Nitroacenaphthene: From Discovery to Modern Uses
Historical Development
People in the early days of organic chemistry always seemed drawn to the strange aromas and smoky vapors rising from naphthalene derivatives. Nitroacenaphthene sprang up out of those dusty laboratories in the late nineteenth century. Scientists eyed the acenaphthene skeleton, with its two fused benzene rings and a five-membered ring, and started adding nitric acid to coax new properties from the base material. This experimentation didn’t happen in a vacuum either; the early pharmaceutical and dye industries had a hunger for molecules that could unlock new colors and pathways. By the turn of the twentieth century, chemists realized that by nitrating acenaphthene, they could produce a compound that would lay the groundwork for everything from pigments to specialty intermediates. Academic papers began stacking up, each one refining the process a little further, chasing both cleaner yields and safer protocols.
Product Overview
You won’t see nitroacenaphthene lining grocery store shelves, but it serves as a reliable stepping stone in plenty of chemical syntheses. The compound usually comes as a light yellow to orange powder, a color that’s hard to mistake if you’ve mixed more than your share of organic chemicals. Its molecular formula, C12H7NO2, captures a simple reality: a nitro group shakes up the electron cloud of plain acenaphthene, creating a reactive and, at times, stubborn intermediate. In practice, manufacturers sell it mainly for research, semiconductors, and complex dye work—rarely as a finished product.
Physical & Chemical Properties
Some properties of nitroacenaphthene stand out right away if you’ve spent time in a synthetic lab. It melts around 140-145°C—a range typical of substituted aromatics. The powder dissolves modestly in polar organic solvents like ethanol and chloroform. Its odor, while not overwhelming, hits with the metallic-tinged scent you find in many nitrated aromatics, reminiscent of vintage chemistry drawers. The nitro group doesn’t just sit on the ring; it draws electrons, setting the stage for further aromatic substitution and reduction. That’s where the compound earns its keep in downstream modifications.
Technical Specifications & Labeling
Chemists expect a high degree of purity, usually upwards of 97%, when ordering from professional suppliers. Labels ought to carry the chemical’s IUPAC name—1-nitroacenaphthene—and indicate the specific isomer, since position matters in this field. You see plenty of warnings about handling and storage right on the packaging. Moisture control, dark storage, and tightly sealed jars are the norm. No sense letting a sensitive aromatic degrade because a lab tech left it out near sunlight or forgot to check the humidity gauge. Hazard labels stay clear: avoid breathing dust, wear gloves, and keep the container closed tight.
Preparation Method
Making nitroacenaphthene in the lab comes down to adding concentrated nitric acid to acenaphthene under controlled temperature, typically below 30°C. That sounds simple until you factor in heat management—nitration’s exothermic, so chemists use ice baths and slow addition rates to keep the mix from running out of control. Sulfuric acid steps in as a catalyst and solvent, encouraging the reaction to favor the desired nitro position. The process takes patience; you wait for the yellow color to deepen, filter out the crystals, and then wash them repeatedly to push out acids and residual reactivity. Skipping purification steps can leave you with a lower yield clogged with tars or unwanted byproducts. Large-scale synthesis replaces glassware with steel, automated stirrers, and jacketed reactors—a jump in technology, but the same principle.
Chemical Reactions & Modifications
Nitroacenaphthene sits as a launch pad for several transformations. Reduction with iron filings and hydrochloric acid strips off the nitro group, making way for amines, a crucial starting point for pharmaceuticals and colorants. Electrophilic aromatic substitution opens the door for further halogenation, sulfonation, or even alkylation. Sometimes, chemists run condensation reactions using aldehydes or anhydrides to append even larger groups for pigment or polymer applications. In years working with these nitroaromatics, I’ve seen how precision—timing, temperature, reagent ratios—makes all the difference between a clean product and a batch that turns dark with side-reactions.
Synonyms & Product Names
Ask for nitroacenaphthene in a global lab supply catalog, and you’ll find a handful of alternative aliases: 1-nitroacenaphthene, α-nitroacenaphthene, or NSC 4271. Some industrial circles abbreviate it for faster database searches—shortening to “NAN” or using chemical indexes. These names help cut through language differences and avoid mistakes in orders or safety checks.
