Nickel Nitrite: A Detailed Perspective
Historical Development
Nickel nitrite didn't emerge overnight. Early chemistry books don't speak much of it because the nitrite salts of transition metals often take a backseat to their better-known nitrate siblings. Most nitrites were first prepared in the 19th century as part of broader research into nitrous acid derivatives. Chemists like Liebig and the next wave of European experimenters helped lay the foundation for this group of compounds, investigating their relationship to agriculture, dyes, and analytical chemistry. Nickel nitrite, though, got added to the list relatively quietly. It wasn’t the star of the show like nickel sulfate in plating baths or nickel carbonate in ceramics, but it slowly found more relevance in academic circles, especially as analytical techniques grew sophisticated through the twentieth century. Researchers saw value in studying its decomposition, coordination chemistry, and impact on transition metal catalysis.
Product Overview
In the lab, nickel nitrite shows up as a green or yellow compound, depending on factors like hydration and purity. It’s not something you see in every storeroom, but analytical chemistry circles keep it around for testing, especially for nitrite determination and as a reagent in certain complexometric titrations. Even though bulk production is limited, specialized chemical suppliers stock nickel nitrite for researchers who need its particular properties. It’s known under names such as Nickel(II) nitrite and Nickelic nitrite, and sometimes pops up in catalogs focused on niche applications.
Physical & Chemical Properties
Nickel nitrite typically crystalizes in greenish to yellowish hues, signifying the paramagnetic nature of divalent nickel paired with the reactive nitrite ion. Its solubility in water helps researchers, though the solution takes on a yellow color that hints at the presence of both the metal ion and the nitrite’s own chromophore. The compound decomposes with heat, producing nitrogen oxides, a detail any experimenter working at scale needs to respect. The nitrite ion already shows sensitivity to light and air, which means samples stored outside of tightly sealed containers degrade quickly. Chemically, nickel nitrite sits in the territory where transition metals interact with ligands in complex, sometimes unpredictable, ways, especially under varying pH or redox conditions.
Technical Specifications & Labeling
Producers assign product numbers and safety designators to samples of nickel nitrite to keep up with regional chemical regulations. Labels outline molecular weight (generally around 148 g/mol for the anhydrous salt), recommended storage procedures (cool, dry, oxygen-free containers), and pictograms warning about toxicity and oxidizing potential. Purity levels matter in sensitive applications, and suppliers usually guarantee at least 97% for research-grade material, though analytical grade goes higher. Labels call out batch information and indicate if the material meets REACH, OSHA, or GHS hazard classification. In my hands, a reliable supplier means fewer headaches when designing analytical runs or material science tests.
Preparation Method
Lab-scale preparation of nickel nitrite doesn’t require exotic techniques. The reaction of nickel(II) nitrate and sodium nitrite in aqueous solution under controlled temperatures yields the salt after careful removal of water and avoidance of acidic byproducts. Some approaches use metathesis reactions, where nickel(II) sulfate swaps partners with barium or calcium nitrite, leaving behind relatively pure nickel nitrite that crystallizes as water evaporates. Doing this work in open air brings exposure to nitrogen oxides, so anyone running this prep keeps the fume hood sash down and ventilation on high. Over the years I’ve seen students underestimate the production of gas during these reactions—easy to do, but that's where safety training proves its value.
Chemical Reactions & Modifications
Chemists often use nickel nitrite as a starting material to explore coordination complexes or catalysis involving nitrite ligands. Heating causes it to decompose, giving off nitrogen dioxide, oxygen, and leaving behind nickel oxide. This process generates large volumes of gas even from a spoonful of material, so scaling up carries risks. Solution chemistry offers another window: nickel nitrite can react with ligands like ammonia or ethylenediamine to create various colored complexes. These transformations matter to researchers in coordination chemistry, helping them understand how metal ions interact with different binding partners. Modifying nickel nitrite’s properties often starts with adding new ligands to generate compounds with different solubility, reactivity, or spectroscopic fingerprints.
Synonyms & Product Names
Nickel nitrite goes by several names in the trade and in chemical literature. Nickel dinitrite, Nickelous nitrite, and Nickel(II) nitrite all mean the same greenish-yellow powder. Some suppliers stick with IUPAC rules and list it simply as Nickel(II) nitrite, while others use catalog designations like Ni(NO2)2 for clarity. Researchers sometimes refer to it in shorthand as “nickel nitrite,” especially in paperwork or lab notes. It shows up in safety databases and regulatory reports in all of these forms, adding an extra step to research if someone isn’t careful in matching synonyms.
