headaches, dizziness, or even more severe poisoning. Skin and eye contact bring irritation, so even indirect exposure means workers need to wear gloves, goggles, and lab coats. I’ve heard stories from old colleagues about headaches after brief exposure in production plants, a clear sign of its immediate health risk.
Environmental issues pop up once production or storage ramps up. Spills or leaks seep into the ground, tainting water supplies. This isn’t the sort of material someone leaves out in an ordinary warehouse; responsible handlers keep it locked down, away from anything that might trigger a chain reaction.
Regulations and Oversight
Manufacturers, defense contractors, and researchers can’t get by with a handshake and a nod. National security laws put controls on transport, sales, and storage. The Chemical Facility Anti-Terrorism Standards (CFATS) in the U.S. include nitroguanidine for good reason. Inspections and paperwork may feel tedious, but they keep both workers and communities safe.
Hazard communication forms part of every job involving energetic chemicals. Teams undergo regular safety drills, emergency equipment stands ready, and clear reporting channels exist for close calls or spills. I’ve seen firsthand how thorough planning and practice lead to fast, capable responses in situations where seconds matter.
Safer Handling and Alternatives
Education stays at the center of good safety culture. Anyone working with nitroguanidine receives detailed instructions—no shortcuts allowed. Routine checks for storage conditions, using grounded equipment to prevent sparks, and keeping incompatible materials separated all add layers of protection.
Ongoing research fuels progress towards less hazardous alternatives. Public funding often supports labs seeking new propellants with reduced toxic fallout. While nitroguanidine holds a special place in some industries, the future points toward safer, more sustainable technology.
How should Nitroguanidine be stored and handled?
The Basics of Nitroguanidine Risk
Nitroguanidine pops up in plenty of settings—labs, munitions factories, agricultural research. Anyone working with it learns the hard way: this chemical isn’t forgiving. It won’t wait around while someone fumbles with the caps or forgets why “cool and dry” means more than just flipping on the air conditioner. Pure nitroguanidine looks like a fluffy white crystal and doesn’t easily ignite, but throw enough energy at it or let it clump with the wrong stuff, and problems start fast.
Storage Isn’t Just a Label on a Drum
In my experience, leaving careful storage for “later” invites trouble. Rooms that house nitroguanidine should stay below 30°C with humidity under control—moisture causes it to clump, turning handling into a risky guessing game. I once saw a barrel exposed to afternoon sun through a warehouse window. Within days, the crystals changed texture and we had to waste time separating sticky masses. Shade and low humidity kept in check by a proper dehumidifier make a difference.
Forget storing this material close to acids, strong bases, or anything that might spark. Incompatibles belong in a different building, not just on another shelf. Someone might shrug off a few feet of distance, but vapors and accidental leaks don’t respect tape lines on a floor. Fire-resistant cabinets, preferably locked and grounded, slow down disaster if something sparks or heats up nearby. Dry powder fire extinguishers should sit within easy reach—water does little to stop the burning, so dry chemical types matter.
Handling Needs More Than Gloves and Good Luck
Gloves, goggles, airtight lab coats—these make up the minimum. I always double-checked valves and lids, because a minor spill sends fine dust into the air, easy to breathe in unexpectedly. Proper extraction systems mean a lot to anyone tired of headaches and coughs at shift’s end. Witnessing someone sweep up spilled powder with a dry broom stuck with me. Static charge alone, built up from fast movements or synthetic bristles, can turn a regular sweep into something ugly. Use conductive equipment; go slow. Ground yourself before touching bulk containers. Safety floors matter, too—concrete cracks can easily hold decades-old residues or catch new spills.
Training shapes habits. Staff who only see Powerpoint slides undervalue risk; hands-on demos where instructors show worst-case scenarios help set priorities straight. Emergency showers and eye wash stations hold a place in every workspace, no exceptions. Place clear signage about storage rules, emergency contacts, and safe cleanup procedures on every wall.
Legal and Environmental Realities
Regulation isn’t some distant paperwork exercise. Rules grow out of past accidents with real costs. Government directives in countries with history in explosives research mandate annual reviews—ignore that, and fines pile up. Local fire departments often request access to storage logs and MSDS documents. They want clear answers on what sits behind those locked doors and if enough ventilation exists. It becomes a practical issue when disposal gets discussed; legal disposal follows strict routes, so pouring leftovers down the drain isn’t just dangerous, it’s criminal.
