N-Propyl Isocyanate: Chemical Progress, Properties, and Future Possibilities
Historical Development
Chemistry keeps finding ways to squeeze more out of molecules, and the story of N-Propyl Isocyanate stretches back to the broader pursuit of isocyanate compounds. Commercial development surged in the postwar era. Back then, chemists explored different alkyl isocyanates, drawn by their reactivity and possible roles in growing industries such as pharmaceuticals and materials science. The late 20th century saw scale-up of isocyanate production for polyurethane foams and specialty chemicals, paving the way for better yields, purities, and handling protocols. N-Propyl Isocyanate became recognized for its unique niche among isocyanates: less volatile than methyl or ethyl isocyanate, but reactive enough for a toolbox of synthetic routes. Much of the fundamental data about its behavior emerged from lab work in the 1960s and 70s, where researchers tested its chemistry and pressed on the boundaries between safe laboratory fumbling and real-world scaling.
Product Overview
Anyone who handles N-Propyl Isocyanate gets introduced to its clear, colorless, pungent liquid form. Its appeal comes from reliable reactivity with nucleophiles, which drives its use in custom synthesis. What sets it apart from its siblings (like methyl isocyanate) is slightly lower volatility and a sharper boiling point, making it somewhat easier to manage for chemists who demand control and predictability. Chemical vendors supply it at high purity for specialty sectors: medicinal chemistry, agrochemical intermediates, and specialty polymer chains. The main selling points line up with reactivity, handling profile, and a favorable balance of boiling and volatility for process safety.
Physical & Chemical Properties
In labs and factories, details matter. N-Propyl Isocyanate carries a molecular weight of 99.13 g/mol, boils at about 88–90°C, and forms a vapor dense enough to demand solid ventilation. Its flash point clocks in at roughly 15°C, so it doesn’t wait long to ignite around open flame or static electricity. Its density—hovering around 0.89 g/mL at room temperature—sets it apart from water, guiding how it separates in spills and washes. Solubility in water runs low, so runoff pools and lingers, raising stakes for environmental concerns. Chemically, its isocyanate functionality opens the door to a range of polymerization and addition reactions. Experienced handlers pay attention to pH, temperature, and airflow, knowing that one slip can lead to dangerous decomposition or release of toxic gases.
Technical Specifications & Labeling
Product quality translates directly to laboratory results. Suppliers deliver N-Propyl Isocyanate at purities above 98%, with labeling that follows GHS and REACH protocols. Labels mark hazard statements, pictograms warning about respiratory sensitization and acute toxicity, and tightly controlled lot numbers. SDS sheets flag both acute and chronic hazards, and spec sheets spell out purity, refractive index, water content, and batch traceability. Packaging often comes in amber glass to block UV light and includes PTFE-lined caps to stop leaks and vapor migration. Workers scan QR codes or barcodes, tying shipments directly to batch analyses and regulatory compliance records on factory floors or within chemical inventory software.
Preparation Method
N-Propyl Isocyanate usually gets synthesized via phosgenation or from carbonylation of related precursors. The traditional route relies on the reaction between n-propylamine and phosgene under controlled temperature and pressure. This process produces HCl as a byproduct, handled with scrubbers and neutralization units. Modern approaches, for safety and compliance, look for non-phosgene paths such as using diphosgene or triphosgene, which offer finer control and less risk of large-scale release. No matter the method, operators work with closed reactors, proactive pressure monitoring, and layered PPE, since exposure to isocyanates often carries lifelong respiratory consequences.
Chemical Reactions & Modifications
The functional group in N-Propyl Isocyanate —N=C=O— doesn’t waste time finding partners. It reacts sharply with amines to form ureas, reacts with alcohols to make urethanes, and combines with water to evolve CO2 and n-propylamine. Organic chemists lean on this for custom synthesis, often adding N-Propyl Isocyanate late in the route to avoid premature reaction. In pharmaceutical research, modifications include attaching various acyl or aryl groups on the nitrogen, swinging the final product’s solubility or reactivity. In polymer science, tweaking the isocyanate chain builds new urethane networks or crosslinked resins, each with its own mechanical and thermal properties. This reactivity makes N-Propyl Isocyanate a go-to reagent for creative synthesis, but also a source of challenge when designing safe process runs.