Safety & Operational Standards
Handling nickel nitrite safely calls for a good understanding of both nickel and nitrite toxicology. Both ions spell trouble when mishandled. Nickel ions cause allergic reactions on skin and, with enough exposure, long-term harm to lungs and kidneys. Nitrites act as strong oxidizers and can form nitrogen oxides during decomposition, which means toxic fumes lurk in the background if accidents occur. Labs stock nitrile gloves and face shields; experienced users keep spill kits ready and treat even small quantities with plenty of respect. Down the years, best practices now include not eating or storing any food in prep areas, regular air monitoring, and specific waste collection protocols. Disposal involves neutralizing solutions to harmless nitrate or nickel carbonate forms before sending them on to qualified waste streams. I’ve seen new researchers learn quickly not to cut corners on PPE—one exposure incident drives the point home far quicker than any written warning.
Application Area
Nickel nitrite rarely features in bulk manufacturing or consumer goods, but the research lab provides a canvas for its usefulness. Analytical chemists use it to develop procedures for nitrite quantification in water and food analysis. Materials scientists investigate how it forms complex ions, especially those relevant to surface chemistry and catalysis. Some researchers pursue its ability to form colored coordination compounds as teaching tools, where student hands mix solutions and learn about ligand behavior in real-time. In bench-scale inorganic synthesis, nickel nitrite gives a source of both transition metal and oxidizing power. Catalysis groups look to its action in promoting oxidation reactions, while others test its impact on electrode stability in electrochemical setups. While not as commercially important as nickel sulfate or chloride, it fills gaps in specific niches where both metal and nitrite anion are needed.
Research & Development
Research on nickel nitrite shows an appetite for understanding how it interacts in multi-metal systems and under rapid redox cycling. Electrochemists take advantage of its ability to undergo changes in oxidation state, mapping out reaction pathways that shed light on transition metal redox chemistry. Coordination chemists, meanwhile, keep pushing new synthetic methods that involve nickel nitrite, forming polynuclear complexes that have applications in electronic materials, catalysis, and small molecule activation. Academic groups chase innovation in how nickel nitrite activates small molecules such as hydrogen peroxide or organic substrates, exploring green chemistry angles and fine-tuning reaction efficiency. Investment into automation in laboratories boosts reproducibility in nickel nitrite–mediated synthesis, making it easier to document results that can be scaled later if commercial applications develop.
Toxicity Research
Both nickel and nitrite ions have lengthy profiles in toxicology databases. Experimental toxicologists study nickel nitrite to understand its impact on aquatic organisms, plant cells, and mammalian tissues. Nickel’s record as a sensitizer and suspected carcinogen puts it high on the list for occupational safety controls. Nitrite ions convert hemoglobin into methemoglobin, diminishing the blood’s ability to transport oxygen, which presents risks both in acute exposure and chronic low-level intake, especially for infants. Modern journals report on how trace amounts of nickel and nitrite in water streams influence local ecosystems, pushing calls for stricter discharge limits and monitoring. Recent findings highlight the need for better detection methods for both ions in the environment, using nickel nitrite as a test standard in calibration and modeling.
Future Prospects
Looking ahead, nickel nitrite will find its role shaped by advances in analytical instrumentation, environmental regulations, and new applications in catalysis or nanoscience. Researchers continue to explore sustainable synthesis and safer ways to handle and dispose of nitrite-based transition metal salts. Computational chemistry, which has transformed understanding of complex coordination chemistry, also shines a light on the unique electron structures in nickel nitrite–based systems. Interest in green chemistry encourages scientists to revisit old compounds with an eye for cleaner transformations—either by replacing more hazardous components or finding new, less polluting applications for nickel nitrite and its relatives. There’s every reason to believe its quiet importance will keep growing, both as a research reagent and as a benchmark for developing safer nitrite chemistry.
What is Nickel Nitrite used for?
Why Chemists and Manufacturers Care About Nickel Nitrite
Nickel nitrite often sits quietly in the background of chemical supply rooms, but this greenish compound grabs attention once you start dealing with specialty reactions and tough industrial tasks. Its true value shines through for those who work with organic synthesis and catalysis. As someone who’s spent time in both research and manufacturing labs, I've seen how specific chemicals become problem-solvers you can't do without—nickel nitrite is one of those.
