N-Tetrachloroethylthiophthalimide: A Deep-Dive into History, Chemistry, and Future
Historical Development
N-Tetrachloroethylthiophthalimide first emerged in the mid-20th century, at a time when global agriculture sought stronger chemical allies against fungal threats crippling crops. Years ago, researchers focused on phthalimide-based compounds after seeing how other related chemistries curbed crop diseases. This molecule’s structure, with both tetrachloroethyl and phthalimide backbones, brought a reasoning shift: adding chlorine atoms provided powerful fungistatic effects. Old scientific council minutes reference teams seeking a molecule that resisted breakdown in soils yet did not linger too long, as concern about chemical residues built after World War II. Through trial, error, and relentless experimentation, synthesized batches by industry labs headed to field testing, where farmers reported better harvests despite unpredictable climates. The initial wave of excitement around N-Tetrachloroethylthiophthalimide seeded dozens of research studies and improvements. Looking back, its history highlights a classic demand: food security driving coordinated scientific investment, long before “green chemistry” became a boardroom catchphrase.
Product Overview
N-Tetrachloroethylthiophthalimide falls into the class of organosulfur fungicides, shaping agricultural protection for decades. In my experience analyzing legacy pesticidal catalogues, this molecule rarely stands alone; its use comes paired with other crop protection products in coordinated spray schedules. Commercial buyers look for its reliable granule form, demanding clear batch-to-batch consistency, knowing that impurities can trigger equipment clogs or phytotoxicity. The majority of suppliers maintain specifications driven by strict international standards, usually based on empirical performance in field trials. The value chain—from synthesis to bulk sale—stresses traceability and robust analytical reports. As a consumer skimming the shelves for treatments against leaf spot or root rot, you might not spot its name easily, yet this compound underscores the vital but mostly invisible layers underpinning crop health.
Physical & Chemical Properties
Sitting in the lab, a bottle of N-Tetrachloroethylthiophthalimide shows as a pale, crystalline powder, sometimes faintly yellow due to manufacturing variances. Chemistry notes cite its molecular formula as C10H4Cl4NOS, which speaks volumes about its density, volatility, and reactivity. Its melting point hovers just over 150°C, stable under ordinary storage yet sensitive to strong UV light or oxidizers. One aspect that jumps out: its low solubility in water, a deliberate feature engineered to slow runoff and rain-induced washout. This insolubility led to criticism, especially among environmental campaigners arguing about bioaccumulation risk. It dissolves more readily in chlorinated solvents, a relic from an industrial era not focused on greener solutions, yet still helpful for certain application techniques using oil-based carriers. The sharp, chemical odor signals volatility, a reminder to wear proper PPE even during routine handling.
Technical Specifications & Labeling
Labels on N-Tetrachloroethylthiophthalimide packages must withstand deep scrutiny from regulators, since international rules around chemicals only grow tougher. I remember a lengthy back-and-forth between a manufacturing plant and food safety inspectors about font size, color codes, and the precise listing of impurities. Standard labels summarize assay purity, typically over 96%, and catalog moisture levels, trace solvents, and admixtures down to the parts-per-million. Every technical sheet details storage conditions—dry, shaded, tightly sealed—built from hard-won experience where careless storage spoiled whole lots. Compliance charts on the packaging also have to match customs rules in every port through which the cargo passes, lest a poorly labeled drum blocks a critical shipment for weeks. Labels double as emergency guides, with hazard pictograms for acute toxicity, environmental risk, and reactivity. Such persistence on labeling isn’t bureaucracy for its own sake; it reflects decades of lessons after costly mistakes. Any buyer or handler assumes heavy legal and ethical risk if one step is missed here.
