Neodymium Nitrate: A Closer Look at Its History, Chemistry, and Real-World Value
Historical Development
Curiosity drove rare earth research long before most modern uses found their place in tech and industry. Neodymium nitrate’s roots trace back to the late 19th century, just after Carl Auer von Welsbach isolated neodymium from the mineral didymium. Rare earths were scientific mysteries in those days, difficult to separate and often misunderstood. Scientists struggled with limited purification techniques and minimal analytical tools, so new compounds like neodymium nitrate seemed almost like novelties. The chemistry advanced with breakthroughs in fractional crystallization and later, ion exchange. By the late 20th century, when magnetics and optics started needing high-purity rare earths, processes had become reliable enough that consistent production became possible on a commercial scale. The journey from rare mineral sample to front-line industrial reagent tells the story of how knowledge, patience, and technical grit gradually unlock new chapters for an element that once gathered dust in a mineral cabinet.
Product Overview
Neodymium nitrate shows up in specialized labs and advanced manufacturing lines alike. Whether powder, crystal, or dry flakes, producers supply it to meet exacting technical standards. It dissolves in water and alcohol, yielding solutions with a faint pinkish hue thanks to its neodymium core. This compound brings precision to industries ranging from high-performance ceramics to laser technology. Markets treat it as a high-value specialty chemical, with catalog listings from Sigma-Aldrich to regional suppliers. The cost isn’t low, reflecting the work needed to isolate neodymium from ores, purify it, and convert it into this functional salt. Users expect consistent purity and carefully graded hydration levels, so careful packaging and documentation matter as much as the powder inside the jar.
Physical & Chemical Properties
A quick look at neodymium nitrate shows pale violet or pink hues, typical for many neodymium compounds. It features the chemical formula Nd(NO3)3·xH2O, usually as a hexahydrate or another hydrated form. With a molar mass that climbs with the number of attached water molecules, its solubility stands high in water, lending itself to liquid-phase reactions and simple preparation. At room temperature, it sits as a crystalline solid, but heat will drive off water and ultimately decompose it to neodymium oxide plus nitrogen oxides. The nitrate group makes it an oxidizer, so storage keeps it away from combustibles or reducing agents. The color signals its rare earth content and purity, and even tiny amounts of iron or other lanthanides can tint it noticeably.
Technical Specifications & Labeling
Labels on bottles of neodymium nitrate show more than just a name. Most producers list chemical formula, hydration degree, batch number, and purity—often above 99.9%, aimed squarely at electronic, laser, or optics-grade buyers. Some offer specific breakdowns, with trace analysis for other rare earths or heavy metals. The packaging includes safety symbols for oxidizers, moisture-sensitive content, and sometimes for environmental hazards. Storage advice usually sits upfront, advising sealed containers and cool, dry conditions. The safety data sheet always follows, showing handling details and emergency info. Buyers in the EU or US look for REACH and TSCA compliance, another layer of confidence for workplaces and regulatory standards.
Preparation Method
There’s no magic shortcut to neodymium nitrate. Producers begin with neodymium oxide or carbonate, dissolving it in nitric acid and filtering out insoluble impurities. The neutralization step matters, so pH gets checked closely to prevent unwanted hydrolysis or the presence of other rare earth nitrates. The product slowly crystallizes as water evaporates, leaving pinkish crystals ready for drying. Modern facilities automate much of this, but the principles stay true to old chemical craftsmanship—smart separation, careful pH control, and rigorous purification at every step. Some manufacturers go further, using ion exchange to boost purity or freeze-drying methods to obtain precise crystalline forms.
Chemical Reactions & Modifications
Neodymium nitrate finds a role both as precursor and as participant in broader chemistry. Adding heat turns it to neodymium oxide, a step toward fabricating magnets, catalysts, or optical ceramics. In solution, neodymium nitrate reacts with agents like sodium carbonate to yield neodymium carbonate and sodium nitrate. Reducing agents, if handled with care, also offer routes to other neodymium salts via controlled reactions. Its oxidizing nature brings value when preparing specialty glass or in certain combustion research setups. Modified forms—such as lightly doped crystals—pop up in research around luminescent materials or laser gain media. Every reaction asks for solid lab technique, as nitrate decomposition generates toxic nitrogen oxides.
