Nitrobenzotrifluoride: A Deep Dive into Its Origins, Properties, and Prospects
Historical Development of Nitrobenzotrifluoride
Nitrobenzotrifluoride carries a legacy that winds back to the explosive expansion of synthetic chemistry through the twentieth century. Early chemists, chasing reliable intermediates for dyes and pharmaceuticals, ran across the electro-withdrawing power in the trifluoromethyl group and noticed its impact on aromatic compounds. Labs focused effort on harnessing this trait for more robust molecules. By the post-war period, nitrobenzotrifluoride had moved from a curious byproduct to a staple in intermediate synthesis. Chemical companies, especially in Europe and the US, capitalized on its stability and made it part of the backbone for agrochemicals and performance polymers that demanded not just potency but also chemical resilience. In my own time around research benches, talk about trifluoromethyl aromatics centers not only on their chemistry but also the legacy left by these early industrial explorers.
Product Overview: Forms and Availability
Every major reagent supplier stocks nitrobenzotrifluoride. The market distinguishes among its isomers—ortho, meta, and para—each serving slightly different niches but sharing core functionalities. Most often, chemists buy the liquid, m-nitrobenzotrifluoride (meta isomer), as it slides easily into reaction setups. Drums of this material appear at big manufacturing plants, while research labs stock its smaller bottles. The need for a clean, consistent trifluoromethyl source explains why chemical supply chains keep this product front and center, both for bulk users and at the research scale.
Physical and Chemical Properties
M-nitrobenzotrifluoride brings together two powerful groups on a benzene ring—a nitro and a trifluoromethyl. This molecule packs a punch both structurally and chemically. It looks like a colorless to pale yellow liquid, avoids much water, and emits a slightly sweet odor with a hint of sharpness. The compound weighs in at 207.1 g/mol with a melting point around -30°C and a boiling point that hovers near 174°C. The low polarity of the trifluoromethyl group, combined with the electron-withdrawing nitro, makes it both chemically stable and a strong attractor or repeller, depending on what shares the reaction flask. The high electronegativity shields some reactions, yet opens doors for nucleophilic aromatic substitutions, lending versatility to the molecule. I remember testing its solubility profile and noting how it leans toward organic solvents, making cleanup straightforward and integration into complex syntheses much simpler.
Technical Specifications & Labeling
Suppliers outline nitrobenzotrifluoride’s purity—mostly above 98% for research and industrial input. Standard specs highlight low levels of related aromatic impurities, plus thresholds for water content and acidity that avoid side-reactions during critical synthesis steps. Labels must note the hazard class, with flammable and toxic tags sitting front and center. The globally harmonized system places nitrobenzotrifluoride within the acute toxicity and irritant categories for transport and handling. Shipping docs reference its UN number, and I find most labs avoid confusion by color-coding storage areas and logging every bottle's expiration date. This attention around technical specification doesn’t just meet regulation; it gives peace of mind to researchers firing up a new reaction route.
Preparation Method
Industry and universities prepare nitrobenzotrifluoride mainly through direct nitration of benzotrifluoride. Chemists blend trifluorotoluene with a mixture of nitric and sulfuric acids, tweak the temperature, and drive the reaction progress toward the preferred isomer. Skilled handling of reaction temperature and ratio between acids tilts the outcome either toward meta or para products. Some advanced shops tweak conditions, like adding acetic anhydride or using solid acid catalysis to control selectivity. Clean-up involves neutralization and extraction, sometimes followed by distillation to squeeze every drop of purity from the batch. Watching nitration reactions on a research team, I notice strict limits on scale and temperature, as runaway exothermicity caused by these conditions can pose real risks to chemists.
Chemical Reactions & Modifications
Nitrobenzotrifluoride’s value multiplies through functional group transformations. Its nitro group opens the door to reduction—yielding potent aminobenzotrifluoride intermediates used widely in pigment and pharmaceutical production. Chemists use catalytic hydrogenation or employ tin and hydrochloric acid to trim away the nitro, granting access to a versatile aniline. The trifluoromethyl group, considered tough, sometimes undergoes halogen exchange or is exploited in further aromatic substitutions. Nucleophiles attack the aromatic ring, swapping out halides for stronger electron-withdrawing partners. I’ve watched postdocs debate whether to start with the nitro or trifluoromethyl installation, always considering the order to avoid harsh conditions that might wreck the structure. Newer green chemistry avenues focus on milder reduction and halogenation, promising less waste and fewer hazardous byproducts.