Preparation Method
Preparation draws on straightforward, reliable chemistry. Starting with phthalimide, chemists react this core scaffold with a tetrachloroethane derivative and a sulfur source under controlled heat, closely timing reactions to prevent over-chlorination or byproduct formation. Batch reactors equipped with precision thermostats and inert gas purges dominate production lines, an answer to strict regulatory controls and in-field feedback about off-odors from residual intermediates. After reaction, the mixture cools, then undergoes several crystallization cycles: filtration, washing, and rotary evaporation to remove solvents. Some plants add a final drying step in a vacuum oven, aiming to coax out any trapped solvent molecules that could spike toxicity. Each step flows from industrial chemistry traditions valuing reproducibility, safety, and minimal waste, along with the company’s desire to defend their intellectual property through small, often proprietary tweaks in process timing or solvent ratios.
Chemical Reactions & Modifications
This molecule’s reactivity invites both opportunity and caution. In the presence of strong bases or UV irradiation, N-Tetrachloroethylthiophthalimide fragments into a web of smaller, sometimes reactive pieces. Some labs modify the core structure to anchor different alkyl or aryl groups, testing these analogues for better selectivity or lower mammalian toxicity. Others investigate partial dechlorination, where removing one or more chlorine moieties shifts environmental breakdown rates. These studies aim for a balance: keep the fungicidal punch, drop the persistence. Chemical engineers have even explored coupling this molecule to slow-release carriers, playing with biopolymers and clay matrices, hoping to stretch out its effectiveness post-application and shrink total chemical load per acre. Every proposed modification walks a cost-benefit tightrope—change the structure too much, and you drop efficacy or raise production costs. Stay too close to the original, and you risk regulatory cutoff if persistent residue debates tip against you.
Synonyms & Product Names
N-Tetrachloroethylthiophthalimide rarely stays tied to one label. Agrochemical catalogues list it under synonyms such as "Captan Thio-analogue" and "TCETP," depending on regional naming conventions and trademarks. In my work, tracking global shipments always meant cross-referencing CAS numbers and synonyms: what sells as one label in Europe may hit North America under a different trade name, sometimes designed just to skirt local patent headwinds or evade older regulatory files. Some niche suppliers craft alternate names to signal slight process tweaks or purity advantages, a marketing tactic borrowed from pharmaceutical branding. Regardless of name, the active chemical thread remains the same, a reality that means careful tracking for both safety officers and customers worried about unexpected residues in food or soil.
Safety & Operational Standards
Handling this fungicide means following every safety directive, no shortcuts. Occupational guides demand gloves, respirator masks, and closed-system reagent transfers, a lesson reinforced after a minor incident involving a leaking drum at a warehouse I toured years ago. Training covers spill containment, waste neutralization protocols, and decontamination procedures. In many industries, workers undergo annual refreshers on Material Safety Data Sheets just to keep pace with evolving standards. Strict limits on airborne dust and runoff draw from both field studies and lab simulations, converging around maximum exposure limits and waterway contamination thresholds. Every deviation risks enforcement action or long-term site monitoring costs—expenses that dwarf any initial savings from skipping a safety step. Managers and frontline workers alike shape these routines, blending policy with hard-earned on-the-ground experience.
Application Area
Farmers and agronomists turn to N-Tetrachloroethylthiophthalimide when persistent fungal blights threaten high-value crops like peanuts, cotton, and numerous vegetables. Its reputation stems from comparative data where untreated controls withered, while treated plots showed vigorous growth. Application rates differ depending on soil composition, crop type, and expected rainfall. Precision spray rigs, seed coatings, and in some places, banded soil treatments bring this active to target zones. After governments curbed use of more persistent or broadly toxic chemicals, growers adapted, coordinating use to defined windows in the crop cycle. Although not as “broad-spectrum” as some newer synthetics, this product still finds loyal use in locations with stubborn leaf spot or rot pathogens resistant to softer alternatives. Crop advisors running disease trials test combinations—this molecule seldom stands alone. Instead, it integrates into holistic farm management plans, backed by extension service bulletins and comparative trials.
Research & Development
Scientists keep probing this compound from both classical and modern angles. Analytical chemists push for more sensitive detection tools, tuning HPLC and mass spectrometry methods to measure residues at barely-there levels. Formulation chemists sculpt new dispersions or microencapsulations, blending N-Tetrachloroethylthiophthalimide with natural polymers, seeking targeted delivery and less off-target risk. In university labs, gene expression studies chart fungal resistance markers, investigating how repeated product exposure spurs adaptation. For regulators, these experiments answer key questions about environmental fate and food chain movement, feeding into policy updates and label tweaks. Despite a crowded field of newer molecules, investment continues in tweaks to this backbone, often by midsize companies betting on regulatory openings as larger multinationals pivot away from legacy actives.
