Nitrogen Dioxide: A Chemical with a Past and an Uncertain Future
Tracing the Journey of Nitrogen Dioxide
Nitrogen dioxide has a history that reflects both scientific discovery and society’s tricky relationship with pollution. Scientists first observed it through laboratory experiments in the early 1800s, but real-world encounters came long before then — think of the brown haze hanging over smoky cities, a signal of coal burning and industrial progress. Chemists pieced together its identity in the laboratory, discovering that mixing nitric oxide and oxygen created this distinctive, reddish gas. By the early 20th century, growing cities noticed the sting in the air and the way outdoor paint faded faster near busy roads. From the industrial revolution to the automobile boom, nitrogen dioxide left its mark on both science labs and city skies.
Getting Familiar With What Nitrogen Dioxide Is
Anyone who’s walked through heavy city traffic has probably breathed in a small amount of nitrogen dioxide. It’s not invisible—at high concentrations, you notice the sharp tang in the air and see a reddish-brown tint, especially on hot, smoggy days. In chemistry speak, it’s a small molecule, just one nitrogen and two oxygen atoms. Far more than a laboratory curiosity, the gas shows up wherever fossil fuels burn, especially car engines and power stations. Factories using nitric acid processes also give off significant amounts. Over time, the public focus shifted from pure science toward questions of air quality, human health, and the practical headaches that come with bad air days.
Diving Into Its Physical and Chemical Side
The smell of nitrogen dioxide is unmistakable — a mix of metallic sharpness and biting acidity. At room temperature, the gas hovers in the air, colored rust-brown, with a knack for irritating lungs and corroding metal. Cold it further, you get a liquid that’s almost as aggressive as its reputation. It weighs more than air and reacts willingly with water to form nitric acid, a chemical that eats away at metal and stone, which explains the blackened edges on old statues and buildings near busy roads. Chemists have measured its reactivity over and over, and its place in acid rain and ozone chemistry is well documented. Nitrogen dioxide doesn't just hang around; it triggers other chemical cascades, shifting the balance of local air and affecting both urban and rural environments.
Real Talk About Technical Specs and Labeling Practices
In practical settings — think factories, research labs, or regulatory checkpoints — nitrogen dioxide comes with flashing lights and bold warnings. Labels focus on concentration limits: not just to keep the workplace safe, but to comply with air quality standards set by regional and international authorities. These numbers matter. Exceeding just a few parts per million can send workers scrambling for fresh air. Over decades, regulatory agencies such as OSHA or the WHO have hammered out exposure limits based on studies tracking city-wide health data. Preparing a tank for industrial use or calibrating an instrument means tracking every decimal place, double-checking purity, and treating any leaks as emergencies, not routine spills.
How It’s Made and Modified
For bulk production, factories usually lean on burning ammonia or oxidizing nitric oxide in oxygen-rich air — the underlying chemistry is far from exotic, but getting high purity is no small feat. Labs can tweak reaction conditions to get the right blend of nitrogen oxides, then cool and compress the gas or dissolve it in water for easier handling. Some researchers explore catalytic methods, hoping to shift traditional production away from old, energy-hungry routes. That’s becoming more urgent as sustainability moves from nice-to-have to absolute must-have in industrial chemistry.
Chemical Transformations: Nitrogen Dioxide at Work
In the natural world and industrial reactors, nitrogen dioxide juggles several roles. It oxidizes, it reduces, it flips between chemical forms depending on pressure, temperature, and what else is around. Its most notorious reaction — with water to form nitric acid — shaped industrial manufacturing for everything from fertilizers to explosives. It also drives parts of the atmospheric chemistry that creates ozone near ground level, turning city air into something out of a chemistry set on summer afternoons. In some reactions, nitrogen dioxide serves as a middleman, helping shuttle atoms from reactants to products. This ability to morph and mediate attracts both admiration from chemists and frustration from environmentalists.
What’s in a Name: Synonyms and Industrial Lingo
In the scientific world, a compound can go by many names. For nitrogen dioxide, “NO₂” stands as the cleanest shorthand; in the trade, people might call it “nitrogen peroxide” or lump it with other “nitrogen oxides” under the generic, not-so-friendly label of NOx. This cluster of similar chemicals makes life tricky for regulators and scientists — cleaning up the air means sorting out which cousin causes which problem. In labeling, safety data sheets often list both formal and informal names, so no one mistakes a potent oxidizer for something milder.