Synonyms & Product Names
Anyone who hunts for neodymium nitrate may find it listed as neodymium(III) nitrate or neodymium trinitrate. Trade catalogs sometimes mark it as neodymium nitrate hexahydrate or give a shorthand like Nd(NO3)3. Some suppliers use inventory codes or highlight purity grades in the label. The name might also reference its hydrate form, such as “neodymium nitrate, hexahydrate, 99.99% trace metals basis.” The synonyms do more than fill catalog space—they help researchers, buyers, and customs officers know exactly what rides inside each shipment.
Safety & Operational Standards
Safety officers take a no-nonsense line with neodymium nitrate. It calls for gloves, splash-resistant eyewear, and well-ventilated spaces because dust or solution splashes threaten eyes and skin. Its oxidizing potential demands respect, so workers store it apart from organic chemicals, phosphorus, and reducing agents. Emergency plans detail what to do if containers tip or if powder releases into the air. Staff training brings practical drills in risk control and proper PPE use. On top of that, transport follows strict hazardous goods regulations—labels shout out oxidizer status, and shippers pad containers against drops or leaks. Disposal has to meet local rules for rare earths and nitrates, typically involving neutralization and careful waste tracking.
Application Area
Neodymium nitrate feeds high-tech industries. Makers of specialty glass blend it for color filters, UV-blocking lenses, and high-intensity lamps. People working in laser technology rely on neodymium compounds for active media—pumping energy into the system for precise cutting or medical uses. For ceramics, it brings controlled color and sometimes magnetic properties. Some chemical syntheses choose neodymium nitrate as a mild oxidant or a precursor to advanced catalysts. Western electric car markets and wind energy both depend indirectly on magnetic materials built from neodymium, and the nitrate stands upstream in those production chains. Researchers open new doors every year, charting new uses for this adaptable rare earth salt.
Research & Development
Labs go deep, exploring what neodymium nitrate can offer. Some teams tweak sol-gel methods to craft better optical fibers or transparent ceramics. Others chase advanced phosphors or improvements in neodymium-doped glass for lasers. New purification processes matter, trimming costs and environmental impact when mining and refining rare earths. Analytical chemists push for ever finer purity, using spectroscopy and ion chromatography to check impurities at the parts-per-billion range. Materials science groups run experiments on how thermal or chemical treatments shift the structure and reactivity of neodymium-based solids. With every research cycle, another layer of use emerges, and the chemical gets smarter—quicker production, higher purity, broader reach.
Toxicity Research
The safety data for neodymium nitrate tells a mixed story. Short-term exposure typically brings mild irritation to eyes, skin, and lungs, handled with standard PPE and washing routines. Chronic or excessive contact raises other questions. Some studies point to rare earths accumulating in the liver or bones, particularly in environments with lots of exposure. Researchers debate the potential for long-term effects, but the consensus leans toward caution, especially for those in manufacturing or research settings. Animal studies test high doses, and most occupational health recommendations call for minimizing exposure and monitoring workplace air. The chemical’s role as an oxidizer means fire risk and decomposition hazards pile on top of toxicity—giving safety managers plenty to handle from purchasing to disposal.
Future Prospects
Neodymium nitrate’s story will keep evolving as demand for rare earths continues. The shift toward clean energy and high-efficiency electronics pulls even more attention to rare earth elements. Innovations in recycling, green chemistry, and alternative synthesis could ease environmental pressures and reduce costs. As magnetic, optical, and laser industries expand, the need for higher purity and safer production methods comes front and center. New regulations about supply chain traceability and environmental impact push producers to adopt advanced monitoring and best practices. At the same time, fundamental research into new uses—whether in quantum computing, biomedical imaging, or smart materials—opens new frontiers for what this compound can achieve.
What is Neodymium Nitrate used for?
More Than A Lab Chemical
Most folks don’t hear about neodymium nitrate at the dinner table. Yet this compound shapes many modern technologies—quietly boosting industries from glass manufacturing to electronics. If you’re wearing sunglasses or using a computer, you’re closer to its reach than you might think. I got curious about it while talking to a friend who shapes specialty glass for scientific equipment. His stories give a look at just how much one compound can matter.