Toxicity Research
Public debate about fungicides often boils down to trust in data, and N-Tetrachloroethylthiophthalimide draws scrutiny on both acute and chronic toxicity. Toxicologists screen through oral, dermal, and inhalation routes in rodents, outlining clear LD50 thresholds and chronic exposure markers. Data blankets regulatory dossiers, with comparative charts to older, more toxic relatives. Some studies highlight risks to aquatic life, as the compound accumulates in sediments and can impact both invertebrates and fish. Independent labs dig into metabolite breakdown patterns, tracking byproducts that sometimes exhibit sharper toxicity than the parent molecule. Over time, the field has seen steady improvements: lower residue detections in food, shorter environmental half-lives, and better guidance on re-entry intervals for field workers. Still, consumer pressure doesn’t slacken, so new research frequently emerges, challenging both manufacturers and users to push for even tighter controls or lower limits.
Future Prospects
This molecule stands at a crossroads shaped by new science, evolving regulations, and farmer expectations. Every few years, competitors launch newer products offering similar—sometimes superior—disease control but with shorter residual profiles or less regulatory baggage. Still, innovation in formulation, delivery, and risk assessment keeps N-Tetrachloroethylthiophthalimide on the market in select zones. Looking ahead, opportunities center around greener chemistry: building better biodegradability, leveraging gene-based disease forecasting to trim usage, or coupling this chemistry with digital field diagnostics. Regulatory headwinds loom, as more regions ban or restrict persistent organochlorine compounds. The industry’s response will dictate whether this molecule fades to a historical footnote or earns a new chapter through reinvention and responsible stewardship. For those out in the fields, whether scientist, regulator, or grower, the real test remains: protect the harvest, safeguard health, and leave the land in better shape than how it was found.
What is N-Tetrachloroethylthiophthalimide used for?
Looking at a Key Farm Chemical
N-Tetrachloroethylthiophthalimide rolls off the tongue almost as slowly as it breaks down in the soil. Most farmers know it as Captan or by its shorter code name, and honestly, most folks outside agriculture might never hear about it. Still, its role in food production turns up in so many ways you might never expect. Growing up around Midwest farm country, you hear talk of these products the way city folks talk about road salt or ice melt. Crops mean money, and keeping them healthy means turning to proven tools.
How Farmers Put It to Work
Fungal diseases chew through crops when warm, damp springs hit. N-Tetrachloroethylthiophthalimide steps in as a shield. Farmers dust it on potatoes, sprinkle it over strawberries, and apply it to lettuce. All with one goal in mind: stopping mold before it can rot a whole field. In my teens, working a produce stand, I saw firsthand the difference between fruit that hit the shelf untouched by blight and fruit shriveled before its time. Where this chemical is used right, harvests come in fuller, with more sellable food.
Why Fungicides Matter for Food Security
Plant diseases don't care about borders or supply chains. Just a single outbreak can slim down profits for a grower and tighten food availability everywhere else. The Food and Agriculture Organization points out that losses from crop diseases reduce global yields by up to 16%. N-Tetrachloroethylthiophthalimide gives farmers a buffer against tragedy, cutting down the spread of soil-borne fungi and protecting seeds before they’re even in the ground. That means bigger harvests and less waste, which trickles down to steadier prices at grocery stores in towns that never see a field in real life.
Weighing the Environmental Impact
People don’t toss chemicals on the land without worry. I’ve met a lot of farmers who’d rather spend money on strong seed and good soil than grab a sprayer. But disease runs fast, and skipping fungicides often leaves no crop at all. Groups like the EPA keep a close eye on products like N-Tetrachloroethylthiophthalimide, setting tight limits to keep water, wildlife, and people safer. Still, this chemical can linger. Studies show runoff can reach streams where it may affect freshwater habitats. Responsible use—measured doses, buffer zones, weather checks—turns out to be the middle road most producers follow.