Safety & Operational Standards
Nitrated aromatics tend to get respect in any lab. Even a quick glance at old incident reports will show why. Nitroacenaphthene is no exception. Dust is hazardous to the lungs; gloves and safety goggles are a must. Labs usually run dedicated hoods during weighing, and chemical spill kits sit nearby. Storage always takes place in containers that prevent static buildup or accidental exposure to sunlight, and there are never open flames anywhere close, due to the low flashpoint and risk of decomposition. Employees train on what to do if someone gets exposed, including access to eyewash stations and showers. Chemical disposal uses licensed contractors; no shortcuts there.
Application Area
You won’t find nitroacenaphthene powering the local recycling plant or showing up in over-the-counter cleaners, but industries value it for the specific roles it plays. Dye and pigment manufacturers use the molecule’s reactivity to develop new shades for textiles and plastics. Electronic materials engineers sometimes employ it as a precursor for certain advanced polymers. Specialty pharmaceutical companies reach for it during the multi-step synthesis of experimental drugs, especially ones requiring aromatic amines with unusual substitution. Research institutions keep small stocks around for studies in physical organic chemistry, looking to unlock more insight on aromatic nitration mechanisms.
Research & Development
Academic labs continue to explore nitroacenaphthene both as a reagent and as an object of study all on its own. Using advanced spectroscopic techniques, scientists probe how the nitro group changes the electron density across the acenaphthene skeleton, seeking clues to both reaction speed and selectivity. Some of the more forward-thinking projects pair traditional wet chemistry with computational modeling, simulating thousands of possible modifications to predict color outcomes in dyes or assess potential biological activity. Research funding often comes from energy, electronics, and pharmaceutical industries hungry for new intermediate scaffolds, giving career chemists regular reason to revisit nitroacenaphthene’s intricacies in grants and proposals.
Toxicity Research
No one wants to handle chemicals blind to the risks. Toxicological research on nitroacenaphthene has shown that inhalation and skin contact create health hazards, in line with many aromatic nitro compounds. Repeated exposure can irritate airways and even affect liver function in high doses. Animal studies led to stricter lab policies requiring minimized exposure and access restrictions. Material safety data sheets (MSDS) include comprehensive tables for safe handling, emergency response, and first-aid directions. Personal experience shows most incidents happen not from large spills, but from complacency during repeated small-scale handling. Regular safety audits and revised standard operating procedures have made a difference, but ongoing vigilance remains key.
Future Prospects
Chemists keep searching for ways to modify the acenaphthene core to unlock new electronic, pharmacological, or optical properties. Potential advances in selective nitration may lead to safer, higher-yielding synthesis routes. As the drive for greener chemistry intensifies, research could point toward using less hazardous nitrating agents, reducing acid waste, or creating recyclable catalyst systems. The semiconductor and specialty dyes sectors are already asking if these advances could make nitroacenaphthene-based products both cleaner and more cost-effective. Most importantly, a new generation of chemists is eager to see how deep this relatively simple molecule’s utility will reach, especially as AI-driven synthesis designs become part of the research landscape.
What is Nitroacenaphthene used for?
A Look at Nitroacenaphthene’s Place in Modern Industry
Few folks outside of chemistry circles hear about nitroacenaphthene, but this compound plays a subtle role in several real-world applications. Having logged plenty of time in material science labs, I have bumped into nitroacenaphthene while tracking down raw materials for dyes and certain chemical syntheses. It acts as a building block in the manufacturing world, and the results show up in more places than most would notice.
Beyond the Lab Bench
Most noticeably, nitroacenaphthene shows up in the world of dyes and pigments. Chemists use it to produce deep and stable colors that withstand light and wear. Many synthetic dyes owe their brightness to chemicals derived from this molecule. In quality control tests I’ve run, products made with these colors outlast many alternatives. That reliability proves valuable for manufacturers seeking longer-lasting consumer goods—from textiles to automotive interiors.
Armed with a molecular structure related to acenaphthene, nitroacenaphthene also matters as a starting material for other specialty chemicals. Organic synthesis often depends on small tweaks to core molecules to deliver completely new properties. I have watched teams in the lab customize pharmaceuticals and agricultural chemicals this way. Small molecular differences can mean new medicines or pest control agents that target problems more effectively. According to industrial chemistry data, carefully managed transformations using nitroacenaphthene contribute to safer products through selectivity and control.