Synonyms & Product Names
Different catalogs and papers may refer to N-Propyl Isocyanate as Propyl Isocyanate, 1-Isocyanatopropane, or n-Propylcarbyl Isocyanate. CAS Registry Number 1134-73-2 sticks as the anchor ID across regulatory and chemical safety documents. Some chemical brands assign internal item codes or abbreviate as n-PrNCO. These synonyms matter for procurement, literature searches, and regulatory tracking, since mix-ups can lead to costly delivery mistakes or unintended process changes.
Safety & Operational Standards
Direct contact spells trouble. N-Propyl Isocyanate irritates skin, eyes, and lungs, and it can trigger asthma-like sensitization after repeated or heavy exposures. Most facilities with experience in isocyanate use closed handling systems, negative-pressure gloveboxes, local fume hoods, and cartridge respirators when transferring open liquids. Eyes, skin, and lungs need full coverage: face shields, nitrile gloves, Tyvek suits, and frequently checked air monitors. Engineers keep spill kits at hand, along with absorbents resistant to isocyanate reactivity. Training sticks out as the weak link—a skilled chemist respects these dangers, and companies that cut corners pay in injuries and fines. Waste handling routes channel residues to incineration and regulated disposal. Risk assessments run before any new process kicks off.
Application Area
Most users bump into N-Propyl Isocyanate in research settings, where it gives medicinal chemists an edge in synthesizing ureas and carbamates that anchor new drug candidates. Its role in competitive agrochemical design helps researchers attach isocyanate-derived tethers for improved delivery or metabolic stability. R&D teams in high-performance materials build prepolymers and cross-linkers, threading N-Propyl Isocyanate into specialty coatings and engineered plastics. Industrial users value its controlled reactivity compared to smaller isocyanates, reducing volatility and accidental emissions in scaled equipment. Its use typically stays niche, but where precision matters in the lab or pilot-scale process, this compound delivers.
Research & Development
R&D stories draw on N-Propyl Isocyanate’s strengths in medicinal chemistry and process innovation. Chemists building libraries of potential enzyme inhibitors or signaling blockers regularly add it as a key step, using its reactive handle and predictable product profiles. Collaboration between academic labs and industry leverages its use for custom labeling of peptides, production of isotopically labeled standards, and one-pot syntheses that cut down purification steps. Teams in green chemistry push for process improvements that avoid hazardous reagents or find milder routes for similar isocyanate intermediates. Blockchain and AI-driven inventory tools help reduce waste and optimize real-time sourcing. As digital labs and smart reactors become common, feedback from early-career scientists shapes improvements in safety and efficiency.
Toxicity Research
N-Propyl Isocyanate, like other low-molecular-weight isocyanates, poses serious risks for those with direct or repeated contact. Animal studies and occupational health surveys both point to acute respiratory and dermal toxicity, with long-term sensitization a regular finding. Inhalation at low levels triggers coughing, shortness of breath, and in severe cases, chemical pneumonitis. Chronic exposure links to occupational asthma—a reality that drives some facilities to overengineer their handling protocols. Toxicology teams delve into downstream metabolites and repair mechanisms after exposure, tracking not only acute effects but also the slow progression of immune system changes. Safe exposure limits trend lower over time, with regulators often playing catch-up to new evidence from human and animal studies.
Future Prospects
Trends in the specialty chemical world point toward smarter, safer, and more sustainable isocyanate production, with N-Propyl Isocyanate’s future tied to these shifts. Demand rises on both the synthesis and the control sides, mixing calls for higher purity and lower residual impurities with sharper focus on closed-loop production and green chemistry metrics. Lab automation and smart reactors improve consistency and safety, letting researchers spend less time on basic syntheses and more time on creative molecular designs. In parallel, deeper toxicology research and stricter environmental controls will shape supply chains and handling standards. As digital tracking, advanced sensors, and process AI chip away at uncertainty, chemists and engineers aim for cleaner, safer, and more innovative chemical routes—N-Propyl Isocyanate stays woven into the story, linking old-school organic chemistry to the next era of precision synthesis.
What is N-Propyl Isocyanate used for?
Understanding the Role of N-Propyl Isocyanate
N-Propyl isocyanate pops up in conversations among chemists and manufacturers who work closely with isocyanates. Most folks don’t realize that every day, products around them rely on raw materials like this one to deliver the features people take for granted. N-Propyl isocyanate serves as a building block in things like pharmaceuticals, agrochemicals, and specialty materials. Anyone with a background in lab work or chemical engineering will likely remember their first encounter with the unmistakable sharp odor—it leaves an impression.