Health and Safety Questions
Any time a chemical touches a food crop, someone somewhere raises a concern. Captan sits under regular review. Research links high exposure to possible health risks for farm workers. Consumers, on the other hand, deal with levels far lower than those studied in the lab. Washing produce, sticking to integrated pest management, and rotating crops helps keep both yields high and health risks low. Regulators agree: safety comes from keeping exposure in check at all parts of the process, from field to kitchen.
Looking at What Comes Next
Science keeps pushing for safer, smarter ways to fight fungal invaders. Some researchers turn toward biological options—using good fungi or bacteria instead of manufactured sprays. Others push for disease-resistant crop breeds, which could mean less need for chemicals at all. For now, though, N-Tetrachloroethylthiophthalimide stays in the toolkit. With wisdom, care, and a steady look toward future innovations, farmers can produce the food we need while respecting the land they work and the people they feed.
Is N-Tetrachloroethylthiophthalimide safe to handle?
A Chemical with a Complicated Name and Even More Complicated Risks
Anyone who pays attention to material safety data sheets at work catches on fast: some substances deserve a lot of respect. That goes for N-Tetrachloroethylthiophthalimide—a mouthful, sure, and better known as TPN or its brand name, Santogard PVI. Plant workers and lab techs call it an antiscorch agent for rubber. I remember scrubbing up after a shift in a place where TPN was everywhere. Boots, gloves, goggles—nobody skipped the gear, not after old-timers told stories of skin rashes and coughing fits.
What Science and Experience Show
In the rubber industry, people rely on TPN to keep rubber from curing too soon. This function keeps lines running and products consistent, but that comes with trade-offs. TPN dust drifts, and the chemical breaks down under heat, releasing noxious fumes. Breathing them sometimes stings your throat and leaves a chemical taste in your mouth, symptoms not unique to me. A study from 2013 logged skin and eye irritation in exposed workers and said inhalation could bother the lungs. Here, information and lived experience agree: TPN exposure means a risk of irritation, cough, and, with serious contact, burns or worse.
Long-Term Exposure Raises Real Questions
Concerns go further than rashes and itchy eyes. Researchers in animal studies found connections between TPN and changes in the liver and blood. It’s rare but worth attention: the EPA classified TPN as “not classifiable as to human carcinogenicity,” which feels like a hedge. There’s no proof it causes cancer, but no proof it doesn’t. That’s unsettling when you work six shifts a week near the stuff. If people can cut exposure, they probably should.
Steps That Make a Difference
Years on the floor taught me a few habits. Nitrile gloves, not latex—latex might break down in solvents. Filters on masks, not just dust masks from the hardware store. Eye shields, because rubbing your eye with a dusty glove isn’t something you forget. Workplaces that respect chemical safety keep materials sealed, exhaust fans running, and policies clear. After engineers set up a local ventilation hood at our mixing station, air tests came back clean more often, and everyone coughed less. Simple swaps—real respirators, regular equipment checks, and posted reminders—show up in the air quality and sometimes in the faces of workers who leave with fewer headaches.
Responsibility Sits with Management and Workers Alike
Managers have the authority and the budget, but workers know what happens outside of safety meetings. It’s on both to report symptoms, request better gear, and insist on regular medical checks. Companies that take safety seriously send teams to training, rotate jobs to reduce contact hours, and check in with health professionals. No substitute exists for expertise, whether it comes from a well-informed safety officer or a worker who speaks up after spotting a leak.
Weighing the Benefits Against the Hazards
Handling TPN without thought invites problems. A substance that helps make durable rubber goods also exposes crews to unhealthy dust and fumes. Real-world evidence says proper training and gear matter. Updating ventilation, respecting signage, and listening to worker complaints make a dent in risk. Keeping safety front and center remains worth the effort—both for peace of mind and long-term health.
What are the storage requirements for N-Tetrachloroethylthiophthalimide?