For anyone wondering if this is another obscure substance, it’s not. Its real-world presence sneaks into more spaces than you might think, from dye manufacturing to forming nickel-based pigments. Chemists turn to it for controlled nitrate introduction in organic reactions. In the right environment, it generates nitric oxide in situ—a strategy that lets teams streamline their processes and minimize waste.
Nickel Compounds and Science’s Tinkering
Manufacturing sectors lean on nickel nitrite for producing specialized glass and enamels. The colors and finishes it can help create give artists and industrial designers more control over how a finished product looks, which means fewer rejects and less wasted material. Additive manufacturing sometimes dabbles with compounds like nickel nitrite to deliver tighter tolerances for certain metal parts, especially those exposed to heat or heavy wear.
The compound also plays a part in surface treatment. Creating corrosion-resistant coatings for machinery isn’t just about longevity—it’s about workplace safety and reducing the frequency of breakdowns. Factories that operate in climates where equipment faces rough conditions can't afford regular replacements or downtime. By supporting nickel plating processes, nickel nitrite quietly saves time, money, and headaches.
Nickel Nitrite and Environmental Caution
Handling nickel nitrite requires a steady hand and strict safety gear. It’s not something you sprinkle around without care. This compound can introduce significant dangers—both as a toxin and a possible carcinogen. Health and safety teams in research institutions and factories establish training sessions dedicated to proper storage, glove selection, and emergency response. Government agencies, like OSHA in the US and REACH in Europe, keep close tabs on its usage, making sure exposure levels stay within strict boundaries.
Disposal stands out as a major sticking point. Nickel nitrite waste doesn’t just vanish safely down a drain. It demands specialized disposal streams to avoid groundwater contamination and soil degradation. Most labs and factories work with certified waste handlers who know how to neutralize or contain residues according to local and international law. Strong protocols aren't just about regulatory fines—they help protect everyone from long-term health risks and environmental fallout.
Building a Safer, More Efficient Future
Despite the risks, nickel nitrite sticks around thanks to its key scientific contributions and industrial payoffs. Future solutions should focus on greener alternatives for the processes where it’s currently essential. Universities and start-ups are already pushing for new catalysts, often based on less toxic metals or organic structures. Factories can upgrade ventilation and automate powder handling to remove humans from danger zones. Real progress will hinge on partnerships between chemists, engineers, safety regulators, and environmental groups. Working together can phase out the roughest chemicals, curb waste, and push technology forward without holding on to outdated hazards.
What is the chemical formula of Nickel Nitrite?
Chemical Formula and Structure
Nickel nitrite combines nickel and the nitrite ion. Its chemical formula is Ni(NO2)2. This formula means each molecule has one nickel atom and two nitrite groups. Unlike more familiar compounds like nickel chloride or nickel sulfate, nickel nitrite isn’t something most people run into daily. Still, the unique combination sets it apart in both chemistry labs and industry.
The nitrite ion, NO2-, brings in its own level of reactivity. It can transform into various compounds in the environment. Throw in nickel, a transition metal, and you get a pairing with real chemistry behind it. Understanding this formula opens the door to a mix of uses and risks, depending on where and how it’s handled.
Nickel Nitrite in Real Life
Nickel nitrite isn’t a mainstay in homes or schools. Still, you’ll find compounds like this in research labs and specialty manufacturing. Manufacturing industries may use nickel nitrite when they want to take advantage of the specific behavior that the nitrite ion offers. It reacts quickly and can act as an oxidizing agent. For researchers, the complexity of nitrite partnering with a metal such as nickel means there’s potential for interesting chemistry—catalysis, synthesis, and sometimes as an intermediate step in other processes.
Its rarity stems in part from safety concerns. Both the nitrite and the nickel carry health risks if handled the wrong way. Nickel can cause allergic reactions and has been classified as possibly carcinogenic. Nitrite ions have their own toxicity profile, sometimes causing blood problems if exposure is high.
Safety and Environmental Factors
Working in any lab, nothing beats the memory of safety briefings on handling nickel-based compounds. I’ve seen a single mishap cause skin rashes. Mixing with nitrite, the stakes go up. Researchers and workers stick to gloves, lab coats, and proper ventilation. Labs that care about both human health and the local sewer system will make sure runoff or spills never reach the outside world.
Nickel nitrite’s environmental impact can’t be ignored. Nickel can persist in soil and water, disrupting plant life and aquatic animals. Nitrite brings a risk of turning groundwater into something unsafe for drinking, especially near industrial sites that don’t keep a close watch on waste.
