N-Propyl N-Butyrate: More Than Just an Ester
Historical Development
Back in the days of early organic chemistry, the synthesis of esters marked a pivotal moment in the evolution of chemical science. Laboratories in the 19th century bubbled with curiosity over the fruity aromas produced by reactions between acids and alcohols. N-Propyl N-Butyrate popped up as both a curiosity and a practical compound, its pleasant scent drawing interest for applications in flavors and fragrances before the word “green chemistry” made headlines. As industrial chemistry grew, so did the drive to create esters reliably and safely. Over time, processes got cleaner, yields got higher, and the understanding of how these molecules interact in mixtures helped shape sectors from food production to industrial solvents.
Product Overview
N-Propyl N-Butyrate stands out for its clear liquid form and distinctive sweet, pineapple-like scent. This ester comes from reacting n-butanol with n-propionic acid. People working in food science, perfumery, and even analytical laboratories know it as a reliable standard and ingredient. In flavors, it boosts the perception of ripeness or tropical aroma in many formulations. It’s not just a treat for the nose—its volatility allows engineers to tune the release of its scent for different uses, and in chromatography, it often serves as a reference marker for calibration.
Physical & Chemical Properties
N-Propyl N-Butyrate packs a punch with a boiling point around 155°C and a melting point well below zero. Its density comes in near 0.86 g/cm³ at room temperature, and it walks the line between hydrophobic and slightly polar, so it rarely mixes with water but dissolves easily in organic solvents. Pour it out and you’ll find it flows freely, clear and without color. Its flash point sits comfortably above many other esters, which matters when you have strict regulations in factories and storage warehouses. Odor is unmistakable—sharp and reminiscent of overripe pineapple.
Technical Specifications & Labeling
Detailed technical sheets provide everything a producer or quality-control chemist cares to know about N-Propyl N-Butyrate. Purity over 99% remains the gold standard for anything heading into regulatory environments like flavor production or certified reference materials. Impurity profiles get tightly monitored using gas chromatography. Labeling follows international standards, with hazard identification under GHS and classic transport labels due to its flammable liquid status. Batches get tracked with lot numbers, and shelf life under correct storage conditions stretches several years. Regulatory assurance leaves no guesswork.
Preparation Method
To make N-Propyl N-Butyrate, labs lean on a standard Fischer esterification. Mix n-butanol and n-propionic acid with a strong acid catalyst—concentrated sulfuric acid or sometimes p-toluenesulfonic acid—then heat under reflux. Water forms as a byproduct and gets driven off to pull the reaction forward, shifting the odds in favor of ester over unreacted starting materials. After cooling, neutralization, and washing steps remove excess acid, while distillation purifies the product. Yields climb past 80% with careful control, letting chemists churn out predictable volumes with consistent properties.
Chemical Reactions & Modifications
The chemistry of N-Propyl N-Butyrate doesn’t stall at simple synthesis. Under basic or acidic hydrolysis, it snaps back to its parent acid and alcohol, a useful trick for recycling or waste processing. In the right hands, the ester group can get swapped or reduced, making this compound a handy building block for more complex esters or even alcohols after reduction. Reactions with nucleophiles open the door to whole fields of organic transformations, feeding into chain elongation or other ester interchange reactions. For teaching labs, this ester often demonstrates the practicality and challenges of isolation and purity in organic synthesis.
Synonyms & Product Names
Across catalogs and regulatory sheets, you’ll spot N-Propyl N-Butyrate listed as Butyric acid n-propyl ester or Propyl butyrate. Some trade names play on its flavor notes or application focus, but the chemical crowd usually sticks to the systematic options. Internationally, EINECS and CAS registry numbers keep things consistent, no matter the market or language.
Safety & Operational Standards
Handling N-Propyl N-Butyrate asks for classic laboratory safety along with industry best practices. Vapor can irritate the eyes and lungs in high concentrations, so well-ventilated workspaces and tight container seals reduce risks during storage and transfer. Flammability brings its own set of challenges—keeping open flames, sparks, or heat sources away becomes essential. Personal protective gear, from chemical splash goggles to nitrile gloves, stays in easy reach. Spill containment and explosion-proof storage solve problems before they start, with staff training closing the loop on accident prevention.