Making Glass With A Purpose
Neodymium nitrate steps up when glassmakers want precise color or special optical effects. Toss a little into molten glass and you end up with a striking purple shade—just the thing for those rose-colored lenses in welding goggles and high-end optics. That color isn’t for show. The glass filters out specific wavelengths, protecting eyes and sharpening images. Some labs even use this glass in lasers, where clarity and reliability can’t get left to chance.
This color talent links back to neodymium’s unusual electronic structure. The ions soak up bits of the light spectrum, which explains the rich tints. I’ve seen that specialty glass pop up in high-speed cameras and science gear—places where a regular windowpane just won’t do. Design teams often rely on neodymium nitrate to keep their optics on target.
Fueling New Tech
Electronics and renewable energy benefit as well. Neodymium plays a big part in the magnets found inside wind turbines, microphones, and headphones. The nitrate doesn’t end up in the magnets themselves; it helps refine the neodymium that goes into them. Chemists turn to this compound while separating and purifying rare-earth elements. The neodymium nitrate dissolves easily, making it easier to pull pure metal out of ore without wasting resource or energy. Cleaner production means less environmental headache down the line.
Medical imaging and research labs also value this nitrate. Neodymium-containing compounds often help prep catalysts or create materials that react predictably under lab conditions. Those reactions form the backbone of procedures in analytical chemistry and biomedicine—fields where accuracy trumps guesswork, and no one wants shortcuts.
Supply, Safety, And The Path Forward
All that usefulness carries risks. Mining and processing rare earth elements—including sources for neodymium nitrate—leave behind waste that can leak into soil and waterways. I’ve seen headlines from mining hotspots in China and the US, where poor planning left long scars. Smart regulation keeps contamination in check, but it takes real investment and attention to get right. Countries that invest in clean extraction can hold their heads high, protecting both workers and neighbors.
Looking ahead, safer production and recycling keep climbing the priority list. Research teams work on better ways to recover neodymium from old electronics and magnets. Reducing new mining and recycling what we already have seems like common sense. After all, neodymium nitrate isn’t going anywhere; demand is only inching up as the world leans into green energy and advanced tech.
Understanding chemical building blocks like neodymium nitrate offers a window into the machinery of modern life—and a reminder that innovation depends on a steady flow of materials from both nature and know-how.
What is the chemical formula of Neodymium Nitrate?
The Formula: A Quick Look
Neodymium nitrate, as found in many chemical labs, carries the formula Nd(NO3)3. This simply means it’s built from one neodymium atom tangled up with three nitrate ions. Neodymium itself gets grouped with rare earth elements, sitting right next to others like praseodymium. The “nitrate” part comes from nitric acid, a common staple in everything from fertilizer production to fireworks.
Why Naming Matters in Chemistry
I remember mislabeling a chemical bottle in college, and my lab partner ended up with a cloud of nasty fumes—nobody wants a repeat. Getting the formula right keeps experiments safe and predictable, not to mention accurate in research and industrial work. In chemistry, one swapped number means a totally different substance, bringing whole new hazards and uses. Ask anyone who’s ever mistaken sodium for potassium in the kitchen; details matter, and so do formulas.
Where Science Meets Everyday Life
Neodymium nitrate isn’t famous on its own, but its elements show up in places people don’t expect. Neodymium powers magnets in headphones, wind turbines, electric cars—technology folks use almost daily. The chemical helps prep pure neodymium for those magnets. But the nitrate part deserves respect too; in chemistry, nitrate salts dissolve easily in water, so they fit into a host of lab processes: purification, extraction, and testing. Neodymium nitrate’s water solubility makes it a handy choice for researchers needing a reliable source of neodymium ions.