Staying Safe and Setting the Rules
Nitrogen dioxide isn’t something to take lightly. Workers in chemical plants use gas detectors with blaring alarms, full-face respirators, and strict protocols for storage and handling. Spills or leaks call for quick evacuation, not curiosity-driven fiddling. Governments set daily limits for urban air, driven by hospital admissions and years of epidemiological research. These rules evolve with new evidence: if long-term exposure links to asthma and heart disease, the allowed limits drop, sometimes to industry’s dismay. Schools near highways monitor outdoor air more closely, often sparking debates that pit public health against transportation infrastructure.
Where Nitrogen Dioxide Shows Up in Real Life
Outside chemistry textbooks, nitrogen dioxide’s fingerprints smudge a surprising range of sectors. Big industrial plants churn it out as a step in mining, fertilizers, and cleaning metals. Power generation, especially from older coal or oil plants, throws up enormous extra clouds when equipment fails or maintenance slips. Cities rate nitrogen dioxide among the key air pollutants that drive smog alerts and trigger bans on wood or trash burning. In research labs, it’s an easy way to simulate pollution under controlled conditions. It even creeps into consumer products through side reactions, especially when combustion or high temperatures are involved.
Current Research and Questions Scientists Keep Asking
Over the past decade, the conversation around nitrogen dioxide shifted. Headlines follow new findings linking urban exposure to slower growth in children, a rise in respiratory illnesses, and changes to cognitive development. Research teams chase elusive links between short bursts of exposure and long-term risk, often sifting through massive city-wide health records to find patterns that blunt headlines miss. Others focus on understanding its role in transforming atmospheric chemistry, with knock-on effects on everything from climate to crop yields. Across university campuses, scientists hunt for cheaper, cleaner production methods and ways to trap or convert the gas before it leaves a smokestack or tailpipe. These aren’t minor side projects — instead, they shape how regulators, industry, and the public talk about clean air and societal health.
Sizing Up the Toxic Side of the Story
As research piled up, nobody could ignore that nitrogen dioxide does real damage. Breathing it in, even at low levels, messes with the lungs, especially for children, the elderly, and people living near busy streets. Doctors see higher asthma rates, more hospital visits, and persistent inflammation among people living in polluted neighborhoods. Animal studies reinforce what’s seen in the clinic — this gas can set off a chain reaction of inflammation and tissue damage. Toxicology experts measure safe exposure in weeks, months, and years, then argue over the right numbers behind closed doors. Policymakers juggle science, economics, and practical limits while setting air quality rules, often finding themselves pressured from all sides. For communities already hit hardest by pollution, these debates turn into urgent local fights for cleaner air and better health infrastructure.
A Hard Look at What’s Next for Nitrogen Dioxide
Today, nitrogen dioxide sits in a strange spot: essential for certain chemical processes, unwanted in urban skies, and tightly regulated. Technology offers hope, such as better catalytic converters, greener chemistry, and smart city air monitoring. Governments rethink transportation and industrial policy to shrink the public health toll. Research into new materials and renewable energy could cut down on emissions, but progress depends on political will as much as chemistry know-how. In my view, real solutions mean treating nitrogen dioxide not just as a molecule but as a social signal — of what we value in public health, city design, and scientific investment. Every smoky day and every news story about rising respiratory illness nudges the conversation forward. There’s no neat answer, only continued work from scientists, policymakers, and communities to shrink exposure while balancing industry’s genuine needs.
What is Nitrogen Dioxide used for?
Real-World Roles of Nitrogen Dioxide
Anyone who’s lived in a city has probably seen nitrogen dioxide in action, even if they didn’t realize it. Picture urban smog, that gauzy brown haze above the skyline—nitrogen dioxide often plays a big role there. This gas usually forms when fuel burns at very high temperatures, like inside car engines or power plants. That’s not where its story ends. Nitrogen dioxide, with its sharp, biting odor, lands itself into all kinds of uses that shape both industry and daily life.
Industrial Importance
Chemists count on nitrogen dioxide in the industrial production of nitric acid. Nitric acid forms the backbone for fertilizers, explosives, and certain plastics. Every time farmers grow wheat or corn for market, there’s a good chance their fertilizer traces back to a process fueled by nitrogen dioxide. Years ago, I toured a plant where the acrid tang of this gas drifted through the air near huge reactors. The workers wore heavy-duty masks. A steady supply of nitrogen dioxide made all the difference for their routine output.