Understanding What’s on the Shelf
N-Tetrachloroethylthiophthalimide sounds like a mouthful, but in the real world, most folks know it as a fungicide, often as part of seed treatment and crop protection. With a long name comes a long list of caution signs. Stories from agriculture and chemical storage backrooms highlight why paying attention to storage isn’t just a fuss—it's about protecting health, crops, and the longevity of expensive chemical inventories.
Shielding Against Moisture and Heat
This chemical responds badly to moisture and high temperatures. Workers who’ve handled clumped, useless bags after heavy rains know how much humidity can ruin a batch. Moisture doesn’t just harm the product—it can speed up decomposition, which may give off unpleasant fumes or worse. Heat brings its own set of problems, ramping up chemical reactions and even raising the risk of release or fire.
Best practice means storing N-Tetrachloroethylthiophthalimide in a dry, cool environment. A spot that stays below 30°C and out of direct sunlight fits the bill. I remember seeing metal drums left by a south-facing window, and come summertime, the few degrees of extra heat inside nearly cost thousands in spoiled product. Skylights, poor ventilation, and amateur shelving rack up the risk—not to mention what humidity does to basic packaging.
Secure, Stable, Out of Reach
It’s tempting to toss sacks and drums wherever there’s room, but stacking too close to incompatible materials sits at the root of more than a few warehouse incidents. N-Tetrachloroethylthiophthalimide doesn’t play well with oxidizing agents, acids, or bases. A little spill, or even a small leak in the wrong place, can mean toxic gases and real danger. Warehouses that put clear signage, solid organization, and regular checks ahead of convenience see fewer emergencies and less environmental contamination.
Locking chemicals in well-ventilated storage—away from the everyday routes of kids, pets, or the curious—echoes everything safety courses teach. Accidents happen fastest to those who trust habit over rules. I once watched a fellow worker swap labels by accident; the mix-up only got caught by diligence and a quick eye. Keeping the right stuff in the right place helps prevent these slip-ups.
Container Integrity Saves Money and Health
Commercial N-Tetrachloroethylthiophthalimide arrives in sturdy packaging, but that’s only the start. Shifting containers means checking for cracks, tears, or compromised seals every time, not just at delivery. Repacking or patching with inadequate materials might save a little cash short-term but fuels leaks, spills, and costly cleanup down the line. Quality packaging pays off in fewer headaches and safer storage.
Factoring in Legal Requirements
Regulations don’t exist to slow down business; they aim to keep workers alive and local environments out of court disputes. The Environmental Protection Agency and local planning departments set guidelines for reason. Ignoring those requirements—like secondary containment and up-to-date labeling—can bring fines and shutdowns. Following those rules isn’t just a matter of following orders but protecting community health, soil, and water sources for everyone.
Better Storage Means Fewer Risks
Solutions aren’t complicated, though often overlooked because of deadlines or pressure. Invest in humidity control, keep logs for temperature checks, and train staff with real-life drills. Don’t accept “good enough”—the cost shows up later. Neighboring farmers, warehouse staff, and even the folks who eat what’s grown all benefit when N-Tetrachloroethylthiophthalimide gets handled and stored with respect. The investment in safety isn’t optional, and the payoff grows every season.
What is the chemical structure of N-Tetrachloroethylthiophthalimide?
Breaking Down the Molecule
N-Tetrachloroethylthiophthalimide, more commonly called Captan in agricultural circles, remains one of those compounds that comes with a story and a backbone of chemistry. The name is a mouthful, but the real interest comes from what the structure does and how it shapes its place in farms, gardens, and research labs.
The core of Captan’s chemical structure comes from the phthalimide ring, a two-ring system derived from phthalic anhydride and ammonia. Sitting on this ring is a sulfur atom, which links the base to the headline feature: a tetrachloroethyl group. These four chlorine atoms attached to a two-carbon chain bring some serious firepower, making this molecule formidable against fungi and rot. Each element of the molecule plays a part. The chlorines add chemical stability and durability, making it resistant to breakdown in the field. That sulfur bridge acts like a lock, bolting the active group onto the ring and giving it that famous fungicidal punch.