Health and Environmental Concerns
Neodymium nitrate, like many rare earth nitrates, irritates the eyes and skin on contact. Inhalation or accidental spillage can cause burns or more serious troubles. I remember scolding a friend for not wearing gloves or goggles in the lab while handling similar compounds—extra caution makes the difference between just another day and a trip to the ER. Waste from these compounds doesn’t always break down the way organic things might; nitrates contribute to water pollution if not kept in check, while rare earth elements, if carelessly disposed, add to the mounting electronic waste problem. This stuff shouldn’t go down the drain or into landfills without proper processing.
Stepping Toward Safer, Smarter Use
Getting safer with neodymium nitrate takes more than a good pair of gloves. Labs and factories investing in proper handling courses see fewer accidents; the payoff is strong. Also, collecting waste liquids, neutralizing hazardous residues, and sending them to licensed recyclers protects workers and local communities. Some companies experiment with closed-loop production, meaning they reclaim as much rare earth element as possible—less mining, less pollution, more resource conservation. Regulators need to keep eyes on nitrate waste limits near manufacturing zones since the leftovers seep into streams and mess with ecosystems long after folks have packed up for the day.
Wrapping Up the Formula’s Importance
The formula Nd(NO3)3 connects the dots from textbooks to real life, helping fuel both science and technology while also calling for responsibility. Understanding what this formula means opens doors to safer research, greener industry, and smarter policy. Every time I see a rare earth compound handled right, I remember how one small formula keeps bigger systems running smoothly.
Is Neodymium Nitrate hazardous or toxic?
Understanding the Substance in Practical Terms
Neodymium nitrate doesn’t show up much in everyday life unless you work in a lab, a chemical plant, or near research sites. Its main gig is to help make magnets, specialty glasses, and ceramics. This compound has no smell, and its striking color makes it easy to spot if you spill it on a white surface. That’s useful—because spills happen, and knowing what you’re dealing with helps you act fast.
Direct Health Risks and Absorption
Lab technicians know neodymium nitrate can get into the body through skin, inhalation, or accidental ingestion. People who handle it risk serious irritation if dust or solution touches their eyes or skin—burning, redness, and pain show up quickly. More exposure, especially over time, can kick up respiratory trouble, coughing, or even damage to lung tissue. Getting some on your hands once isn’t likely to wreak havoc, but long-term or careless use causes real harm.
Researchers see that rare earth metal compounds like this one can linger in the body, especially in the lungs and bones. The World Health Organization explains how rare earth elements, including neodymium salts, can build up and trigger pulmonary fibrosis if inhaled for too long. Anyone working with powders or concentrated solutions in tight, poorly ventilated places runs a higher risk.
Environmental Impact and Waste
Factories and research centers that use neodymium nitrate produce liquid and solid waste. Flushing these down the drain or tossing them into ordinary trash invites problems. These nitrates can travel through soil and water, posing health risks for plants, aquatic life, and people who depend on local water sources. Animals exposed to rare earth elements in water show stunted growth and unusual behavior, which rings alarm bells for folks who study food chains. Nitrate itself also contributes to water pollution and can cause “blue baby syndrome” in infants when it spikes in drinking water.
Reinforcing Responsible Practices at Work and Home
Gloves, goggles, and masks aren’t nuisances—they mean fewer emergency room visits and a safer workplace. Too often, people skip these basics, especially during routine tasks. Training and reminders turn into stronger habits. Good engineering controls help too—fume hoods, solid ventilation, and careful storage cut down risk. Neodymium nitrate burns well and can fuel a fire if it meets the wrong chemicals. Mixing without checking labels has caused explosions and injuries, so slow, deliberate handling saves lives.
Better Oversight for Long-Term Protection
Laws around the world set exposure limits for hazardous chemicals, but they work best when checked and enforced. Research published by the Agency for Toxic Substances and Disease Registry links long-term exposure and rare cancers, though data isn’t as deep as for lead or mercury. Agencies suggest frequent checks, recordkeeping, and rapid cleanup of all spills.
Communities can demand better tracking of use, storage, and disposal, especially near schools and neighborhoods. That puts power into local hands to protect water and air. Personal vigilance—reporting odd smells, leaks, or symptoms—supports a circle of safety for everyone involved.