Application Area
In food science, N-Propyl N-Butyrate shows up in candy, soft drinks, baked goods—anywhere a burst of fruity aroma lifts experience. Perfumers use its volatile profile for top notes in fragrances. In industrial analysis, it acts as a calibration standard for various instruments. Even pesticide and pharmaceutical industries rely on its ester group for studying break-down products or synthesizing related molecules. Its use in learning environments also teaches future chemists essential skills in synthesis, purification, and analytical techniques.
Research & Development
Researchers dig deep into improving both green manufacturing and novel functional properties tied to N-Propyl N-Butyrate. Enzyme-catalyzed syntheses offer lower energy input and waste compared to old-school acid catalysis. Studies explore new uses for biodegradable solvents drawn from esters, focusing on safety and minimal environmental impact. Analytical chemists devise quicker, cleaner methods for quantifying N-Propyl N-Butyrate in everything from flavored syrups to environmental air samples. Academic groups sometimes follow how the smell profile blends with other esters as they chase the holy grail of naturalistic artificial flavors.
Toxicity Research
Over the past few decades, N-Propyl N-Butyrate has faced scrutiny from toxicologists and safety regulators. Acute oral or dermal toxicity stays low—studies in rats and rabbits show few risks at concentrations far beyond typical exposure, and metabolic breakdown products match those found in many common foods. Prolonged high-dose exposure may cause mild liver effects, though at levels not normally approached in food or industrial use. Airborne concentrations in flavor or fragrance plants need monitoring, since headaches or irritation can flare up from vapors. Regulatory agencies, from the FDA to EFSA, track emerging data to set safe thresholds in consumer products and workplaces.
Future Prospects
Esters like N-Propyl N-Butyrate look poised to ride the wave of consumer demand for “natural” and sustainable ingredients in food and personal care. Biocatalysis promises cleaner production. As the marketplace shifts toward eco-friendly solvents, esters may replace halogenated or petroleum-derived chemicals. The ongoing hunt for authenticity in flavors places renewed value on molecules with a known safety pedigree and friendly regulatory record. Data-driven approaches keep refining how these compounds get made, analyzed, and incorporated so supply stays stable, safe, and satisfying for both producers and consumers alike.
What is N-Propyl N-Butyrate used for?
Long chemical names have a way of making people glaze over. N-Propyl N-Butyrate sounds like something only a chemist would care about, but this colorless liquid changes the way everyday things taste and smell. I’ve spent years working around food production, and flavors never just “happen”—manufacturers rely on molecules like this to put the right note in a berry yogurt or add sweetness without sugar. Few people realize how many of the flavors enjoyed daily spring from such ingredients.
The Hidden Flavor Builder
In the lab, N-Propyl N-Butyrate draws a crowd for its distinct fruity aroma. It mimics the essence of pineapple or pear, and that’s no accident. Flavorists and perfumers reach for this ester to bring high-impact notes to candies, drinks, ice cream, and baked goods. Often, “natural flavor” on a label means a blend of compounds like this one, whether made from plant fermentation or built from scratch. I learned early in my career that using too much dulls a product, while the right touch keeps a strawberry sparkling or a soft drink refreshing. This chemical strikes that balance, building complexity where nature fell short or faded in storage.
Industrial Roles Beyond the Kitchen
Perfume makers aren’t the only ones using these fruity notes. N-Propyl N-Butyrate works well as a solvent for resins and cellulose derivatives. Factories lean on it for coatings and inks, since it evaporates cleanly and leaves little residue. I remember watching a batch of printing ink thicken perfectly thanks to careful use of this solvent. The formula would have stalled without it, clumping instead of coating smoothly. This shows its versatility—one day in candy, another in packaging facilities that print millions of snack wrappers each hour.