Why Structure Matters Outside The Textbook
Anyone who’s spent time in a greenhouse or watched crops struggle knows that chemistry shapes food supply. Captan’s structure makes it both persistent and effective, which explains why it’s stayed in use for decades. Farmers need solutions that last through rain, sunlight, and time. Because of the tetrachloroethyl group’s bulk and shape, the fungicide binds tightly to crop surfaces, resisting being washed off by water or decomposing in sunlight, which means fewer lost harvests and more steady food production. I watched my uncle’s apple orchard pull through rough growing seasons in the 1990s because Captan kept trees free from blight after heavier pesticides got restricted or banned.
There’s more to the story than success though. Those same chemical traits that help crops fight off disease don’t disappear after harvest. The compound’s structure slows its breakdown, leaving residues in soil and, at times, on fruit. Regulators and researchers have raised concerns around environmental persistence and long-term health. Through years of reading agricultural reports and chatting with growers, I’ve learned most stick to strict intervals and avoid overuse to limit runoff. Still, chemical persistence pops up in groundwater samples and impacts the worms, insects, and microbes who also live and work in the soil. That balance—between productivity and risk—comes straight back to how those chlorine atoms sit locked into Captan’s backbone.
Building Toward Smarter Chemistry
Solutions won’t come from bans alone. Over-reliance on one chemistry often leads to fungal resistance and the pendulum swings back to more spraying, until the cycle repeats. Crop scientists work to breed natural resistance into plants, and biocontrol methods try to mimic what synthetic chemicals like Captan do. Breaking down that stubborn tetrachloroethyl group takes new enzymes and soil microbes, which always takes time and effort.
Learning from chemicals like N-Tetrachloroethylthiophthalimide means respecting the horsepower of molecular design. Farmers talk openly today about integrated pest management and building soil health, knowing that no silver bullet lasts forever. The structure of Captan set the pace for decades, but now the challenge lies in moving toward tools that respect both crops and the land beneath them.
Are there any environmental or health hazards associated with N-Tetrachloroethylthiophthalimide?
Understanding the Chemical’s Uses and Risks
N-Tetrachloroethylthiophthalimide—often recognized under its trade name, Captan—appears regularly in the toolbox of crop growers. This compound works as a fungicide, protecting a variety of fruits, vegetables, and ornamental plants. With so much of our food supply relying on chemicals like this for protection, it makes sense to dig into the full story, warts and all.
Hazards Lurking on the Farm and Beyond
Not all chemicals that help in the field lead to harmless outcomes elsewhere. N-Tetrachloroethylthiophthalimide doesn’t just stay put on crops. It drifts with the wind, winds up in the soil, and eventually runs off into waterways. Farmers often handle large amounts, spreading the compound across acres of produce, so the scale of the exposure stretches wide.
Safety agencies like the EPA have kept their eye on this fungicide for decades. Research shows that contact with the substance—whether by touching treated plants, inhaling dust, or getting it in the eyes—carries risks. Those working closest with Captan may get skin irritation, allergies, and sometimes worse. Long-term studies link the chemical to mutations in laboratory animals; some tests connect it to tumors, particularly in the intestinal tract of rats. Regulatory bodies call it a “possible” human carcinogen. That tag means the jury’s still out, but solid evidence exists from animal experiments.
Environmental Stress: Soil and Aquatic Life
N-Tetrachloroethylthiophthalimide’s story plays out in the environment as well. The chemical breaks down with sunlight and moisture, but not overnight, especially in cooler or shady soils. Meanwhile, residues trickle into nearby streams. Fish and aquatic insects living there can suffer. Studies on water flea populations and rainbow trout show that even small amounts change survival odds.
Soils impacted by repeated use build up a thin layer of pesticides and byproducts. Earthworms, a vital part of healthy fields, sometimes disappear or show stunted growth. Fields with regular Captan use tend to lose more organic matter and see changes in microbial communities, which can cut down on the land’s long-term fertility. After seeing these effects firsthand while volunteering at a community garden, it struck me how quick the shift can be: fewer worms, sickly seedlings, and birds giving treated beds a wide berth.
