Nickel Nitrate: Moving Through Science, Industry, and Innovation
Tracing the Historical Arc
Nickel nitrate has steadily woven itself into industrial and academic narratives over the past two centuries. Some of its earliest encounters came about as chemists explored nickel’s sulfate and carbonate cousins, learning just how diverse the metal’s chemistry could be. Eventually, methods improved, evolving from small-scale alchemy into large-scale nitric acid reactions that laid the path for bigger batch productions. By the end of the twentieth century, this compound found a home in laboratories and factories in a way early pioneers could only guess at. The story of nickel nitrate runs parallel to advances in chemical engineering, where increased demands for specialty catalysts and electroplating intermediates gave this vibrant green salt a permanent place in chemical catalogs.
Product Overview
Usually sold as either a crystalline hydrate or a concentrated solution, nickel nitrate delivers consistent results for both small labs and major manufacturers. Its unmistakable emerald-green color, easy solubility in water, and predictable reactivity let it slide into a number of roles. Markets often flag it for its purity, moisture content, and the absence of corrosive side-products, drawing a clear line between industrial and analytical quality. Whether it's dolled up in a glass bottle for the teaching bench or packed in drums for large-scale factory runs, you know you’re handling something with a long-standing reputation for reliability in chemical processes.
Physical and Chemical Properties
In its hexahydrate form (Ni(NO3)2·6H2O), nickel nitrate forms bluish-green crystals that don’t just look pretty—these are easy to handle, melt just above room temperature, and dissolve almost instantly in water. The aqueous solution itself has a straightforward ionic character. Its nitrate groups show strong oxidizing behavior, which chemists have used to their advantage for decades. The compound gives a characteristic green color in flame tests and gives off toxic nitrogen oxides if heated too much. With a molecular mass above 290 g/mol and distinct density values for solid and solution, its handling requires a practical approach rooted in the fundamentals of inorganic chemistry.
Technical Specifications and Labeling
Suppliers label nickel nitrate with technical clarity, marking out specifications like purity (usually above 98%), hydrate content, and levels of trace metals, since even a small contamination with elements like cobalt or copper causes issues in some applications. Safety data includes not just the CAS number (13478-00-7 for the hexahydrate), but dense hazard information pointed at researchers and production managers alike. Labels always warn about strong oxidizer properties, the need for gloves and goggles, and the requirement for careful storage away from organic materials or reducing agents. Quality specs often reference international standards, including REACH and GHS guidelines, aiming to keep everyone on the same safe page whether they’re in a university or an industrial plant.
Preparation Method
Manufacturers produce nickel nitrate using straightforward chemistry. Nickel metal, powder or oxide reacts with concentrated nitric acid, forming hydrated nickel nitrate and giving off nitrogen oxides. The resulting solution can be crystallized through careful evaporation, yielding the characteristic hexahydrate crystals at cooler temperatures. Scaling up involves closed systems and scrubbing towers to control NOx emissions, which aren't just environmentally harmful—they can jeopardize worker health. The chemistry itself has not changed much since the 1800s, but the scale, control, and focus on minimizing environmental footprints reflect today’s chemical industry priorities.
Chemical Reactions and Modifications
The reactivity of nickel nitrate often gets called upon in redox and coordination chemistry. In organic synthesis, it works as a source of Ni2+ for catalyst preparation and sometimes gets used to oxidize alcohols. When heated, the salt decomposes, giving nickel oxide, nitrogen dioxide, and oxygen, a strategy still used for producing high-purity nickel oxides in the lab. Applying reducing agents like hydrogen or hydrazine converts it back to metallic nickel, a process that shows up in powder metallurgy and electronics manufacturing. Chemists tinker with the nitrate ion, exchanging it for other anions to yield a variety of nickel salts, each suited to a different set of tasks.
Synonyms and Product Names
Nickel nitrate appears under a handful of aliases: nickelin, nickelous nitrate, and sometimes just plain “Ni nitrate.” International markets recognize it by the same chemical identifiers, but suppliers might use differing product codes depending on the hydrate level or purity. These names help buyers dodge confusion, especially among compounds with similar appearances but very different behaviors—something I learned the hard way years ago when a mislabeled bottle muddied up an entire experiment.