Addressing the Challenges
Nickel and nitrite both have regulations tied to them. Countries keep limits on how much nickel can end up in drinking water and soil. Companies handling nickel nitrite face growing pressure to switch to closed-loop systems, recycling as much as possible, and finding less hazardous substitutes where chemistry allows.
Teaching safe handling starts from day one in science courses: always label, always store securely, don’t cut corners with cleaning. In my experience, the difference between a safe workplace and a risky one often boils down to who’s willing to take these steps seriously every day.
Where to Go Next
Nickel nitrite isn’t about to disappear, but there’s momentum towards using less risky alternatives, especially in academic settings. Industry trends focus on minimizing waste, updating standards when new science shows something safer, and keeping a closer eye on long-term environmental impact. For anyone who works with or around specialty chemicals, knowing the formula is just the start. The real test comes in what choices get made every day to keep people and the planet safer.
Is Nickel Nitrite hazardous or toxic?
Digging into What Nickel Nitrite Means for Health
Nickel nitrite is not a chemical you see every day outside of specific industrial settings. It rarely makes headlines, but it deserves a closer look because the risks stack up in laboratories and workplaces. Most people have heard about nickel allergies from jewelry or buttons. Combine that metal with nitrites, which bring their own health warnings, and you start to see why extra caution is essential.
Understanding the Dangers
Handle nickel nitrite carelessly, and you’re inviting trouble. Nickel nitrite can irritate skin and eyes—it only takes a brief moment for burning or redness to show up. Longer exposure can push things into more serious territory. Workers in chemical plants know that inhaling nickel dust or fumes does more than tickle the throat: chronic exposure raises lung cancer risks because both nickel ions and nitrite compounds play rough with DNA over time. Evidence from the International Agency for Research on Cancer points directly to nickel compounds as known human carcinogens.
The questions don’t stop at cancer. Nickel nitrite sparks allergic reactions for many, bringing out eczema and other skin rashes. For folks with asthma or chemical sensitivities, even a sniff of nickel-laced air can mean breathing troubles or agitated lungs.
Other Hazards—Not Just to People
Chemicals that harm people can trash the environment as well. If nickel nitrite lands in rivers or soil, it can poison aquatic life and stress out entire ecosystems. Small organisms show malformations or die off faster, and as the chemical drifts through the food chain, problems climb. Regulators in the US and Europe have flagged nickel, nitrite, and many of their cousins for special containment. This isn’t just paranoia. Real-world spills have killed fish, wiped out bugs, and triggered long cleanup campaigns.
Room for Caution and Better Habits
Handling chemicals always brought a sense of caution into my early lab days. Gloves, eye protection, and solid ventilation cover the basics, but so much depends on habits. If a beaker tips or powder scatters, even seasoned scientists can get hurt. Safety data sheets for nickel nitrite practically shout about immediate clean-up, double-bagging of waste, and strict storage rules away from acids and organics—too many fires and explosions start with poor housekeeping. For anyone around this compound, showing up unprepared leaves no margin for error.
Building Solutions that Work
The real fixes land upstream. Substituting safer chemicals is the gold standard. Industry already looks for nickel-free options or better storage systems for reactive compounds. Regular health surveillance for workers can catch trouble early, and training programs teach folks how to handle incidents before small leaks grow into major exposures. At a policy level, tighter regulations encourage companies to limit emissions and watch water quality downstream from their facilities.
Awareness solves half the problem. Most lab workers or factory staff know nickel nitrite isn’t harmless, yet mistakes still happen—fatigue, rushing, slip-ups. Cultivating a strong safety mindset takes leadership, practice, and a clear sense that everyone—not just managers—shares responsibility for a clean, safe workspace. Only then can the risks tied to nickel nitrite shrink to manageable size without stifling the benefits that responsible chemical use brings to modern science and technology.
How should Nickel Nitrite be stored?
Handling a Compound with Real Risks
Working with chemicals as unpredictable as nickel nitrite pushes you to care about details the average person might overlook. This isn’t table salt or baking soda. Nickel nitrite comes with some heavy baggage in terms of safety. During my early years in research labs, we treated every storage bottle of reactive compounds like a potential hazard—and nickel nitrite requires exactly that mindset.