Maintenance and documentation matter. Double-check seals and containers periodically. Rotate stock; old product turns unreliable. Inspecting storage areas as part of daily routines limits the risk of surprise. Keep backup supplies of protective gear, absorbents, and containment bins. Trust the wisdom of routines established after decades of learning from mistakes, not shortcuts crafted in the name of convenience.
What are the safety precautions when working with Nitroguanidine?
Why Nitroguanidine Gets Extra Attention
Nitroguanidine isn’t just another chemical tossed around in a lab. It shows up in explosives, some propellants, and even specialty labs. Its reputation for energetic reactions comes from its structure — that combination of nitro and guanidine packs a punch. Stories from the field point to real risks: a small oversight can lead to a big mess, both for equipment and the people in the room.
Personal Protection Starts Simple
Getting decked out with safety glasses, solid gloves, and a lab coat feels routine, but these basics matter with nitroguanidine. Standard nitrile gloves resist most chemical splash, and keeping goggles snug blocks the stray splash that could cause a nasty burn or worse. Folks I’ve worked with keep extra gloves close by. They swap them at the first sign of wear, because one pinhole could be the difference between a quiet shift and a trip to the nurse.
Know the Air You Breathe
Nitroguanidine doesn’t fool around with dust. It tends to release fine particles, making the air you breathe a concern. A lab with good, local exhaust ventilation keeps particles off your lungs. Not every workshop can afford a fancy setup, but even a basic fume hood makes handling safer. A mask rated for particulates adds another layer for bigger jobs. Respirators shouldn’t get locked up and forgotten — I’ve seen folks stash backup cartridges in their locker, just in case the main one gets bogged down with dust.
Handling: Steady, Calm, and Organized
People reach for scoops and spatulas out of habit, not realizing metal tools can set off a spark. Plastic or wood cuts risk. Nitroguanidine also reacts to shocks, so no tossing containers or banging them on the bench. If someone drops a jar, they call it out and everyone gives space until it’s dealt with by the designated cleanup crew. A lot of places use anti-static mats and ask crew to ground themselves before sitting down to work. Small details save lives.
Storage Makes a Difference
Dryness keeps nitroguanidine stable. Moisture invites bad news. Sealed, clearly labeled containers make it easy to spot what’s what in storage rooms. Folks stack containers only one layer high, and often separate them from acids or strong oxidizers. I remember one old-timer who stored a spill kit and a bucket of sand near the chem shelf and checked them every week. His routine seemed fussy, but that kit turned out useful the one time a container leaked.
Clear Communication and Training
Rules on paper don’t matter if nobody reads them. I’ve seen crews gather for Friday morning check-ins, talking through close calls and what went right or wrong. That openness builds trust — if anyone hears a strange noise or smells something odd, they speak up fast. Training drills stick with people much longer than a slide deck. Practice with an emergency shower or a spill kit once a month keeps reflexes sharp for the day they’re really needed.
Respect for the Substance, Respect for Each Other
Caring for each other pushes safety past rules and toward real culture change. Small reminders, fresh gloves, and keeping clutter off the bench show respect for friends and coworkers. Nitroguanidine doesn’t forgive accidents easily, but with steady routines and shared responsibility, teams can work safely and get the job done right.
Where can I purchase Nitroguanidine?
Navigating Chemical Purchases in Today’s World
Nitroguanidine draws interest from both research and industrial communities. Its place in chemical research is well-known, and it also shows up in some military and energetic material discussions. If you’re looking into buying it, understanding why it’s so closely regulated isn’t just about jumping through bureaucratic hoops—it’s about public safety and legal responsibility.
Regulated Access and Strict Oversight
Buyers sometimes assume chemical sourcing is as simple as ordering from a big distributor. But nitroguanidine isn’t a casual purchase. In the United States and many other countries, it lands on watchlists because it can be used to make explosives and other dangerous products. That means suppliers won’t just check your credit—they’ll want paperwork, licenses, and a clear reflection of your intended use.
Major chemical suppliers—names like Sigma-Aldrich, Fisher Scientific, and Tokyo Chemical Industry—keep it behind serious customer vetting processes. They sell to research labs, universities, and businesses with a proven track record. No established supplier wants to risk a regulatory crackdown or unwanted headlines because of a careless transaction. If a seller skips the due diligence, that should be a signal to walk away. This is one of those products that brings regular people face-to-face with policy and oversight.
Why All the Attention?