Better Mitigation, Safer Outcomes
People recognize hazards best through education, not just posted warnings. Regular training, clear chemical safety talks, and emergency drills build a sense of responsibility. For anyone working with neodymium nitrate, treating it with real respect keeps personal health, co-workers, and neighbors safer every day.
How should Neodymium Nitrate be stored?
Neodymium Nitrate: Not Your Average Chemical
Neodymium nitrate comes off as just another salt at first glance. Purple crystals, a neat formula, used in industries that range from optics to magnet manufacturing. Underneath the surface, though, you've got a compound that plays rough with moisture and doesn’t like getting warm. That’s not a combination anyone takes lightly in a storage room.
The Real Dangers
Most folks only think about big spills or accidents. In my years working with rare earth salts in labs, I've seen careless storage cause problems long before a real “accident.” Poorly closed bottle, next thing you know, you are contending with a mess on the shelf and acid fumes drifting through shared space. Neodymium nitrate absorbs water faster than you can blink—which turns it sticky, drives corrosion on metal shelving, and, more importantly, raises the risk for chemical reactions you didn’t plan on.
Safe Storage: Not Just a Best Practice, But a Hard Rule
If you’ve spent time working with oxidizing agents, the rules get seared into your brain: keep them dry, keep them cool, keep them isolated. Neodymium nitrate ticks all those boxes. Leaving a bottle next to organics or, worse, flammables means rolling the dice with strong oxidizers. It feels convenient to group chemicals by owner, but these shortcuts always come back to bite you. At a university lab where I worked, a lazy afternoon move to consolidate shelves led to a minor scare—just a puff and a scare, but the cleanup wasted a whole day.
Packing for the Long Run
Moist air turns these purple crystals into mush. Forget Tupperware—the only real protection comes from sealed glass bottles or strong plastic containers with tight-fitting lids. I used to double-wrap the top of each container with parafilm before closing it up, then stash silica gel packs right in the storage cabinet to soak up stray moisture. You could get away with less, but the risk just isn’t worth it.
Temperature plays against you, too. A friend once left some on a shelf facing the window, and weeks later found the label unreadable with the cap rusted shut. Neodymium nitrate wants a cool, dark, low-humidity spot—preferably in a vented cabinet made for oxidizers, behind a locking door.
Communication Beats Luck
Labels matter. A faded label turns a routine restock into a headache because you can't place half the bottles you find. Every lab tech I know, myself included, keeps a logbook with date in, who used it last, any oddities noticed. Clear labeling, regular checks, those things save your skin.
With neodymium nitrate you can’t rely on a basic “chemical closet and forget it” routine. People think chemical management systems only matter for big industry or for folks with OSHA breathing down their neck, but small-scale carelessness racks up silent costs in damage, lost material, and risk.
Staying Ready for Trouble
Accidents look boring until they aren’t. I keep a spill kit close—neutralizers, gloves, a face shield. The first time you see a drop of neodymium nitrate drag brown lines across steel shelving, you know it’s no joke. Good habits—tight seals, cool storage, keeping incompatibles far apart—aren’t just for the textbook. They’re the difference between a safe lab and a story you’d rather not star in.
What is the purity and appearance of Neodymium Nitrate?
Neodymium Nitrate in the Real World
Neodymium nitrate turns up in research labs and advanced manufacturing, often hidden away in chemical bottles marked for those who know what to look for. Purity matters a great deal because anything less than the proper grade can throw off lab results or cause hiccups in hi-tech equipment. I’ve had my own brush with this compound while tinkering with rare earth salts for electronics, and just a trace of an unexpected element brought results no one could trust.
Appearance Speaks Volumes
Anyone who’s spent time in a chemistry workspace recognizes neodymium nitrate by its pale violet or even lavender color. That shade doesn’t just look pretty on the glass; it’s a sign that neodymium’s in its expected oxidation state, and there are no obvious contaminants that stain the powder. The compound usually comes as a crystalline solid or sometimes as white chunks with a hint of purple. Moisture picks up fast, and crystals can start to clump if not sealed tightly. If you ever spot sudden discoloration or cloudiness, chances are the sample dealt with air or water, and you’ll spend extra time purifying it all over again.