This compound tends to come up most in synthetic organic chemistry. Let’s say a researcher is trying to create a custom molecule for a new medication or crop treatment. N-Propyl isocyanate steps in to bring nitrogen, carbon, and a functional group to the table that helps build more complex molecules. In some cases, it reacts with amines to produce substituted ureas—these compounds show up in herbicides and antidiabetic drugs. Researchers value it for its reliability. You get a predictable result, which matters when the stakes are high.
Risks on the Job and in Production
Handling N-Propyl isocyanate brings challenges that shouldn’t be ignored. I remember being instructed to check every valve and seal before starting any reaction involving isocyanates. Skin contact or inhalation can cause irritation or trigger allergic reactions. The safety data sheets don’t exaggerate—proper gloves, eye protection, and fume hoods turn a risky experiment into everyday lab work.
In manufacturing, larger volumes magnify the risks. It takes strong protocols, staff training, and a commitment from management to invest in proper containment. The industry has faced scrutiny in the past after leaks or spills led to acute exposure incidents. Lessons learned from events like these led to process improvements: better ventilation, real-time air monitoring, and strict access control in facilities handling volatile chemicals like N-Propyl isocyanate.
Connecting the Dots to Your Daily Life
Many consumers never hear the term “N-Propyl isocyanate,” but its presence lingers in footprints left by science and industry. Foam in car seats, the glues holding shoes together, pills that keep blood sugar stable—behind each of these sits a quiet dance of chemistry where isocyanates make things possible.
In agriculture, crop protection products depend on carefully designed molecules. Isocyanate chemistry supports the synthesis of herbicides and fungicides that target pests and diseases without wiping out everything green. In the pharmaceutical world, flexibility in chemical design allows scientists to tweak drug candidates for higher effectiveness or fewer side effects, often leaning on the versatility of isocyanates.
Supporting Safe and Responsible Use
Knowledge makes the difference between responsible progress and accidents waiting to happen. Those using N-Propyl isocyanate should push for ongoing training, clear safety signage, and regular audits. Industry leaders have started investing in greener alternatives and process improvements to cut down hazardous waste and emissions. That reduces environmental impact and improves worker health in the long run.
Regulators play a heavy role, setting exposure limits and holding companies to task through inspections. Community engagement also matters—neighbors living near manufacturing sites deserve transparency about what chemicals get used and what steps keep them safe. These measures don’t just protect people today; they help set a standard for the chemists and engineers to come.
What are the safety precautions when handling N-Propyl Isocyanate?
Risk Isn’t Just Theory: N-Propyl Isocyanate Is Real Stuff
Years of working around chemicals have a way of teaching you what safety signs on a drum really mean. N-Propyl Isocyanate isn’t a household name, but its hazards belong on every chemist’s radar. Even one whiff tells you it’s not something to shrug off. Short-term exposure can burn your eyes, throat, and lungs. Extended contact gives skin rashes that linger for days. Sometimes, folks start wheezing and coughing and have trouble breathing, just because a seal leaked on the fume hood.
Skin, Lungs, Eyes: Guard Every Gate
Some workers trust thin latex gloves or regular safety glasses, thinking those are enough for jobs around harsh chemicals. Not here. Splash-resistant goggles, nitrile gloves, and a face shield make a real difference. Lab coats must cover arms, and shoe covers go over sneakers. N-Propyl Isocyanate can pass through some materials, so don’t cut corners by grabbing worn-out gloves from the bottom of the box.
I’ve seen smart people get careless. One guy wiped up a tiny spill with a bare hand, thinking a little soap would fix it. His skin turned red and itchy almost right away. Even worse, spills can make toxic vapors that standard room fans won’t push away. Good local exhaust ventilation matters. Real fume hoods save lungs, especially for weighing or transferring this stuff.
Preparation: More Than Just a Safety Sheet
Reading a safety data sheet is just one piece of the puzzle. Real preparation means knowing your emergency showers work, fire extinguishers stand nearby, and every person in the building understands what to do if something goes wrong. I always double-check my eyewash station is clear of clutter and test that water once a week. Would you want to scramble for the nearest working shower with chemicals in your eyes?
Respecting the Chain: Storage and Disposal
This chemical doesn’t store well with acids, water, or other reactive compounds. Keep it in a cool, dry place with clear labeling. More than once, I found mismatched lids or mysterious containers, which can lead to disaster if someone adds it to the wrong waste drum. Use sealed, compatible containers and make sure lids actually fit. Labeling should shout the hazards, not hide them in tiny print.