A Cool, Dry Place Isn't Just a Suggestion
Nickel nitrite reacts badly to heat and humidity. Left in a warm, moist environment, it can decompose or even trigger a fire thanks to runaway oxidation. A few years ago, our lab learned the hard way. A simple error—leaving it near a radiator—meant the entire batch had to be disposed of before anyone got hurt. So, a temperature-controlled chemical cabinet, away from steam pipes and sunlight, became non-negotiable. I see many people forget how quickly temperature and moisture in an older building can shift, but for nickel nitrite, a few degrees make all the difference.
Keep It Sealed, Keep It Clean
Air and moisture creeping into storage containers spell trouble. Nickel nitrite doesn’t just dislike water; it breaks down. To keep it stable, use tightly sealed glass or HDPE containers. Sometimes, I watched junior lab mates eyeball an old plastic jar and think it’d be safe. Tight seals win every time. Cross-contamination needs watching, too. Dust from other reactive salts drifting in can cause serious, unpredictable reactions.
Segregate from Incompatible Substances
Lab experience taught me a painful lesson about storage compatibility. Pairing nickel nitrite near organic solvents, acids, or reducing agents kicks up the risk of explosions or toxic gas release. Keeping it on its own shelf, with clear labels and away from possible chemical antagonists, worked much better. Even accidental spills or careless stocking during a rush can lead to dangerous surprises.
Clear Labels and Records Save Lives
Every jar, every flask, needs a label. It sounds basic, but the day a substitute instructor almost tipped a sample onto a hot plate thinking it was something benign, I realized just how much clear communication matters. I learned to respect the professionals who insist on documenting every batch’s origin, condition, and age. Good record-keeping is the backbone of chemical safety: not a bureaucratic hurdle, but the reason I sleep better after locking up a hazardous stockroom.
Personal Protective Equipment: No Exceptions
Pulling nickel nitrite from storage isn’t a casual affair. Lab coat, gloves, and eye protection are the bare minimum. It stains everything it touches, so skin contact isn’t just a cleanup problem; it’s a health risk. Regular safety drills in my lab drilled this home, and I never saw anyone regret being too careful.
Why All the Fuss About Storage?
Neglecting safe storage for nickel nitrite doesn’t just invite regulatory headaches; it endangers every person in the vicinity. Nickel compounds often carry carcinogenic or allergenic risks, and nitrites release nitrogen oxides during breakdown. Local authorities aren’t just nitpicking when they demand rigorous standards—stories from poorly managed storerooms make news for the wrong reasons all the time.
Better Solutions Go Beyond Bare Minimums
Hands-on experience showed me that chemical storage isn’t something to just “tick off” a compliance list. Simple changes—like investing in well-maintained, explosion-proof cabinets and running regular training—raise the bar for everyone’s safety. Management support helps, but personal vigilance, double-checking labels, and keeping open lines of communication with peers and supervisors can prevent disasters. Storing nickel nitrite right isn’t just about protecting property; it keeps people healthy and workplaces running—two things everybody values.
What are the handling precautions for Nickel Nitrite?
Understanding the Risks of Nickel Nitrite
Nickel nitrite brings more than its share of hazards to the workplace. People who spend time around this compound know it stands out for its toxicity and reactivity. Many don’t realize how easily it can dust up or dissolve in water, raising the risk not only for inhalation and skin contact, but also for environmental spread. Repeated exposure, even to small amounts, can cause serious health issues like lung irritation, allergic skin reactions, and even cancer. Nickel in any form raises red flags among safety officers, but nitrites add another dimension, with fire and explosion hazards on the table too.
Proper Storage Makes a Difference
Safety starts with smart storage. Nickel nitrite reacts badly with acids, reducing agents, and other common chemicals. People who deal with chemicals often forget the risk of mixing incompatible substances, but the potential outcome—fire, release of toxic gases, or even an explosion—can turn an ordinary workday into a disaster. Store this compound in a cool, dry place, far from direct sunlight and away from oxidation-prone or flammable materials. Keep it in tightly sealed containers, labeled in clear language. Never store near food, drinks, or personal items. Small changes in layout or old habits can save lives and prevent bigger headaches later.
Personal Protection Isn’t Optional
Working with nickel nitrite is not a job for bare hands or open faces. Gloves, long sleeves, and well-fitted chemical goggles make a strong frontline defense. Many older facilities stopped at basic gloves or dust masks, but that approach invites trouble. Use gloves made from resistant materials—think nitrile or neoprene—and never gamble on thin latex. Full-face shields and barrier aprons cut the odds of splashes making contact. Good hygiene matters too; wash hands and arms after handling, don’t smoke or eat in workspaces, and keep contaminated clothes out of regular laundry cycles.