Nitrogen dioxide also steps in for bleaching and oxidation jobs in specialty labs. Some companies use it to treat flour, giving bread its lighter texture and whiter crumb. In the electronics field, controlled amounts of this gas etch micro-patterns onto silicon chips. These chips drive the phones, tablets, and laptops that see us through each day. Tech giants depend on tightly monitored nitrogen dioxide flows to avoid costly defects.
Medical and Research Uses
Hospitals and research labs sometimes put nitrogen dioxide to work, but under strict rules. Controlled doses serve during sterilization and disinfectant routines when certain resistant spores threaten public health. For researchers, nitrogen dioxide gives clues about how air pollution damages lungs. Decades of studies point to its role in triggering asthma, coughing, and more serious conditions. After a grueling summer commute, I’ve felt my chest tighten—more than once, studies suggest nitrogen dioxide in city air tips the scales.
Concerns and Solutions
Strong evidence links nitrogen dioxide to health problems in high concentrations, especially among children and older adults. A 2023 report from the World Health Organization estimates nitrogen dioxide exposure causes thousands of respiratory hospital admissions each year. Short-term spikes around highways or schools hit hardest. Even at levels below legal limits, some scientists believe children’s lungs don’t escape unscathed.
People have worked to cut down emissions by developing cleaner fuels and stricter engine standards. It works—cities with newer buses and cars report clearer air. I’ve seen local officials push for green spaces and electric vehicle incentives after nitrogen dioxide readings creeped past safe lines. While no magic bullet erases the stuff overnight, cleaner transit and tougher rules make a dent where people rally around the science.
Looking Forward
The future of nitrogen dioxide use sits at a crossroads. Industry still needs it for critical processes, yet the health risks can’t be shrugged off. A realistic approach means balancing industrial demand, stronger filtration, and enforcement of air quality targets. Routine monitoring, rapid warnings, and steady investment in alternative chemical methods will help lighten health and environmental tolls. As more people recognize the risks, the drive for practical solutions only gets stronger—both on factory floors and community streets.
Is Nitrogen Dioxide hazardous to health?
Real Risks that Can’t Be Brushed Aside
Standing on a city street in heavy traffic, you can taste the air—there’s a tingle, a heaviness, almost metallic. That isn’t just smog; nitrogen dioxide does a number on your lungs even if you feel healthy. I grew up near a busy route, and I used to think itchy eyes and an irritated throat were just normal. Later, I learned that our bodies signal trouble before doctors do.
Countless studies link nitrogen dioxide to asthma flare-ups and respiratory illnesses, especially in kids. The World Health Organization points to higher rates of bronchitis among children living in polluted neighborhoods, and those statistics show up over and over across the globe. Even perfectly healthy adults breathing in too much of this gas won’t get a pass; it quietly chips away at lung function and ramps up the number of days people skip work or land at urgent care.
Evidence from Everyday Observation and Hard Data
If the science feels remote, personal stories draw the threat closer. Parents see their children cough throughout winter, teachers notice more absences, and emergency rooms track a bump in visits after days with heavy traffic emissions. Across America, cities with stubborn air quality problems usually have higher nitrogen dioxide measurements, often traced straight back to cars, buses, and trucks.
The harm goes beyond a sore throat. Breathing in nitrogen dioxide inflames airways and lowers the body’s defenses, making viral infections harder to fight off. In my own family, relatives with asthma always fared worse during rush hour times and on days when air felt thick. I saw how medication and careful planning only went so far—without cleaner air, attacks became more common, school days were missed, and the feeling of missing out grew.
No Silver Lining in Uncontrolled Exposure
Jobs and family life often pin us to polluted places. Living near highways, industrial sites, or major streets means no escape from chronic exposure unless something changes in the environment. Nitrogen dioxide comes from burning fossil fuels and from everyday things like gas stoves, not just tailpipes. That matters when thinking about indoor air too. Cooking with gas at home, especially without a good vent, can spike nitrogen dioxide levels way above the thresholds health experts recommend.