Focus on Safety and Quality
Safety shapes every stage of producing and using such chemicals. The food industry sets strict rules for flavoring agents. Each batch must meet specific purity standards, and traceability matters if manufacturers want to sell worldwide. Growing global concern about allergens and food fraud pushes companies to document sources and batch histories. I’ve seen rigorous audits in plants, with inspectors checking supply chains before any case reaches grocery shelves. On the industrial side, workers use protective gear and ventilation, since inhaling vapors or splashing on skin could irritate. Good ventilation, regular chemical safety training, and accessible first aid can cut down on workplace mishaps.
Looking Ahead: Balancing Taste, Safety, and Sustainability
The push for “natural” products brings demand for new ways of making flavor compounds. Biotechnologists use yeast or bacteria to produce N-Propyl N-Butyrate in a lab, cutting down on harsh solvents or reliance on petrochemical feedstocks. This shift can lower greenhouse gas emissions and waste, especially if companies use renewable agricultural byproducts. Given my background, I see value here—not just for the environment, but for transparency. Telling customers how a flavor was made, and that it isn’t tied to rare fruits or petroleum, builds trust. Continued investment in safer, cleaner synthesis pays off for everyone from factory workers to the families eating the finished foods.
N-Propyl N-Butyrate might sound like just another synthetic. If you look closer, its story weaves through global industry, flavor science, and sustainability efforts, all to help companies deliver consistent products in a changing world.
Is N-Propyl N-Butyrate safe to handle?
Practical Concerns in the Workplace
Plenty of industrial labs and even flavor manufacturing facilities call on N-Propyl N-Butyrate for its fruity aroma. It brings a pleasant pear-like scent, a trait that often masks the fact this compound comes with genuine safety considerations. Most people working around industrial esters learn early that something smells good doesn’t mean it’s harmless. The sweet smell draws the nose, but caution remains the best partner in any workspace handling chemicals like this.
Known Hazards and Exposure Risks
Direct contact with N-Propyl N-Butyrate can irritate skin and eyes, sometimes causing lasting discomfort. If a spill splashes on bare hands, redness or even blistering may show up. Breathing in the vapor over a long shift might bring headaches and some dizziness. At higher concentrations, this compound can sting the lungs or cause a scratchy throat. I’ve seen coworkers ignore its low hazard rating on a material safety data sheet, thinking it safe as long as it doesn’t burn or fume. That casual attitude led to ruined afternoons and unnecessary first-aid.
This ester burns easily. An open flame, a spark near a solvent drum, or a poorly ventilated workbench brings the kind of trouble best avoided. People working in paint labs or flavor houses share stories about small fires starting from just a few spilled milliliters, especially if containers stay open or wrists slip. No amount of fondness for its fruity aroma is worth a late-night emergency cleanup.
Protective Measures That Actually Work
Rubber gloves and nitrile aprons become everyday wear for anyone working with N-Propyl N-Butyrate beyond a classroom demonstration. Tight-fitting goggles protect against the accidental splash while pouring to smaller bottles. Fume hoods aren’t just for stronger acids—esteemed industrial hygienists recommend their use with most esters, especially with repeated exposure.
Good ventilation in processing and research spaces saves time and health. Proper air changes and local exhausts prevent vapor buildup and clear out the scent before it settles in your lungs. From personal experience, I hold my breath topping off containers, but nothing beats an exhaust system that works as designed. Fire extinguishers rated for alcohol and solvent fires (Class B) should stay in reach. Every safety training, the instructor impresses how spilled esters spread quickly—mopping them up with sand or absorbent pads goes much farther than water.
Training and Culture Around Chemical Handling
A workplace that builds real, practical training into the routine makes a world of difference. Reviewing the labels, storing containers away from open flames, and preventing cross-contamination do not depend on company size or budget. A supervisor who insists on safety shows everyone pays attention to the details for a reason. From summer interns to seasoned chemists, no one skips the gloves or leaves the lid off a drum. I’ve coached young technicians, and I tell them: even a mild skin reaction teaches you more about the necessity for protection than a week of online courses.
Solid Steps Toward Safe Use
Regulators and industry bodies, such as OSHA and the European Chemicals Agency, publish recommendations for handling esters like N-Propyl N-Butyrate. Following those rules is straightforward—wear suitable gloves, use goggles, ventilate the work area, and store the material correctly. The stories shared in breakrooms carry weight, too. Most people learn safety through the shared wisdom of mishaps and the discipline of respecting every chemical, no matter how sweet it smells.