The controls on nitroguanidine aren’t about making life hard for scientists or manufacturers. They come from a long record of unsafe handling, theft, and misuse. Chemical incidents spark real-world harm. Bomb squads and hazmat teams don’t enjoy overtime, either. The paper trail tightens because lives actually depend on it.
If you’re part of a university, industrial lab, or a government project, your purchasing department knows the path. The checks may feel tedious, especially when you’ve already spent years acquiring credentials and security clearances. Each step, from concept to delivery, matches a gap in trust that regulators and companies have seen abused before. Buying chemicals outside official channels courts trouble you won’t see coming—think cancelled grants, lost jobs, or legal charges. That risk seldom balances out whatever shortcut you think you’re taking.
Supporting Legitimate Users
Scientists and engineers work best with clear, accessible rules and suppliers who respect safety. Nitrogauanidine doesn’t live in a vacuum. As a practitioner, I’ve gone through the audit process and annual site checks. The paperwork feels never-ending, but it matches the nature of work where you’re trusted with dangerous tools. The best solution has always been streamlining honest buyers into verified supplier networks, not erasing controls entirely. Some folks clamor for looser rules, but loosening security because some people don’t like paperwork can lead to big regrets.
Retail stores and e-commerce shops don’t carry this one for good reasons. Regulators have a duty to protect, and not every chemical belongs in a drop-down shopping cart. If you think your project or research can’t proceed without it, reach out to institutional purchasing staff. Ask your supplier what paperwork and screening they require. Don’t expect anonymity or overnight shipping on something with nitroguanidine’s risk profile. If you see sources promising hassle-free sales to the general public, their offers should prompt caution—both for your safety and for those around you.
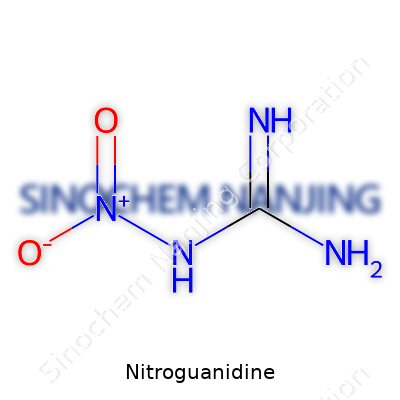
| Names | |
| Preferred IUPAC name | N-(Diaminomethylidene)nitramide |
| Other names |
Picrite NGu Picrignit |
| Pronunciation | /ˌnaɪ.trəʊˈɡwɑː.nɪˌdiːn/ |
| Identifiers | |
| CAS Number | 556-88-7 |
| Beilstein Reference | 14626 |
| ChEBI | CHEBI:7554 |
| ChEMBL | CHEMBL1239 |
| ChemSpider | 66171 |
| DrugBank | DB11337 |
| ECHA InfoCard | 100.006.692 |
| EC Number | 206-438-9 |
| Gmelin Reference | 6046 |
| KEGG | C06509 |
| MeSH | D009596 |
| PubChem CID | 9576 |
| RTECS number | MN2800000 |
| UNII | OM9447LW8N |
| UN number | UN0144 |
| CompTox Dashboard (EPA) | DTXSID7020822 |
| Properties | |
| Chemical formula | CH4N4O |
| Molar mass | 104.08 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.62 g/cm3 |
| Solubility in water | Moderately soluble |
| log P | -3.19 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 12.03 |
| Basicity (pKb) | 11.08 |
| Magnetic susceptibility (χ) | -7.5·10⁻⁷ |
| Refractive index (nD) | 1.437 |
| Viscosity | Low |
| Dipole moment | 3.48 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -87.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -487 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | N01AX10 |
| Hazards | |
| Main hazards | Explosive, toxic if swallowed, causes eye and skin irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS01,GHS03 |
| Signal word | Warning |
| Hazard statements | H272, H302, H315, H319, H335 |
| Precautionary statements | P210, P264, P280, P370+P378, P402+P404, P501 |
| NFPA 704 (fire diamond) | 2-1-2 |
| Autoignition temperature | 310 °C |
| Explosive limits | Explosive limits of Nitroguanidine: 0.2–2.0 vol % |
| Lethal dose or concentration | LD50 (oral, rat): 1250 mg/kg |
| LD50 (median dose) | LD50 (median dose): 5500 mg/kg (oral, rat) |
| NIOSH | TTJ |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Nitroguanidine: "Not established |
| REL (Recommended) | 50 mg/m³ |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Guanidine Methyl nitroguanidine Aminoguanidine Dinitroguanidine Nitroguanine |