Researchers from leading agencies, including the EPA and WHO, warn against prolonged contact. Average urban levels regularly exceed limits set for safety. Too many communities sit near busy roads, and kids who walk to school along these corridors don’t pick where they grow up. Families renting in city centers rarely get a say in cleaner air or the location of truck routes. These stories don’t appear in academic studies, but they describe real costs—missed paychecks, growing medical bills, and the helpless worry that comes with chronic illness.
Common-Sense Solutions
Electrifying city buses, building safer walking routes away from busy roads, and improving public transportation could keep nitrogen dioxide levels down. In homes, switching to induction stoves or making sure kitchens get proper ventilation gives folks back some control, even if they rent. Local governments could push again for clean air zones or expand parks to buffer neighborhoods from traffic congestion, something proven to work in cities like London.
People deserve air that doesn’t make them sick or limit opportunities. For now, understanding the problem arms us for conversations with policymakers, doctors, and neighbors. The first step stays simple: knowing these invisible threats matter just as much as what we can see.
How should Nitrogen Dioxide be stored?
A Hazard with a Long Shadow
Anyone who’s stepped into a chemical stockroom will recognize that sharp, biting smell of nitrogen dioxide. This is not just another bottle sitting on the shelf. It’s a substance that can color the air a deep, rust-brown, especially on a humid afternoon or when someone’s gotten careless with the cap. This isn’t just a matter of housekeeping—it’s about workers getting home safely, lungs intact, headaches avoided.
What Makes Nitrogen Dioxide Tricky?
Nitrogen dioxide likes to keep people on their toes. It's a powerful oxidizer, quick to attack organic material and even metals under the right conditions. Sometimes it’s a gas, sometimes a liquid. Above 21°C (70°F), you see mostly gas—cooler than that, it starts to liquefy. That means storage tanks face pressure swings as temperatures change. It turns out the laws of physics don’t take a day off in a chemical plant.
There’s another side to all this: nitrogen dioxide reacts with water to make nitric acid. Any slip—an open valve, a damp gasket, a sweaty palm—can trigger not just splatters or puddles, but corrosive clouds and injured hands.
No Ordinary Drum Will Do
The right containers set the first line of defense. Steel tanks, lined against corrosion, deliver the strength needed to hold back both the acid and oxidizing action. Copper, many plastics, or anything with a bit of moisture will fail. A friend who’d seen storage mishaps always said, “Overbuild it; you’ll only regret what you skimped.” Double-walled tanks and automatic pressure relief valves have saved more than a few budgets and reputations.
Good engineers know storage areas can’t just be a quiet alcove at the back. Ventilation does the heavy lifting, whisking away rogue gases before anyone inside gets a headache or worse. In every facility I’ve visited, you can hear the fans running. Noisy, yes. But those fans mean you keep breathing easy.
Practical Steps for the Real World
Every time someone moves a cylinder, there’s potential for a dropped bottle and an emergency call. Securing tanks upright and chaining them to walls, not just using a piece of rope, keeps people safer. Regular leak checks and maintenance stop problems early. At one plant, a simple routine—wiping valves and fittings with ammonia-soaked rags—caught tiny leaks you couldn’t even smell.
Nobody can ignore regulations: OSHA and EPA standards put hard numbers on air monitoring, storage pressure, and employee training. In factories I’ve known, folks aren’t shy about asking for fresh respirators or shutting down a line if they detect a leak. Real safety culture grows when managers listen, not just post rules in a break room.
Thinking Beyond the Tank
Nitrogen dioxide doesn’t just challenge us in the lab or plant. Shipping brings another layer of risk: containers go on trucks over bumpy asphalt, through summer heat, or winter cold snaps. People loading and unloading need the right protective clothing. If a cylinder breaks en route, rapid response counts more than apologies. Clear labels, strong training, and robust emergency drills form a triad everyone relies on. We owe that to the drivers, handlers, and neighbors living near these transit routes.
Ultimately, handling nitrogen dioxide is about respect—for the science, for the risk, and most of all, for the people nearby. Getting it right means more than passing inspections. It’s about protecting lives each day the tanks stand waiting for use.
What safety precautions are required when handling Nitrogen Dioxide?
Growing Concern in Everyday Labs
Nitrogen dioxide stands out as a chemical folks don’t want lingering in the air. This brownish gas smells sharp, hits the throat hard, and roughs up lung tissue in no time. Years back, I watched a technician scramble for fresh air after knocking over a bottle he didn’t notice was damaged. That day never left the rest of us. Getting careless even for a moment turns a regular day into a health scare, all because exposure moved faster than anyone expected.