What are the storage conditions for N-Propyl N-Butyrate?
Everyday Experience with Chemical Storage
N-Propyl N-Butyrate sits in the company of volatile esters. Anyone who’s spent time in a laboratory, whether in the food and fragrance world or a classic chemistry lab, knows you respect the quirks of anything with a fruity scent and a flashpoint below 100°C. Mistakes in storage can be costly—ruined batches, possible fires, and headaches you feel all the way to your toes. Clear knowledge and respect for safe handling keep everyone out of trouble.
Essential Facts: What Happens If You Ignore Proper Storage
N-Propyl N-Butyrate evaporates fast. Leave it open, you notice that sweet smell filling the room, but also watch precious material disappear into the air. Add a stray spark or a hot plate nearby, and you are looking at the perfect recipe for danger. This is no time for guessing. The stuff catches fire at around 37°C. In a regular storeroom during a hot summer, temperatures climb high enough to push its vapor into the danger zone.
A simple, well-ventilated, flame-proof cabinet does the job. No fancy bells or whistles—just good sense and equipment that stands up to the material. Steel shelves, closed doors, and clear labelling usually mean you bring control to potential chaos. One summer, a neighbor chemistry teacher left an ester bottle on a sunny shelf. Their clean-up effort turned into weeks of paperwork and a strong whiff that lingered like old gossip.
Keep Out Moisture and Heat
Esters like N-Propyl N-Butyrate break down with water and acids, forming butyric acid and propanol. Neither smells much better than a gym locker. If humidity gets inside a loosely closed container, you end up with a soup that’s tough to recover. Anyone who has cleaned up a spill of decomposed ester in an academic store room doesn’t forget the odor or the stress. Every label, lid, and gasket calls for regular checks.
Direct sunlight stresses containers; plastic breaks down, thins out, and then you have leaks. Metal containers rust inside when damp air or water sneaks in. Even a tiny pinhole messes up stock and safety. This chemistry really counts on dry, stable spaces. Simple tricks help: silica gel packets near the bottles, storage in dry rooms, and careful log-ins at each inventory check.
Fire, Fume, and Friendship: Respect for Rules
Local fire regulations and building codes don’t just exist to please inspectors. Safety sheets for N-Propyl N-Butyrate—drawing from trusted resources like PubChem and science handbooks—insist on spark-free fridges for larger storage, and clear escape aisles. No smoking signs mean business. Responding with speed and honesty about a spill or ruined bottle prevents larger accidents.
Workers and community members count on clear rules and practices for the sake of health and focus. Simple training—how to use a spill kit, where to find the fire blanket, how to check a chemical label—keeps the workplace calm and confident. Knowing the limits of ventilation and investing in regular equipment checks always pays off.
Steps for Smarter Storage
Cool and dry storage wins every time. Keep N-Propyl N-Butyrate in small, clearly labelled original containers, tight-fitting lids, away from oxidizers, acids, and sources of ignition. Fridges for organics need proper seals and ignition-proof switches. Chemistry teachers and industrial operators alike have stories proving the worth of these simple, common-sense steps.
My own storage routine comes down to respect for the material, a careful hand, and regular check-ins. You store it right, you save money, time, and plenty of headaches.
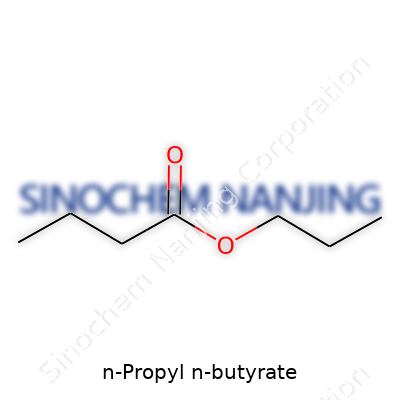
What is the chemical formula of N-Propyl N-Butyrate?