Safety and Operational Standards
Nickel nitrate’s oxidizing nature means safe storage and handling are non-negotiable. Facility protocols call for robust ventilation, separation from organic material and combustibles, and emergency procedures that account for chemical burns and inhalation exposure. Concentrated forms can burn skin, and accidental spills can quickly migrate through a damp lab. Larger installations invest in spill containment, regular staff drills, and automated monitoring for nitrogen oxide buildup in the air. Respirators, gloves, face protection, and full material safety data sheets must stay at arm’s reach wherever it is used. The border between research and production gets blurry, but the rules keep everyone out of trouble.
Application Area
Nickel nitrate finds itself at the center of several industrial processes. It plays a major part in catalyst preparation, especially for hydrogenation reactions and petrochemical plants. Plating shops rely on it to deposit nickel coatings on everything from machine parts to jewelry, where corrosion resistance matters. In ceramics, it takes on the role of a colorant, and in battery production, it contributes as a precursor to nickel-based cathode materials. Despite being simple in form, it winds up at multiple stopping points along the supply chains for electronics, glass, coatings, and specialty alloys. In my experience, its versatility means you can bump into it whether you work in a chemistry classroom, an R&D facility, or a full-scale manufacturing plant.
Research and Development
Recently, a surge in battery and catalysis research pushed nickel nitrate back under the microscope. Labs worldwide examine how to fine-tune its conversion into nickel-based nanomaterials for fuel cells and high-performance batteries. Complexes derived from nickel nitrate serve as starting points for a whole spectrum of coordination compounds, finding use in magnetism studies, material science, and even environmental remediation. Industry sponsors support projects that reduce its toxicity footprint, experiment with greener alternatives for nitric acid, and develop recyclable processes for its waste streams. As regulatory and economic pressures tighten, research groups look for new, less hazardous ways to synthesize the compound itself.
Toxicity Research
Nickel nitrate has a tricky reputation. Studies dating back to the 1970s show clear links between exposure and respiratory or skin issues, with occupational asthma and dermatitis rising among factory workers. It acts as a suspected carcinogen, with authorities like IARC putting it in Group 1 for its links to cancer in humans. Modern scientists dig deeper, tracing how nickel ions cross cell membranes and interact with DNA, and figuring out exposure risks even at low concentrations. Wastewater from nickel-based manufacturing needs special care, since nickel can persist in ecosystems and accumulate in aquatic life. Turning lab safety guidelines into daily habits—frequent skin checks, proper fume hoods, careful labeling—keeps danger at bay. Frankly, a healthy respect for the risks beats any short-term cost savings.
Future Prospects
Nickel nitrate still finds itself central to many of tomorrow’s technologies. As the transition to renewable energy ramps up, demand for efficient catalysts and battery components keeps growing. Researchers work on improving selectivity, durability, and recyclability by starting with well-characterized nickel nitrate precursors. Regulatory shifts in Europe and North America push for low-toxicity formulations and closed-loop recovery processes, so forward-looking companies invest in greener routes and waste minimization. From its humble crystal forms in glass jars to its strategic position in clean energy initiatives, nickel nitrate shows no sign of losing relevance, even as the need for safety and sustainability dominates the conversation around industrial chemicals.
What is Nickel Nitrate used for?
Nickel Nitrate in Everyday Products
Nickel nitrate hides behind the scenes in plenty of factories and workshops. It has a simple job on the surface—providing a source of nickel. After working in an industrial lab, I’ve seen first-hand why companies need it. This green, water-soluble salt plays a crucial role in producing coatings and alloys we see around us all the time.
Look at faucets, car bumpers, and bike frames with a shiny silvery finish. Those slick surfaces get their appearance from electroplating. In that process, manufacturers run an electric current through a bath where parts and nickel nitrate meet. As current flows, nickel leaves the solution and sticks to the product’s surface. The goal? Rust resistance, longer life, and a clean look. No matter how advanced production becomes, nickel nitrate remains a reliable choice for plating thanks to its ability to dissolve quickly and deliver nickel in a form that spreads evenly.