Don’t Assume—Train for the Worst
Just because you’ve worked with N-Propyl Isocyanate before doesn’t mean the next batch acts the same. Fresh containers sometimes leak. Transfer lines dry out and crack. I’ve seen incidents start because someone forgot the basic rule: treat every chemical as if it’s dangerous. Annual safety drills help. So does open conversation in the lab or plant. People avoid mistakes when the right procedures feel second nature, not like a school quiz they forgot to study for.
A Better Way Forward
PPE supplies cost less than a hospital bill. Good ventilation saves lives, not just comfort. Sharing close calls and lessons learned—without blaming—creates a work culture where new hires respect N-Propyl Isocyanate’s risks right away. Small steps, taken seriously, protect every worker. I’ve seen accidents, and I remember the faces. Nobody forgets firsthand why safety rules matter after that.
What is the chemical formula and structure of N-Propyl Isocyanate?
Getting to Know N-Propyl Isocyanate
Few synthetic chemicals grab attention like isocyanates. These compounds shape the backbone of some of the world’s most impactful industrial products. N-Propyl isocyanate stands out among them. Its chemical formula—C4H7NO—points toward a specific arrangement of atoms. The structure, written out as CH3CH2CH2NCO, makes its identity clear. That NCO group sitting at the end gives this molecule its telltale reactivity.
What Makes Its Structure Unique
To break it down, the backbone comes from three carbon atoms in a chain, making up the propyl group. The isocyanate group (-N=C=O) attaches to the end carbon. Chemists often draw N-Propyl isocyanate with stick figures: a straight chain linking to a tightly bonded nitrogen and carbonyl oxygen. This shape leads to an uneven charge distribution. Polar bonds set the stage for quick reactions, especially with water or anything containing hydrogen.
Why Does It Matter?
I’ve watched small manufacturing shops handle isocyanates with extra caution. A whiff of the stuff can make eyes water. This stems from the reactive isocyanate group. It latches onto moisture so fast that even the air’s humidity can trigger strong responses. In workspaces where coatings, adhesives, and foams take shape, knowing the exact chemistry pays off. If someone forgets the aggressive nature of this chemical, the results can mean unsafe work environments or failed batches.
Authorities like the Occupational Safety and Health Administration (OSHA) pay keen attention to compounds like N-Propyl isocyanate. Short-term exposure commonly leads to symptoms like coughing, breathing trouble, and even asthma-like conditions in some workers. For someone new to chemical handling, that hard-to-pronounce formula turns into more than a tongue-twister—it becomes a vital safety lesson.
Responsible Use Calls for Clarity
Most reading about chemicals in the news crave more than jargon. Genuine experience in labs, whether during long hours in college or later in industry, shows how formulas shift from textbook facts into real-world risks and solutions. Protective gloves, goggles, and proper ventilation guide safe handling. Engineering controls prevent vapors from ever reaching lungs. Not everyone trusts just labels or fact sheets. Seeing the three-carbon chain and that NCO group in person leaves a much more lasting mark in memory.
Where Chemistry Meets Regulation and Progress
The world’s appetite for new materials keeps demand for specialized chemicals alive. Product designers count on properties of isocyanates—like N-Propyl isocyanate’s sharp reactivity—to create heat-resistant foams or fast-setting adhesives. At the same time, anyone invested in safety watches for alternatives and improvements. Closed systems, better operator training, and direct substitution with less hazardous reactants tackle health risks while allowing industries to keep pace with modern needs.
Through every step, grounding these changes in solid chemistry understanding ensures workers, bystanders, and the environment don’t shoulder unnecessary risk. Each molecule’s structure, from the humble propane backbone to the reactive NCO group, shapes everything from product quality to personal safety.
How should N-Propyl Isocyanate be stored?
Why Safety Starts With Where You Put the Drum
Anyone who’s handled chemicals long enough knows the ugliest mistakes often come from cutting corners. N-Propyl isocyanate isn’t the sort of compound to take lightly. This is one of those cases where experience, training, and pure respect for chemistry mix together. You smell trouble, literally. The compound’s pungent odor is only one warning sign. A sniff alone reminds you it belongs nowhere near a casual shelf or crowded workspace.