Understanding N-Propyl N-Butyrate
N-Propyl N-Butyrate, sometimes showing up in labs and food flavors, sparks interest because it brings together two familiar groups: propyl and butyrate. You look at its name and see a pattern—propyl hints at a three-carbon group, and butyrate points to a four-carbon chain that started as butyric acid. Combined, this molecule makes a pretty simple ester.
The Chemistry That Connects Us
Getting to the chemical formula means following the building blocks. Start with butyric acid, which is C4H8O2. Swap the hydrogen on the acid group for a propyl group—just as you would in a classic esterification reaction. That propyl group brings three more carbons and adds seven hydrogens. Put it together: you get C7H14O2.
Why It Matters
In the world of flavors and fragrances, N-Propyl N-Butyrate earns its keep. Its scent leans toward pear and pineapple—a sweet, fruity note that quietly helps lift fruit flavors in hard candy, drinks, and other foods. It’s not just about the taste, though. Chemists turn to this compound for research into reactions, especially in organic synthesis. Having a clear formula—C7H14O2—lets professionals track its purity and safety.
On a more personal note, N-Propyl N-Butyrate reminds me of the link between simple molecules and memorable moments. The smell of certain candies, the tang of tropical drinks—they all weave back to esters like this one. That small connection plays a role in why we feel nostalgia or pleasure from foods that feature such ingredients.
What Science Says About Safety
Consumers rely on experts to guarantee safety in food and fragrance. Food chemists stay vigilant about the compounds that go into products. Regulatory agencies, such as the US Food and Drug Administration, provide guidelines after reviewing data on toxicity and human health. For N-Propyl N-Butyrate, studies have shown it breaks down easily in the body, thanks to our enzymes. In routine food use, this molecule doesn’t show health warnings at concentrations used for flavor enhancement. This comes from peer-reviewed science, not marketing hype.
Challenges and Solutions
There’s always a need to monitor quality and sourcing. Whenever synthetic molecules join the food chain, there’s possible risk of contamination or misuse. To address that, food producers and chemical suppliers invest in frequent lab testing. Advances in chromatography make it easier to spot impurities or hazardous byproducts. Strong regulations further push companies toward transparency and accountability.
Sustainability calls for new pathways, too. Petroleum-based chemistry has often dominated ester production, which means chemists search for bio-based routes. By turning to renewable feedstocks or green catalysts, manufacturers reduce their environmental footprint. Science-driven problem solving keeps both people and the planet in mind.
Unlocking the Value in Everyday Chemistry
N-Propyl N-Butyrate sits at the intersection of research, flavor design, and responsible manufacturing. Its formula, C7H14O2, connects us back to fundamentals in organic chemistry while also reminding us why authenticity and safety matter. As regulations become stronger and technology evolves, focus stays on delivering high standards in every finished product.
Does N-Propyl N-Butyrate have any specific hazards?
Looking at the Full Picture
N-Propyl N-Butyrate pops up in laboratories, industrial plants, and sometimes as a flavor ingredient. It brings a rich, fruity scent that can make it valuable in perfumery or as a solvent. The story readers rarely get lies in the handling risks hidden beneath this pleasant surface.
The Hazards Hiding in Plain Sight
Spending time in the field has shown me that routine chemicals can surprise those who underestimate them. N-Propyl N-Butyrate ranks as a flammable liquid. Left near heat or open flames, it reacts quickly and burns clean but fiercely. Many lab fires trace back to solvents like this left in the wrong place at the wrong time.
The vapors can sneak up on you. Somebody might open a bottle in a closed room and catch a faint, sweet smell. Not much later, headaches emerge, maybe dizziness, sometimes even disorientation. Long exposure without ventilation leaves people blaming fatigue or poor breakroom coffee, never suspecting what hung in the air. Toxicity doesn’t always announce itself loudly. Small spills, poorly capped containers, and lackluster ventilation work together, turning minor mistakes into bigger hazards.
Skin contact brings another set of problems. It doesn’t take much for irritation to start. I’ve seen someone with a red rash after brushing up against a countertop wiped with this compound an hour earlier. Wearing gloves helps, and lab coats do more than protect from splashes; they keep lingering residue off clothing and skin.