Industry Benefits and Hazards
Plenty of companies value nitroacenaphthene’s ability to create stability in synthetic resins and polymers. High-performance plastics sometimes require additives that can survive tough conditions. Nitro-containing compounds often deliver heat and chemical resistance. In the polymer pilot plants I’ve visited, even small changes to additives alter how a resin handles stress. Nitroacenaphthene gives engineers an important tool for getting performance just right for demanding applications, such as electronics insulation or engine components.
Experiences in safety training brought up another side of this chemical: handling risks. Nitro-organic compounds come with red flags for health and the environment. Overexposure in the workplace can lead to skin and respiratory issues. More importantly, improper disposal means introducing persistent contaminants to soil and water. Statistics from safety regulators urge rigorous containment and disposal procedures for all nitro-aromatics, not just nitroacenaphthene. Watching lab teams suit up in gloves and masks turned classroom warnings into immediate reality. Protecting workers and ecosystems sits among my top concerns working in industrial chemistry.
Safer Paths and Industry Responsibility
Cleaner production methods have grown out of research into alternatives and safer chemistries. Several companies have tried to replace nitroacenaphthene in certain colorant and resin applications with less hazardous options. Life cycle assessments suggest that moving away from nitro compounds helps decrease toxic byproducts and lower long-term environmental risks. At industry conferences, experts push for tighter controls and regular checks on all aromatic nitro compounds in the supply chain.
Consumer awareness also shapes the future for chemicals like nitroacenaphthene. More buyers want transparency and safe ingredients in goods ranging from plastics to clothing. Regulations in Europe and the US place limits on nitro-aromatic residues in finished products, nudging producers toward safer practices. Switching to greener chemistry or investing in recycling and waste treatment stands as a responsible move—something leaders in this space discuss as more than a compliance step.
What are the safety precautions when handling Nitroacenaphthene?
Direct Contact Can Lead to Health Problems
Nitroacenaphthene doesn’t play around. Touch it bare-handed and you risk absorbing a potent irritant. I’ve spent enough hours in labs to know how quickly a hand-wipe with strong chemicals turns into raw, cracked skin or worse, chemical burns. You want strong nitrile gloves, not the cheap latex kind that plenty of folks bring out for basic jobs. Eyes and lungs catch even more heat. Splash goggles make a difference. If you're without them, expect stinging, redness, or worse—cornea damage. A snug-fitting lab coat and splash-resistant apron aren’t overkill, especially if you have a clumsy streak.
Hydration, Heat, and Sparks: A Dangerous Mix
Leaving even a smudge of Nitroacenaphthene near a heating element or spark can be life-changing. Its nitro-compound nature means it reacts at lower temperatures than most organic chemicals. Your workspace has to be cool, dry, and well-ventilated. Forget the idea of sipping a coffee where you work with Nitroacenaphthene or letting your phone spark on a metal tray.
Ventilation Isn't a Luxury
Spend just half an hour handling this compound in a closed-off room and headaches hit fast. Its fumes drift in the air, and regular windows won’t clear them out. Fume hoods, those big whirring machines I’ve leaned into for years, are non-negotiable. Even after twenty minutes in a good hood, I get why people push for them in new labs: breathing toxic fumes on the job sets you up for a lifetime of problems.
Chemical Storage and Labeling Still Get Ignored
Too many storage rooms become disasters in a week—bottles toppling in dark cabinets, barely legible labels, no separation between volatile chemicals and acids. Nitroacenaphthene needs its own sealed container, kept far from oxidizers and strong acids. I nearly tossed a nitro compound on the same shelf as bleach once. It only takes one mix-up to set off a chain reaction you'd never outrun. Label everything, date it, and add hazard symbols big enough for anyone in a rush to notice.
What to Do When Things Go Wrong
Spills mean chaos. The time to search for spill kits isn't after things tip over. In my first year on the job, I watched three senior staff freeze while a bottle of chemicals poured out from a shelf. Absorbent pads, proper neutralizers, gloves, and splash goggles need to be within grabbing distance. The worst mistake is underestimating small spills—no shorts or sandals, either.