Real World Risks Cannot Be Ignored
With my years in chemical labs, I saw what can go wrong—and how quickly—if you treat volatile isocyanates like standard solvents. Most folks worry about fire, and for good reason. N-Propyl isocyanate flashes at about 27 °C (80 °F). Mild ambient heat, sparks, or even static charge can tip things into chaos. If it leaks, reacts with water, or gets into the air, don’t expect just a nasty smell; it brings health hazards like respiratory irritation and potential sensitization. People think a fume hood is always enough. With this chemical, it might not forgive a single exposure.
No Room For Complacency—What the Science Demands
Storing N-Propyl isocyanate in a dry, ventilated, and flame-proof area isn’t just about obeying regulations. Moisture triggers dangerous reactions, creating carbon dioxide and pressure until a sealed drum booms or bursts open. I once walked a warehouse, cardboard soft with humidity, and spotted crusty residue around sealed drums. The manager shrugged, unfamiliar with isocyanate hydrolysis. I asked who checks for leaks, who tests for vapors, who logs temperature. Just blank faces.
Just tucking it on the chemical rack can turn a minor spill into a facility-wide evacuation. Fire codes, insurance demands, and lab protocols mark these storage areas with big warnings, sometimes chains and locks. Insurance companies don’t make exceptions—too many claims trace back to sloppy storage.
Storage Solutions: Practical Tips from the Field
Concrete floors with secondary containment are your friends. Steel safety cabinets rated for flammables provide the best safeguard, far better than open shelving or improvised corners. Temperature control matters—think of every warm storage area as an invitation for vapor release or even combustion. Ventilated spaces, never sealed closets: more airflow, less buildup.
I learned to double up: proper labels facing out, containers always sealed tight, and desiccant packs nearby to fight humidity. Never stack heavy drums or let them sit where forklifts or foot traffic threaten. No water anywhere close—an accidental splash turns into an emergency.
Beyond Checklists—Why Experience Matters
Training the team pays off more than another inspection checklist. I’ve watched new techs freeze when confronted with a leak. Anyone working with N-Propyl isocyanate needs hands-on hazard drills, not just signs on the wall. Spill kits, eye wash stations, immediate access to respirators—those basics should never slip below top priority.
It’s tempting to ease restrictions or “temporarily” store things the wrong way, especially in busy labs. The reality: safe storage of hazardous chemicals isn’t negotiable. Too many stories end with an anecdote about the day someone skipped a step. Safety depends on the habits you build, the risks you respect, and the lessons you refuse to ignore.
What are the potential health hazards of exposure to N-Propyl Isocyanate?
Walking into Chemical Risks
Breathing easy never feels as simple around chemicals like N-Propyl Isocyanate. This stuff shows up in industrial settings, where workers shape plastics, coatings, and adhesives. The sharp, acrid smell alone sends a loud message—don’t shrug off the warning signs. Many folks don’t realize how fast trouble can begin just by breathing in the vapors.
Breathing and Skin—The Front Lines of Exposure
Everyday people might feel detached from the topic of workplace chemical safety, but plenty of cases end with someone rushing to a sink or medical tent. N-Propyl Isocyanate can irritate the airways, leaving your nose running and your throat scratchy within minutes. Inhaling a decent amount could even make you wheeze or trigger asthma, especially if your lungs grew sensitive over years of dust and fumes. One study by OSHA classified isocyanates among substances most likely to provoke occupational asthma. Skin opens another risky pathway—this chemical can seep in and leave you with burns or rashes that don’t clear up quickly. Toss in the fact that some people develop allergies to isocyanates after just a few exposures, and you see trouble snowballing.
Eyes and Long-Term Effects
Eyes often pay the price in environments where tiny droplets hang in the air. Get a whiff too close or a splash on unprotected skin, and burning sets in quickly. Redness, blurry vision, even chemical conjunctivitis—all real possibilities among workers dealing with isocyanates. Repeated exposure, even at low levels, can stack up. Industrial health surveys show increased asthma rates and eczema among plant employees using these chemicals every day. Long-term risks extend beyond allergy and asthma. There’s early research asking if chronic low-dose exposure could play into immune or nervous system problems down the road, though hard links need more science.