Catalysts and Chemistry
Nickel nitrate doesn’t just deliver good looks. Companies mining raw materials or making specialty chemicals often call on it as a precursor for catalysts. Catalysts speed up reactions without getting used up, and many contain nickel. When a business wants to turn raw oil into fuel or break down pollution, nickel nitrate often steps in as the start of the reaction chain. After a few steps, it helps form nickel oxide or nickel metal, both of which set the stage for these processes. The fuel you pump and the clean air you breathe can both trace their journeys back to this compound.
The pigment industry also values nickel nitrate. Mixed with other metals, it helps create vivid greens or subtle blues that color ceramics and glass. Glassmakers depend on nickel compounds to protect their glass from taking on unwanted hues. What surprises people is how a powder can melt into glass or glaze, bringing color and quality control in each finished piece.
Risks and Solutions
As valuable as nickel nitrate may be, it brings serious health risks. Most plants keep it locked away from regular workers. Its dust and mist can damage breathing and cause skin irritation. If it enters waterways, plants and fish struggle. I remember wearing heavy gloves and a mask while handling it—we didn’t take chances because of strict rules set by workplace safety agencies like OSHA and environmental authorities.
To limit harm, companies train staff, use sealed systems, and collect waste for proper treatment. Factories treat rinses and spills so nickel doesn’t reach the environment. While accidents happen, tough monitoring and emergency drills help keep people safe. The message from regulations is clear: treat nickel nitrate with respect or pay a heavy price.
Looking Ahead
Safer handling matters as nickel demand rises for batteries and electronics. Researchers have started developing greener ways to recover nickel and reuse it instead of relying only on fresh nickel nitrate. In my own work, I’ve seen more companies looking for new processes that recycle nickel from spent products. The more progress we make, the cleaner and safer our industries can become—without losing the benefits that nickel nitrate brings.
Is Nickel Nitrate hazardous or toxic?
Nickel Nitrate at Workplaces and in Daily Life
I’ve spent a good chunk of time around industrial areas, where you catch a whiff of something unfamiliar and wonder what it could do to your health. Nickel nitrate pops up wherever metal finishes, catalysts, or batteries get manufactured. Despite its bright green color, this stuff is far from harmless.
What Makes Nickel Nitrate a Concern?
This compound looks innocent enough if you’ve never heard of it — but it carries serious risks. Breathing in nickel nitrate at work or after a spill can lead to chronic breathing problems. Short exposure can cause skin rashes, itchiness, and sore eyes. During my visits to older plants, I heard more than a few stories about strange rashes that stumped doctors until someone checked for nickel. Nickels salts, such as nickel nitrate, have been flagged as a cause of allergic reactions or “nickel itch” in people working long hours without protective gear.
Science backs this up. According to the International Agency for Research on Cancer (IARC), nickel compounds, including nitrate, are classified as carcinogenic. This means that routine, unprotected contact isn’t just uncomfortable — it can set the stage for cancer, especially in the lungs or nasal passages. When you see a metallic green powder, you’re not just looking at a workplace hassle, you’re seeing a proven health threat.
Environmental Toxicity and Community Impact
Farms near industrial zones sometimes report crop damage or odd water tastes after a spill. Nickel nitrate dissolves easily, slipping into groundwater and moving quickly. Once in soil or streams, it doesn’t just go away. The Environmental Protection Agency (EPA) points out risks to water life, which can face reproductive problems and even die-offs from nickel contamination. Over time, villagers and farm workers get exposed, often without warning about the source of the problem.
Why Talking About It Matters
Too many places play catch-up once an accident happens. Nickel nitrate isn’t new in factories, but knowledge alone won’t protect families or employees. I’ve noticed that sites with strong safety leadership do better: they train workers on chemical spills, install proper ventilation, and make gloves and masks part of the daily routine. Skipping these steps leaves workers at risk, but smart operators set clear paths for chemical handling and disposal long before regulators announce a visit.
Better Choices and Safer Habits
Some companies have switched to less toxic alternatives where possible. Not every process needs nickel nitrate, and creative teams have found different compounds that finish the job without the same health drag. But swapping materials only solves part of the picture. Safety must run deeper than material choice. Well-labeled containers, regular medical checks, and spill drills make a real difference. Good ventilation and access to running water help, too. Even simple steps — like closing containers tightly or reporting small spills — can cut big risks.