Training Never Stops
Any team member—rookie or veteran—has to revisit the hazards of Nitroacenaphthene. Those safety data sheets some folks ignore have real value. I make sure to run drills and refreshers every six months, not because the boss asks, but because muscle-memory in tense moments determines who stays safe.
Solutions Rely on Teamwork and Clear Communication
One careless person can break everyone’s safety. I lean on my team to double-check setups, flag damaged PPE, and question uncertain behaviors. That simple habit of calling out unsafe moves, or pausing the group when procedures slip, turns “close calls” into tales instead of headlines.
Smart Handling Saves Lives
Working with a tricky chemical like Nitroacenaphthene always means thinking ahead, sticking to strong habits, and respecting its dangers. Most accidents don’t come from freak events—they creep in through shortcuts. The must-haves—proper gear, ventilation, dedicated storage, spill prep, and refresher training—stand between every worker and the worst-case scenario.
What is the chemical formula of Nitroacenaphthene?
Understanding the Formula of Nitroacenaphthene
Nitroacenaphthene carries the chemical formula C12H7NO2. The structure emerges from the acenaphthene backbone, a polycyclic aromatic hydrocarbon, adorned with a nitro group (–NO2) replacing one hydrogen atom. That small swap changes the substance in a big way. In my college days, a chem professor once said, "Switch out just a single group, and you’ve got a different wild animal entirely." That’s definitely the case here.
Acenaphthene itself looks like two benzene rings fused together, with a cyclopentene bridge. Add a nitro group, and you see obvious shifts in chemical reactivity. Now you’re not dealing with another inert hydrocarbon—suddenly you’re looking at new potential for use in synthesis and analysis. C12H7NO2 stands apart in that it brings oxidative abilities, higher electron-withdrawing capacity, and a host of safety implications. That nitro group isn’t just for show.
Relevance in Industry and Research
The growth of synthetic dyes and specialized polymers often relies on nitroarenes like nitroacenaphthene. This compound has featured in academic papers focused on the environmental breakdown of aromatic pollutants. The stability of its polycyclic structure tends to resist rapid degradation. For researchers digging into how aromatic nitro compounds interact with soils or living organisms, nitroacenaphthene turns into a key reference point. Some studies draw attention to its persistence, making it a talking point in environmental chemistry courses.
I’ve worked in a lab where aromatic nitro compounds had to be handled with care. Even a brief contact with skin required washing up right away. The risks aren’t just about toxicity—these compounds can also change over time under heat or light, forming more hazardous byproducts. This has lent urgency to developing safe handling protocols, particularly for students and lab techs early in their careers.
Addressing Environmental and Health Concerns
Nitro aromatics such as nitroacenaphthene have raised red flags in environmental safety circles. Data gathered by the EPA and similar bodies points toward the persistence and possible risks tied to bioaccumulation. While direct consumer contact remains rare, improper disposal can have sweeping effects on soil and water health. Policies push hard for secure waste treatment, often at an industrial level. The introduction of advanced remediation techniques, like photocatalysis and bioremediation, helps minimize these concerns. Researchers have demonstrated bacteria strains that break down nitro groups—an exciting prospect for cleaning up legacy contamination.
Toward Safer Synthesis and Use
Scientists and industry professionals have begun exploring greener synthetic routes for nitroacenaphthene and other complex nitro compounds. Catalytic systems using less hazardous reagents—sometimes even moving away from classic nitration with fuming acids—point the way forward. There’s also an ongoing push to monitor workplace exposure. In my own lab experience, routine air sampling and closed systems went a long way to reduce risks. No one wants to even approach chronic exposure levels found in some industrial accidents from decades past.
Each discovery tied to compounds like nitroacenaphthene pushes chemistry forward, but without careful attention, that progress can bring fresh problems. It comes down to knowing your molecule as well as your mission in the lab—something old pros always make sure to pass down to the next generation.
How should Nitroacenaphthene be stored?
Why It Matters
Anyone who’s handled chemicals knows every bottle comes with a different set of rules, and Nitroacenaphthene doesn’t cut any corners in that department. Growing up in a family where a shed full of solvents and fuel could double as a science experiment, I learned early that shortcuts can turn minor mistakes into real problems. You can’t just toss Nitroacenaphthene onto a dark shelf and forget about it.