Trust Personal Protection Over Luck
No one tries to prove their toughness around nitrogen dioxide. Even in small amounts, this stuff attacks respiratory systems. A lab coat means nothing without gloves and tight-fitting goggles. I don’t bother with plain safety glasses; chemical splash goggles make more sense. Respirators fill in the gap when air is anything less than pristine.
Folks like to cut corners, but you can’t get away with regular latex gloves for tasks like refilling cylinders or cleaning up drips. Butyl rubber or a double-glove routine works best, keeping corrosion far from skin. I keep extra gloves nearby, because I trust sweat more than delayed reactions.
Keep Air Moving, Don’t Rely on Hope
Labs shouldn’t feel stuffy, especially around nitrogen dioxide. Fume hoods transform the workspace. I make sure exhaust fans hum loud, and I never act surprised if air monitoring sensors chirp. Nobody wins a prize for ignoring alarms. I always want to know parts per million, not guess at how safe the room feels.
After one scary spill, our lab locked down a policy: store nitrogen dioxide in ventilated storage, away from sunlight and any heat. Cool and dry wins every time. Push containers back from edges, mark them with bold labels, and keep an eye on anything that looks rusty or dented.
Handling Spills: Every Second Counts
Spills make everyone nervous, but it’s the speed that matters. No one pauses to “assess the situation” for too long. I reach for the emergency respirator first, then tackle the cause. I never forget the eyewash and drench shower stations. They earn their keep during actual emergencies, not just inspections. Draining the lungs and washing the skin off takes priority, and someone always calls emergency responders sooner rather than later.
I never handle nitrogen dioxide solo. Two sets of eyes double the odds someone remembers the procedure. We drill on evacuation routes and emergency numbers until it feels rote. Sometimes basic training saves lives in a way fancy policies simply can’t.
Prevention: Thoughtful Practices Over Rushed Routines
I’ve learned that accidents favor the hurried and the distracted. Double-checking labels, reviewing the day’s tasks, and warning newcomers about the risks keeps everyone alert. Clear training works better than huge binders of forgotten procedures.
Regular checks on ventilation, protective gear, and evacuation plans reduce mistakes. Posting emergency info where everyone sees it prevents confusion, especially for folks new to the job. People support safe habits when the reasons ring true, not just because regulations demand it.
Nitrogen dioxide doesn’t make room for half-measures. Every safety step matters, from storing cylinders upright to putting on thicker gloves. I’d rather over-prepare than explain away a preventable scare or hospital visit. Respect for chemicals starts with each person—every shift, every bottle, every time.
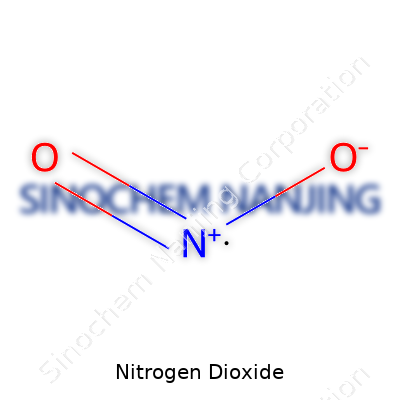
What is the chemical formula and physical properties of Nitrogen Dioxide?
Getting to Know Nitrogen Dioxide
Nitrogen dioxide, with the chemical tag NO2, hardly slips under the radar in any chemistry or environmental science class. This gas draws plenty of attention because it shows up in the air we breathe, in car exhaust, near industrial stacks, and wherever fuel finds a spark to burn. Over the years, working near city traffic and living close to busy roads has made me listen more carefully to the stories scientists share about the stuff swirling around us.
How It Looks and Smells
NO2 doesn’t bother hiding. Anyone standing near a tailpipe or an old power plant chimney might notice the sharp, biting odor or catch a glimpse of its reddish-brown tint. This gas weighs in heavier than air and tends to hover in low-lying places instead of drifting up and away. It’s one thing to read about NO2’s color and smell in books, but catching that strange metallic tang on a foggy morning drive stays with you.