Nickel nitrate isn’t the only hazardous chemical in industry, but its potential for harm feels close to home for folks living near factories or earning a paycheck on the production line. Awareness, solid training, and a little extra investment in safety can protect not just workers but whole neighborhoods for years to come.
What is the chemical formula of Nickel Nitrate?
Navigating Chemistry’s Simple Truths: Ni(NO3)2
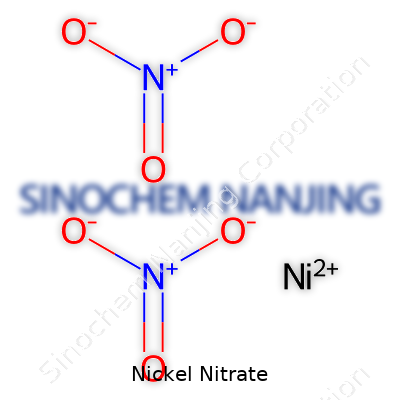
The chemical formula for nickel nitrate is Ni(NO3)2. That little group of numbers and letters hangs around in any proper chemistry textbook and pops up in unexpected corners of industry. I’ve written about chemical compounds before, but few have as much going on, both in safety and practical importance, as nickel nitrate. As someone digging into science stories, it’s easy to skip past formulas, but these small details shape entire industries—and they definitely change how we think about safety, sustainability, and health.
Nickel, Nitrate, and their Chemistry
Nickel sits in the periodic table with atomic number 28. Its ions often show up in green solutions, easily reacting with other compounds. Nitrate—NO3—carries a negative charge and has a habit of making many metals more soluble. So, Ni(NO3)2 tells you directly about the bonds forming in solution: two nitrate ions balance one nickel ion. This formula speaks to the entire identity of the compound—how it dissolves in water, why it stains glassware green, how it can help or harm depending on the job at hand.
Why It Matters on the Shop Floor and in the Lab
Nickel nitrate lands on shelves where batteries are assembled, pops up in metal plating shops, and sometimes in places you wouldn’t expect, like as a catalyst in big chemical plants. Plating experts reach for it when they want a smooth, nickel-rich coating. Chemists like it because it dissolves easily, giving a controlled supply of nickel ions for reactions. It moves through global supply chains and eventually ends up in all sorts of gadgets and tools.
Safety stories unfold around compounds like this. Even though Ni(NO3)2 looks harmless on paper, anyone breathing its dust or splashing it on skin is taking a risk. Chronic exposure without protective gear isn’t just risky—it’s dangerous, putting workers at higher odds for skin conditions and even cancer. I’ve seen regulations ramp up in real-time over the last decade, with workplaces trading old habits for gloves, respirators, and new training sessions. Regular audits matter more than ever.
Environmental Responsibility and Long-Term Challenges
This formula might look simple, but its breakdown in the environment carries weight. Spilled nickel nitrate contributes nickel ions to waterways, making it toxic to some plants and aquatic life. Nitrate, another part of this duo, pushes up the risk of harmful algal blooms. People sometimes trust wastewater treatment to fix everything, but these treatment plants can’t always grab every ion out of solution. The real fix comes from reducing waste at the source, recycling wherever possible, and investing in cleaner technologies. Some companies now reclaim spent nickel from plating baths, turning a pollution risk into a resource.
What We Can Do with This Knowledge
Reading Ni(NO3)2 isn’t just for exams. Knowing what it is, where it goes, and how it behaves makes a difference in keeping workplaces safer and water cleaner. Strong policies back this up, but personal responsibility in handling and disposal cuts risks even further. I’ve learned not to take any compound for granted—every time someone pays attention to the formula and its story, things improve a little. That’s the real value hiding in those letters and numbers.
How should Nickel Nitrate be stored?
Understanding the Stakes
Nickel nitrate doesn’t sound all that intimidating until you look up the safety data sheet. As someone who’s spent time in both research labs and maintenance supply closets, nickel nitrate always demanded respect. Breathing in its dust or letting the liquid touch your skin can lead to all sorts of health issues. It’s not something you want leaking into your shop drain or drifting around in the air.