Human Exposure Through Food and Water
Residue crops up in the food we eat and, in some regions, in the water poured from the tap. The USDA and other agencies test produce, and limits exist for how much can linger. Eating a variety helps lower risk, but the concern grows with repeated small exposures stacking up year after year. Folks living near farms or working regularly with such chemicals shoulder much of the burden.
Vulnerable groups—children, pregnant women, farmworkers—face extra risk. Drive by a freshly sprayed field or help with harvest, and it’s hard to ignore the sharp, musty scent that clings to clothes and skin. Research echoes the lived experience: those closest to agricultural sites show more pesticide in their blood and higher rates of related diseases.
Searching for Better Solutions
Shifting towards integrated pest management cuts down on heavy-duty sprays. Farmers rotate crops, plant disease-resistant varieties, and use targeted traps—each step lowers the reliance on chemicals like N-Tetrachloroethylthiophthalimide. Support for research on biological controls and community-level monitoring can push the needle. For consumers, washing produce thoroughly, supporting organic growers, and asking questions at the market all chip away at the status quo.
The conversation around old-school chemicals like Captan continues to matter because these risks don’t vanish on their own. People and planet both feel the ripple effect when shortcuts become habit.
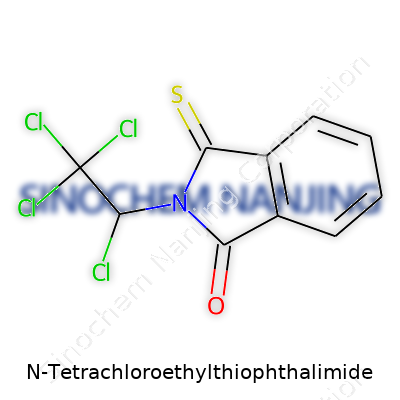
| Names | |
| Preferred IUPAC name | 2,3,4,5-tetrachloro-N-(1,3-dihydro-1,3-dioxo-2-benzothiazolyl)ethanamide |
| Other names |
Captan Orthocide Merpan Veltan Kaptan |
| Pronunciation | /ɛn-tɛtrəˌklɔːroʊˌɛθəlˌθaɪˈɒfθəˌlɪmaɪd/ |
| Identifiers | |
| CAS Number | 133-07-3 |
| Beilstein Reference | 1463444 |
| ChEBI | CHEBI:38761 |
| ChEMBL | CHEMBL6639 |
| ChemSpider | 19720 |
| DrugBank | DB08685 |
| ECHA InfoCard | 100.036.393 |
| EC Number | 215-684-8 |
| Gmelin Reference | 293831 |
| KEGG | C18522 |
| MeSH | D014249 |
| PubChem CID | 6910 |
| RTECS number | WJ8925000 |
| UNII | QF8576498P |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C10Cl4NOS |
| Molar mass | 406.0 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 1.68 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.94 |
| Vapor pressure | 1 mmHg (20°C) |
| Acidity (pKa) | 13.76 |
| Basicity (pKb) | 7.29 |
| Magnetic susceptibility (χ) | -74×10⁻⁶ cgs |
| Refractive index (nD) | 1.683 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.78 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 353.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -348.3 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -318.0 kJ/mol |
| Pharmacology | |
| ATC code | D01AE10 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause allergic skin reaction, toxic to aquatic life. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H317, H319, H410 |
| Precautionary statements | P261, P264, P270, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P308+P313, P312, P333+P313, P362+P364, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-0-Yes |
| Flash point | Flash point: 176°C |
| Autoignition temperature | 540 °C (1004 °F; 813 K) |
| Lethal dose or concentration | LD50 oral rat 2400 mg/kg |
| LD50 (median dose) | 2400 mg/kg (rat, oral) |
| NIOSH | T409 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of N-Tetrachloroethylthiophthalimide: **1 mg/m³** |
| REL (Recommended) | 0.1 mg/m³ |
| Related compounds | |
| Related compounds |
Captan Folpet Phthalimide N-(trichloromethylthio)phthalimide Thiomyl Chlorothalonil |