Personal Experience—Why Prevention Beats Treatment
Years ago, I visited a site where safety slipped after a ventilation fan broke. Workers noticed the sweet-bitter smell, but many pushed on, ignoring early coughing fits or stinging eyes. It only took an afternoon for some to develop tightness in their chests—a reminder that simple steps, like checking exhaust systems, can spare a week or two off work and trips to the clinic. Once in a while, old-timers in industry will shrug at mild exposure, but there’s no badge of honor in suffering an asthma attack or repeated skin rashes. From personal conversations, most regret ignoring symptoms.
Building Healthier Habits at Work
Preventing harm calls for a handful of steady habits. Wear proper gloves and goggles, use respirators when working in confined spaces, and remember that local exhaust fans pull dangerous vapors away fast. Supervisors who walk the floor looking for leaks or engineering problems help more than safety posters ever could. Proper training does more for safety than rules alone. Learning how fast symptoms show up, and cutting short exposure, protects against lasting harm. OSHA and NIOSH guidelines recommend not just personal gear, but medical checkups and exposure monitoring. These steps keep workers healthy and reassure the families waiting at home.
The Takeaway—Respect the Risks
Few chemicals trigger immediate symptoms like N-Propyl Isocyanate, making it a stark reminder for companies and workers to take protection seriously. Factory floors don’t have to feel dangerous, and health issues tied to exposure are preventable. Simple changes—better ventilation, protective clothing, mandatory training—make a difference. Small efforts can keep lungs clear, skin unscarred, and nerves steady for years to come.
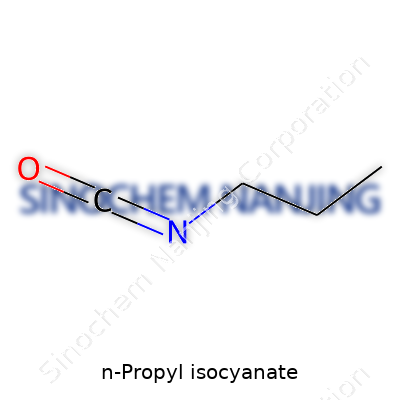
| Names | |
| Preferred IUPAC name | N-propyl isocyanate |
| Other names |
Isocyanic acid, n-propyl ester n-Propylisocyanate 1-Isocyanatopropane |
| Pronunciation | /ɛn-ˈprəʊpɪl aɪsə(ʊ)ˈsaɪəneɪt/ |
| Identifiers | |
| CAS Number | 1118-71-4 |
| 3D model (JSmol) | ``` isocoCCC ``` |
| Beilstein Reference | 1209247 |
| ChEBI | CHEBI:51766 |
| ChEMBL | CHEMBL16326 |
| ChemSpider | 141112 |
| DrugBank | DB04209 |
| ECHA InfoCard | 100.005.800 |
| EC Number | 203-714-2 |
| Gmelin Reference | 8409 |
| KEGG | C14322 |
| MeSH | D017346 |
| PubChem CID | 79596 |
| RTECS number | UK8575000 |
| UNII | 2PV08IF084 |
| UN number | UN2480 |
| CompTox Dashboard (EPA) | CATSUBST-00004524 |
| Properties | |
| Chemical formula | C4H7NO |
| Molar mass | 85.11 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent odor |
| Density | 0.857 g/mL at 25 °C (lit.) |
| Solubility in water | Reacts slowly |
| log P | 0.86 |
| Vapor pressure | 3 mmHg (20 °C) |
| Acidity (pKa) | 15.4 |
| Basicity (pKb) | 13.38 |
| Magnetic susceptibility (χ) | -55.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.384 |
| Viscosity | 0.613 mPa·s (20°C) |
| Dipole moment | 3.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -51.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2010.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H226, H315, H317, H319, H330, H334, H335, H410 |
| Precautionary statements | P210, P260, P261, P273, P280, P285, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P310, P337+P313, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Flash point | 12 °C (closed cup) |
| Autoignition temperature | 460 °C |
| Explosive limits | Explosive limits: 2.3–10.4% |
| Lethal dose or concentration | LD50 oral rat 640 mg/kg |
| LD50 (median dose) | 640 mg/kg (rat, oral) |
| NIOSH | NIOSH: SAJ6650000 |
| PEL (Permissible) | PEL: 0.005 ppm |
| REL (Recommended) | REL: 3 ppm (10 mg/m³) |
| IDLH (Immediate danger) | IDLH: 6 ppm |
| Related compounds | |
| Related compounds |
Methyl isocyanate Ethyl isocyanate n-Butyl isocyanate Phenyl isocyanate Isopropyl isocyanate |