Synonyms & Product Names
Chemical catalogs and patents reference nitrobenzotrifluoride by several handles. The base name splits up into 2-nitrobenzotrifluoride, 3-nitrobenzotrifluoride, and 4-nitrobenzotrifluoride, based on the placement of nitro against the CF3 group. You’ll run into names like trifluoromethyl nitrobenzene or methyltrifluoronitrobenzene depending on the literature’s age or the catalog’s regional naming conventions. CAS numbers—often 98-46-4 for the meta version—help avoid confusion, especially across global supply webs. Reading research papers, old and new, I’ve learned that using clear, standardized names matters far more than most expect, especially if translation or international collaboration enters the mix.
Safety & Operational Standards
Lab and plant workers handle nitrobenzotrifluoride with clear respect for its health and fire dangers. Splashing a drop on skin produces quick irritation; inhaling vapors stings the nose and throat within seconds. Standard safety protocols line up: nitrile gloves, splash goggles, and solid ventilation come as everyday gear. Storage goes in cool metal flasks, behind locked doors, and away from acids or bases. Spills demand sand or inert absorbents, with careful disposal of waste to meet local environmental laws. Training courses on chemicals like this stress the balance between reactivity and toxicity—I remember seeing firsthand how improper handling led to both local health incidents and, in extreme negligence cases, larger environmental fines. Strict protocols pay off: good records, protection equipment, and standardized labeling mean safe and productive work.
Application Area
Many sectors chase the properties of nitrobenzotrifluoride. Agrochemical makers pull on these molecules to design herbicide and pesticide scaffolds that resist breakdown in field conditions. Electronics manufacturers add trifluoromethyl aromatics into liquid crystals and photoresists, seeking robust electronic properties and environmental durability. Pharmaceutical developers look for unique aromatic intermediates, often choosing this group for drugs where metabolic stability and solubility shifts can make or break a candidate molecule. In pigments and dyes, this compound provides high brightness and long fade resistance. My work in a polymer R&D group prompted repeated study of trifluoromethyl aromatics as key pieces of high-performance plastics for lightweight electronics and aerospace use, chasing ever higher strength-to-weight ratios.
Research & Development
Academic and industrial chemists keep nitrobenzotrifluoride on the research radar for both synthesis development and fundamental property exploration. Automated chemistry platforms test new ways to tune selectivity or lower energy consumption during nitration. Catalysis groups dig into green and solid-phase approaches, hoping to eliminate harsh acids and volatile solvents. Physicists collaborate to insert these compounds into novel materials, studying how the powerful electron-withdrawing groups can modulate light absorption or dielectric properties in working devices. From time in joint industry-university seminars, I can say that more funding flows each year toward fluorinated chemistry and that much curiosity centers around not just creating new molecules, but upcycling or recycling older ones. These efforts will extend the lifetime and scope of this chemistry and answer growing environmental and economic pressures.
Toxicity Research
Scientists pay special attention to the toxicity of nitrobenzotrifluoride. Both rodent studies and in vitro cell cultures show acute toxicity at certain exposure levels. Skin contact leads to irritation and sometimes dermatitis; vapor exposure causes headaches and long-term nervous system effects after repeated incidents. Environmental impact looms large, as nitrobenzotrifluoride products sometimes wash downstream, affecting aquatic life. Regulations demand clear reporting of accidental releases and precise tracking of workplace exposure to meet occupational limits. Having reviewed tox reports and sat through industrial hygiene seminars, I find ongoing research into detection, remediation, and lower-toxicity substitutes provides hope that the next generation of trifluoromethyl aromatics might sidestep many past hazards.
Future Prospects
The path ahead for nitrobenzotrifluoride tracks both opportunity and challenge. Global demand grows as the electronics and pharmaceutical fields steer toward fluorinated intermediates that can deliver the necessary combination of performance and staying power. Environmental concerns push producers to prioritize new synthetic strategies, pushing research into cleaner routes, reliable recyclability, and improved toxicity profiles. Green chemistry approaches, guided by both regulation and better science, aim to phase out outdated methods and embrace renewable inputs. From industry panels in 2023, optimism runs high about advanced catalysis and bio-derived feedstocks gaining traction. These changes could shape a market both safer for workers and lighter on the planet, if companies and governments work hand in hand. My years in chemistry have shown that innovation in specialty chemicals never stops, and nitrobenzotrifluoride will stick around—its chemistry, adaptability, and constant reinvention define the heart of progress.
What is Nitrobenzotrifluoride used for?