Safe Storage Means Fewer Headaches
If nickel nitrate ends up in the wrong place, it can wreak havoc. Think respiratory problems or skin rashes, or worse, triggering bigger risks for people who already deal with asthma. From what I've seen, storing industrial chemicals improperly leads to accidents more often than carelessness in their use.
Anyone working with it must have a designated space, away from everyday foot traffic. It needs a sturdy container with a tight-fitting lid. Once, in a lab I worked in, an old glass bottle broke simply because it sat too close to the edge of a shelf. After that mess, we switched to thick polyethylene containers, and the worry dropped significantly. Those containers resist corrosion from the nitrate, and there's less chance of them fracturing if someone bumps into a shelf.
Humidity and Fire Risks: Not Just Small Details
Nickel nitrate loves moisture, meaning humidity can make it clump or cause it to seep through cracks, and it's also an oxidizer. Mixing that risk with oily rags or things like sawdust can invite a nasty fire. Fire codes in many areas require that chemical oxidizers like nickel nitrate shouldn't be cozied up next to flammable materials. Sitting nickel nitrate beside solvents or fuel is asking for trouble, and sometimes you just see it jammed in with everything else out of habit, not malice.
Air conditioning or a simple dehumidifier in a storage area goes a long way. I’ve watched a dehumidifier in a maintenance room, working overtime in a Florida summer, and the nickel nitrate stayed powdery and stable.
Clear Labeling and Training—Not Just Red Tape
Anyone using nickel nitrate needs straightforward labels in clear, bold fonts. During a busy shift, it's all too easy to snatch the wrong container or miss a "danger" warning in faded ink. I once saw an intern pour an oxidizer into a cleaning solution, thinking it was a harmless salt—luckily the fume hood protected him, but things could have spiraled.
Ongoing training cuts down mistakes. Real-world drills, rather than just passing out safety sheets, drive home the stakes. People remember scenarios they've acted out, not just the fine print.
Emergency Prep: A Must, Not an Option
Even the most careful setup can go sideways. I’ve always pushed for keeping spill kits, gloves, and goggles nearby. Quick access to running water helps, too. Clear evacuation plans show everyone what to do, instead of scrambling for answers as panic sets in.
Keep It Simple, Keep It Safe
Proper nickel nitrate storage takes some basic work—good containers, dry rooms, clear labels, and hands-on training. These steps don’t just meet regulations—they keep people healthy and workplaces running. I’ve seen the mess from letting standards slide, and I wouldn’t risk it again.
What are the safety precautions when handling Nickel Nitrate?
A Personal Look at Nickel Nitrate Hazards
Nickel nitrate earns special attention in any chemistry lab or industrial setting. I remember my own first direct experience with this compound—the sharp green crystals, the caution from a senior colleague, and the real worry I felt about getting even a little on my skin. This isn’t just unnecessary anxiety. Nickel nitrate can trigger asthma-like symptoms, skin rashes, and eye irritation. More troubling, there’s a strong link between nickel compounds and cancer after long-term exposure. Anyone working with this chemical holds responsibility for not just their own health, but everyone around them.
Personal Protective Equipment Matters
The first and most obvious step is the gear. I’ve seen people skip the nitrile gloves because they “just need to pour a little.” That kind of shortcut courts disaster. Nitrile or neoprene gloves stand up better to nickel nitrate than the regular latex ones. Safety goggles are a non-negotiable, as this chemical stings badly in the eyes. In places where dust or mist might form, a well-fitted respirator cuts down the odds of inhaling toxic particles. Don’t forget the lab coat or chemical apron. Splashes happen quicker than you think.
Controls and Good Habits
Using nickel nitrate calls for more than good intentions. Work should happen in a chemical fume hood. I’ve seen too many people run tests on the open bench out of convenience, ignoring the slow buildup of toxic fumes. A fume hood with solid airflow keeps exposure down and stops vapors from drifting. In my own lab, no one eats, drinks, or touches their phone until gloves come off and hands get washed. It might sound strict, but accidental ingestion or transfer to your face becomes real after long hours.