Lessons from Real Situations
Not everyone who handles chemicals reads safety data sheets. I once watched an intern mop up a small spill, barehanded, before anybody could stop him. The reaction took minutes: stinging, redness, and quick regret. Even one-time exposure can provoke discomfort. Part of the reason comes from N-Propyl N-Butyrate’s ability to cut through natural skin oils, leaving skin defenseless and dry.
Indoors, vapors concentrate near floors because the compound is heavier than air. Picture a bottle tipped over near ground level. Invisible fumes begin creeping along baseboards and under benches, building up until someone flips a switch, and a spark does the rest. Disaster often starts this way. My own habits changed after seeing what poor airflow and carelessness could do.
Safer Steps Forward
Clear rules, real training, and practical layouts form the front line against risk. Simple choices make powerful differences: storing chemicals in a cool, ventilated area, sealing containers tightly, avoiding all sources of sparks or static. Chemical-resistant gloves and eye protection belong on everyone in the workspace. Education works best through stories and repetition, not by rote memos or posters that get ignored.
Beyond workplaces, anyone bringing chemical flavors home for DIY projects or basement labs should keep their guard up. Dispose of rags, paper towels, or cleaning cloths that soaked up this compound in approved containers—never toss them into trash bins where vapors linger and fires can start. Open windows, use fans, and give fumes nowhere to settle if spills happen.
The Bottom Line
Working with N-Propyl N-Butyrate doesn’t demand fear—just respect. Fire and health risks live alongside benefits, so each bottle deserves attention. Direct experience and attention to detail turn potential hazards into manageable risks. Read labels, trust good habits, and remember that vigilance on a normal day keeps the headlines away.
| Names | |
| Preferred IUPAC name | propyl butanoate |
| Other names |
Butyric acid n-propyl ester n-Propyl butanoate Butanoic acid propyl ester Propyl butanoate Propyl n-butyrate |
| Pronunciation | /ɛn-ˈproʊpɪl ɛn-ˈbjuːtɪreɪt/ |
| Identifiers | |
| CAS Number | *590-01-2* |
| Beilstein Reference | **1209246** |
| ChEBI | CHEBI:87477 |
| ChEMBL | CHEMBL3184791 |
| ChemSpider | 12711 |
| DrugBank | DB14106 |
| ECHA InfoCard | 03a447b1-2a3e-4367-9f38-5ea6f73f8adf |
| EC Number | 203-567-4 |
| Gmelin Reference | 7735 |
| KEGG | C18670 |
| MeSH | D015150 |
| PubChem CID | 8095 |
| RTECS number | UF9625000 |
| UNII | J0WWH5A0U9 |
| UN number | UN2541 |
| CompTox Dashboard (EPA) | DTXSID1020648 |
| Properties | |
| Chemical formula | C7H14O2 |
| Molar mass | 130.186 |
| Appearance | Colorless liquid |
| Odor | Fruity |
| Density | 0.868 g/mL at 25 °C (lit.) |
| Solubility in water | 1.6 g/L |
| log P | 1.97 |
| Vapor pressure | 0.43 mmHg (20 °C) |
| Acidity (pKa) | pKa ≈ 25 |
| Magnetic susceptibility (χ) | -7.22e-6 |
| Refractive index (nD) | 1.418 |
| Viscosity | 1.8 mPa·s (25 °C) |
| Dipole moment | 2.55 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 365.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -399.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3534.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 48 °C |
| Autoignition temperature | 430 °C |
| Explosive limits | Upper: 7.4% ; Lower: 1.1% |
| Lethal dose or concentration | LD50 (oral, rat): 9350 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 8300 mg/kg |
| NIOSH | WA8575000 |
| PEL (Permissible) | PEL (Permissible) for N-Propyl N-Butyrate: 25 ppm (125 mg/m3) as an 8-hour TWA (OSHA) |
| REL (Recommended) | 5 ppm (20 mg/m3) |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Butyric acid Propyl alcohol Ethyl butyrate Isobutyl butyrate Methyl butyrate |