A Chemical Behind Everyday Products
Growing up near a small industrial town, I remember seeing railcars marked with chemical names I could barely pronounce, and one of them was Nitrobenzotrifluoride. Years later, I learned that this compound moves quietly through a range of industries, from medicine to electronics. Most people have never heard of it, but it shapes a lot of what shows up in pharmacies and hardware shops.
Shaping Pharmaceuticals, Paints, and More
Drug makers depend on intermediates—the ‘in-between’ chemicals that let them stitch together complex medicine molecules. Nitrobenzotrifluoride acts like a key building block. Because the trifluoromethyl group packs in stability and changes how other pieces snap together, chemists use this compound to start or speed up reactions. The pain relievers and allergy meds on store shelves, as well as certain antidepressants, would be a lot harder to make without it.
It’s not only about medicine. Nitrobenzotrifluoride finds its way into dyes and pigments. I once spoke with a friend in the coatings business who explained why their latest paints last longer outdoors—the secret lies in chemical tweaks that start with molecules like Nitrobenzotrifluoride. This compound helps paint makers craft brighter, more fade-resistant colors, which hold up to sun and rain much better than old-school options.
Pushing Technology Forward
Anyone who follows trends in electronics knows how tough it is to make gadgets smaller and faster without frying them. Engineers now demand materials that handle heat better, resist chemical damage, and always function reliably. Here, Nitrobenzotrifluoride provides key starting points for the polymers that go into sleek smartphone shells and sturdy circuit boards. Adding fluorine atoms gives these plastics a real boost: they shrug off solvents, keep their shape in heat, and stand up to harsh cleaning.
Environmental and Health Concerns
Chemicals like Nitrobenzotrifluoride bring big perks, but they also raise questions—ones that communities near factories, like my hometown, care about. During production, the risk of leaks or vapor exposure needs serious attention. Research shows that high levels might cause skin or eye irritation, and there’s ongoing study about longer-term effects on health. In the end, workers and neighbors deserve strict monitoring and clear information, not guesses or industry jargon.
Safer Use and Smarter Alternatives
Striking a balance means better safety gear in factories, but there’s more to the story. Green chemistry has moved from buzzword to the front lines; if scientists can design reactions that skip Nitrobenzotrifluoride or cut waste, it’s a win for both business and health. Investment in process improvement—using less of any hazardous chemical or swapping in milder substitutes—pushes the industry forward.
People might never see Nitrobenzotrifluoride directly, yet its fingerprints cover a surprising number of products. Awareness, strong rules, and new ideas ensure its benefits don’t come with unwanted costs—whether in our bodies or the natural world right outside the factory fence.
What are the safety precautions when handling Nitrobenzotrifluoride?
Getting to Know Nitrobenzotrifluoride
Dealing with chemicals in the lab has always put my senses on high alert, and nitrobenzotrifluoride fits right into that category. This compound sees regular use in making dyes, pharmaceuticals, and some pesticides. It's got three fluorine atoms in its molecular mix, which isn’t just trivia for a quiz night — it means the compound is tougher on both gear and people. Breathing in its vapor or getting it on bare skin can trigger serious side effects, and it doesn’t take much to feel the impact. Knowing the facts about what it can do makes the precautions real — not just a list on a sheet pulled off the internet.
Personal Protective Equipment is Non-Negotiable
I still remember my earliest lab days, where a lax attitude meant a pair of goggles hanging uselessly on a hook. One whiff of nitrobenzotrifluoride fixes that sort of mistake fast. Gloves with real chemical resistance — not latex, but nitrile or neoprene — have to cover all skin. Splash goggles with indirect vents and a face shield stop anything from making contact with sensitive areas. Lab coats should button up to the neck, sleeves pushed over gloves to leave no open inches. I'd never turn my back on proper gear.
Good Air is Not Optional
Fume hoods aren't just for looks. Every time I’ve decanted nitrobenzotrifluoride, I put it inside a certified hood, checked the airflow gauge, and let others know what I’m doing. This isn’t just a CYA moment — the vapors can irritate airways and in large enough doses, affect the nervous system. Anyone working with it should review the fume hood’s last inspection date. No exceptions. If the hood’s busted or you smell anything sharp or chemical, pack up and tell the supervisor. Fixing a ventilation system costs less than a hospital trip.