Risks You Can’t Ignore
Nitroacenaphthene catches fire more easily than most people expect. Researchers and industrial workers have documented its volatile nature—this stuff does not cooperate with sloppy storage. The Centers for Disease Control flag Nitroacenaphthene due to its ability to give off hazardous vapors, cause chemical burns, and ignite under the right (or wrong) conditions. We’ve seen what unexpected chemical reactions can do, from minor lab incidents to tragic warehouse fires.
Storing volatile chemicals without real respect for their fire risk or how their vapors can build up indoors gives trouble an open invitation. Personal protective equipment means nothing if the risk is baked right into the setup of your workspace.
How to Store It Right
The first thing I learned handling hotheaded chemicals: Keep them in a cool, dry place, away from direct sunlight or heat sources. Nitroacenaphthene proves this point. It deserves its own spot, away from oxidizers and acids, far from any heat or spark. Fire-retardant cabinets built specifically for flammable chemicals offer solid peace of mind—locking doors and steel construction stop disaster from spreading.
A simple cardboard box won’t cut it. Secondary containment—like tubs that catch leaks—can keep spills local rather than letting them make their way across a floor. It’s the difference between a minor cleanup and an emergency call to hazmat. I’ve watched a drip pad do its job, and watched someone pay for skipping this step.
Label Everything, Every Time
Too many people trust their memories or scribble shorthand on a bottle label. Every container should say what’s inside, the hazard level, and the date it went into storage. If you walk into a room and see “N.A.N.” written in faded marker, that’s a recipe for confusion or worse. Clear, honest, straightforward labeling gives everyone in that space the information they need, including emergency crews.
Ventilation Isn’t Optional
No matter how careful you are, vapors sneak out. Chemical storage rooms need good air flow—ventilation hoods and fans help sweep out invisible dangers before they build up. Regular checks for smell or unusual air quality help stop a bad situation before it grows legs. A cramped, stale room is just asking for a health incident.
Training and Access Control
Rules mean nothing if the people in your space haven’t heard them. Anyone who gets close to Nitroacenaphthene requires proper training. I remember classmates who thought safety training was a hurdle, not a tool. They’re the reason you lock these chemicals away—restricted access keeps thoughtless moments from turning into accidents.
Fixing What’s Broken
If you spot cracked containers, leaking lids, or out-of-date stock, solve it right away. Broken storage is not a tomorrow problem. Old containers that look swollen or faded should get moved to disposal by a qualified team. Accidents happen when folks assume the last person in the storeroom actually checked everything.
Keeping Nitroacenaphthene stored safely isn’t about gold-plated procedures or new gear each year—it’s about sticking to the basics, tuning into reality, and not letting anyone slide on the small stuff.
Is Nitroacenaphthene hazardous to health or the environment?
Looking Under the Hood
It’s not hard to understand why chemical names like nitroacenaphthene cause concern. Industry uses it to make dyes and pigments; it gets released during fossil fuel burning. The everyday person doesn’t buy a bottle of nitroacenaphthene off the shelf, but it still manages to turn up in the places people work and the air they breathe.
What the Science Says
Research flags nitroacenaphthene as a suspected carcinogen. Animal tests point to increased tumor risks after exposure. No one likes to play lab rat, so those results lead health agencies to label this substance as hazardous. Inhaling fine particles or touching contaminated surfaces raises short-term risks. Dizziness, headaches, and skin irritation rank as common complaints. Long-term exposure relates to potential harm in the liver and kidneys, not to mention the possibility of cancer.
The problem runs deeper in workplaces where safety rules sometimes slip. A story comes to mind from my days working in an industrial clean-up crew outside Pittsburgh. I watched crews mix pigments in poorly ventilated spaces. People trusted worn-out respirators, or skipped gloves in the summer when hands sweated under rubber. Supervisors knew the guidelines. But rushed schedules and budget cuts led to “just this once” moments. Folks wouldn’t feel sick right away, but over the years, the toll showed in clinic visits and leave records. These experiences turned the invisible threat of a chemical into a routine worry.