Safe Storage Counts
How nickel nitrate gets stored can be the difference between an ordinary day and an emergency. The chemical likes moisture and will pull water from the air, so it belongs in a tightly sealed, corrosion-resistant container. Store it away from anything flammable, since it acts as a strong oxidizer. Some metal shelves will corrode or discolor if spills go unnoticed, so chemical-grade shelving pays off in the long run. Always label the storage area in plain language; I once had a colleague who mistook nickel nitrate for a safer salt and nearly mixed the wrong solution. Clear labeling with danger symbols keeps things obvious to anyone walking by.
Preparedness for Spills and Exposure
Every lab I’ve worked in runs spill drills at least once per year. These skills stick with you. Clean-up involves using inert absorbents, shielding your skin, and never skipping the goggles. Once the chemical is swept up, careful neutralization and proper disposal keep it from ending up in the regular waste stream. After a spill, check for contamination on clothes or shoes. Trust me, you don’t want to track this outside the lab. If contact happens, rinse the skin right away with plenty of water and seek medical advice even if irritation seems mild.
Workplace Culture Shapes Safety
Getting safety right with nickel nitrate relies on more than rules on paper. The best outcomes come from habits built over time. It helps when leaders actually follow the same protocols and push for continuing education. New staff need hands-on training, not just a sheet of warnings. Reporting problems without blame, sharing updates about safety incidents, and keeping communication open make a huge difference. In my own circles, we swap stories about close calls so the details stick with everyone. That culture of looking out for one another beats any list of regulations.
| Names | |
| Preferred IUPAC name | Nickel(II) nitrate |
| Other names |
Nickel(II) nitrate Nickel dinitrate |
| Pronunciation | /ˈnɪk.əl ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 13138-45-9 |
| Beilstein Reference | 353702 |
| ChEBI | CHEBI:78041 |
| ChEMBL | CHEMBL1520841 |
| ChemSpider | 14118 |
| DrugBank | DB14567 |
| ECHA InfoCard | ECHA InfoCard: 034-009-00-5 |
| EC Number | 028-009-00-5 |
| Gmelin Reference | 66832 |
| KEGG | C01759 |
| MeSH | D009576 |
| PubChem CID | 10196 |
| RTECS number | QR9350000 |
| UNII | VI46X33980 |
| UN number | UN2725 |
| Properties | |
| Chemical formula | Ni(NO3)2 |
| Molar mass | 182.703 g/mol |
| Appearance | Green crystals |
| Odor | Odorless |
| Density | 2.05 g/cm³ |
| Solubility in water | Very soluble |
| log P | -0.52 |
| Acidity (pKa) | 3.6 |
| Basicity (pKb) | 6.7 |
| Magnetic susceptibility (χ) | +1520.0e-6 cm³/mol |
| Refractive index (nD) | 1.61 |
| Viscosity | 1.23 cP (25 °C) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 290.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -207.0 kJ/mol |
| Pharmacology | |
| ATC code | V07AV04 |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes skin and serious eye irritation, may cause allergic skin reaction, may cause cancer, may cause damage to organs through prolonged or repeated exposure, very toxic to aquatic life. |
| GHS labelling | GHS07, GHS09, GHS08, GHS05 |
| Pictograms | GHS05,GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H302, H317, H334, H350, H360D, H410 |
| Precautionary statements | P210, P220, P221, P264, P273, P280, P301+P312, P302+P352, P305+P351+P338, P306+P360, P308+P313, P332+P313, P337+P313, P362 |
| NFPA 704 (fire diamond) | 2-3-0-OX |
| Autoignition temperature | 400°C |
| Lethal dose or concentration | LD50 oral rat 175 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Nickel Nitrate: "175 mg/kg (rat, oral) |
| NIOSH | 0146 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Nickel Nitrate: "0.1 mg/m³ (as Ni, OSHA PEL, TWA) |
| REL (Recommended) | 0.03 mg/m³ |
| IDLH (Immediate danger) | 10 mg/m3 |
| Related compounds | |
| Related compounds |
Nickel(II) oxide Nickel(II) chloride Nickel(II) sulfate Nickel(II) carbonate |