Labeling and Storage Keep Trouble Away
Mislabeling chemicals has led to mix-ups I won’t soon forget. Nitrobenzotrifluoride doesn’t belong anywhere near acids, oxidizers, or other strong reactants. At the end of the day, I lock it in a flammable storage cabinet, keeping it upright in a sealed, clearly labeled bottle. Storing it below eye level makes cleanup easier if something goes wrong. Each container gets a fresh label, showing both the name and hazard info — lazy shortcuts can lead to real risk.
Spills and Waste Should Never Be Improvised
If a beaker breaks or a drop goes astray, I reach for absorbent pads rated specifically for hazardous organics. Trained staff handle the cleanup, wearing both gloves and respirators as needed. I keep emergency contact details and spill kits on hand, always easy to spot. Used materials get sealed in a designated hazardous waste container — not down a drain, not in the regular trash. Training sessions hit this point over and over, and for good reason.
Emergency Procedures Demand Practice
Panic makes small mistakes big in a hurry. I review the emergency shower and eyewash locations at the start of every lab stint. Everyone should know the fire exit and who dials for help. SDS sheets don’t sit in a drawer; they stay at arm’s reach. In my experience, quick responses come from practice, not panic.
Building a Culture of Safety
Complacency breeds trouble. Staff briefings, regular safety drills, and honest debriefs after near misses keep nitrobenzotrifluoride’s hazards in focus. I share hard-learned lessons with new team members, hoping they’ll do the same for someone else down the line. Regulations matter, but respect matters more. It’s worked so far — and I plan to keep it that way.
What is the chemical formula of Nitrobenzotrifluoride?
Getting to Know Nitrobenzotrifluoride
Nitrobenzotrifluoride carries the chemical formula C7H4F3NO2. This name may sound heavy for folks not elbow-deep in a chemistry lab, but the formula packs a real-world punch far beyond a textbook quiz answer. The skeleton of this molecule comes from benzene—a ring of six carbon atoms—decorated with three fluorine atoms and a nitro group, making it unique and versatile in both chemistry and industry.
Why It Shows Up in Industry and Labs
My time spent shoulder-to-shoulder with chemical engineers has taught me the value of a sturdy chemical backbone. Nitrobenzotrifluoride stands out. It’s not as famous as sodium chloride or even aspirin, but it fills a crucial niche. The tight bonds between carbon and those three fluorines aren’t just quirks of structure; they create chemical stability and influence how other substances react around it.
Industries turn to Nitrobenzotrifluoride for tasks requiring a mix of stability and reactivity. This compound gets used for making agricultural chemicals, specialty polymers, dyes, and pharmaceutical ingredients. The trifluoromethyl group—the trio of fluorines—amplifies the molecule’s resistance to breakdown and shapes its interactions during synthesis. Even small changes to this formula create compounds with new properties, a favorite trick for chemists designing new molecules or tweaking drug activity.
Real-World Impact and Risks
Trust in a chemical depends not just on its usefulness but on the safety practices wrapped around it. My experiences managing chemical stocks show that Nitrobenzotrifluoride asks for respect. It’s stable under normal conditions because of the fluorine bonds, yet the nitro group brings some risk. Contact with skin or inhalation can be hazardous, and spills bring challenges for both the person and the environment. Clear labeling, good ventilation, and strong protocols matter every day in facilities handling this substance.
Looking Forward: Solutions for Safety and Sustainability
The chemical world keeps searching for ways to balance utility and safety. One hard-learned lesson comes from seeing how strict training and updated equipment prevent most accidents. Wearing gloves, using fume hoods, and choosing less hazardous alternatives whenever possible is no news to those on the ground. Encouraging transparency in sourcing and tracking Nitrobenzotrifluoride also blocks a lot of opportunities for mismanagement.
Sustainable chemistry isn’t just a catchphrase. Teams working in research and manufacturing push for methods that lower emissions, recycle solvents, and invent new molecules with built-in safeguards. Watching companies follow green chemistry guidelines shows this isn’t out of reach.
The Takeaway
Chemical formulas like C7H4F3NO2 stick with you if you’ve ever broken a sweat making something work better or safer in the lab. Nitrobenzotrifluoride isn’t making headlines, but understanding its structure and uses—and treating it with healthy caution—gives both chemists and consumers a better shot at safety and progress.
Is Nitrobenzotrifluoride hazardous to health?
What Is Nitrobenzotrifluoride?
Nitrobenzotrifluoride pops up in conversations about industrial chemicals, especially in places that crank out paints, pharmaceuticals, and agrochemicals. With its tongue-twisting name, it seems about as removed from daily life as anything could get. Still, step into a lab or chemical plant, and you’ll find that it’s not rare at all. Workers deal with it in the form of a colorless to yellow liquid that packs a distinctive, sharp odor. It comes loaded with a trifluoromethyl group attached to a nitrobenzene backbone — a blend that brings unique chemical properties, including the ability to dissolve otherwise stubborn compounds.