Purity: Not Just a Number on Paper
People often ask how pure neodymium nitrate ought to be for sensitive jobs. For optical work or in manufacturing magnets, purity above 99.9% (so-called "triple nine") isn’t just some gold standard for show—it means you aren’t running into headaches from stray metals that sneak their way in during production. For comparison, even a sodium or iron trace can mess up the entire process, especially when you’re making lasers or glass tinting that depends on precise coloration effects. In my years working alongside analytical chemists, I’ve seen how even parts-per-million differences can set off hours of recalibrating machinery.
Quality Checks and Real Risks
Suppliers sometimes tout impressive purity numbers, but those claims mean nothing if there’s no supporting data. Reliable labs turn to techniques like ICP-MS (inductively coupled plasma mass spectrometry) to measure trace elements and put numbers to claims. A clean certificate of analysis tells users how much trust to invest—which can make or break projects spinning off from government grants or private startups. Once, a project I shadowed in a university setting nearly lost months of progress after a supplier’s “high purity” batch failed to meet the promised standard, and it took weeks to source a verified product.
Getting It Right: Solutions for the Real World
There’s rarely room for error in high-tech materials science. Modern supply chains that focus on full traceability and batch-level documentation cut down risks. For labs or small manufacturers, building long-term relationships with proven suppliers pays off. On the practical front, proper storage—airtight containers with silica gel—preserves the look and value of fine neodymium nitrate for the long haul. Regular testing and visual inspection catch issues early, which beats scrambling to fix problems downstream. Paying attention to both purity and that telltale color does more than check a box for compliance—it keeps projects on track and lets teams build on solid ground.
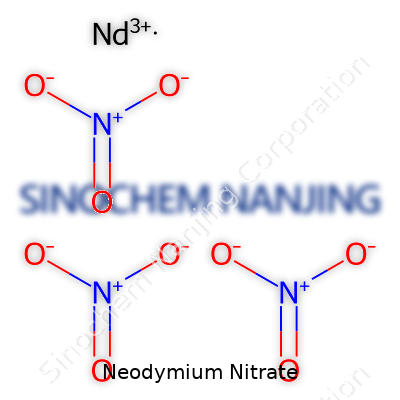
| Names | |
| Preferred IUPAC name | neodymium(3+) nitrate |
| Other names |
Neodymium(III) nitrate Neodymium trinitrate Neodymium nitrate hexahydrate |
| Pronunciation | /niː.oʊˈdɪm.i.əm ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 16454-60-7 |
| Beilstein Reference | 3196417 |
| ChEBI | CHEBI:52723 |
| ChEMBL | CHEMBL4295371 |
| ChemSpider | 21317 |
| DrugBank | DB16409 |
| ECHA InfoCard | ECHA InfoCard: 100.041.272 |
| EC Number | 233-793-8 |
| Gmelin Reference | 72399 |
| KEGG | C101513 |
| MeSH | D017829 |
| PubChem CID | 159393 |
| RTECS number | QU8400000 |
| UNII | K96PG2Y794 |
| UN number | UN1515 |
| CompTox Dashboard (EPA) | DTXSID4041943 |
| Properties | |
| Chemical formula | Nd(NO3)3 |
| Molar mass | 438.22 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.13 g/cm³ |
| Solubility in water | soluble |
| log P | -4.46 |
| Basicity (pKb) | 8.41 |
| Magnetic susceptibility (χ) | +1210e-6 cm³/mol |
| Refractive index (nD) | 1.558 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 215.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1540 kJ/mol |
| Pharmacology | |
| ATC code | V07AA |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, causes serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS03,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H272, H314, H319, H335 |
| Precautionary statements | Precautionary statements for Neodymium Nitrate: "P210, P220, P221, P280, P305+P351+P338, P337+P313, P370+P378, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Lethal dose or concentration | LD50 oral rat 2,800 mg/kg |
| LD50 (median dose) | > 2,000 mg/kg (rat, oral) |
| NIOSH | NA9108 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Neodymium Nitrate: Not established |
| REL (Recommended) | 300 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Neodymium(III) oxide Neodymium(III) chloride Neodymium(III) fluoride Neodymium(III) sulfate |