Physical Traits
NO2 starts letting people know it’s around at surprisingly low exposures. The gas feels cold below freezing and quickly switches back to a gas above 21°C (70°F). Its boiling point sits right at about 21°C, so on a cool day, it might lurk as a liquid in a sealed bottle, but in the open, it floats as a brownish gas. Science turns practical as well: in a chemistry lab, watching NO2 collect and change color with the temperature hits home that chemistry isn’t just for textbooks.
A harsh and acrid smell makes this gas impossible to ignore, even at low parts per million. NO2 also dissolves into water, though not easily. When it finally does, it forms a highly acidic mix, producing both nitric and nitrous acids. That shift matters a lot: emotions run high in places where acid rain threatens crops or historical buildings, and nitrogen dioxide has a hand in that chain of events.
Hazards and Impact on Daily Life
Living near crowded streets means nitrogen dioxide isn’t just an abstract concern. Exposure, even for just an hour, pulls at lungs and stirs up trouble for kids, older adults, and anyone managing asthma. I spent a summer living near a major highway and learned quickly that extra coughing or itchy eyes don’t come out of nowhere. It’s personal for people in dense cities or neighborhoods boxed in by heavy industry.
Data lines up with lived experiences. The EPA has linked rises in outdoor NO2 to increases in respiratory diseases. Cities measure NO2 as a marker for traffic-related air pollution—one reason so many environmental groups push for cleaner engines, better public transportation, and planting dense green buffers. These actions go far beyond paperwork; they try to cut the everyday harm that rises from the streets.
Moving Toward Cleaner Skies
Staying informed on the makeup and properties of nitrogen dioxide helps shape choices at home and in city halls. Anything that trims traffic emissions—simple steps like walking, carpooling, or choosing a bike—chips away at the brown clouds that build up on calm days. Cities switching buses from diesel to electric see the purple-brown haze start to clear up. It’s about recognizing the chemical, not just in labs, but in the crowded, sunlit avenues where people live, work, and breathe.
| Names | |
| Preferred IUPAC name | nitrogen dioxide |
| Other names |
Nitrogen peroxide Nitrogen(IV) oxide Deutoxide of nitrogen Nitro dioxide |
| Pronunciation | /naɪˈtrəʊdʒən daɪˈɒksaɪd/ |
| Identifiers | |
| CAS Number | 10102-44-0 |
| Beilstein Reference | 3587154 |
| ChEBI | CHEBI:17996 |
| ChEMBL | CHEMBL113355 |
| ChemSpider | 991 |
| DrugBank | DB09142 |
| ECHA InfoCard | 100.028.763 |
| EC Number | 233-272-6 |
| Gmelin Reference | Gmelin Reference: **1085** |
| KEGG | C00296 |
| MeSH | D009585 |
| PubChem CID | 3032552 |
| RTECS number | QW9800000 |
| UNII | 3M66J8JZ5F |
| UN number | UN1067 |
| Properties | |
| Chemical formula | NO2 |
| Molar mass | 46.01 g/mol |
| Appearance | Reddish-brown gas |
| Odor | Harsh, pungent |
| Density | 1.88 g/L |
| Solubility in water | 7.9 g/100 mL (20 °C) |
| log P | -0.32 |
| Vapor pressure | 720 mmHg (20°C) |
| Acidity (pKa) | Acidity (pKa) of Nitrogen Dioxide: -3.0 |
| Basicity (pKb) | 7.39 |
| Magnetic susceptibility (χ) | Paramagnetic |
| Refractive index (nD) | 1.448 |
| Viscosity | Gas: 0.014 cP at 15°C |
| Dipole moment | 0.316 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 240.1 J·K⁻¹·mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 33.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | 33.9 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AN02 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS04,GHS05,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H270, H314, H331 |
| Precautionary statements | P261, P271, P273, P280, P304+P340, P305+P351+P338, P308+P311, P312, P320, P330, P337+P313, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-0-OX |
| Autoignition temperature | 150 °C |
| Lethal dose or concentration | LDLo oral rat 86 mg/kg |
| LD50 (median dose) | 110 mg/m³ (human, inhalation, 4 hours) |
| NIOSH | RS206 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Nitrogen Dioxide: "5 ppm (ceiling) |
| REL (Recommended) | 0.08 ppm |
| IDLH (Immediate danger) | 20 ppm |
| Related compounds | |
| Related compounds |
Dinitrogen tetroxide Nitrous acid Nitric acid Nitric oxide Nitrite |