Health Hazards: What’s at Stake?
Over the years, safety experts have flagged nitrobenzotrifluoride for several health concerns. Breathing in its vapor in large amounts can irritate the nose, throat, and lungs. Reports tell of headaches, dizziness, and in stubborn cases, more severe nervous system effects. Splash it on your skin, and irritation or even cracked, red patches may set in. Contact with the eyes brings a much sharper sting and potential for long-lasting problems.
I’ve talked with folks who spend their shifts around volatile chemicals, and they always mention how easily fumes build up in poorly ventilated spaces. One slip — a splash, an unnoticed leak — can mean a trip to the medical station. Documentation from government authorities like the U.S. National Institute for Occupational Safety and Health (NIOSH) shows nitrobenzotrifluoride can get absorbed through the skin, amplifying the risk if gloves or protective suits aren’t part of the routine. The long-term picture matters just as much. Animal studies suggest repeated exposure may affect organs such as the liver and kidneys. Nobody wants to gamble with that kind of uncertainty in their career.
What Makes It So Concerning?
The mix of fast-acting effects and possible longer-term problems demands respect. Nitrobenzotrifluoride doesn't top the list of the most notorious industrial villains like asbestos or benzene, but its toxicity is still real enough that nobody should shrug off the risks. Once inside the body, it can break down into byproducts that do their own damage, sometimes quietly over time. I’ve seen older manufacturing sites, where safety measures lag behind today’s standards, raise serious alarms about lingering contamination in the air and water. The idea that exposure sneaks up on workers, sometimes after years of routine contact, makes strong safety protocols absolutely essential.
How To Tackle The Hazards
Reducing risk starts with education. Workers must know what they're handling, including the hidden dangers. Labels, warnings, and training go far, but only work if people take them seriously. Proper gear — gloves, goggles, and fitted respirators — can stop skin and lung contact. Ventilation systems can keep air quality in check, especially in busy work areas. I’ve watched some companies roll out personal exposure monitoring devices, so employees know right away if the chemical hangs around at risky levels. These investments pay off not just in fewer accidents or sick days, but in peace of mind.
Finally, companies need routine health checks for people exposed to nitrobenzotrifluoride. Blood tests, liver panels, and other screenings can help spot issues early. It sends the message that their health matters for the long haul. That kind of culture doesn’t happen by accident — it grows from leadership who sees workers as more than a line item. For anyone working with chemicals like nitrobenzotrifluoride, safety isn’t just a box to tick but a habit to keep, every single day.
How should Nitrobenzotrifluoride be stored?
Straight Talk on Chemical Safety
Storing nitrobenzotrifluoride doesn’t leave much room for shortcuts. Whether you’re running a small shop or handling chemicals at a large facility, the risks hang heavy without proper steps. I’ve spent years around dusty chemical stockrooms, and the one thing you learn fast: some substances call for real respect. Nitrobenzotrifluoride fits squarely in that category.
Fire Fears and Vapors: Not Just Theoretical
Nitrobenzotrifluoride ignites pretty easily. A surprising number of accidents happen because someone put it next to a steam line or figured, “It will be fine here for a few hours.” That sense of casualness disappears fast after you’ve smelled solvents where they shouldn’t be. Its vapors also irritate the nose and lungs, which means good ventilation saves headaches and worse. Stuffy closets or packed storerooms create a hazard zone. So I’d always recommend keeping it in a well-ventilated, cool space—away from direct sun or steam lines. No matter how drained the maintenance budget looks, skimping on airflow leads to trouble.
Materials Matter: Metal Risks and Seepage Worries
Over time, I’ve noticed some folks packing this compound in random drums. Certain metals react badly with aggressive chemicals. Steel drums eventually corrode or leak—leading to small puddles that take hours to clean up and raise a stink in the workroom. Sturdy, tightly sealed containers, built for chemical storage, offer real peace of mind. Relying on good-quality plastics—with labels that don’t peel in three months—often costs less down the line, especially after factoring in potential fines and cleanup bills.