Ripple Effects on the Environment
Nitroacenaphthene leaves a mark outdoors, too. It doesn’t break down quickly, sticking around in soil or water. Fish and small critters take it up. Scientists have measured concentrations in rivers downstream from old factories. Studies showed lower reproductive rates and genetic changes in exposed animal populations. Nobody needed years of scientific training to see how run-off ruined fishing spots once abundant with wildlife. Local news covered lakes closed to recreation, which hit small tourism businesses hard.
The issue tracks back to waste controls and enforcement. My own cousins grew up near a coal-fired power plant, where rain runoff carried residue into neighborhood streams. Parents warned kids to stay away from certain banks, even though city officials repeated that everything “met allowable limits.” Years later, EPA testing confirmed higher cancer risks in the area. People didn’t wait for government action; they demanded stricter spill monitoring and funded community soil studies using grant money.
Ways Forward
Improving worker safety comes down to more than guidelines printed in a manual. Respirator fit checks, regular medical screenings, and real whistleblower protection slow down accidents. Wastewater treatment plants need stronger filtration setups, not temporary fixes for compliance. Local groups play a role, too. Community science and grassroots reporting reveal new pollution and push governments to act.
Safer chemical alternatives attract research dollars. Some labs seek out substitutes to nitroacenaphthene in pigments and paints. The price tag sometimes delays adoption, but persistent demand from end users—companies making toys, packaging, and food containers—forces suppliers to adjust. Clear product labeling could help families make better buying choices.
Building public awareness starts with better news coverage and access to plain-language information. No parent wants a lesson on chemistry jargon. They want facts and clear options, especially if they live close to industrial sites or busy highways.
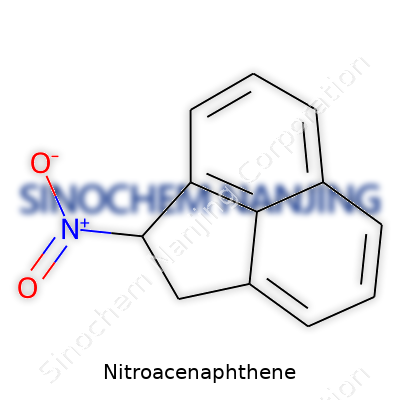
| Names | |
| Preferred IUPAC name | 2-nitroacenaphthene |
| Other names |
Acenaphthene, nitro- 1-Nitroacenaphthene |
| Pronunciation | /ˌnaɪtroʊˌæsəˈnæfθiːn/ |
| Identifiers | |
| CAS Number | 602-60-8 |
| Beilstein Reference | 110680 |
| ChEBI | CHEBI:34480 |
| ChEMBL | CHEMBL128055 |
| ChemSpider | 16469 |
| DrugBank | DB08799 |
| ECHA InfoCard | 100.017.682 |
| EC Number | 208-814-5 |
| Gmelin Reference | 6134 |
| KEGG | C14653 |
| MeSH | D009617 |
| PubChem CID | 68524 |
| RTECS number | HN9625000 |
| UNII | JK49PA0X9E |
| UN number | UN1660 |
| CompTox Dashboard (EPA) | DTXSID5022339 |
| Properties | |
| Chemical formula | C12H9NO2 |
| Molar mass | 245.24 g/mol |
| Appearance | Yellow needles. |
| Odor | Odorless |
| Density | 1.25 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.98 |
| Vapor pressure | 1.5E-4 mmHg |
| Acidity (pKa) | 6.57 |
| Basicity (pKb) | 9.50 |
| Magnetic susceptibility (χ) | -75.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.6750 |
| Viscosity | 1.55 cP (25°C) |
| Dipole moment | 3.76 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 347.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 51.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5485 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | D05BB22 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P270, P271, P280, P301+P312, P304+P340, P312, P330, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-2 |
| Flash point | Flash point: 182°C |
| Autoignition temperature | 530°C |
| Lethal dose or concentration | LD50 (oral, rat): 800 mg/kg |
| LD50 (median dose) | 1100 mg/kg |
| NIOSH | WA8575000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Nitroacenaphthene: "0.3 mg/m3 |
| REL (Recommended) | 0.2 mg/m3 |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Nitroacridine Nitrofluorene Nitronaphthalene |