Air Quality and Ventilation Keep Everyone Safer
Breathing nickel nitrite dust or vapor can set off coughing fits or worse. Labs and workrooms need local exhaust ventilation, the kind that snatches fumes close to the source before anyone else gets a whiff. Open windows or basic fans never match up to a well-designed hood. Employees should look for telltale signs—yellow dust lining vents or a sharp, acrid odor creeping into corridors. If symptoms like headaches or irritation crop up, reassess airflow or upgrade your protection. Employers have a duty to monitor air quality using real-time detectors, not outdated guesswork.
Spill Control and Emergency Protocols
Accidents happen, but a botched cleanup can turn a minor spill into an emergency. Absorb loose nickel nitrite using mineral oil or inert absorbent, clean up with non-sparking tools, and double-bag waste in containers labeled for hazardous disposal. Never wash residue down the drain or toss it with regular trash. Teams should review spill drills every quarter, making sure emergency eyewash stations and showers work as intended. Local fire departments often have helpful guides tailored for chemical spills, and calling for advice beats improvisation every time.
Looking Out for Each Other
A strong safety culture gives everyone a stake in the process. Ongoing training, clear signage, and easy access to safety data sheets can trim down risks and build confidence. Employees who voice concerns about handling practices play an important role in keeping everyone out of harm’s way. Respect for rules and a habit of double-checking storage, personal protection, and equipment go a long way in ensuring nickel nitrite stays useful—and not dangerous.
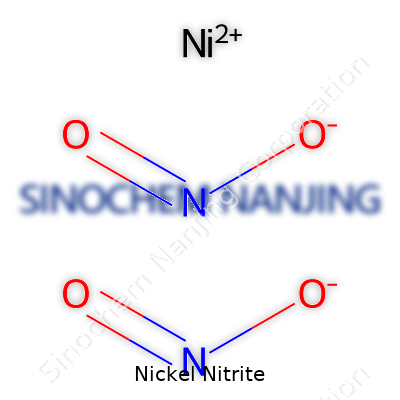
| Names | |
| Preferred IUPAC name | Nickel dinitrite |
| Other names |
Nickelous nitrite Nickel(II) nitrite |
| Pronunciation | /ˈnɪkəl ˈnaɪˌtraɪt/ |
| Identifiers | |
| CAS Number | 13462-90-3 |
| Beilstein Reference | 1202170 |
| ChEBI | CHEBI:86462 |
| ChEMBL | CHEMBL1201647 |
| ChemSpider | 21437 |
| DrugBank | DB11459 |
| ECHA InfoCard | ECHA InfoCard: 100.029.160 |
| EC Number | 236-764-8 |
| Gmelin Reference | Gmelin Reference: **1512** |
| KEGG | C01775 |
| MeSH | D009601 |
| PubChem CID | 24553 |
| RTECS number | QR9625000 |
| UNII | 3953OT5WXD |
| UN number | UN1456 |
| Properties | |
| Chemical formula | Ni(NO2)2 |
| Molar mass | 122.70 g/mol |
| Appearance | Yellow crystals |
| Odor | Odorless |
| Density | 3.41 g/cm³ |
| Solubility in water | slightly soluble |
| log P | -1.24 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 8.55 |
| Magnetic susceptibility (χ) | +368.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.89 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 107.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -151.2 kJ/mol |
| Pharmacology | |
| ATC code | V07AY08 |
| Hazards | |
| Main hazards | Oxidizer, toxic if swallowed, may cause allergic skin reaction, suspected of causing cancer, very toxic to aquatic life |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H301, H317, H334, H341, H350, H360D, H410 |
| Precautionary statements | P260, P261, P264, P270, P271, P273, P280, P284, P301+P310, P302+P352, P304+P340, P308+P313, P311, P314, P321, P330, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Lethal dose or concentration | LD50 (oral, rat): 175 mg/kg |
| LD50 (median dose) | LD50 (median dose) for Nickel Nitrite: Oral-rat LD50: 100 mg/kg |
| NIOSH | 0140 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Nickel Nitrite: "0.1 mg/m³ (as Ni, OSHA PEL) |
| REL (Recommended) | REL (Recommended): 0.1 mg(Ni)/m³ |
| Related compounds | |
| Related compounds |
Nickel nitrate Nickel(II) oxide Nickel(II) sulfate |