A Little Separation Goes a Long Way
Piling incompatible supplies together just causes needless risks. Nitrobenzotrifluoride doesn’t belong anywhere near strong oxidizers, acids, or open flames. I’ve seen too many storage areas where acids and solvents sit shoulder to shoulder. One dropped drum or careless splash could set off a real mess. Just a simple dividing wall or separate shelf makes all the difference. Even with tight storage space, creative shelving or clear signage keeps chemicals apart in a way that protects both people and property.
Training: The Step That Gets Missed
Storing chemicals safely doesn’t just happen. A locked cabinet or a “hazard” sticker offers little help if folks in the shop don’t know what they’re reaching for. Most of the time, problems crop up when new hires or an overworked crew forget which bottle’s which. Regular reminders—or quick five-minute refreshers at shift changes—help keep everyone sharp. Chemical safety sheets shouldn’t just sit in a binder nobody opens. Easy access to real-world guidance beats unread paperwork every single time.
Keeping Tabs On Every Barrel
It’s not enough just to store nitrobenzotrifluoride in the right place. Knowing exactly how much is on hand, along with the last date of inspection, can head off a lot of issues. In my experience, a simple record log—paper or digital—catches slow leaks or missing barrels before they turn into big problems. Checking for corrosion, bulging containers, or weird smells takes maybe ten minutes a week and protects both health and inventory.
Better Practices, Fewer Headaches
Chemicals like nitrobenzotrifluoride highlight the importance of common sense, attention to detail, and regular habits. Respect for storage rules isn’t about red tape—it’s about safety and responsibility. Investing time and resources into proper containers, clear signage, and short, real training pays off in safer days and fewer close calls.
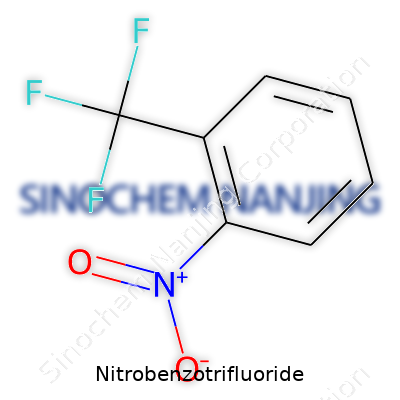
| Names | |
| Preferred IUPAC name | 1-nitro-4-(trifluoromethyl)benzene |
| Other names |
1-Nitro-4-(trifluoromethyl)benzene p-Nitrobenzotrifluoride 4-Nitrobenzotrifluoride p-(Trifluoromethyl)nitrobenzene 1-Nitro-4-(trifluoromethyl)phenyl 4-Nitro-α,α,α-trifluorotoluene |
| Pronunciation | /ˌnaɪ.troʊ.bɛn.zoʊˈtrɪ.fluː.ə.raɪd/ |
| Identifiers | |
| CAS Number | 98-16-8 |
| Beilstein Reference | 492125 |
| ChEBI | CHEBI:81844 |
| ChEMBL | CHEMBL135598 |
| ChemSpider | 14413 |
| DrugBank | DB14167 |
| ECHA InfoCard | 03c4b1e2-7f82-4e93-8e6a-82b2ea8399b9 |
| EC Number | 208-770-8 |
| Gmelin Reference | 79314 |
| KEGG | C14135 |
| MeSH | D009574 |
| PubChem CID | 7417 |
| RTECS number | CU5950000 |
| UNII | 81J3F7P6EY |
| UN number | UN3334 |
| Properties | |
| Chemical formula | C7H4F3NO2 |
| Molar mass | 213.12 g/mol |
| Appearance | Colorless liquid |
| Odor | Aromatic |
| Density | 1.353 g/cm³ |
| Solubility in water | insoluble |
| log P | 3.43 |
| Vapor pressure | 1 mmHg (20°C) |
| Acidity (pKa) | 11.0 |
| Magnetic susceptibility (χ) | -69.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.481 |
| Viscosity | 1.25 mPa·s (25 °C) |
| Dipole moment | 3.68 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 353.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -211.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5194.8 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P261, P264, P271, P301+P310, P305+P351+P338, P312, P337+P313, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 2, Instability: 0, Special: - |
| Flash point | 56 °C |
| Autoignition temperature | 540 °C |
| Lethal dose or concentration | LD50 oral rat 13,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 4550 mg/kg |
| NIOSH | WA8400000 |
| PEL (Permissible) | 100 ppm (ACGIH TWA) |
| REL (Recommended) | 250 ppm |
| IDLH (Immediate danger) | IDLH: 500 ppm |
| Related compounds | |
| Related compounds |
Benzotrifluoride Nitrobenzene Trifluorotoluene Trifluoromethylstyrene |

